Back to Journals » Therapeutics and Clinical Risk Management » Volume 19
Clinical Utility of Deucravacitinib for the Management of Moderate to Severe Plaque Psoriasis
Authors Jin JQ , Spencer RK, Reddy V , Bhutani T, Liao W
Received 29 March 2023
Accepted for publication 15 May 2023
Published 18 May 2023 Volume 2023:19 Pages 413—423
DOI https://doi.org/10.2147/TCRM.S388324
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Garry Walsh
Joy Q Jin,1,2 Riley K Spencer,2 Vidhatha Reddy,2 Tina Bhutani,2 Wilson Liao2
1School of Medicine, University of California at San Francisco, San Francisco, CA, USA; 2Department of Dermatology, University of California at San Francisco, San Francisco, CA, USA
Correspondence: Joy Q Jin, Box 1212, Floor 01, Room 101, 2340 Sutter Street, San Francisco, CA, 94115, USA, Tel +1 415-353-7800, Fax +1 415-502-4126, Email [email protected]
Introduction: Psoriasis is a chronic, immune-mediated skin condition with significant detriments to physical/mental health. While systemic therapies are available for the treatment of moderate-to-severe psoriasis, patients can experience therapeutic failure, loss of efficacy, or medical contraindications that require other therapeutic options.
Objective: With the recent approval of deucravacitinib, a first-in-class TYK2 small molecule inhibitor administered orally for psoriasis patients, we reviewed data from randomized controlled trials (RCTs) to synthesize its clinical utility. To our knowledge, this is the first systematic review and meta-analysis of deucravacitinib comparing its clinical efficacy to placebo in psoriasis.
Methods: A literature search was conducted in PubMed (MEDLINE), Embase, and the Cochrane Central Register of Controlled Trials to identify RCTs studying deucravacitinib in human patients with moderate-to-severe psoriasis.
Results: One placebo-controlled Phase II RCT and two placebo-controlled/active-comparator Phase III RCTs were included for review. Patients (N=1953) treated with deucravacitinib 6 mg daily showed marked improvement in disease severity (Psoriasis Area and Severity Index (PASI), static Physician Global Assessment (sPGA) and quality-of-life outcomes compared to patients administered comparator (apremilast) and placebo. Clinical improvement given deucravacitinib was noted for scalp psoriasis but not fingernail psoriasis. Meta-analysis (deucravacitinib, n=888; placebo, n=466) comparing rates of clearance (sPGA 0/1) demonstrated superior efficacy of deucravacitinib compared to placebo (odds ratio, 12.87; 95% confidence interval, 8.97– 18.48; χ2=4.08, I2=51%). Deucravacitinib was well-tolerated, with similar rate of occurrence and type of adverse events reported among patients treated with placebo or apremilast at Week 12– 16. No cardiovascular events, serious infections, or lab abnormalities were noted.
Conclusion: Deucravacitinib possesses good efficacy, with no report of safety concerns associated with prior JAK inhibitors used for psoriasis. Meta-analysis demonstrated deucravacitinib’s superiority compared to placebo, indicating its promising clinical utility. Further studies are needed to observe long-term safety and efficacy, and to compare deucravacitinib to existing treatments.
Keywords: apremilast, deucravacitinib, meta-analysis, placebo, plaque psoriasis, systematic review
Introduction
Psoriasis is a chronic inflammatory disorder of the skin and joints that affects 8 million Americans and 2–3% of the population globally.1 Psoriasis has a profound impact on both the physical and psychosocial health of those affected. Patients are subject to increased risk of developing comorbid systemic disease, including cardiovascular disease, diabetes, anxiety, depression, and all-cause mortality.2
A variety of therapies are available for the treatment of psoriasis, including topical medications, phototherapy, oral and biologic agents. Oral immunosuppressants such as methotrexate and cyclosporine may be highly effective for some patients, but such treatments have significant potential for adverse effects.3 For individuals with a more severe psoriatic disease course, treatment with a systemic therapy such as a biologic agent is often required. Recent advancements surrounding new targeted agents have yielded promising results; here, we review the clinical potential of deucravacitinib (ie, BMS-986165, SotyktuTM), a new oral small molecule approved by the US Food and Drug Administration (FDA) in September 2022 for the treatment of moderate-to-severe psoriasis.
TYK2 Signaling and Psoriasis Pathogenesis
The pathogenesis of psoriasis is characterized by aberrant keratinocyte differentiation and excessive growth of the epidermis, leading to the formation of erythematous patches and plaques with thick overlying scale.4 Psoriasis pathogenesis involves a complex interplay of genetic (eg, susceptibility alleles) and environmental factors, which can combine to trigger systemic inflammatory cascades leading to disease presentation.4 While psoriatic immune dysregulation is complex and not fully elucidated, T helper 17 (Th17) cells are known to be a central component that, when aberrantly activated, produce important effector cytokines acting in a positive feedback loop to recruit additional immune cells and accelerate psoriasis development.5,6
Involvement of the interleukin (IL)-23/IL17 pathway mediates psoriasis via the activation and promotion of keratinocyte proliferation.5 Cytokines like TNF-α, IL-17, and IL-23 are the targets of biologic agents used in psoriasis.7,8 Many of these same cytokines, including IL-23, bind to type I and II cytokine receptors, which possess no inherent catalytic activity and must rely on Janus kinase (JAK) proteins to mediate their effects.7 Tyrosine kinase 2 (TYK2) is one of four members of the JAK family of proteins.7
JAK/STAT signaling refers to a system comprised of a dimeric transmembrane cytokine receptor, a pair of intracellular JAKs, and a family of Signal Transducers and Activators of Transcription (STATs).7 Upon binding of a cytokine to its receptor, a conformational change in the receptor proteins occur, leading to autophosphorylation of intracellular JAKs.7 This enables another conformational change leading to the phosphorylation of STATs, which then dissociate from the receptor complex before translocating to the nucleus and acting as transcription factors.7
TYK2 pairs with other JAK family members to mediate the signaling of IL-12 and IL-23 receptors, as well as type I IFN receptors; TYK2 inhibition leads to reduced Th17 cell polarization, increased suppressive functions of regulatory T cells, and additional downstream effects protective against psoriasis development.9–12 Given TYK2’s role downstream of current biologic targets such as IL-12 and IL-23, TYK2 inhibition may serve as a promising strategy that can address existing challenges in the treatment of moderate-to-severe psoriasis.12
Deucravacitinib
In September 2022, deucravacitinib—an oral, first-in-class, small molecule, selective allosteric inhibitor of TYK2—was approved by the FDA for the treatment of psoriasis in the U.S.13 Deucravacitinib binds to the catalytically inactive pseudokinase regulatory domain of TYK2 and stabilizes an inhibitory interaction between the regulatory and catalytic domains.13 Through this method, TYK2 is inactivated and cannot interact with other receptors to lead to downstream signal transduction.11,14 In preclinical studies, deucravacitinib was revealed to inhibit TYK2 with high selectivity and minimal off-target effects on other JAK family members,11,14 suggesting that deucravacitinib may possess an improved safety profile compared to less specific JAK inhibitors, which have been associated with hyperlipidemia, increased risk of infections, and other systemic changes.14–16
Given recent FDA-approval and promising clinical data, we aimed to investigate deucravacitinib’s clinical utility for the treatment of moderate-to-severe psoriasis. To our knowledge, no systematic review has been conducted—here, we performed a systematic review with meta-analysis to synthesize the findings from randomized controlled trials (RCTs) studying deucravacitinib for psoriasis.
Materials and Methods
As a review, all data used were non-identifiable and publicly available; institutional review board approval was not required at the University of California, San Francisco. The study protocol and design are reported according to the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols 2020 guidelines.17 A literature search was conducted in PubMed (MEDLINE), Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) in March 2023 using a combination of the terms (“deucravacitinib” OR “sotyktu”) AND (“psoriasis”).
Study Design and Eligibility Criteria
The efficacy of new psoriasis treatments is measured in clinical trials via standardized, objective disease severity metrics, including the Psoriasis Area and Severity Index (PASI) and Physician Global Assessment (PGA).18,19 These tools utilize grading scales to stratify disease severity based on clinical characteristics including body surface area involvement and degree of erythema, induration, and scaling. Given the impact psoriatic disease has on patients’ psychosocial health and quality of life, concomitant assessment of these domains with tools such as the Dermatology Life Quality Index (DLQI) is appropriate and often co-reported in clinical trials or post hoc studies.19
Studies included in this review were RCTs investigating human subjects with moderate-to-severe psoriasis (thus, only Phase II trials and above, as Phase I trials were conducted in healthy participants),20 defined in clinical trials as static PGA (sPGA) ≥3, PASI ≥12, and body surface area (BSA) ≥10%, treated with deucravacitinib. RCTs that studied psoriatic arthritis but not psoriasis were excluded.21 Study characteristics including clinical trial name/number, number of patients, intervention dose and frequency, treatment duration, clinical efficacy, and safety outcomes were obtained using a standardized table tailored to this review.
Study Selection and Data Extraction
Initial screening of studies was performed manually by two independent reviewers (J.Q.J., R.K.S.). Any queries in eligibility criteria were resolved via adjudication by an additional reviewer (W.L.). Data abstraction was performed by two independent reviewers (J.Q.J., R.K.S.). All randomized studies included for analysis were assessed for risk of bias by two independent authors (J.Q.J., R.K.S.) using the Critical Appraisal Skills Programme (CASP) checklist for RCTs.22
Primary outcomes sought for the purpose of this review included an sPGA of 0 or 1 (indicating clear or almost clear disease). Secondary outcomes included an sPGA of 0, a 75%, 90%, or 100% improvement in the PASI score (ie, PASI 75, PASI 90, or PASI 100), a DLQI score of 0 or 1, scalp-specific PGA (ss-PGA) of 0 or 1, and a PGA of Fingernail Psoriasis (PGA-F) of 0 or 1. The final endpoint was determined to be Week 12–16, as all included studies reported efficacy measures within this timepoint.
Statistical Analysis
Meta-analysis was performed using the Cochrane Review Manager 5.4 application comparing the sPGA 0/1 rates of deucravacitinib versus placebo. Only data from patients receiving the FDA-approved dose of deucravacitinib (6 mg per day) or placebo were included for meta-analysis. An odds ratio (OR) was calculated using the Mantel-Haenszel fixed-effects method, which was chosen over the Peto method as the latter is better suited for rare event occurrences.23 Significance of heterogeneity was assessed using the χ2 test (P < 0.1 set as statistically significant) and presented as the I2 test (I2 > 50% indicates significant heterogeneity, I2 < 25% indicates non-significant heterogeneity).
Results
Following the application of inclusion and exclusion criteria (PRISMA diagram shown in Figure 1), three RCTs were included for review, including one Phase II placebo-controlled trial (NCT02931838)24 and two Phase III placebo-controlled and active-comparator (apremilast) RCTs (POETYK PSO-1, POETYK PSO-2).25,26 The three RCTs were composed of a total of 1953 patients with moderate-to-severe psoriasis—including 1065 treated with deucravacitinib, 422 treated with apremilast, and 466 who received placebo. Overall, deucravacitinib patients showed marked improvement in disease severity and quality-of-life outcomes compared to apremilast and placebo groups; deucravacitinib patients with scalp psoriasis demonstrated marked improvement compared to apremilast and placebo groups, but improvements in fingernail psoriasis measures were not significant (Table 1). The risk of bias assessment of all studies is presented in Table 2.
 |
Table 1 Clinical Outcomes Reported in Deucravacitinib Randomized Controlled Trials for Moderate-to-Severe Psoriasis |
 |
Table 2 Risk-Bias Assessment of Included Studies |
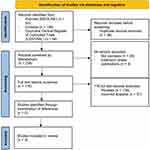 |
Figure 1 PRISMA diagram showing study selection. |
Clinical Outcomes of Included Studies
NCT02931838 was a 12-week, randomized, placebo-controlled, dose-ranging Phase II clinical trial that included 267 adults with moderate-to-severe plaque psoriasis (sPGA ≥3, PASI ≥12, and BSA ≥10%; mean baseline PASI was 18).24 The primary clinical outcome assessed was PASI 75 at Week 12 compared to baseline. Patients were randomly assigned to one of six groups to receive placebo medication or deucravacitinib orally at the following frequencies: 3 mg every other day (QOD), 3 mg daily (QD), 3 mg twice daily (BID), 6 mg BID, or 12 mg QD. Detailed clinical outcomes can be found in Table 1; improvements in PASI scores were associated with higher doses of deucravacitinib and were improved compared to placebo groups. Nearly 70% of the cohort that received deucravacitinib 3 mg BID (closest to the FDA-approved dosage of 6 mg once daily) achieved PASI 75 at Week 12, compared to 6.7% of the placebo cohort. Improvements in clinical outcomes were correlated with biomarker changes and patient-reported quality-of-life (percent of patients who achieved DLQI 0/1).27
POETYK PSO-1 (NCT03624127) and POETYK PSO-2 (NCT03611751) were randomized, double-blind, double-dummy Phase III trials that compared the efficacy and safety of deucravacitinib versus an active-comparator (apremilast) and placebo.25,26 A total of 666 patients (PSO-1) and 1020 patients (PSO-2) were randomized 2:1:1 to deucravacitinib 6 mg QD, apremilast 30 mg BID, or placebo. All participants receiving placebo were crossed over to receive deucravacitinib at Week 16; patients receiving apremilast who did not achieve PASI 50 (PSO-1) or PASI 75 (PSO-2) by Week 16 were also switched to the deucravacitinib group. In PSO-2, deucravacitinib patients achieving PASI 75 at Week 24 were re-randomized 1:1 to deucravacitinib at the same dosing schedule or placebo for the remainder of the study. If the newly switched placebo patients exhibited disease relapse, they were re-started on deucravacitinib.
Detailed clinical outcomes for both studies are reported in Table 1; briefly, deucravacitinib was shown to be more effective than both comparator and placebo at Week 16 for both primary endpoints assessed (PASI 75 and sPGA 0/1). The percent of patients who achieved PASI 75 in PSO-1 and PSO-2 (vs apremilast, placebo) were 58.7% (vs 35.1%, 12.7%) and 53.6% (vs 40.2%, 9.4%), respectively. The percent of patients who achieved sPGA 0/1 in PSO-1 and PSO-2 (vs apremilast, placebo) were 53.6% (vs 32.1%, 7.2%) and 50.3% (34.3%, 8.6%), respectively.
Safety Outcomes of Included Studies
In all included studies, deucravacitinib was well-tolerated, with similar percentages and types of adverse events (AEs) reported among patients treated with placebo or comparator drugs at Week 12 or 16.24–26 The occurrence of any AE by Week 12 or 16 in NCT02931838, POETYK PSO-1, and POETYK PSO-2 were 64% (vs 51% in placebo group), 53.0% (vs 42.4% in placebo group, 55.4% in apremilast group), and 57.5% (vs 54.3% in placebo group, 59.1% in apremilast group), respectively. In all trials, the most frequently reported AEs associated with deucravacitinib were nasopharyngitis (6.3–11%) and upper respiratory tract infection (2–6.3%). Other common AEs reported in the POETYK trials included headache (4.3–4.8% vs 3.0–5.5% in placebo group vs 10.1–11.0% in apremilast group), diarrhea (3.9–4.7% vs 3.6–7.5% in placebo group vs 10.1–13.0% in apremilast group), and nausea (1.2–2.1% vs 1.4–2.4% in placebo group vs 9.1–11.3% in apremilast group), which occurred at similar frequencies to placebo and generally decreased frequencies compared to the apremilast treatment group. Across the three studies, no significant changes in mean blood counts (including neutrophil and platelet levels), serum lipids (including total cholesterol), creatinine, creatine phosphokinase, liver enzymes, or immunoglobulins were reported. Among all patients treated with deucravacitinib, no serious cases of herpes zoster leading to discontinuation occurred; additionally, no opportunistic infections or tuberculosis were reported.
Meta-Analysis Results
Meta-analysis of the three placebo-controlled RCTs comparing the rates of clearance (sPGA 0/1) in patients with moderate-to-severe psoriasis (N = 1354; deucravacitinib, n = 888; placebo, n = 466) demonstrated superior efficacy of deucravacitinib compared to placebo (OR, 12.87; 95% confidence interval (CI), 8.97–18.48) (Figure 2). Heterogeneity was determined as significant (χ2 = 4.08, I2 = 51%) (Figure 2).
Discussion
Psoriasis is a systemic, immune dysregulatory condition that has a significant detrimental impact on a patient’s overall health and quality of life. While a range of biologic therapies are available for the treatment of more severe disease—including agents that target TNF-α, IL-12/IL-23, IL-17, and IL-23—certain biologics can be contraindicated for individuals based on comorbid conditions, safety concerns, or insurance coverage issues. Furthermore, patients with more severe psoriasis are more likely to experience biologic failure, including the sequential failure of multiple biologics, despite adequate time attempting each agent.28–30 Thus, there remains a need to develop new targeted therapeutics—particularly those that can act via a different mechanism of action than existing systemic agents—to treat patients with moderate-to-severe psoriasis.
While the development of psoriasis is complex and involves an interplay between multiple immune signaling pathways, JAK/STAT signaling has been shown to hold a central dysregulatory role in psoriasis pathogenesis for years.16 The importance of such signaling in psoriasis was further emphasized after a recent study found methotrexate to inhibit the JAK/STAT pathway as a potential secondary mechanism of action, particularly relevant in psoriatic arthritis.31 Unfortunately, first-generation JAK inhibitors targeting JAK2 and JAK3 (eg, tofacitinib, baricitinib) experienced limited success for psoriasis as a disease indication due to safety concerns, despite effective associated clinical outcomes (eg, PASI reduction).32,33 For example, incidence of major adverse cardiovascular events and cancer were found to be higher in tofacitinib-treated groups in a dose-dependent fashion for rheumatoid arthritis patients.34 Thus, JAK inhibitors were previously approved only for psoriatic arthritis or for off-label use in certain psoriasis patients who had not responded to conventional systemic therapies.33
Deucravacitinib represents a major advancement as a first-in-class systemic therapy that differs from prior JAK inhibitors and other psoriasis biologics in several ways. First, deucravacitinib is a small molecule, meaning it can be administered by a variety of routes, including orally—as opposed to injected or infused, as many psoriasis biologics are.35 Because of deucravacitinib’s oral bioavailability and simpler dosing regimens, adherence to the intended treatment plan may be easier to achieve for patients, and access to first-line therapy for moderate-to-severe psoriasis may be expanded due to lower drug costs.35 Additionally, small molecules may also hold a reduced risk of immunogenicity compared to biologics, which can translate to a longer period of efficacy in individual patients.35 Finally, our systematic review of RCTs found that psoriasis patients treated with deucravacitinib did not experience major AEs at rates significantly different from patients treated with apremilast or placebo.26,36 In our review of the three included RCTs, the most frequently reported AEs tended to occur at similar rates compared to the placebo group and at lower frequencies compared to apremilast treatment. None of the three included RCTs reported significant changes in blood count, cholesterol levels, or opportunistic infections among patients treated with deucravacitinib, which were all concerns that hampered the approval of JAK inhibitors for psoriasis treatment in the past.37,38 Taken together, these results suggest that deucravacitinib may have favorable safety features as a selective inhibitor of TYK2 in the JAK family—although results should be interpreted with caution due to the limited follow-up periods reported. Further head-to-head comparative studies should be conducted.
The meta-analysis conducted in our study is, to our knowledge, the first performed that compares plaque psoriasis patients treated with deucravacitinib versus placebo. The results (Figure 2) indicate that the primary endpoint of sPGA 0/1, a validated tool providing a global estimate of a patient’s psoriatic disease severity, was achieved significantly more frequently in deucravacitinib compared to placebo treatment groups.18 These promising results of clinical efficacy bode well for other TYK2 inhibitors in clinical development for psoriasis, including rapsacitinib, brepocitinib, NDI-034858, and ESK-001.33 Overall, our systematic review and meta-analysis found deucravacitinib to yield positive improvements for multiple efficacy endpoints, including clinical outcomes (eg, sPGA, PASI) and patient-reported quality of life (via DLQI).
Conclusions
Deucravacitinib is an effective, oral small molecule that possesses good efficacy and safety features, indicating its potential to serve as a first-line treatment for moderate-to-severe psoriasis. Patients with plaque psoriasis showed significant improvements in objective, disease-specific clinical parameters, with meta-analysis of Phase II and III RCTs demonstrating the superiority of deucravacitinib compared to placebo. Within individual RCTs, deucravacitinib was also found to yield increased rates of disease improvement and reduced AE incidence compared to apremilast. Thus, deucravacitinib achieved clinical efficacy while maintaining a favorable safety profile—an important barrier to prior JAK inhibitor use in psoriasis—consistent with its unique mechanism of action and selectivity for TYK2. While further studies should be conducted to evaluate the long-term safety and efficacy of deucravacitinib and compare it to existing biologics, deucravacitinib holds promising clinical utility and represents an important step forward as a first-in-class treatment option for psoriasis patients.
Abbreviations
AE, adverse event; BID, twice daily; CASP, Critical Appraisal Skills Programme; DLQI, Dermatology Life Quality Index; FDA, Food and Drug Administration; IL, interleukin; JAK, Janus kinase; PASI, Psoriasis Area and Severity Index; PGA, Physician Global Assessment; PGA-F, PGA of Fingernail Psoriasis; QD, daily; QOD, every other day; RCT, randomized controlled trial; sPGA, static PGA; ss-PGA, scalp-specific PGA; STAT, Signal Transducers and Activators of Transcription; Th17, T helper 17; TYK, tyrosine kinase.
Data Sharing Statement
All data analyzed in this systematic review can be searched in publicly available databases including PubMed and Embase.
Ethics Approval and Informed Consent
This is a review article of published studies; ethics approval was not required by our Institutional Review Board.
Consent for Publication
No identifiable patient information was included in this article.
Acknowledgments
We are thankful to all the reviewers who contributed to this article.
Author Contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript. All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work. Conception: JQJ, VR, WL. Methodology: all authors. Formal analysis and investigation: JQJ, RKS. Writing—original draft preparation: JQJ, RKS. Writing—review and editing: all authors. Supervision: WL.
Funding
This study was not funded.
Disclosure
J.Q.J. has received research grant funding from the National Psoriasis Foundation and institutional funding from the University of California, San Francisco. T.B. has received research grant funding from Novartis and Regeneron and is a principal investigator for trials sponsored by Abbvie, Castle, CorEvitas, Dermavant, Galderma, Mindera, and Pfizer. T.B. has also served as an advisor for Abbvie, Arcutis, Boehringer-Ingelheim, Bristol Myers Squibb, Janssen, Leo, Lilly, Novartis, Pfizer, Sun, and UCB. W.L. has received research grant funding from Abbvie, Amgen, Janssen, Leo, Novartis, Pfizer, Regeneron, and TRex Bio. The authors report no other conflicts of interest in this work.
References
1. Armstrong AW, Mehta MD, Schupp CW, Gondo GC, Bell SJ, Griffiths CEM. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157(8):940–946. doi:10.1001/JAMADERMATOL.2021.2007
2. Yan D, Blauvelt A, Dey AK, et al. New frontiers in psoriatic disease research, part II: comorbidities and targeted therapies. J Invest Dermatol. 2021;141(10):2328–2337. doi:10.1016/J.JID.2021.02.743
3. Heydendael VM, Spuls PI, Opmeer BC, et al. Methotrexate versus cyclosporine in moderate-to-severe chronic plaque psoriasis. N Engl J Med. 2003;349(7):658–665. doi:10.1056/NEJMOA021359
4. Greb JE, Goldminz AM, Elder JT, et al. Psoriasis. Nat Rev Dis Prim. 2016;2. doi:10.1038/NRDP.2016.82
5. Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: a review. JAMA. 2020;323(19):1945–1960. doi:10.1001/JAMA.2020.4006
6. Rendon A, Schäkel K. Psoriasis pathogenesis and treatment. Int J Mol Sci. 2019;20(6):1475. doi:10.3390/IJMS20061475
7. Kisseleva T, Bhattacharya S, Braunstein J, Schindler CW. Signaling through the JAK/STAT pathway, recent advances and future challenges. Gene. 2002;285(1–2):1–24. doi:10.1016/S0378-1119(02)00398-0
8. Hawkes JE, Yan BY, Chan TC, Krueger JG. Discovery of the IL-23/IL-17 signaling pathway and the treatment of psoriasis. J Immunol. 2018;201(6):1605. doi:10.4049/JIMMUNOL.1800013
9. Howell MD, Kuo FI, Smith PA. Targeting the Janus kinase family in autoimmune skin diseases. Front Immunol. 2019;10. doi:10.3389/FIMMU.2019.02342
10. Muromoto R, Shimoda K, Oritani K, Matsuda T. Therapeutic advantage of Tyk2 inhibition for treating autoimmune and chronic inflammatory diseases. Biol Pharm Bull. 2021;44(11):1585–1592. doi:10.1248/BPB.B21-00609
11. Burke JR, Cheng L, Gillooly KM, et al. Autoimmune pathways in mice and humans are blocked by pharmacological stabilization of the TYK2 pseudokinase domain. Sci Transl Med. 2019;11:502. doi:10.1126/SCITRANSLMED.AAW1736
12. Abduelmula A, Gooderham MJ. TYK2 inhibition: changing the treatment landscape for psoriasis? Expert Review of Clinical Immunology. 2021;18(3):185–187. doi:10.1080/1744666X.2022.2008240
13. Hoy SM. Deucravacitinib: first approval. Drugs. 2022;82(17):1671–1679. doi:10.1007/S40265-022-01796-Y
14. Chimalakonda A, Burke J, Cheng L, et al. Selectivity profile of the tyrosine kinase 2 inhibitor deucravacitinib compared with janus kinase 1/2/3 inhibitors. Dermatol Ther (Heidelb). 2021;11(5):1763–1776. doi:10.1007/S13555-021-00596-8
15. Gadina M, Chisolm DA, Philips RL, McInness IB, Changelian PS, O’Shea JJ. Translating JAKs to jakinibs. J Immunol. 2020;204(8):2011–2020. doi:10.4049/JIMMUNOL.1901477
16. Schwartz DM, Kanno Y, Villarino A, Ward M, Gadina M, O’Shea JJ. JAK inhibition as a therapeutic strategy for immune and inflammatory diseases. Nat Rev Drug Discov. 2017;16(12):843–862. doi:10.1038/NRD.2017.201
17. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372. doi:10.1136/BMJ.N71
18. Robinson A, Kardos M, Kimball AB. Physician Global Assessment (PGA) and Psoriasis Area and Severity Index (PASI): why do both? A systematic analysis of randomized controlled trials of biologic agents for moderate to severe plaque psoriasis. J Am Acad Dermatol. 2012;66(3):369–375. doi:10.1016/J.JAAD.2011.01.022
19. Mattei PL, Corey KC, Kimball AB. Psoriasis Area Severity Index (PASI) and the Dermatology Life Quality Index (DLQI): the correlation between disease severity and psychological burden in patients treated with biological therapies. J Eur Acad Dermatol Venereol. 2014;28(3):333–337. doi:10.1111/JDV.12106
20. Catlett IM, Aras U, Hansen L, et al. First-in-human study of deucravacitinib: a selective, potent, allosteric small-molecule inhibitor of tyrosine kinase 2. Clin Transl Sci. 2023;16(1):151–164. doi:10.1111/CTS.13435
21. Mease PJ, Deodhar AA, Van Der Heijde D, et al. Efficacy and safety of selective TYK2 inhibitor, deucravacitinib, in a phase II trial in psoriatic arthritis. Ann Rheum Dis. 2022;81(6):815–822. doi:10.1136/ANNRHEUMDIS-2021-221664
22. CASP CHECKLISTS - CASP - critical appraisal skills programme. Available from: https://casp-uk.net/casp-tools-checklists/.
23. Cochrane Training. Chapter 10: analysing data and undertaking meta-analyses. Available from: https://training.cochrane.org/handbook/current/chapter-10.
24. Papp K, Gordon K, Thaçi D, et al. Phase 2 trial of selective tyrosine kinase 2 inhibition in psoriasis. N Engl J Med. 2018;379(14):1313–1321. doi:10.1056/NEJMOA1806382/SUPPL_FILE/NEJMOA1806382_DATA-SHARING.PDF
25. Armstrong AW, Gooderham M, Warren RB, et al. Deucravacitinib versus placebo and apremilast in moderate to severe plaque psoriasis: efficacy and safety results from the 52-week, randomized, double-blinded, placebo-controlled Phase 3 POETYK PSO-1 trial. J Am Acad Dermatol. 2023;88(1):29–39. doi:10.1016/J.JAAD.2022.07.002
26. Strober B, Thaçi D, Sofen H, et al. Deucravacitinib versus placebo and apremilast in moderate to severe plaque psoriasis: efficacy and safety results from the 52-week, randomized, double-blinded, phase 3 program for evaluation of TYK2 inhibitor psoriasis second trial. J Am Acad Dermatol. 2023;88(1):40–51. doi:10.1016/J.JAAD.2022.08.061
27. Thaçi D, Strober B, Gordon KB, et al. Deucravacitinib in moderate to severe psoriasis: clinical and quality-of-life outcomes in a phase 2 trial. Dermatol Ther (Heidelb). 2022;12(2):495–510. doi:10.1007/S13555-021-00649-Y
28. Mastorino L, Roccuzzo G, Dapavo P, et al. Patients with psoriasis resistant to multiple biological therapies: characteristics and definition of a difficult-to-treat population. Br J Dermatol. 2022;187(2):263–265. doi:10.1111/BJD.21048
29. Hadeler E, Kumar S, Yeroushalmi S, et al. Factors associated with multi-biologic use in psoriasis patients at an academic medical center and review of biologic survival. J Psoriasis Psoriatic Arthritis. 2022. doi:10.1177/24755303221131259
30. Shalom G, Cohen AD, Ziv M, et al. Biologic drug survival in Israeli psoriasis patients. J Am Acad Dermatol. 2017;76(4):662–669.e1. doi:10.1016/J.JAAD.2016.10.033
31. Gremese E, Alivernini S, Tolusso B, Zeidler MP, Ferraccioli G. JAK inhibition by methotrexate (and csDMARDs) may explain clinical efficacy as monotherapy and combination therapy. J Leukoc Biol. 2019;106(5):1063–1068. doi:10.1002/JLB.5RU0519-145R
32. Jo CE, Gooderham M, Beecker J. TYK 2 inhibitors for the treatment of dermatologic conditions: the evolution of JAK inhibitors. Int J Dermatol. 2022;61(2):139–147. doi:10.1111/IJD.15605
33. Loo WJ, Turchin I, Prajapati VH, et al. Clinical implications of targeting the JAK-STAT pathway in psoriatic disease: emphasis on the TYK2 pathway. J Cutan Med Surg. 2022. doi:10.1177/12034754221141680/ASSET/IMAGES/LARGE/10.1177_12034754221141680-FIG2.JPEG
34. Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386(4):316–326. doi:10.1056/NEJMOA2109927/SUPPL_FILE/NEJMOA2109927_DATA-SHARING.PDF
35. Makurvet FD. Biologics vs. small molecules: drug costs and patient access. Med Drug Discov. 2021;9:100075. doi:10.1016/J.MEDIDD.2020.100075
36. Armstrong AW, Gooderham M, Warren RB, et al. Deucravacitinib versus placebo and apremilast in moderate to severe plaque psoriasis: efficacy and safety results from the 52-week, randomized, double-blinded, placebo-controlled phase 3 POETYK PSO-1 trial. J Am Acad Dermatol. 2022. doi:10.1016/J.JAAD.2022.07.002
37. Canada.ca. Health Canada safety review finds link between the use of Xeljanz and Xeljanz XR (tofacitinib) and increased risk of serious heart-related issues and cancer. Available from: https://recalls-rappels.canada.ca/en/alert-recall/health-canada-safety-review-finds-link-between-use-xeljanz-and-xeljanz-xr-tofacitinib.
38. FDA. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death.
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Efficacy and Safety of Low-Dose Brexpiprazole for Acute Schizophrenia: Meta-Analysis of Randomized Placebo-Controlled Trials
Zhao M, Qin B, Mao Y, Wang H, Wang A, Wang C
Neuropsychiatric Disease and Treatment 2022, 18:1705-1713
Published Date: 11 August 2022
The Efficacy and Safety of Acupuncture for Tinnitus-Associated Insomnia: A Protocol for a Systematic Review and Meta-Analysis
Fan L, Gao Y, Zhou J, Hu H, Gao H
Journal of Pain Research 2022, 15:2957-2965
Published Date: 21 September 2022
A Systematic Review and Meta-Analysis Protocol on How Best to Use Non-Pharmacologic Therapies to Manage Chronic Low Back Pain and Associated Depression
Guo Y, Ma Q, Zhou X, Yang J, He K, Shen L, Zhao C, Chen Z, Tan CIC, Chen J
Journal of Pain Research 2022, 15:3509-3521
Published Date: 4 November 2022
Tezepelumab for Patients with Severe Uncontrolled Asthma: A Systematic Review and Meta-Analysis
Zoumot Z, Al Busaidi N, Tashkandi W, Aljohaney AA, Isse S, Vidyasagar K, Ukwaja KN
Journal of Asthma and Allergy 2022, 15:1665-1679
Published Date: 18 November 2022
The Analgesic Effect and Potential Mechanisms of Acupuncture for Migraine Rats: A Systematic Review and Meta-Analysis
Sun S, Liu L, Zhou M, Liu Y, Sun M, Zhao L
Journal of Pain Research 2023, 16:2525-2542
Published Date: 24 July 2023

