Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 17
Patient-Reported Burden of Severe Alopecia Areata: First Results from the Multinational Alopecia Areata Unmet Need Survey
Authors Bewley A , Figueras-Nart I, Zhang J, Guerreiro M, Tietz N , Chtourou S, Durand F, Blume-Peytavi U
Received 22 October 2023
Accepted for publication 16 March 2024
Published 29 March 2024 Volume 2024:17 Pages 751—761
DOI https://doi.org/10.2147/CCID.S445646
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Rungsima Wanitphakdeedecha
Anthony Bewley,1 Ignasi Figueras-Nart,2 Jainzhong Zhang,3 Mariana Guerreiro,4 Nicole Tietz,4 Sami Chtourou,4 Frederick Durand,4 Ulrike Blume-Peytavi5
1Department of Dermatology, The Royal London Hospital & Queen Mary University, London, UK; 2Department of Dermatology, University Hospital de Bellvitge, Barcelona, Spain; 3Department of Dermatology, Peking University People’s Hospital, Beijing, People’s Republic of China; 4Eli Lilly and Company Ltd, Indianapolis, IN, USA; 5Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Department of Dermatology, Venereology and Allergology, Berlin, Germany
Correspondence: Anthony Bewley, Department of Dermatology, The Royal London Hospital & Queen Mary University, London, UK, Tel +44 020 7767 3200, Email [email protected]
Purpose: Alopecia areata (AA) is an autoimmune disease characterized by hair loss that has significant psychosocial implications. This study aims to describe the patient-reported burden of severe AA, coping mechanism and information needs using data from the multinational AA Patient Satisfaction and Unmet Need Survey.
Patients and Methods: Participants with current or previous ≥ 50% scalp hair loss (n = 747) were recruited from 11 countries and completed a web-based survey that assessed demographics, clinical characteristics, disease burden and psychosocial impact. Data were stratified according to sex, current age, disease duration and current severity of scalp hair loss.
Results: The mean (SD) age of participants was 43.8 (7.1) years, 55.3% were women, and 63.5% reported AA symptoms within 6 months of diagnosis. Most participants had black or brown hair (88.4%), reported a disease duration of 2 years or more (75.6%) and had current scalp hair loss of ≥ 50% (87.4%). Severe hair loss also extended to eyebrow (46.9%), eyelash (48.7), beard (61.5%) and body hair (73.2%). Participants commonly reported comorbidities such as anxiety (26.1%), depression (18.1%) and sleep problems (28.1%). The Dermatology Life Quality Index revealed a severe impact on quality of life; 86.2% of participants scored > 10. Mental health/mood was significantly affected; 55.8% of participants reported a substantial impact. Long-term effects included decreased self-esteem (32.9%), poor mental health (28.1%) and challenges in day-to-day activities (27.2%). Information needs were centered around treatment expectations, mental health, and available treatment options. More severe symptoms and a greater daily impact were reported by women and those with a longer disease duration.
Conclusion: The study emphasizes the substantial burden, including impaired quality of life and psychological well-being, of severe AA on the lives of surveyed participants. The findings highlight the importance of comprehensive disease management strategies that address both physical and psychosocial aspects of AA.
Plain Language Summary: Alopecia areata (AA) is a disease that results in hair loss and can greatly affect quality of life and well-being. The authors wanted to understand how this condition affects people’s lives and what they need to cope with it. A survey was completed by adults from 11 different countries who had current or past severe AA. The participants were asked about their demographics, their experiences with the condition and how it impacted their daily lives. The results showed that AA has a severe impact on their quality of life, including their mental health and daily activities. Participants also experienced decreased self-esteem and faced challenges in their relationships and daily routines, and many reported feeling anxious, depressed, and having trouble sleeping. Participants found different ways to cope with their condition and expressed a need for realistic expectations about treatment results, information about mental health, and treatment options. The study also found that women and those with a longer duration of AA tended to have more severe symptoms and the impact on their lives was greater. Overall, this study shows that current or previous episodes of severe AA had a significant impact on people’s lives, including their mental health and daily activities. It emphasizes the need for information about the condition and treatment options with realistic expectations. The findings help to better understand the experiences of people with AA and may aid the provision of appropriate support and information.
Keywords: burden of disease, quality of life, disease management
Introduction
Alopecia areata (AA) is an inflammatory autoimmune disease characterized by nonscarring hair loss that can range in severity from small patches to complete hair loss on the scalp and/or body.1,2
AA has considerable psychosocial sequelae, negatively impacting quality of life (QoL), and is associated with the presence of anxiety, depression, and work absenteeism/unemployment.2 In fact, the burden of AA extends to all aspects of life, including stigmatization, low self-esteem, and confidence, and difficulties in relationships (romantic, family, and friends) and performing daily activities.3,4
AA disease management focuses on treatments for hair regrowth, concealment of hair loss, and psychological support.5–7 To date, traditionally used treatments have shown limited efficacy and effectiveness for hair regrowth and many are associated with safety concerns.5,7,8 Overall, patients and dermatologists considered their experience with commonly used traditional therapies to be unsatisfactory.3,7,9 There have therefore been numerous calls for more effective treatment options.2,10,11 With a greater understanding of disease pathogenesis, the development of new treatments for AA have been successful and approved.12,13
In 2022, an oral selective Janus kinase (JAK)1/JAK2 inhibitor, baricitinib, became the first therapy to be approved by the European Medicines Agency, and the US Food and Drug Administration for adults with severe AA.14,15
Previous research has described the broad impact that AA can have beyond hair loss; however, data on the lived experience of severe AA are more limited.16 To address this gap, the large multinational cross-sectional AA Patient Satisfaction and Unmet Need Survey was conducted in 11 countries and across three continents.
This analysis describes the patient-reported burden of severe AA, using data from the AA Patient Satisfaction and Unmet Need Survey.
Materials and Methods
Participants and Setting
Participants were recruited via internet panels from February to April 2023 in 11 countries (Brazil, China, France, Germany, Italy, Japan, South Korea, the Netherlands, Spain, Sweden, and the United Kingdom). The recruitment goal was 730 patients. Eligible participants were aged ≥18 years, with self-reported physician confirmed (dermatologist or, in Sweden, primary care physician) AA, and self-reported hair loss involving ≥50% of the scalp (currently or previously). Participants were excluded if they had ever received a JAK inhibitor for the treatment of AA, or any other condition.
Survey
Data were collected using market research methodology and following all applicable codes of conduct for Market Research via a web-based, 25-minute cross-sectional survey. The research was conducted in compliance with the International Council for Harmonisation (ICH) Declaration of Helsinki. The survey was developed in collaboration with four of the authors who are experts in the management of AA (AB, IFN, JZ, UBP). Survey clarity and understanding were checked using three online pilots conducted in English. Ethical approval was not required for this research.
Collected data included patient-reported demographics, clinical characteristics, disease burden, and psychosocial impact, as well as coping mechanisms, sources of support, and information needs. Participants assessed their severity of scalp hair loss with the Scalp Hair Assessment Patient-Reported Outcome™ (a 5-point response scale, with higher scores indicating greater hair loss).17 Existing patient-reported outcome (PRO) measures of current eyebrow loss, eyelash loss, eye irritation and nail appearance were used by participants to report the severity of AA signs/symptoms beyond scalp hair loss (4-point scales, with higher scores indicating greater severity hair loss).18 The degree of stubble/beard and body hair loss (including intimate body hair) was assessed by participants using a similar approach to the existing Scalp Hair Assessment PRO™ hair loss measures (5-point scales, with higher scores indicating greater hair loss). Participants assessed their QoL and burden of disease using the Dermatology Life Quality Index (DLQI; 10 questions; range: 0–30, with higher scores indicating greater impairment of QoL).19,20 A seven-point scale was used to assess the day-to-day impact of AA in participants who were receiving a treatment for AA at the time of the survey (1–7), with higher scores indicating a greater burden. The long-term impact of AA was measured using responses to the question: “How, if at all, has your life changed as a result of your AA?”. Coping skills were assessed by asking: “Thinking about your AA in the last 6 months, please select what is helping you cope with your AA”, and information needs were measured by responses to the question: “What, if any, information or support would you like to have in relation to your AA?”. Details of the PRO measures for AA signs and symptoms used in the survey are presented in Table S1.
The survey was translated into local languages, as applicable, and validated translations were used for the standardized scales. Hall & Partners Europe Ltd (London) conducted the survey on behalf of the sponsor, Eli Lilly and Company. Patients provided informed consent to share their health data before participating in the survey and all survey answers were reported anonymously.
Statistical Analysis
Data from the AA Patient Satisfaction and Unmet Need Survey are presented overall for the 11 countries. In addition, participants were stratified according to sex (female, male), current age (≤40 years, 41–50 years, >50 years), duration of AA since diagnosis (<2 years, 2–4 years, >4 years), and current severity of scalp hair loss (<50%, 50–94%, ≥95%).
Data were analyzed descriptively. Continuous variables were described using mean and standard deviation (SD). Categorical variables were reported as the frequency and percentage within each category. No imputation of missing data was conducted. Analyses were performed using STATA/SE 13.1 software (StataCorp, College Station, TX 77845, USA).
Results
Demographics and Clinical Characteristics
A total of 747 adults with self-reported severe AA were recruited and completed the survey in Brazil (n = 72), China (n = 90), France (n = 80), Germany (n = 70), Italy (n = 76), Japan (n = 62), South Korea (n = 51), the Netherlands (n = 50), Spain (n = 71), Sweden (n = 20), and the UK (n = 105).
Demographics and clinical characteristics at the time of data collection are summarized in Table 1 for the overall population. Baseline characteristics revealed that participants had a mean (SD) age of 43.8 (7.1) years, with 55.3% of them being women. Natural hair color was self-reported as black or brown (dark, mid, or light) by 88.4% of participants. The mean (SD) time since being diagnosed with AA was 4.8 (6.3) years and 75.6% of participants reported a time since diagnosis of ≥2 years. Most participants (63.5%) reported a duration of symptoms before diagnosis of 6 months, and initial hair loss developed over a day to a month in 63.9% of participants. At the time of data collection, 87.4% of participants had current scalp hair loss ≥50% and moderate to severe levels of eyebrow, eyelash, stubble/beard, and body hair loss were reported by 46.9%, 48.7%, 61.5% and 73.2% of participants, respectively. In addition, moderate-to-severe eye irritation and nail symptoms (eg, pitted, rough, brittle or split) were reported by 44.9% and 41.8% of participants, respectively. Nearly half the participants (47.3%) reported a previous or current medical diagnosis of anxiety, depression, and/or sleep problems.
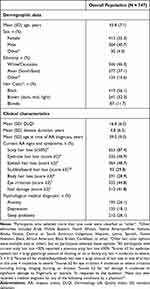 |
Table 1 Patient Demographics and Clinical Characteristics of Adults with AA |
Demographics and clinical characteristics of the selected subgroups are presented in Table S2. AA signs and symptoms were generally more severe in women, in participants aged >50 years, and in those with a disease duration >4 years than in the other respective comparator groups. Participants with <50% scalp hair loss were more likely to be men (57.5%) and those with ≥50% hair loss were more likely to be women (57.6%). Individuals experiencing <50% scalp hair loss also had fewer moderate or severe AA signs and symptoms, with the exception of stubble/beard hair loss, compared to those with ≥50% scalp hair loss. Anxiety, depression and/or sleep problems were reported across the range of current scalp hair loss severity, anxiety being most common in those with <50% scalp hair loss (34.0%) and sleep problems showing higher rates in those with ≥95% scalp hair loss (32.1%). However, the current severity of scalp hair loss did not appear to greatly affect the negative psychological effects, which were similar across the three scalp hair loss groups.
Disease Burden and Psychosocial Impact
The mean (SD) DLQI for the overall population was 16.8 (6.0) (Table 1) and 86.2% of participants reported severe QoL impairment (DLQI >10) (Figure 1). The impact of AA on daily life was measured in patients receiving AA treatment at the time of the survey. These participants experienced the greatest impact in mental health/mood, with 55.8% of participants reporting a substantial impact (score ≥6) (Figure 2). For all other categories—except the day-to-day impact of AA on participants’ religious identity—a substantial impact was reported by more than 40% of respondents.
When asked about the long-term burden of AA, most participants (79.9%) reported at least some AA-related impact on their lives (Figure 3). The most frequently reported long-term effects of AA were decreased self-esteem (32.9%), and negative impact on mental health (28.1%) and day-to-day activities (27.2%).
Disease burden and psychosocial impact in the selected subgroups are reported in Table S3. Men had a lower mean (SD) DLQI of (15.3 [6.0]) than women (17.9 [5.9]). However, in both groups, large proportions of participants had severely impaired QoL (DLQI >10 in 79.9% of men and 90.6% of women). Severe day-to-day and long-term impacts of AA were more frequently reported by women than men. Mean (SD) DLQI was similar across age groups (16.6 [5.8]–16.9 [6.0]). Younger participants (aged 40 years) reported a greater impact of AA on most day-to-day dimensions than those aged 41–50 years or >50 years, but no specific trend could be seen regarding the long-term burden of AA.
Across the three groups defined by disease duration, the mean (SD) DLQI was lower (but still >10) in participants with a disease duration >4 years (15.6 [6.4]) compared to <2 years (17.8 [5.6]) and 2–4 years (17.2 [5.8]). Among these three disease duration groups, those with AA for <2 years reported the highest day-to-day impact as well as the greatest long-term impact of AA. The mean (SD) DLQI was lower in participants with <50% scalp hair loss (14.6 [6.7]) than in participants with ≥50% scalp hair loss (16.6 [5.0], 50–94% and 17.6 [6.7] ≥95% scalp hair loss). Nevertheless, the mean DLQI was >10 irrespective of current scalp hair loss. The day-to-day impact of AA was generally reported to be substantial by greater proportions of participants with ≥95% scalp hair loss than those with 50–94% or <50% scalp hair loss; no clear trends were seen across the current hair-loss groups for long-term impact of AA.
Coping Mechanisms and Information Needs
Talking to others, particularly to a healthcare professional or other people with AA, and doing exercise or sport, were the most frequently selected coping mechanisms overall (Table 2).
 |
Table 2 Coping Mechanisms of Adults with AAa |
When asked about the information they would like to have concerning their AA, participants reported that realistic treatment result expectations (39.1%), AA and mental health (37.5%), and prescription-based treatment options (34.0%) were their most important needs (Figure 4).
Coping mechanisms and information needs in the selected subgroups are reported in Tables S4 and S5. Overall, men and women reported different coping mechanisms and information needs. Women reported a much greater need for all types of information, most notably for online support groups/forums (34.4% vs 26.6%) and information relating to the risk/benefits of different treatments (36.1% vs 26.3%). Women were also more likely to talk to other people with AA, talk to friends and family, get involved with religious or patient organizations, use social media channels, and wear wigs or experiment with make-up.
Looking at the age subgroups, a greater percentage of participants aged >50 years reported talking in person to others with AA as a coping mechanism. A smaller proportion of this age group reported talking to friends and family or healthcare professionals, or using social media, whereas those aged <40 years were more likely than the other age groups to do exercise or sport or get involved in religious activities or patient organizations. Participants aged 41–50 years were the least likely to increase their alcohol consumption and most likely to talk online with others with AA. The youngest age group was most likely to want information related to AA interventions other than prescription-based options.
Talking to others, irrespective of who or the method used, and doing exercise or sport were coping mechanisms less often selected by participants with a disease duration >4 years in comparison to those with a disease duration of <2 years or 2–4 years. However, in participants with AA for more than 4 years, 37.7% still used exercise and sport to cope. In contrast, a lower percentage of participants in this group selected increasing alcohol consumption (10.3%) or smoking (8.7%) as coping strategies. Nevertheless, these numbers were still higher than other disease duration groups. Indeed, more participants with a disease duration >4 years also reported not using any coping strategies than the other disease duration groups. Participants with a disease duration <2 years were the least likely to not want additional information or support regarding their AA.
Finally, a higher percentage (49.8%) of participants with ≥95% scalp hair loss selected talking to other people with AA as a coping mechanism when compared with the two other groups based on scalp hair loss, 39.1% (scalp hair loss 50–94%) and 25.5% (scalp hair loss <50%). Individuals experiencing <50% scalp hair loss were more inclined to use food (11.7%) or experiment with makeup (10.6%) and hair loss concealers (4.2%) as coping strategies, when compared to the other scalp hair loss groups. For all groups of scalp hair loss, talking with others, doing exercise, and using social media channels were the most frequently selected coping mechanisms. However, information needs were generally higher in participants with the greatest scalp hair loss across the range of information categories considered.
Discussion
The results of this large multinational survey highlight the substantial burden of disease associated with severe AA (defined as current or previous ≥50% scalp hair loss). The characteristics of the overall population are aligned with findings from previous studies.3,10 In our analysis, QoL was severely impaired in most participants (86.2% with DLQI >10), with a mean DLQI of 16.8 for the overall population. This is similar to the mean DLQI (13.5) reported in another study that included 300 adults with severe AA (defined as >40% scalp hair loss and >10 patches scattered all over the body).21 Also previously reported is that patients with AA experience poor QoL, which is associated with a greater extent of scalp involvement,22,23 and that women experienced a greater impact on QoL than males,24 aligning with our results.
A diagnosis of anxiety and depression was reported by 26.1% and 18.1% of participants, respectively, and is comparable to findings of a systematic literature review that reported rates of 27.1% anxiety and 18.9% depression in patients with AA.25 Indeed, AA has a considerable psychosocial impact,26 as was reported by participants in our survey. We found that a substantial day-to-day mental health burden of AA was reported by 55.8% of participants. The impact on mental health or decreased self-esteem were among the most frequently reported long-term effects of AA, reported by 28.1% and 32.9% of participants, respectively. These findings confirm those of an earlier study conducted in adults with self-reported moderate-to-severe AA (78% with hair loss on more than one-third of the scalp and 22% with hair loss on less than one‑third), which showed a marked impact of AA on self-esteem that did not abate over time.3
Overall, nearly 80% of participants reported at least some AA-related long-term impact on their life, with a negative effect on day-to-day activities reported as a long-term effect of AA by 27.2% of participants. Of concern, AA showed a negative impact on all aspects of day-to-day life in patients who were receiving treatment for AA at the time of survey completion, affecting cultural, religious, and personal identity as well as mental health, physical health, concentration, and sleep. Findings of another survey involving participants with severe AA (totalis or universalis) suggest that cultural associations with hair and hair loss are pervasive, and may drive social avoidance and camouflage behaviors in people with AA.27
Importantly, in this survey, 51.5% of participants reported that talking in person with a healthcare professional helped them cope. In addition, family members and friends were used as support pillars, as well as other people with AA. However, healthcare professionals may not (as yet) be as facilitative as patients would like them to be; participants with AA from the United Kingdom reported in another survey that healthcare professionals failed to offer support. Rather support was found from family and friends, other people with AA, and patient associations.27 Here it is pertinent to highlight variations in healthcare systems where there is a limited number of dermatologists, and general practitioners may not be the most suitable group of professionals to provide adequate information on this specialized disease. That survey also reported that the Alopecia UK website and the National Alopecia Areata Foundation were valuable sources of reassurance and information to help reduce isolation and to provide emotional relief.27 In the present survey, only 11.0% of participants identified a patient organization as an important resource to help them cope with AA, although online conversations with others with AA were frequently used. In our analysis population, information needs were primarily related to treatment options and result expectations, as well as mental health.
Of interest, most participants (88.4%) in the survey self-reported black or brown hair color (46.3% were white/Caucasian). While the exact cause of AA is unknown, several epitopes related to pigment production in melanocytes have been proposed as potential autoantigens in AA28,29 and result in the preferential loss of pigmented hairs.30
Data were further analyzed according to sex, age, duration since AA diagnosis, and severity of scalp hair loss. The severity of AA signs, symptoms, and burden tended to be higher in women when contrasted with men. Similar results have been reported in a previous study, but only in females with an AA diagnosis before age 16 years.31 Participants older than 50 years at the time of data collection also generally reported more severe AA signs and symptoms. However, the burden of AA tended to be lower in these older participants than in those aged 50 years or less, although the impact on QoL was substantial (DLQI >16) across all age groups. Other studies have identified that adults with AA aged between 20 and 50 years are at high risk for poor QoL and that AA-related healthcare visits peak in those aged 30–59 years.32
The severity of signs and symptoms tended to be higher in participants with a disease duration >4 years in our analysis, but the highest burden of AA was observed in participants with a disease duration <2 years, possibly reflecting a higher impact of AA during the active phase of the disease or possibly lower disease acceptance and insufficient coping mechanisms.10
We found that increasing severity of scalp hair loss was generally associated with greater severity of other AA signs and symptoms. While the mean DLQI was lower in participants with <50% than those with ≥50% scalp hair loss at the time of this survey, all scalp hair loss groups were experiencing a severe impact on their QoL. Not surprisingly, those with ≥95% scalp hair loss experienced the greatest day-to-day impact and long-term burden of AA. Importantly, all participants with <50% scalp hair loss had previously experienced a scalp hair loss of ≥50%, which may have impacted our findings. Previous studies have reported conflicting results on the relationship between the severity of hair loss and the burden of AA.11,22
Subgroups of participants reported different coping mechanisms and information needs. Women tended to talk more with others who experienced AA, join religious or patient organizations, use social media channels, and wear wigs and make-up. Participants aged >50 years generally reported fewer information needs than younger participants, and those with a disease duration >4 years reported employing fewer coping mechanisms. Participants with a scalp hair loss of ≥95% tended to enlist more coping mechanisms than other scalp hair loss categories and had a greater need for information.
This analysis of AA disease burden could contribute to increasing awareness and an understanding of the pressures that patients with AA experience in daily living, and inform the design of patient-centered care strategies. There is a growing interest in considering the complexity of personalized care needs when setting patient goals for AA in health and life, while simultaneously minimizing the burden of disease.33
On a clinical level, personalized treatment and support options for AA are currently limited by the efficacy, safety, and cost of traditional therapy.12,34 For example, access to camouflaging agents, like wigs, used by many participants in this survey are often cost-prohibitive and not covered by health insurance plans.34 To effectively manage the burden of AA and reduce the negative stigma associated with AA, additional program development and funding from national organizations will likely be needed.34 On a broad scale, further exploration into AA PROs could help to increase disease advocacy and awareness and enhance accessibility to tools that facilitate navigating life with AA.34 There is also a need to support efficacious, less burdensome AA treatments that meet the physical, psychological, and psychosocial needs of patients with AA.3
These analyses are subject to the limitations associated with surveys such as the potential for sampling, selection, and recall bias. Furthermore, the cross-sectional design, the absence of a comparator population, and the descriptive nature of the analyses do not allow us to conclude on causality and limit the interpretation of some of the results. Other limitations include the self-reported severity of AA signs and symptoms (although validated instruments have been used for most assessments), the low number of participants in some participating countries, and the exclusion of patients exposed to a JAK inhibitor or a biologic drug (previously, or at the time of data collection) for the treatment of AA or any other condition. At the time the survey was conducted, JAK inhibitors had only recently been approved for the treatment of severe AA, and it was anticipated that their use would vary significantly across countries. Furthermore, it was not known whether individuals who had used these drugs may have had different experiences with AA compared to those using other systemic therapies. Excluding individuals who had taken JAK inhibitors was part of the study design to focus on a more homogeneous subset of the population and better control for confounding variables. Future studies should further examine the patient-reported burden of severe AA in patients exposed to JAK inhibitors. Lastly, because the DLQI method was not created expressly for AA, some of the questions, such as those about itch, pain, and physical activity, might not be appropriate for evaluating the impact of AA.10
Conclusion
Successful long-term management of AA can be improved by understanding the burden of the condition on those affected. This comprehensive survey provides real-world data on the burden of severe AA collected from a large sample of adults in 11 countries across three continents. The impact of severe AA reaches far beyond hair loss, and is associated with severe impairment in QoL and a substantial psychosocial burden regardless of demographic or clinical characteristics. These results show that severe AA is a condition associated with persistent high unmet medical needs originating from diverse aspects. To address the persistently high unmet medical needs identified, future strategies could encompass integrated healthcare models, targeted patient education focusing on treatment expectations and mental health, holistic support programs beyond conventional medical interventions, advancements in treatment paradigms, and global advocacy efforts. This comprehensive approach aims to mitigate the burden of severe AA and enhance overall well-being, offering a roadmap for proactive and compassionate disease management.
Abbreviations
AA, alopecia areata; DLQI, Dermatology Life Quality Index; JAK, Janus kinase; n/a, not applicable; PRO, patient-reported outcome; QoL, quality of life; SD, standard deviation.
Data Sharing Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Informed Consent
This study conformed to market research codes of conduct and in compliance with the International Council for Harmonisation (ICH) Declaration of Helsinki.
Acknowledgments
The sponsors acknowledge the participation of all volunteers and the study site personnel involved in the original research. Medical writing support was provided by Clare Koning and Caroline Spencer (Rx Communications, Mold, UK), funded by Eli Lilly and Company.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This paper was funded by Eli Lilly and Company.
Disclosure
Anthony Bewley reports consultation/advisory and/or lecturing fees and/or travel support from: AbbVie, Almirall, BMS, Eli Lilly, Galderma, Janssen, Leo-Pharma, Novartis, Pfizer, Sanofi, UCB and is a member of the Medical Advisory Board for: National Eczema Society, Ichthyosis Support Group, Psoriasis Association and reports royalties from Practical Psychodermatology (Wiley 2014). Ignasi Figueras-Nart consultation/advisory and/or lecturing fees and/or travel support from: AbbVie, Eli Lilly, Leo-Pharma, Novartis, Pfizer, Sanofi, Vifor Pharma. Ulrike Blume-Peytavi has served on advisory boards and/or is a consultant and/or is a clinical trial investigator for AbbVie, Amryt, Bayer, Boots Healthcare, Cassiopeia, CeraVe, Concert Pharmaceuticals/Sun Pharma, Dermocosmétique Vichy, Eli Lilly and Company, Galderma, Laboratoires Bailleuil LEO Pharma, Novartis, Pfizer, Pierre Fabre, Sanofi Regeneron, Timber. Mariana Guerreiro, Nicole Tietz, Sami Chtourou, and Frederick Durand are employees and minor shareholders of Eli Lilly and Company. The authors report no other conflicts of interest in this work.
References
1. Lintzeri DA, Constantinou A, Hillmann K, Ghoreschi K, Vogt A, Blume‐ Peytavi U. Alopecia areata – current understanding and management. JDDG. 2022;20:59–90.
2. Muntyanu A, Gabrielli S, Donovan J, et al. The burden of alopecia areata: a scoping review focusing on quality of life, mental health and work productivity. J Eur Acad Dermatol Venereol Epub. 2023;37(8):1490–1520. doi:10.1111/jdv.18926
3. Mesinkovska N, King B, Mirmirani P, Ko J, Cassella J. Burden of illness in alopecia areata: a cross-sectional online survey Study. J Investig Dermatol Symp Proc. 2020;20(1):S62–S68. doi:10.1016/j.jisp.2020.05.007
4. Burns LJ, Mesinkovska N, Kranz D, Ellison A, Senna MM. Cumulative life course impairment of alopecia areata. Int J Trichol. 2020;12(5):197–204. doi:10.4103/ijt.ijt_99_20
5. Lai VWY, Chen G, Gin D, Sinclair R. Systemic treatments for alopecia areata: a systematic review. Australas J Dermatol. 2019;60(1):e1–e13. doi:10.1111/ajd.12913
6. Messenger AG, McKillop J, Farrant P, McDonagh AJ, Sladden M. British association of dermatologists’ guidelines for the management of alopecia areata 2012. Br J Dermatol. 2012;166(5):916–926. doi:10.1111/j.1365-2133.2012.10955.x
7. Rossi A, Muscianese M, Piraccini BM, et al. Italian guidelines in diagnosis and treatment of alopecia areata. G Ital Dermatol Venereol. 2019;154(6):609–623. doi:10.23736/S0392-0488.19.06458-7
8. Fukuyama M, Ito T, Ohyama M. Alopecia areata: current understanding of the pathophysiology and update on therapeutic approaches, featuring the Japanese dermatological association guidelines. J Dermatol. 2022;49(1):19–36. doi:10.1111/1346-8138.16207
9. Fridman M, Ray M, Gandhi K, Shy ME, Mostaghimi A. Treatment patterns and treatment satisfaction among adults with alopecia areata in the United States. Adv Ther. 2022;39(12):5504–5513. doi:10.1007/s12325-022-02338-4
10. Andersen YMF, Nymand L, DeLozier AM, Burge R, Edson-Heredia E, Egeberg A. Patient characteristics and disease burden of alopecia areata in the Danish skin cohort. BMJ Open. 2022;12:e053137.
11. Edson‐Heredia E, Aranishi T, Isaka Y, Anderson P, Marwaha S, Piercy J. Patient and physician perspectives on alopecia areata: a real-world assessment of severity and burden in Japan. J Dermatol. 2022;49(6):575–583. doi:10.1111/1346-8138.16360
12. Yan D, Fan H, Chen M, et al. The efficacy and safety of JAK inhibitors for alopecia areata: a systematic review and meta-analysis of prospective studies. Front Pharmacol. 2022;13:950450. doi:10.3389/fphar.2022.950450
13. Pourang A, Mesinkovska NA. New and emerging therapies for alopecia areata. Drugs. 2020;80(7):635–646. doi:10.1007/s40265-020-01293-0
14. European Medicines Agency. Olumiant, baricitinib EPAR. Available from: https://www.ema.europa.eu/en/medicines/human/EPAR/olumiant.
15. Food and Drug Administration. Olumiant (baricitinib). Highlights of prescribing information. Available from: https://pi.lilly.com/us/olumiant-uspi.pdf.
16. Aldhouse NVJ, Kitchen H, Knight S, et al. “You lose your hair, what’s the big deal?’ I was so embarrassed, I was so self-conscious, I was so depressed:” a qualitative interview study to understand the psychosocial burden of alopecia areata. J Patient-Rep Outcomes. 2020;4(1):76. doi:10.1186/s41687-020-00240-7
17. Wyrwich KW, Kitchen H, Knight S, et al. Development of the scalp hair assessment PRO™ measure for alopecia areata. Br J Dermatol. 2020;183(6):1065–1072. doi:10.1111/bjd.19024
18. Wyrwich KW, Kitchen H, Knight S, et al. Development of Clinician-Reported Outcome (ClinRO) and Patient-Reported Outcome (PRO) measures for eyebrow, eyelash and nail assessment in alopecia areata. Am J Clin Dermatol. 2020;21(5):725–732. doi:10.1007/s40257-020-00545-9
19. Finlay A, Khan G Dermatology Life Quality Index (DLQI). Available from: https://www.nhsfife.org/media/32589/dermatology-life-quality-index-dlqi.pdf.
20. Hongbo Y, Thomas CL, Harrison MA, Sam Salek M, Finlay AY. Translating the science of quality of life into practice: what do dermatology life quality index scores mean? J Invest Dermatol. 2005;125(4):659–664. doi:10.1111/j.0022-202X.2005.23621.x
21. Al-Mutairi N, Eldin ON. Clinical profile and impact on quality of life: seven years experience with patients of alopecia areata. Indian J Dermatol Venereol Leprol. 2011;77(4):489–493. doi:10.4103/0378-6323.82411
22. Liu LY, King BA, Craiglow BG. Health-related quality of life (HRQoL) among patients with alopecia areata (AA): a systematic review. J Am Acad Dermatol. 2016;75(4):806–812.e3. doi:10.1016/j.jaad.2016.04.035
23. Abedini R, Hallaji Z, Lajevardi V, Nasimi M, Karimi Khaledi M, Tohidinik HR. Quality of life in mild and severe alopecia areata patients. Int J Womens Dermatol. 2018;4(2):91–94. doi:10.1016/j.ijwd.2017.07.001
24. Senna M, Ko J, Glashofer M, et al. Predictors of QOL in patients with alopecia areata. J Invest Dermatol. 2022;142(10):2646–2650.e3. doi:10.1016/j.jid.2022.02.019
25. Lee S, Lee H, Lee CH, Lee WS. Comorbidities in alopecia areata: a systematic review and meta-analysis. J Am Acad Dermatol. 2019;80(2):466–477.e16. doi:10.1016/j.jaad.2018.07.013
26. Toussi A, Barton VR, Le ST, Agbai ON, Kiuru M. Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: a systematic review. J Am Acad Dermatol. 2021;85(1):162–175. doi:10.1016/j.jaad.2020.06.047
27. Davey L, Clarke V, Jenkinson E. Living with alopecia areata: an online qualitative survey study. Br J Dermatol. 2019;180(6):1377–1389. doi:10.1111/bjd.17463
28. Pratt CH, King LE, Messenger AG, Christiano AM, Sundberg JP. Alopecia areata. Nat Rev Dis. 2017;3:17011.
29. Messenger AG, Bleehen SS. Alopecia areata: light and electron microscopic pathology of the regrowing white hair. Br J Dermatol. 1984;110(2):155–162. doi:10.1111/j.1365-2133.1984.tb07461.x
30. Jia W-X, Mao Q-X, Xiao X-M, Li Z-L, Yu R-X, Li C-R. Patchy alopecia areata sparing gray hairs: a case series. Adv Derm Allergol. 2014;2:113–116.
31. Tan E, Tay YK, Giam YC. A clinical study of childhood alopecia areata in Singapore. Pediatr Dermatol. 2002;19(4):298–301. doi:10.1046/j.1525-1470.2002.00088.x
32. Villasante Fricke A, Miteva M. Epidemiology and burden of alopecia areata: a systematic review. Clin Cosmet Invest Dermatol. 2015;8:397–403. doi:10.2147/CCID.S53985
33. Spencer-Bonilla G, Quiñones AR, Montori VM. Assessing the burden of treatment. J Gen Intern Med. 2017;32(10):1141–1145. doi:10.1007/s11606-017-4117-8
34. Alhanshali L, Buontempo MG, Lo Sicco KI, Shapiro J. Alopecia areata: burden of disease, approach to treatment, and current unmet needs. Clin Cosmet Invest Dermatol. 2023;16:803–820. doi:10.2147/CCID.S376096
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Assessing the Burden of Osteoarthritis in Africa and the Middle East: A Rapid Evidence Assessment
Al Saleh J, Almoallim H, Elzorkany B, Al Belooshi A, Batouk O, Fathy M, Vainstein N, Kaki AM
Open Access Rheumatology: Research and Reviews 2023, 15:23-32
Published Date: 15 March 2023
Managing Diagnosis, Treatment, and Burden of Disease in Hereditary Angioedema Patients with Normal C1-Esterase Inhibitor
Jones D, Zafra H, Anderson J
Journal of Asthma and Allergy 2023, 16:447-460
Published Date: 22 April 2023