Back to Journals » Veterinary Medicine: Research and Reports » Volume 12
The Use of Autologous Protein Solution (Pro-Stride®) and Leukocyte-Rich Platelet-Rich Plasma (Restigen®) in Canine Medicine
Authors King W , Cawood K, Bookmiller M
Received 15 October 2020
Accepted for publication 1 March 2021
Published 19 March 2021 Volume 2021:12 Pages 53—65
DOI https://doi.org/10.2147/VMRR.S286913
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Young Lyoo
William King,1 Kevin Cawood,2 Megan Bookmiller2
1Owl Manor, Warsaw, IN, USA; 2Indian Creek Veterinary Hospital, Fort Wayne, IN, USA
Correspondence: William King
Owl Manor, 720 E Winona Ave, Warsaw, IN, 46580, USA
Tel +1 574 267 4391
Fax +1 866 602 5275
Email [email protected]
Abstract: The use of autologous orthobiologics is an emerging area of interest in veterinary medicine. In this retrospective study, we reviewed the clinical results achieved using two orthobiologics devices to address orthopedic injuries and tissue repair. Leukocyte (White blood cell)-rich platelet-rich plasma (L-PRP) devices produce outputs containing high concentrations of growth factors from venous blood. The Autologous Protein Solution (APS) device produces an orthobiologic containing high concentrations of growth factors and anti-inflammatory cytokines. L-PRP has commonly been used to address soft tissue injuries. APS has been injected into the joint to address osteoarthritis. In the last five years, our practice has treated 35 dogs (38 treatments) with L-PRP and 98 dogs (108 treatments) with APS. Our group has used L-PRP and APS to address orthopedic conditions including osteoarthritis, bursitis, tendinitis, tendon/ligament rupture/repair procedures, post-surgical femoral head osteotomy/tibial-plateau-leveling osteotomy tissue repair, lumbosacral stenosis, patellar luxation, joint laxity, and osteochondral dissecans. The results achieved with L-PRP and APS have been favorable (observed pain improvement and minimal adverse reactions), but sometimes have not achieved complete pain relief or tissue repair. The most common application for L-PRP was patellar luxation and the most common application for APS was injection post-ACL surgery. Canine OA has been successfully managed in several patients with repeat injections of APS over the course of several years. Future studies on specific conditions are ongoing and including efforts to further characterize these products in canine medicine.
Keywords: APS, PRP, canine, anti-inflammatory, orthopedics, osteoarthritis
Introduction
There has been a growing unmet need in veterinary medicine to provide solutions that do more than just address symptoms. To address this unmet need, autologous devices which process and concentrate a patient’s own blood have been explored. While some have called this area of practice “regenerative medicine” we prefer to use the term “orthobiologics” as it more accurately describes the devices and their outputs. Whole blood contains growth factors and cytokines which play a role in inflammation, tissue, and joint healing. There are distinct classes of blood concentrating devices in veterinary medicine including Platelet-Rich Plasma (PRP),1 Autologous Conditioned Serum (ACS),2 and Autologous Protein Solution (APS) devices.3 They isolate and concentrate different factors from blood and have been historically used for varying applications. Traditionally, PRP has been used for tendon and ligament disease or injuries in human and veterinary medicine.4–7 PRP devices have been explored and used in canine medicine for the longest time period, particularly in dental reconstruction research.8–10 ACS devices were introduced for injection into the joint to address osteoarthritis (OA).11,12 In typical ACS device processing, blood is clotted with glass beads in an incubator for up to 24 hours. The device is then processed in a centrifuge and the serum is extracted. ACS devices have been extensively explored in large animal medicine, and a study characterizing the cytokines in canine ACS has recently been published.13 ACS clinical impressions in equine medicine have been disappointing and now it appears that ACS is mainly used as a “maintenance” injection in mild lameness and performance problems.14 The APS device was designed to address the inflammation associated with osteoarthritis. The APS device has been adopted in large animal medicine, notably equine, and a more recent addition to the canine veterinarian’s tool-kit.3,15–18 We have incorporated leukocyte-rich PRP (L-PRP) and APS devices into our practice since 2012 with good clinical outcomes for a variety of orthopedic applications.
PRP in canine medicine is produced by commercial devices or laboratory centrifuge tubes to concentrate platelets in a small volume of plasma. Some devices also concentrate white blood cells (leukocytes).19–21 PRP is a catch-all term that could include many different formulations. Using these different cell separation systems, PRP provides growth factors to aid in tissue repair by stimulating cell proliferation, migration, differentiation, and blood vessel growth. Platelets deliver more than just growth factors: they also deliver chemokines which recruit white blood cells. Inflammation is part of the wound healing process and the first step of tissue repair involves platelets releasing their growth factors and cytokines to recruit white blood cells as the next step in tissue healing (Figure 1).22
 |
Figure 1 The timing and cell types involved in wound repair demonstrating the role of WBC (including neutrophils, macrophages, and lymphocytes) in successful healing. Note: Adapted from King W, Toler K, Woodell-May J. Role of White Blood Cells in Blood-and Bone Marrow-Based Autologous Therapies. BioMed Res Int. 2018;2018. Creative Commons license and disclaimer available from: http://creativecommons.org/licenses/by/4.0/ legalcode.22 |
Several cell culture in vitro studies have suggested that L-PRP could contain inflammatory cytokines.23–27 However, these in vitro studies have not translated to adverse clinical outcomes in human28–30 or veterinary clinical studies.18,31,32 In human clinical studies, OA patients who had higher concentrations of WBC in their autologous concentrates were more likely to be OMERACT-OARSI high pain responders indicating they had the most improvement.33 Some PRP systems in canine medicine market that they make a “pure” PRP (P-PRP) containing only platelets. However, removing neutrophil and monocytes from PRP actually lowers the concentration of anti-inflammatory cytokines like interleukin-1 receptor antagonist (IL-1ra or IRAP).34 This is because IL-1ra comes from monocytes and neutrophils.35 Indeed, studies have shown that P-PRP does not have high concentrations of IL-1ra because it does not contain WBCs.36 IL-1ra is present (in high concentrations) in formulations containing leukocytes (L-PRP21 and APS) or from ACS devices in which leukocytes are cultured with glass beads for 6–24 hours to stimulate the production of IL-1ra.37 There currently is no correlation between platelets or cell concentrations and clinical outcomes known in canine medicine. Research should seek to characterize the relationship between the concentration of platelets and cells in canine medicine.
The L-PRP device we use in our clinic was designed to capture a high concentration of platelets and white blood cells to aid in the tissue repair process (Restigen® PRP Device, Owl Manor, in veterinary medicine; GPS III® Platelet Concentration System, Zimmer Biomet, in human medicine). This device has been used in human medicine since 2007 and is the most clinically studied PRP.38 The concentrations of growth factors in PRP are lower than their recombinant forms in drug products and are thought to still induce tissue repair by their combinatorial effect. Key growth factors in PRP are shown below in Table 1.
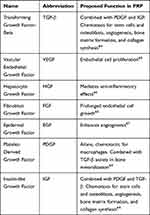 |
Table 1 Growth Factors and Their Proposed Functions in PRP |
PRP has been explored in small animal medicine for a wide variety of applications including surgical bone repair,39,40 soft tissue repair,41 osteochondral injuries,42 and osteoarthritis.43,44 Although PRP has had an excellent safety profile in these studies, the efficacy of PRP across different indications have been mixed. These mixed clinical results could be attributed to the varying outputs of different devices, patient-to-patient differences, and different biochemical requirements for different diseases and injuries. Most small animal studies are also limited by a small number of study subjects (< 25). Therefore, a goal of this manuscript was to compile our clinical experience with L-PRP to provide evidence to small animal veterinarians about areas we have had clinical success and to suggest areas of future research for randomized and controlled clinical studies.
OA was traditionally thought of as a purely mechanical disease. The last 20 years of research has shown that there is a strong biochemical component of OA driven primarily by inflammation. Inflammation in canine OA can be induced by trauma or genetics. Inflammatory cytokines bind to cells in the joint and induce the secretion of matrix metalloproteases (MMPs). MMPs degrade extracellular matrix (ECM) in the joint. The resulting ECM fragments bind to cells in the joint and induce the secretion of even more inflammatory cytokines. This creates a destructive feed-forward cycle that drives osteoarthritis.45,46 A tool to address this feed-forward cycle would require both anti-inflammatory cytokines and growth factors.
The APS device was designed to rapidly produce anti-inflammatory cytokines and growth factors to block the inflammatory cycle that drive OA (Pro-Stride® APS Device, Owl Manor, in veterinary medicine; nSTRIDE® APS Kit, Zimmer Biomet, in human medicine). APS contains anti-inflammatory cytokines in addition to the growth factors from PRP (Appendix Table 1). APS has been shown to block the release of MMPs from chondrocytes,15 inflammatory cytokines from macrophages,17 and prevent ECM molecule release in inflammatory cell-culture conditions.16 In a rat meniscal tear OA model, APS decreased collagen and cartilage degeneration, resulting in a significantly improved total joint score compared to saline control.47 APS has shown to decrease osteoarthritis pain in canine,18,32 equine,31 and human28–30,33 clinical trials. In summary, APS has blocked inflammation in cell culture in vitro studies, experimental animal studies, and decreased pain in clinical trials.
There have been three randomized and controlled trials using APS in veterinary medicine. In the first trial, 40 horses with naturally occurring OA were injected with either APS (n = 20) or saline control (n = 20). Two weeks post-injections, APS treated horses had statistically significant reductions in lameness compared to saline controls. The 20 horses originally injected with saline received APS and then all 40 horses were sent home. These horses still had statistically improved lameness, according to owner surveys, one year after APS injection.31 In the first canine clinical trial of APS, 21 dogs with OA and single limb lameness in their stifles or elbows were injected with either APS or saline. Compared to pre-treatment values, APS treated dogs showed a significant improvement in pain scores, lameness scores, and peak vertical force 12 weeks post-injection.18 In the second canine APS clinical trial, five dogs with bilateral hip dysplasia were injected in one hip with APS and in the contralateral hip with saline. One month after injection, hips treated with APS improved significantly in their total pressure index and put significantly more weight than the hips treated with saline indicating pain relief.32 Later in this manuscript we will describe our clinical experience with APS to address different orthopedic conditions to encourage further research in canine medicine.
Materials and Methods
Lameness Evaluations
In this study, lameness evaluations were performed by reviewing a patient’s pertinent medical history including the following survey given to owners below in Table 2.
 |
Table 2 Survey for Canine Owners at Beginning of Lameness Evaluations |
In this study, physical examinations were performed using the steps in Table 3 as well as neurological examinations.
 |
Table 3 Physical/Neurologic Examination Steps |
Neurological conditions were discerned from orthopedic conditions if there appeared to be no nerve deficits and loss of proprioception. Diagnostics such as radiographs, CT/MRI scans were used to determine the origin of the condition. Radiographs, MRI and CT scans were used to look for bone and soft tissue involvement. Bloodwork was used to help diagnose tick borne diseases, and infections. Biopsies were used to help diagnose types of masses that could have caused lameness. Each limb was evaluated, as dogs can present with lameness in more than one limb.
Synovial fluid was evaluated by sending the fluid to the laboratory. The technician observed the thickness of the fluid, color, and evaluated the white and red blood cells (RBC) under a microscope. The presence of crystals or bacterial infections were also evaluated. Nucleated cell counts, RBC, glucose, proteins, uric acid, and lactic dehydrogenase were measured.
Blood Draw and Device Processing
Autologous blood was obtained by performing a peripheral blood draw mixed with anti-coagulant citrate dextrose solution A (ACD-A) (Citra Labs, Braintree, MA). The blood draw volume was based on the technology used, technique, and output volume desired. For canine patients in this study, blood draws were either 30 or 60mL (based on the device used).
Blood was drawn by clipping an area over the jugular vein and then the skin was aseptically cleaned. The ACD-A anti-coagulant was pre-loaded in a syringe and connected to a winged-blood collection needle-tube set. The anti-coagulant was drawn down the syringe and then up to the tube so that all blood contacting surfaces had been coated with anti-coagulant. The syringe was gently inverted while pulling blood and after removing the needle to prevent clotting. If possible, sedation was performed after the blood draw to maintain the patient’s blood pressure. One technician drew the blood and one technician held and comforted the patient (Figure 2). Canine patients were sedated using 5 μg/kg Dexmedetomidine (Zoetis, Parsippany-Troy Hills, NJ) and 0.2 mg/kg of Butorphanol (Zoetis, Parsippany-Troy Hills, NJ) administered intravenously while the device was processed. A pain free and immobile patient was required.
30mL or 60mL devices (Restigen® PRP Device, Owl Manor, Warsaw, IN) were used to prepare L-PRP. The amount of blood and ACD-A anti-coagulant was decreased when blood was drawn from patients who were less than 10kg. The APS device (Pro-Stride® APS Device, Owl Manor, Warsaw, IN) was only available in a 60mL version. The PRP device took 15 minutes to process (Figure 3) and the APS device used an additional 2-minute processing step. The 60mL L-PRP device output 6mL of PRP. The 30mL L-PRP device output 3mL of PRP. The APS device processed 60mL of blood through a two-step procedure that produced 2.5mL of APS (Figure 4). Both devices are processed using the Owl Manor Centrifuge (Owl Manor, Warsaw, IN). The entire process from starting a blood draw to being ready to use the PRP or APS took about 30 minutes and was performed by veterinary technicians in our practice.
The volume of L-PRP used depended on the size of the joint. Very commonly we divided the output of the kit to address multiple joints (ie bilateral stifles, bilateral elbows, elbow and a shoulder, etc).
The volume of APS used per condition was chosen by the volume of the joint and the number of joints injected. For most bilateral OA, the output of the APS kit was divided in half and half was injected in both joints. In post-surgical applications, the surgery was completed and the APS was injected right before final bandaging.
Joint Injections
Gloves were worn and the injection site was prepared like surgery. The joint was entered with an appropriately sized needle attached to a syringe (Table 4). Placement in the joint was always confirmed by aspiration of all available joint fluid which was submitted for laboratory testing if desired. While the needle was still in the joint, the first syringe was replaced with a second syringe containing the L-PRP or APS. The injections were smooth and without resistance. For intra-articular (IA) injections approaches we followed guidance from Fossum et al.48 Owners were instructed to restrict the patient’s activity for 5–7 days post-injections before resuming normal activity level. We also recommended an NSAID (Carprofen, Deracoxib, or Meloxicam) for at least 7 days post-injections.
 |
Table 4 Procedures Used During Canine Joint Injections |
Tendon/Ligament/Subcutaneous Injections
Tendon or ligament cases started with tendons or ligaments that had mechanical integrity but were not compromised beyond general use. Patients who received tendon/ligament injections failed standard treatments like rest, physical therapy, or NSAIDs. For subcutaneous (SQ) injections the skin was palpated and anatomical landmarks were used to locate the soft tissue to inject (the areas of maximal tenderness). The skin was prepared for injection and then the tissue was locally blocked. The PRP or APS was then slowly injected using either a 22 or 25-G needle using a peppering technique in which small volumes of PRP or APS were placed in the tendon or fascia covering the tender area. Owners were instructed to restrict patient’s activity for 5–7 days post injections before resuming normal activity level. It was also recommended for patients to take an NSAID (Carprofen, Deracoxib, or Meloxicam) for at least 7 days post-tendon/ligament/SQ tissue injection. Owners were given post-surgical discharge sheets to follow.
Over the reviewed period, 5 years, L-PRP and APS devices have been used in 146 treatments on 133 dogs. Before treatment with L-PRP or APS owners were counseled on expected outcomes and rehab procedures, as well as the importance of compliance with post-injection protocols. From February – March 2020 medical records were reviewed and owners were followed-up with. Results reported in this study were based on veterinarian and owner evaluations (See Appendix Tables 2–3). Outcomes were segmented as: 1) the patients were doing “well” (complete improvement), 2) had “mixed results” (improvement but still on non-steroidal anti-inflammatory drugs (NSAIDS) after the transient post-injection period), or 3) had unsatisfactory results (no improvement). The safety profile of L-PRP and APS was also evaluated with owners by asking if their dogs had any short-term or long-term complications post-treatment. Clinical success was evaluated in these cases by performing a follow up call on all patients 24–48 hours after surgery as well as a recheck exam 6–8 weeks post operatively to access how the patient was recovering. Owners were contacted periodically, including at suture removal in 2 weeks, to get updates early on and further out to record how long the clinical benefits of the injections last.
Results
We have used L-PRP in our clinic for a broad spectrum of orthopedic conditions listed below in Table 5. The average age of patients treated with L-PRP was 6.4 ± 3.5 years old (Range 0.5–12 years old). Of the patients treated with L-PRP, 15 were spayed, 16 were neutered, 1 was female, and 3 were male. Our most common applications for L-PRP included IA and SQ injections for patellar luxation, lumbosacral pain, ACL surgery (lateral imbrication surgery), and bicipital tendonitis. Post ACL surgery, both stifles were injected with APS to reduce pain and inflammation, improve healing time, and return to function quicker, as well as minimize the chances of the opposite stifle from tearing its anterior cruciate ligament. We do the same process for our patients who have TPLO surgery to reduce pain and inflammation and help with healing of the TPLO site. While the number of cases presented here are relatively small per condition, many of these are the first published cases using an orthobiologic in canine medicine. The data presented in this manuscript could be used to statistically power randomized and controlled trials.
 |
Table 5 Frequency of L-PRP Applications in Our Practice Including Tendon/Ligament Applications, OA, and Joint Applications |
In the cases with unsatisfactory outcomes, patients typically presented with severe pathologies. L-PRP injections have been able to “tighten” soft tissues with SQ injections. There has been limited success in the distal limbs with SQ tarsus and carpal injections (Table 5). L-PRP has had positive clinical outcomes for tendon/ligament applications including patellar luxation and after ACL surgery. Repeat injections have been performed on some patients. For example, in one patient, L-PRP was injected SQ around both stifles which produced satisfactory results for 21 months before the patient was injected again. In a separate patient with bilateral stifle OA, satisfactory results were observed for 13 months before a second injection of L-PRP.
The average age of patients treated with APS was 6.8 ± 3.3 years old (Range 1.3–15 years old). Treatments were performed on, 48 spayed, 44 neutered, 3 female, and 3 male patients. The most common applications for APS included injection post-ACL surgery (lateral imbrication surgery), bilateral hip OA, and bilateral stifle OA (Table 6). In some cases, repeat injections have been performed on the same patient. One 3 year-old patient with bilateral hip OA was injected with APS and responded well. Over time more NSAIDs were required and a second injection was performed 6 months later. One 11 year-old patient with bilateral stifle OA was injected with APS and responded well for 15 months and was re-injected with APS. In another case, starting at age 4, a patient received yearly APS injections in its stifles to manage its OA for the last 4 consecutive years (Table 6).
 |
Table 6 Description of APS Use in for a Variety of Orthopedic Conditions Including Tendon/Ligament Applications, OA, and Joint Applications |
In general, L-PRP and APS treatments were well-tolerated with transient inflammation that resolved over several days without intervention. No serious adverse events including infection or complications that required surgical intervention were recorded.
Discussion
The results of this case series suggested that L-PRP and APS have been clinically beneficial for OA in canine joints, tendon/ligament repair, and in post-surgical applications based on the number of positive owner-reported performance and safety outcomes. In general, a year or more of durable pain relief is seen after treatment with L-PRP or APS. Although there has been published research on L-PRP with the system used in this retrospective study in large animal medicine,4,5 this is the first study to characterize clinical outcomes in canine medicine. Historically, L-PRP has been used successfully in tendon/ligament applications.4,5 The results of this case series indicate that L-PRP is beneficial in tendon/ligament applications like patellar luxation and post-TPLO surgery. The data from this case series could be used to statistically power a randomized and controlled clinical trial with better defined endpoints.
Previous APS research has shown its positive short-term clinical benefits in addressing canine OA.18,32 Our case review shows that APS may have longer-term benefits in canine OA with patients with mild to moderate OA; with some cases receiving repeat injections yearly to manage symptoms. This mirrors the year-long benefit observed for equine subjects in a randomized and controlled study of APS.31 Although mostly successful, there have been some cases where APS has not produced clinically successful results. In most of these unsuccessful cases, patients presented with severe OA, as was noted in the records of 3/4 unsatisfactory bilateral hip patients. Similar results have been observed in horses,31 suggesting that APS is best suited for patients with mild to moderate OA. Additional studies are indicated to determine if these modalities are superior to other clinically relevant treatments and better define the durability of L-PRP and APS in longer-term randomized and controlled trials.
The management of canine OA with NSAIDs is standard practice for many veterinarians. However, their continued use is not without drawbacks. Common adverse effects are associated with the gastrointestinal (GI) tract, renal and hepatic adverse effects have been reported less frequently, while lethargy and inhibition of coagulation have been infrequently reported.49 Long-term NSAID use requires routine blood tests to check for these side effects. Studies reporting long-term safety data are lacking and adverse event reporting may be incomplete.50 These concerns have been cited as a barrier to their use by veterinarian surgeons.51 Furthermore, veterinary surgeons have described difficulty in recognizing, managing, and avoiding adverse events associated with NSAIDs.52 Survey data has indicated that there is a link between owner safety concerns and poor compliance.53 There are pragmatic concerns on the oral delivery of medications including palatability concerns by the patient and therefore, challenging delivery of the medication for the owner.54 These combined drawbacks have motivated many to seek alternative treatments to NSAIDs for canine osteoarthritis. L-PRP and APS do not require daily delivery of an oral medication, blood tests, and they have had an excellent safety profile in clinical trials. Although there have not been any head-to-head studies comparing the safety and effectiveness of the L-PRP or APS, we have had clinical success using these autologous orthobiologics as alternatives to NSAIDs. Future statistically powered clinical trials will be required to ultimately determine the differential safety and efficacy of the different modalities available to veterinarians.
There have been efforts to provide standardizing nomenclature to classify the outputs of orthobiologics devices. Most standardization systems classify products based on their centrifuge processing parameters (ie hard vs soft spin), if the product is clotted, platelet concentration, and white blood cell concentration.55–57 The outputs of orthobiologic devices can be altered with freezing, sonication, and other mechanical disruption methods.58 Further confounding classification, device output from the same individual can vary from the same patient if they have been exercising,59 have underlying health conditions,3 or have been taking medications.60 The products discussed in this paper were L-PRP, ACS, and APS devices. The L-PRP device in this study produces an output with a high concentration of platelets and WBC with a normal concentration of plasma proteins.61,62 ACS devices produce an output of a serum that contains plasma proteins and the WBC secretome when incubated in a clot with glass beads. ACS devices contain very little/no platelets or WBC.13 APS devices produce outputs with concentrated platelets, white blood cells and plasma proteins.62 Each one of these products contain different concentrations of different cells, cytokines, and growth factors.
There are a few limitations to our review. First, this study was a retrospective analysis of the procedures performed using L-PRP and APS in our practice over the last 5 years and was not a prospective or controlled study. Also, the evaluation was not quantitative but rather based on our evaluation and owner feedback. The time from treatment to follow-up varies from months-years in our review and is not standardized. For many of the conditions we used L-PRP or APS there are limited case numbers which makes it challenging to perform statistical analysis. Low sample number comparisons are susceptible to bringing people to incorrect decisions across disciplines.63 For some of the conditions we injected IA, SQ, or both depending on pathology. Finally, although most patients were instructed to stay off NSAIDs and other medications it is possible owners had their dogs on non-prescription medications we were unaware of during follow-up, started seeing different veterinarians, or moved. Despite these limitations, we feel this retrospective analysis adds additional information about the clinical application of L-PRP and APS in canine medicine.
Conclusion
This retrospective case series study demonstrated that L-PRP and APS have had beneficial clinical outcomes in canine medicine. The logistics of performing blood draws and joint injections are relatively simple and could be adopted into many small animal veterinary practices. In several patients OA has been managed with yearly injections of APS. Future randomized and controlled studies will be required to demonstrate the superiority of L-PRP and APS to other clinically relevant options in canine medicine.
Abbreviations
ACD-A, anticoagulant Citrate Dextrose Solution, Solution, A; ACL, anterior cruciate ligament; ACS, Autologous Conditioned Serum, APS, Autologous Protein Solution; CT, computed tomography; extracellular matrix, ECM; gastrointestinal, GI; IA, intra-articular; interleukin-1 receptor antagonist, IL-1ra or IRAP; L-PRP, leukocyte-rich platelet rich plasma; matrix metalloproteases, MMPs; magnetic resonance imaging, MRI; non-steroidal anti-inflammatory drugs, NSAIDS; OA, osteoarthritis; OCD, osteochondral dissecans; OMERACT-OARSI, Outcome Measures in Rheumatology - Osteoarthritis Research Society International; PRP, Platelet -rich plasma; P-PRP, pure platelet-rich plasma; RBC, red blood cells; SQ, subcutaneous; TPLO, tibial-plateau-leveling osteotomy; WBC, white blood cells.
Acknowledgments
We thank the staff at Indian Creek for supporting this study and enabling treatment of our clients.
Disclosure
The authors declare that this retrospective analysis received funding from Owl Manor. The funder was involved in reviewing and approving the study. WK is an employee of Owl Manor. KC is a consultant veterinarian for Owl Manor and has received research support from multiple orthopedic and regenerative medical companies. MB is a Registered Veterinary Technician consultant for Owl Manor with extensive interest and experience in surgical management and regenerative medical procedures. The authors report no other conflicts of interest in this work.
References
1. Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10(4):225–228. doi:10.1097/00008505-200110000-00002
2. Wehling P, Moser C, Frisbie D, et al. Autologous conditioned serum in the treatment of orthopedic diseases. BioDrugs. 2007;21(5):323–332. doi:10.2165/00063030-200721050-00004
3. O’Shaughnessey K, Matuska A, Hoeppner J, et al. Autologous protein solution prepared from the blood of osteoarthritic patients contains an enhanced profile of anti-inflammatory cytokines and anabolic growth factors. J Orthop Res. 2014;32(10):1349–1355. doi:10.1002/jor.22671
4. Bosch G, van Schie HT, de Groot MW, et al. Effects of platelet-rich plasma on the quality of repair of mechanically induced core lesions in equine superficial digital flexor tendons: a placebo-controlled experimental study. J Orthop Res. 2010;28(2):211–217. doi:10.1002/jor.20980
5. Bosch G, Moleman M, Barneveld A, van Weeren PR, Van Schie H. The effect of platelet-rich plasma on the neovascularization of surgically created equine superficial digital flexor tendon lesions. Scand J Med Sci Sports. 2011;21(4):554–561. doi:10.1111/j.1600-0838.2009.01070.x
6. Ortved KF. Regenerative medicine and rehabilitation for tendinous and ligamentous injuries in sport horses. Vet Clin Eq Pract. 2018;34(2):359–373.
7. Ho LK, Baltzer WI, Nemanic S, Stieger-Vanegas SM. Single ultrasound-guided platelet-rich plasma injection for treatment of supraspinatus tendinopathy in dogs. Can Vet J. 2015;56(8):845–849.
8. Yamada Y, Ueda M, Naiki T, Takahashi M, Hata K-I, Nagasaka T. Autogenous injectable bone for regeneration with mesenchymal stem cells and platelet-rich plasma: tissue-engineered bone regeneration. Tissue Eng. 2004;10(5–6):955–964. doi:10.1089/1076327041348284
9. Kim S-G, Chung C-H, Kim Y-K, Park J-C, Lim S-C. Use of particulate dentin–plaster of paris combination with/without platelet-rich plasma in the treatment of bone defects around implants. Int J Oral Maxillofac Implants. 2002;17(1):86–94.
10. Suba Z, Takács D, Gyulai-Gaál S, Kovács K. Facilitation of β-tricalcium phosphate-induced alveolar bone regeneration by platelet-rich plasma in beagle dogs: a Histologic and Histomorphometric Study. Int J Oral Maxillofac Implants. 2004;19(6):832–838.
11. Frisbie DD, Kawcak CE, Werpy NM, Park RD, McIlwraith CW. Clinical, biochemical, and histologic effects of intra-articular administration of autologous conditioned serum in horses with experimentally induced osteoarthritis. Am J Vet Res. 2007;68(3):290–296. doi:10.2460/ajvr.68.3.290
12. Baltzer A, Moser C, Jansen S, Krauspe R. Autologous conditioned serum (Orthokine) is an effective treatment for knee osteoarthritis. Osteoarthritis Cartilage. 2009;17(2):152–160. doi:10.1016/j.joca.2008.06.014
13. Sawyere DM, Lanz OI, Dahlgren LA, Barry SL, Nichols AC, Werre SR. Cytokine and growth factor concentrations in canine autologous conditioned serum. Vet Surg. 2016;45(5):582–586. doi:10.1111/vsu.12506
14. Smith RK. Regenerative medicine in equine orthopaedics: what and when? Vet Equine. 2020;4(1):8–13. doi:10.12968/ukve.2020.4.1.8
15. Woodell-May J, Matuska A, Oyster M, Welch Z, O’shaughnessey K, Hoeppner J. Autologous protein solution inhibits MMP-13 production by IL-1β and TNFα-stimulated human articular chondrocytes. J Orthop Res. 2011;29(9):1320–1326. doi:10.1002/jor.21384
16. Matuska A, O’shaughnessey K, King W, Woodell-May J. Autologous solution protects bovine cartilage explants from IL-1α-and TNFα-induced cartilage degradation. J Orthop Res. 2013;31(12):1929–1935. doi:10.1002/jor.22464
17. O’Shaughnessey KM, Panitch A, Woodell-May JE. Blood-derived anti-inflammatory protein solution blocks the effect of IL-1β on human macrophages in vitro. Inflamm Res. 2011;60(10):929. doi:10.1007/s00011-011-0353-2
18. Wanstrath AW, Hettlich BF, Su L, et al. Evaluation of a single intra-articular injection of autologous protein solution for treatment of osteoarthritis in a canine population. Vet Surg. 2016;45(6):764–774. doi:10.1111/vsu.12512
19. Franklin SP, Garner BC, Cook JL. Characteristics of canine platelet-rich plasma prepared with five commercially available systems. Am J Vet Res. 2015;76(9):822–827. doi:10.2460/ajvr.76.9.822
20. Franklin SP, Birdwhistell KE, Strelchik A, Garner BC, Brainard BM. Influence of cellular composition and exogenous activation on growth factor and cytokine concentrations in canine platelet-rich plasmas. Front Vet Sci. 2017;4:40. doi:10.3389/fvets.2017.00040
21. Carr BJ, Canapp JSO, Mason DR, Cox C, Hess T. Canine platelet-rich plasma systems: a prospective analysis. Front Vet Sci. 2016;2:73. doi:10.3389/fvets.2015.00073
22. King W, Toler K, Woodell-May J. Role of white blood cells in blood-and bone marrow-based autologous therapies. BioMed Res Int. 2018;2018:1–8. doi:10.1155/2018/6510842
23. McCarrel TM, Minas T, Fortier LA. Optimization of leukocyte concentration in platelet-rich plasma for the treatment of tendinopathy. JBJS. 2012;94(19):e143. doi:10.2106/JBJS.L.00019
24. Cavallo C, Filardo G, Mariani E, et al. Comparison of platelet-rich plasma formulations for cartilage healing: an in vitro study. JBJS. 2014;96(5):423–429. doi:10.2106/JBJS.M.00726
25. Anitua E, Zalduendo M, Troya M, Padilla S, Orive G. Leukocyte inclusion within a platelet rich plasma-derived fibrin scaffold stimulates a more pro-inflammatory environment and alters fibrin properties. PLoS One. 2015;10(3):e0121713. doi:10.1371/journal.pone.0121713
26. Browning SR, Weiser AM, Woolf N, et al. Platelet-rich plasma increases matrix metalloproteinases in cultures of human synovial fibroblasts. JBJS. 2012;94(23):e172. doi:10.2106/JBJS.K.01501
27. Sundman EA, Cole BJ, Fortier LA. Growth factor and catabolic cytokine concentrations are influenced by the cellular composition of platelet-rich plasma. Am J Sports Med. 2011;39(10):2135–2140. doi:10.1177/0363546511417792
28. van Drumpt RA, van der Weegen W, King W, Toler K, Macenski MM. Safety and treatment effectiveness of a single autologous protein solution injection in patients with knee osteoarthritis. BioRes Open Access. 2016;5(1):261–268. doi:10.1089/biores.2016.0014
29. Hix J, Klaassen M, Foreman R, et al. An autologous anti-inflammatory protein solution yielded a favorable safety profile and significant pain relief in an open-label pilot study of patients with osteoarthritis. BioRes Open Access. 2017;6(1):151–158. doi:10.1089/biores.2017.0027
30. Kon E, Engebretsen L, Verdonk P, Nehrer S, Filardo G. Clinical outcomes of knee osteoarthritis treated with an autologous protein solution injection: a 1-year Pilot Double-Blinded Randomized Controlled Trial. Am J Sports Med. 2018;46(1):171–180. doi:10.1177/0363546517732734
31. Bertone AL, Ishihara A, Zekas LJ, et al. Evaluation of a single intra-articular injection of autologous protein solution for treatment of osteoarthritis in horses. Am J Vet Res. 2014;75(2):141–151. doi:10.2460/ajvr.75.2.141
32. Franklin SP. Pilot study assessing treatment of canine hip dysplasia using autologous protein solution. Front Vet Sci. 2019;In Submission:1–9.
33. King W, van der Weegen W, Van Drumpt R, Soons H, Toler K, Woodell-May J. White blood cell concentration correlates with increased concentrations of IL-1ra and improvement in WOMAC pain scores in an open-label safety study of autologous protein solution. J Exp Orthop. 2016;3(1):9. doi:10.1186/s40634-016-0043-7
34. King W, Steckbeck K, O’shaughnessey K, Woodell-May J Effect of preparation technique on anti-inflammatory properties of autologous therapies.
35. Lorenz HP, Longaker MT. Wounds: biology, pathology, and management. In: Norton J, Bollinger RR, Chang AE, et al, Eds. Essential Practice of Surgery Basic Science and Clinical Evidence. New York, NY: Springer; 2003:77–88.
36. Cassano JM, Kennedy JG, Ross KA, Fraser EJ, Goodale MB, Fortier LA. Bone marrow concentrate and platelet-rich plasma differ in cell distribution and interleukin 1 receptor antagonist protein concentration. Knee Surg Sports Traumatol Arthrosc. 2018;26(1):333–342. doi:10.1007/s00167-016-3981-9
37. Marques-Smith P, Kallerud AS, Johansen GM, et al. Is clinical effect of autologous conditioned serum in spontaneously occurring equine articular lameness related to ACS cytokine profile? BMC Vet Res. 2020;16:1–9. doi:10.1186/s12917-020-02391-7
38. Chahla J, Cinque ME, Piuzzi NS, et al. A call for standardization in platelet-rich plasma preparation protocols and composition reporting: a systematic review of the clinical orthopaedic literature. J Bone Joint Surg Am. 2017;99(20):1769–1779. doi:10.2106/JBJS.16.01374
39. Franklin SP, Burke EE, Holmes SP. The effect of platelet-rich plasma on osseous healing in dogs undergoing high tibial osteotomy. PLoS One. 2017;12(5):e0177597. doi:10.1371/journal.pone.0177597
40. Souza TF, Andrade AL, Ferrreira G, et al. Healing and expression of growth factors (TGF-β and PDGF) in canine radial ostectomy gap containing platelet-rich plasma. Vet Comp Orthop Traumatol. 2012;25(06):445–452. doi:10.3415/VCOT-10-10-0146
41. Murray MM, Spindler KP, Devin C, et al. Use of a collagen-platelet rich plasma scaffold to stimulate healing of a central defect in the canine ACL. J Orthop Res. 2006;24(4):820–830. doi:10.1002/jor.20073
42. Franklin SP, Stoker AM, Bozynski CC, et al. Comparison of platelet-rich plasma, stromal vascular fraction (SVF), or SVF with an injectable PLGA nanofiber scaffold for the treatment of osteochondral injury in dogs. J Knee Surg. 2018;31(07):686–697.
43. Pashuck TD, Kuroki K, Cook CR, Stoker AM, Cook JL. Hyaluronic acid versus saline intra-articular injections for amelioration of chronic knee osteoarthritis: a canine model. J Orthop Res. 2016;34(10):1772–1779. doi:10.1002/jor.23191
44. Cook JL, Smith PA, Bozynski CC, et al. Multiple injections of leukoreduced platelet rich plasma reduce pain and functional impairment in a canine model of ACL and meniscal deficiency. J Orthop Res. 2016;34(4):607–615. doi:10.1002/jor.23054
45. Goldring SR, Goldring MB. The role of cytokines in cartilage matrix degeneration in osteoarthritis. Clin Orthop Relat Res. 2004;427(Suppl):S27–S36. doi:10.1097/01.blo.0000144854.66565.8f
46. Goldring MB. The role of the chondrocyte in osteoarthritis. J Arthritis Rheumatism. 2000;43(9):1916–1926. doi:10.1002/1529-0131(200009)43:9<1916::AID-ANR2>3.0.CO;2-I
47. King W, Bendele A, Marohl T, Woodell-May J. Human blood-based anti-inflammatory solution inhibits osteoarthritis progression in a meniscal-tear rat study. J Orthop Res. 2017;35(10):2260–2268. doi:10.1002/jor.23528
48. Fossum TW, Duprey LP, Hedlund CS, Duncan L. Small Animal Surgery; Mosby; 1997.
49. KuKanich B, Bidgood T, Knesl O. Clinical pharmacology of nonsteroidal anti-inflammatory drugs in dogs. Vet Anaesth Analg. 2012;39(1):69–90. doi:10.1111/j.1467-2995.2011.00675.x
50. Innes JF, Clayton J, Lascelles BDX. Review of the safety and efficacy of long-term NSAID use in the treatment of canine osteoarthritis. Vet Rec. 2010;166(8):226–230. doi:10.1136/vr.c97
51. Bell A, Helm J, Reid J. Veterinarians’ attitudes to chronic pain in dogs. Vet Rec. 2014;175(17):428. doi:10.1136/vr.102352
52. Belshaw Z, Asher L, Dean RS. The attitudes of owners and veterinary professionals in the United Kingdom to the risk of adverse events associated with using non-steroidal anti-inflammatory drugs (NSAIDs) to treat dogs with osteoarthritis. Prev Vet Med. 2016;131:121–126. doi:10.1016/j.prevetmed.2016.07.017
53. Zoetis I. Rimadyl chewable tablets: compliance unleashed; 2013. Available from: https://cupdf.com/document/rimadyl-unleashed.html.
54. Thombre AG. Oral delivery of medications to companion animals: palatability considerations. Adv Drug Deliv Rev. 2004;56(10):1399–1413. doi:10.1016/j.addr.2004.02.012
55. Ehrenfest DMD, Rasmusson L, Albrektsson T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte-and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009;27(3):158–167. doi:10.1016/j.tibtech.2008.11.009
56. DeLong JM, Russell RP, Mazzocca AD. Platelet-rich plasma: the PAW classification system. Arthroscopy. 2012;28(7):998–1009. doi:10.1016/j.arthro.2012.04.148
57. Rossi L, Murray I, Chu C, Muschler G, Rodeo S, Piuzzi N. Classification systems for platelet-rich plasma. Bone Joint J. 2019;101(8):891–896. doi:10.1302/0301-620X.101B8.BJJ-2019-0037.R1
58. McClain AK, McCarrel TM. The effect of four different freezing conditions and time in frozen storage on the concentration of commonly measured growth factors and enzymes in equine platelet-rich plasma over six months. BMC Vet Res. 2019;15(1):1–9. doi:10.1186/s12917-019-2040-4
59. Anz AW, Parsa RS, Romero-Creel MF, et al. Exercise-mobilized platelet-rich plasma: short-term exercise increases stem cell and platelet concentrations in platelet-rich plasma. Arthroscopy. 2019;35(1):192–200. doi:10.1016/j.arthro.2018.06.043
60. Mannava S, Whitney KE, Kennedy MI, et al. The influence of naproxen on biological factors in leukocyte-rich platelet-rich plasma: a prospective comparative study. Arthroscopy. 2019;35(1):201–210. doi:10.1016/j.arthro.2018.07.030
61. Hessel L, Bosch G, Van Weeren P, Ionita JC. Equine autologous platelet concentrates: a comparative study between different available systems. Equine Vet J. 2015;47(3):319–325. doi:10.1111/evj.12288
62. Muir SM, Reisbig N, Baria M, Kaeding C, Bertone AL. The concentration of plasma provides additional bioactive proteins in platelet and autologous protein solutions. Am J Sports Med. 2019;47(8):1955–1963. doi:10.1177/0363546519849671
63. Tversky A, Kahneman D. Belief in the law of small numbers. Psychol Bull. 1971;76(2):105. doi:10.1037/h0031322
64. Pietrzak WS, Eppley BL. Platelet rich plasma: biology and new technology. J Craniofac Surg. 2005;16(6):1043–1054. doi:10.1097/01.scs.0000186454.07097.bf
65. Anitua E, Andia I, Ardanza B, Nurden P, Nurden AT. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91(01):4–15. doi:10.1160/TH03-07-0440
66. Zhang J, Middleton KK, Fu FH, Im HJ, Wang JH. HGF mediates the anti-inflammatory effects of PRP on injured tendons. PLoS One. 2013;8(6):e67303. doi:10.1371/journal.pone.0067303
67. Matsui M, Tabata Y. Enhanced angiogenesis by multiple release of platelet-rich plasma contents and basic fibroblast growth factor from gelatin hydrogels. Acta Biomater. 2012;8(5):1792–1801. doi:10.1016/j.actbio.2012.01.016
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



