Back to Journals » Journal of Inflammation Research » Volume 15
Systemic Immune-Inflammation Index Predicts Long-Term Outcomes in Patients with Three-Vessel Coronary Disease After Revascularization: Results from a Large Cohort of 3561 Patients
Authors Zhao J, Lv H , Yin D, Zhou X, Zhu H , Guo L , Wang J
Received 21 August 2022
Accepted for publication 9 September 2022
Published 12 September 2022 Volume 2022:15 Pages 5283—5292
DOI https://doi.org/10.2147/JIR.S385990
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Ji Zhao, Haichen Lv, Da Yin, Xuchen Zhou, Hao Zhu, Lei Guo, Junjie Wang
Department of Cardiology, the First Affiliated Hospital of Dalian Medical University, Dalian City, People’s Republic of China
Correspondence: Lei Guo; Junjie Wang, Department of Cardiology, the First Affiliated Hospital of Dalian Medical University, 222 Zhongshan Road, Dalian City, 116011, People’s Republic of China, Tel +86 411-83635963, Email [email protected]; [email protected]
Objective: This study aimed to investigate the prognostic value of systemic immune inflammation index (SII) concerning long-term outcomes in patients with the three-vessel disease (TVD) after revascularization in a large cohort.
Methods: In total, 3561 TVD patients who had undergone revascularization between 2013 and 2018 were included in the study. Patients were divided into the low SII (< 694.3 × 109/L) (n = 2556, 71.8%) and the high SII (≥ 694.3 × 109/L) group (n = 1005, 28.2%). The C-index, net reclassification improvement (NRI), and integrated discrimination improvement (IDI) were calculated to assess whether the addition of SII to a baseline model with traditional risk factors improved the accuracy of cardiac event prediction. The primary outcome was the frequency of major adverse and cerebrovascular events (MACCE). The secondary outcome was the incidence of all-cause death.
Results: After 2.4 years of follow-up, the Cox proportional hazard regression model analysis displayed that high SII was independently associated with an increased risk of developing future MACCE (hazard ratio [HR] 1.65, 95% confidence interval [CI] 1.23– 2.21, p = 0.001) and all-cause death (HR: 2.96; 95% CI: 1.19– 7.32, p = 0.019). The addition of SII significantly improved the reclassification beyond the baseline model with traditional risk factors (MACCE: NRI, 0.115; p = 0.0001; all-cause death: NRI, 0.369; p = 0.0001). Reclassification with the addition of SII also demonstrated an IDI of 0.0022 (p = 0.006) in MACCE and 0.0033 (p = 0.014) in all-cause death.
Conclusion: In TVD patients after revascularization, increased SII is an independent prognostic factor for long-term outcomes of MACCE and death. Compared to traditional risk factors, SII improved the risk prediction of major cardiovascular events in TVD patients who underwent revascularization.
Keywords: three-vessel disease, revascularization, systemic immune inflammation index, outcomes, coronary artery disease
Introduction
As a distinct and severe type of coronary artery disease (CAD), three-vessel disease (TVD) presents in nearly 30% of CAD patients. Characterized by significant stenosis in all three major coronary arteries, TVD is one of the biggest challenges in coronary intervention.1 TVD Patients are at the highest risk for cardiovascular adverse events in CAD.2 Revascularization has been found to lead to long-term benefits among TVD patients.3
Scientific evidence has depicted that inflammation plays an important role in atherosclerotic CAD pathogenesis. Recently, emerging data have shown that some inflammation and immune indices based on the complete blood count, such as platelet-lymphocyte ratio (PLR) and neutrophil-lymphocyte ratio (NLR), provide useful information for predicting future adverse cardiovascular events in patients with CAD.4,5 Systemic immune inflammation index (SII) is a new parameter for assessing inflammation. Essentially, it is a composite inflammatory indicator that combines three significant immune cells, namely, neutrophils, lymphocytes, and platelets. Recently, SII has been proposed to assess both inflammatory and immune status,6 is considered an excellent indicator of local immune response and systemic inflammation. Initially, SII was used to assess the prognosis of cancer patients.7 Recently, several studies have reported SII to be associated with adverse outcomes in cardiovascular disease patients, such as chronic heart failure, atrial fibrillation, and peripartum cardiomyopathy.8–10
Compared with single-vessel disease, TVD patients have a more prominent inflammatory state. Additionally, TVD confers an almost two-fold higher risk of mortality, which is a significant risk factor for worse outcomes.11,12 However, the relationship between SII and clinical outcomes in TVD patients was still unexplored. Therefore, this study aimed to investigate the prognostic value of SII concerning long-term outcomes in TVD patients after revascularization in a large cohort.
Methods
Study Population
In our institution, 13,890 patients underwent coronary angiography between January 2013 and December 2018. Out of these 13,890 patients, 4308 (31.0%) were TVD patients. The exclusion criteria comprised patients receiving medical therapy alone, patients with malignant tumors, immune disease, severe liver diseases, and renal failure, as well as clinically evident acute or chronic inflammation. In total, 3561 TVD patients undergoing revascularization were included in this study. Revascularization includes percutaneous coronary intervention (PCI) or coronary artery bypass graft surgery (CABG).2 Study participants were categorized into 2 groups according to preprocedural SII (694.3 × 109/L) as determined by the receiver operating characteristics (ROC) curve analysis.13,14 Baseline demographics, procedural characteristics, and in-hospital outcome data were collected from medical records and compiled in a dedicated database. Data on long-term clinical outcomes were obtained by telephone interviews with patients or their relatives, outpatient visits, and hospital readmissions.
Ethics
This single-center, retrospective observational study was approved by the Ethics Committee of the First Affiliated Hospital of Dalian Medical University (Ethics No: YJ-KY-FB-2021-10). Study protocols were consistent with the Declaration of Helsinki. All participants provided their written informed consent.
Procedures
Coronary interventions were performed according to the current standard guidelines. All patients were pre-treated with aspirin and clopidogrel prior to catheterization. During the procedure, unfractionated heparin (100 U/kg) was administered to all patients; the use of glycoprotein IIb/IIIa inhibitors was left to the discretion of the medical team. Post dilatation was performed with additional balloons to ensure optimum stent apposition. All deployed stents were drug-eluting stents (DES). After DES implantation, dual-antiplatelet medications were administered for a minimum of 12 months.15,16 CABG procedures could be done with or without extracorporeal circulation, and use of arterial conduits was encouraged.
Definition and Endpoints
TVD was defined as angiographic stenosis of ≥ 50% in all three main coronary arteries, including the left anterior descending, left circumflex, and right coronary arteries, with or without the involvement of the left main artery. SII was calculated as absolute peripheral neutrophil count × platelet count/lymphocyte count.6 The primary endpoint was set as major adverse cardiac and cerebrovascular events (MACCE), consisting of all-cause death, myocardial infarction (MI), repeat revascularization, stroke, and readmission for angina pectoris or heart failure. All-cause death was the secondary outcome.
Statistical Analysis
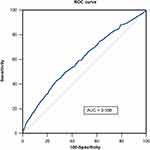
Kolmogorov–Smirnov test was used to test normality. Continuous variables were expressed as mean ± standard deviation or median and interquartile ranges and compared using Student’s t-test or Mann–Whitney U-test. Categorical variables were expressed as numbers and percentages. Chi-square or Fisher’s exact tests were used to evaluate the differences. Survival-free of adverse events was calculated by the Kaplan-Meier analysis and compared using the Log rank test. Three models were used to evaluate potential confounders’ effects on the association between SII level and long-term outcomes. Model 1 adjusted for age and sex; Model 2 adjusted for age, sex, hypertension, diabetes, and chronic kidney disease (CKD); and Model 3 adjusted for age, sex, hypertension, diabetes, CKD, lipid profile, left ventricular ejection fraction (LVEF), and acute coronary syndrome (ACS). The ROC curve analysis was used to evaluate the sensitivity and specificity of the SII for predicting MACCEs (Figure 1). The area under the curve (AUC) was utilized as a measure of the predictive accuracy of SII. To assess for improvements in the accuracy of adverse cardiac event predictions after adding SII to a baseline model with traditional risk factors (ie, age, sex, hypertension, and diabetes), the C-index, net reclassification improvement (NRI), and integrated discrimination improvement (IDI) were calculated. All two-sided P values < 0.05 were considered statistically significant. All statistical analyses were conducted using SPSS version 24.0 and R version 4.1.3.
Results
Baseline and Procedural Characteristics
The study participants were divided into low SII (<694.3 × 109/L) (n = 2556, 71.8%) and high SII (≥694.3 × 109/L) groups (n = 1005, 28.2%). 3401 (95.5%) patients underwent PCI and 160 (4.5%) patients were treated with CABG. Table 1 summarizes the characteristics of the cohort. Patients with higher SII had a significantly higher incidence of CKD, ACS, and in-hospital death, as well as higher baseline white blood cell (WBC), neutrophil, lymphocyte, platelet levels, NLR and PLR on admission, but a lower incidence of smoking and a history of peripheral artery disease. However, they had lower levels of blood pressure, glucose, LVEF, low-density lipoprotein cholesterol (HDL-C), triglyceride, and hemoglobin. There were no differences between the groups regarding the prevalence of comorbidities, including smoking, hypertension, diabetes, and dyslipidemia. Additionally, insignificant differences were found in C-reactive protein (CRP) and the SYNTAX score.
 |
Table 1 Baseline Clinical, Angiographic, and Procedural Characteristics and in-Hospital Outcome in Patients with TVD After Revascularization According to SII |
Association Between SII and Outcomes
The median follow-up period was 2.4 (1.1 to 4.1) years. In the higher SII group, there were 268 (26.7%) MACCEs, comprising 25 (2.5%) all-cause deaths, 101 (10%) MI, 170 (16.9%) repeat revascularizations, 117 (11.6%) angina pectoris readmissions, 86 (8.6%) heart failure readmissions, and 14 (1.4%) nonfatal strokes. The Cox proportional hazard regression model analysis revealed that high SII was independently associated with increased risk of developing future MACCE (hazard ratio [HR] 1.65, 95% confidence interval [CI] 1.23–2.21, p = 0.001), all-cause death (HR: 2.96; 95% CI: 1.19–7.32, p = 0.019), repeat revascularization (HR: 1.95; 95% CI: 1.38–2.76, p < 0.001), pectoris readmission (HR: 1.77; 95% CI: 1.19–2.63, p = 0.004), heart failure readmission (HR: 1.78; 95% CI: 1.02–3.10, p = 0.041) and stroke (HR: 5.81; 95% CI: 1.69–19.97, p = 0.005) except for MI (HR: 1.27; 95% CI: 0.71–2.27, p = 0.420) after adjusting for age, gender, hypertension, diabetes, CKD, lipid profile, LVEF, and the presence of ACS during enrolment according to the multivariable Model 3 (Table 2). Furthermore, the association between MACCE and SII remained significant in multivariable Model 1 and Model 2. Moreover, Kaplan-Meier analysis revealed that patients with high SII had higher MACCE rates than patients with low SII in the long-term follow-up (Table 2, Figure 2).
 |
Table 2 Association Between SII and Adverse Events in Patients with TVD After Revascularization |
Adding SII to Baseline Model for Prediction of Outcomes
The addition of SII to a baseline model with traditional risk factors led to improvements in the prediction of MACCE (p < 0.001), all-cause death (p < 0.001), MI (p < 0.001), repeat revascularization (p = 0.003) and heart failure readmission (p < 0.001), as demonstrated by the significantly increased C-index. The addition of SII significantly improved reclassification beyond the baseline model with traditional risk factors (MACCE: NRI, 0.115; p = 0.0001; all-cause death: NRI, 0.369; p = 0.0001). Reclassification adding SII also indicated an IDI of 0.0022 (p = 0.006) in MACCE and 0.0033 (p = 0.014) in all-cause death, suggesting that the addition of SII leads to a significantly improved predictive value than traditional risk factors in TVD patients after revascularization (Table 3).
 |
Table 3 Evaluation of Predictive Models for Adverse Events Using the C-Index, NRI and IDI |
Discussion
To the best of our knowledge, this is the first study analyzing the ability of SII to predict clinical outcomes among patients with TVD after revascularization. Based on the findings, increased SII is directly related to the incidence of MACCEs and long-term mortality rates in these patients.
Chronic inflammation is known to be a major contributor to atherosclerotic CAD. As the first inflammatory cell involved in plaque formation, neutrophils attract leukocytes and promote foam cell formation.17 Additionally, peripheral lymphopenia during MI has been noted as a sign of severe response to MI. Higher platelet counts may be related to worse outcomes by demonstrating the increased release of inflammatory mediators. Increased platelet activation leads to destructive inflammatory responses and a prothrombotic state.18 In particular, neutrophil extracellular traps can induce numerous prothrombotic effects, including platelet adhesion, activation, and aggregation,19 which may lead to thrombosis and increase the risk of acute complications such as stroke. Evidence shows that platelet adhesion to vessel walls enhances leukocyte recruitment and precedes atherosclerotic plaque invasion by leukocytes, initiating atheroprogression.20
The NLR and the PLR have been introduced as novel markers. NLR has been reported as an independent predictor of cardiovascular events and mortality in ST-segment elevation myocardial infarction (STEMI).4 A meta-analysis demonstrated that in STEMI patients who were treated with PCI, higher PLR before the procedure was associated with poor in-hospital and long-term prognosis.21 In addition to the NLR and PLR, platelets play an important role in the pathogenesis of CAD and ACS. Neutrophils, platelets and the cytokines they produce are mainly related to non-specific immune responses, while lymphocytes are considered to be mainly related to specific immune pathways. Remarkably, the SII, a relatively novel inflammatory marker combining platelet, neutrophil, and lymphocyte counts, can better reflect the immune and inflammatory state of the body than NLR and PLR which are typically developed from two cells or any one of these other markers in isolation.6 Therefore, SII is superior to other systemic inflammation index in prognostic assessment of CAD. CRP has been proven to be associated with increased relative risks of cardiovascular events in ACS patients.22 However, CRP is not specific and is affected by many factors. The SII is thought to be more specific than CRP.7 In addition, the SII has been widely reported to be associated with poor outcomes in patients with various malignancies.
Among CAD patients, those with TVD have the highest risk for cardiovascular adverse events. Based on multivariable analysis, our previous studies have reported that clinical descriptors such as diabetes mellitus, low LVEF of 40%, and left main coronary artery disease were independent predictors of MACCE in TVD patients. Recently, several studies have reported that SII was independently associated with contrast-induced nephropathy development after PCI in patients with non-STEMI, coronary collateral circulation formation in stable CAD or mortality in patients with chronic heart failure.10,23,24 However, in the current literature, there is no prior clinical study demonstrating the prognostic value of SII for long-term outcomes in TVD patients after revascularization. An early, simple, inexpensive clinical or laboratory marker must be identified for these high-risk patients.
In this study, various multivariable Cox regression analysis models were generated to assess the independent effects of the SII and outcomes of TVD patients after revascularization after adjusting for other potential confounders. The addition of SII to the clinical model improved the predictive power for adverse cardiovascular events in TVD patients after revascularization via the C-index. Additionally, it improved the reclassification of the subjects into different risk categories by performing the NRI and IDI. Based on our findings, the inflammatory status indicated by SII is an effective parameter in predicting MACCEs in TVD patients after revascularization. The present study might be the first to demonstrate the good predictive value of SII for MACCEs in TVD patients undergoing revascularization.
Aspirin and statins have been displayed to decrease thrombosis and inflammation indirectly. Therefore, these are often used before procedures to reduce cardiovascular events.25 Recently, Ridker et al reported that anti-inflammatory therapy targeting interleukin-1β significantly reduced the rate of cardiovascular events in patients with previous MI.26 Further studies are required to determine whether anti-inflammatory therapy can reduce the occurrence of clinical adverse events in TVD patients with a high level of SII after revascularization.
As a simple and inexpensive index that can be easily calculated from complete blood count (CBC) parameters, SII is a practical and reliable indicator. Therefore, it can be widely used to predict recovery rates and assist clinicians in rapidly identifying high-risk patients with worse prognoses for adverse cardiovascular events. Our study suggests that TVD patients with a high SII count after revascularization should be carefully evaluated for further clinical monitoring and treatment.
Limitations
First, the major limitation of this study was its single-center observational design. Despite the use of multivariable analysis, there may have been certain unmeasured confounders that could have affected the study results. Second, we calculated SII only once at admission and did not monitor changes of SII during the study period.
Conclusion
In TVD patients after revascularization, increased SII is an independent prognostic factor for long-term outcomes of MACCE and death. Compared to traditional risk factors, SII improved the risk prediction of major cardiovascular events in TVD patients who underwent revascularization. Therefore, as an easy and practical indicator, SII has the potential to help clinicians identify high-risk patients.
Abbreviations
SII, systemic immune-inflammation index; TVD, three-vessel disease; PCI, percutaneous coronary intervention; MACCE, major adverse and cerebrovascular events; CAD, coronary artery disease.
Data Sharing Statement
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgment
The authors acknowledge all participants of the study and the interventional cardiologists and surgeons from the First Affiliated Hospital of Dalian Medical University.
Funding
This study was supported by the the National Natural Science Foundation of China (82170252), Dalian Medical Science Research Project (2112002) and Dalian Youth Science and Technology Star Support program (2021RQ006).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bradley SM, Spertus JA, Kennedy KF, et al. Patient selection for diagnostic coronary angiography and hospital-level percutaneous coronary intervention appropriateness: insights from the National Cardiovascular Data Registry. JAMA Intern Med. 2014;174(10):1630–1639. doi:10.1001/jamainternmed.2014.3904
2. Liu Y, Zhu Y, Wang J, et al. Gender-based long-term outcomes after revascularization for three-vessel coronary disease: a propensity score-matched analysis of a large cohort. Clin Interv Aging. 2022;17:545–554. doi:10.2147/CIA.S362027
3. Thuijs DJ, Kappetein AP, Serruys PW, et al. Percutaneous coronary intervention versus coronary artery bypass grafting in patients with three-vessel or left main coronary artery disease: 10-year follow-up of the multicentre randomised controlled SYNTAX trial. Lancet. 2019;394(10206):1325–1334. doi:10.1016/S0140-6736(19)31997-X
4. Han YC, Yang TH, Kim DI, et al. Neutrophil to lymphocyte ratio predicts long-term clinical outcomes in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Korean Circ J. 2013;43(2):93–99. doi:10.4070/kcj.2013.43.2.93
5. Arbel Y, Finkelstein A, Halkin A, et al. Neutrophil/lymphocyte ratio is related to the severity of coronary artery disease and clinical outcome in patients undergoing angiography. Atherosclerosis. 2012;225(2):456–460. doi:10.1016/j.atherosclerosis.2012.09.009
6. Hu B, Yang XR, Xu Y, et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin Cancer Res. 2014;20(23):6212–6222. doi:10.1158/1078-0432.CCR-14-0442
7. Yang R, Chang Q, Meng X, Gao N, Wang W. Prognostic value of Systemic immune-inflammation index in cancer: a meta-analysis. J Cancer. 2018;9(18):3295–3302. doi:10.7150/jca.25691
8. Luo Y, Zhang J, Liu T, et al. The systemic-immune-inflammation index predicts the recurrence of atrial fibrillation after cryomaze concomitant with mitral valve surgery. BMC Cardiovasc Disord. 2022;22(1):45. doi:10.1186/s12872-022-02494-z
9. Zhang Y, Liu W, Yu H, et al. Value of the Systemic Immune-Inflammatory Index (SII) in predicting the prognosis of patients with peripartum cardiomyopathy. Front Cardiovasc Med. 2022;9:811079. doi:10.3389/fcvm.2022.811079
10. Seo M, Yamada T, Morita T, et al. P589Prognostic value of systemic immune-inflammation index in patients with chronic heart failure. Eur Heart J. 2018;39:
11. Min JK, Dunning A, Lin FY, et al. Age- and sex-related differences in all-cause mortality risk based on coronary computed tomography angiography findings results from the international multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: an International Multicenter Registry) of 23,854 patients without known coronary artery disease. J Am Coll Cardiol. 2011;58(8):849–860. doi:10.1016/j.jacc.2011.02.074
12. Zhao X, Jiang L, Xu L, et al. Predictive value of in-hospital white blood cell count in Chinese patients with triple-vessel coronary disease. Eur J Prev Cardiol. 2019;26(8):872–882. doi:10.1177/2047487319826398
13. Leu HB, Yin WH, Tseng WK, et al. Identification of new biosignatures for clinical outcomes in stable coronary artery disease - The study protocol and initial observations of a prospective follow-up study in Taiwan. BMC Cardiovasc Disord. 2017;17(1):42. doi:10.1186/s12872-017-0471-z
14. Yang YL, Wu CH, Hsu PF, et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur J Clin Invest. 2020;50(5):e13230. doi:10.1111/eci.13230
15. Guo L, Wang J, Ding H, et al. Long-term outcomes of medical therapy versus successful recanalisation for coronary chronic total occlusions in patients with and without type 2 diabetes mellitus. Cardiovasc Diabetol. 2020;19(1):100. doi:10.1186/s12933-020-01087-4
16. Guo L, Zhong L, Chen K, Wu J, Huang RC. Long-term clinical outcomes of optimal medical therapy vs. successful percutaneous coronary intervention for patients with coronary chronic total occlusions. Hellenic J Cardiol. 2018;59(5):281–287. doi:10.1016/j.hjc.2018.03.005
17. Carbone F, Nencioni A, Mach F, Vuilleumier N, Montecucco F. Pathophysiological role of neutrophils in acute myocardial infarction. Thromb Haemost. 2013;110(3):501–514. doi:10.1160/TH13-03-0211
18. Koupenova M, Clancy L, Corkrey HA, Freedman JE. Circulating platelets as mediators of immunity, inflammation, and thrombosis. Circ Res. 2018;122(2):337–351. doi:10.1161/CIRCRESAHA.117.310795
19. Warnatsch A, Ioannou M, Wang Q, Papayannopoulos V. Inflammation. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science. 2015;349(6245):316–320. doi:10.1126/science.aaa8064
20. Czyz A, Kołacz E, Angerer D, Zawilska K. Expression of activation antigens on lymphocyte surface and circulating platelet-leukocyte aggregates in ischaemic heart disease. Kardiol Pol. 2005;62(3):
21. Willim HA, Harianto JC, Cipta H. Platelet-to-lymphocyte ratio at admission as a predictor of in-hospital and long-term outcomes in patients with st-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention: a systematic review and meta-analysis. Cardiol Res. 2021;12(2):109–116. doi:10.14740/cr1219
22. He LP, Tang XY, Ling WH, Chen WQ, Chen YM. Early C-reactive protein in the prediction of long-term outcomes after acute coronary syndromes: a meta-analysis of longitudinal studies. Heart. 2010;96(5):339–346. doi:10.1136/hrt.2009.174912
23. Kelesoglu S, Yilmaz Y, Elcık D, Kalay N. Systemic immune inflammation index: a novel predictor for coronary collateral circulation. Perfusion. 2021;37(6):605–612. doi:10.1177/02676591211014822
24. Kelesoglu S, Yilmaz Y, Elcık D, et al. Systemic immune inflammation index: a novel predictor of contrast-induced nephropathy in patients with non-st segment elevation myocardial infarction. Angiology. 2021;72(9):889–895. doi:10.1177/00033197211007738
25. Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med. 1997;336(14):973–979. doi:10.1056/NEJM199704033361401
26. Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–1131. doi:10.1056/NEJMoa1707914
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Elevated HsCRP in Chronic Obstructive Pulmonary Disease: A Prospective Study of Long-Term Outcomes After Percutaneous Coronary Intervention
Zheng Y, Qi Y, Seery S, Yang J, Li C, Wang W, Gao J, Meng X, Shao C, Tang YD
International Journal of Chronic Obstructive Pulmonary Disease 2022, 17:2517-2528
Published Date: 7 October 2022
Prognostic Value of Fibrinogen-to-Albumin Ratio in Coronary Three-Vessel Disease
Li X, Wang Z, Zhu Y, Lv H, Zhou X, Zhu H, Liu J, Guo L
Journal of Inflammation Research 2023, 16:5767-5777
Published Date: 1 December 2023