Back to Journals » Patient Preference and Adherence » Volume 16
Relation Among Anxiety, Depression, Sleep Quality and Health-Related Quality of Life Among Patients with Systemic Lupus Erythematosus: Path Analysis
Authors Chen HJ, Wang H, Qiu LJ, Ling HY, Wu LL, Wang TR, Zhou Y, Xue Y, Ye DQ, Wang B ![]()
Received 18 March 2022
Accepted for publication 10 May 2022
Published 25 May 2022 Volume 2022:16 Pages 1351—1358
DOI https://doi.org/10.2147/PPA.S366083
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Naifeng Liu
Hui-Juan Chen,1,2,* Hong Wang,1,2,* Li-Juan Qiu,1,2 Hua-Yun Ling,1,2 Ling-Ling Wu,1,2 Ting-Rui Wang,1,2 Ying Zhou,1,2 Yu Xue,1,2 Dong-Qing Ye,1,2 Bin Wang1,2
1Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University, Hefei, People’s Republic of China; 2The Key Laboratory of Major Autoimmune Diseases, Anhui Medical University, Hefei, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Bin Wang, Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University, 81 Meishan Road, Hefei, Anhui, 230032, People’s Republic of China, Email [email protected]
Purpose: This study aimed to examine the relationship between anxiety, depression, sleep quality and health-related quality of life among systemic lupus erythematosus (SLE) patients in China.
Patients and Methods: After ethical approval and obtaining participants’ informed consent, a cross-sectional study was conducted in The First Affiliated Hospital of Anhui Medical University between October 1, 2021 and January 30, 2022. The data comprised demographic information, number of SLE symptoms, Hospital Anxiety and Depression Scale (HADS), Pittsburgh Sleep Quality Index (PSQI) and Systemic Lupus Erythematosus-Specific Quality of Life Questionnaire (SLEQoL). We performed descriptive statistics, Spearman or Pearson correlations, and multiple linear regression. And Path analysis was performed to examine direct and indirect associations between these variables and health-related quality of life.
Results: A total of 580 patients were recruited and 513 met our target criteria. Our final model fitted the data well: goodness-of-fit index (GFI) =0.996; adjusted goodness-of-fit index (AGFI) =0.974; comparative fit index (CFI) =0.998; root mean square error of approximation (RMSEA) =0.043. This model explained 57.3% of the variance on health-related quality of life (HRQoL) in patients with SLE and all the hypothesized paths reached significance (P< 0.05). Anxiety, depression, sleep quality, income/family, and number of SLE symptoms were related to health-related quality of life, and anxiety had the most influence on HRQoL (β=0.561).
Conclusion: The study model helps to explain the relation among anxiety, depression, sleep quality and health-related quality of life in patients with SLE. It also suggests that health care professionals should be aware of factors such as anxiety, sleep quality, number of SLE symptoms, and depression in their care for HRQoL of SLE patients.
Keywords: anxiety, sleep, depression, quality of life, systemic lupus erythematosus
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease whose relapse-remission process can involve multiple organs or systems.1 Although over the decades, a sharp increase of patient survival has been observed, morbidity and mortality rates remain high.2 The disease causes significant economic costs as well as unquantifiable costs such as reduced health-related quality of life (HRQoL).3
Patients with SLE have poorer health-related quality of life (HRQoL), which may be worse than patients with other chronic conditions, such as congestive heart failure, myocardial infarction, and diabetes.4 Paying attention to HRQoL in SLE patients is of great importance in evaluating SLE treatment and predicting mortality.5
Health-related quality of life (HRQoL), a multi-dimensional concept that focuses on the patient’s overall perception of the impact of a disease,6 should be one of the comprehensive descriptions of patients with SLE according to the recommendation of the Systemic Lupus International Collaborating Clinics (SLICC) group.7 During the long-term process, many patients experience emotional burdens and poor sleep quality. The prevalence of depression and anxiety in SLE ranged from 8.7% to 78.6% and 1.1% to 71.4%.8 And SLE patients without major neuropsychiatric symptoms have high symptoms of depression and anxiety.9 Depression and anxiety, which lead to a severe decrease in quality of life in patients with SLE, were also significant negative predictors.10 Sleep disorders are common in patients with SLE, with prevalence ranging from 55% to 85%.11 Poor sleepers had impaired HRQoL in almost all domains of the Lupus Quality of Life than good sleeper. However, previous studies have paid attention to depression, anxiety and sleep quality as independent predictors for HROoL. There were few published literatures that explore the direct and indirect effects of various factors on HRQoL in patients with SLE using a structural equation modeling (SEM) framework. Path analysis, a statistical analysis method used in structural equation modeling (SEM) can measure causality between variables as well as direct and indirect effects.12 This study constructed a SEM framework and used path analysis to investigate the direct and indirect effects of various factors on HRQoL in patients with SLE.
Patients and Methods
The ethical approval was obtained from Biomedical Ethics Committee of Anhui Medical University (approval number: 20150111). After obtaining participants’ informed consent, a cross-sectional study was conducted in The First Affiliated Hospital of Anhui Medical University between October 1, 2021 and January 30, 2022. Participants of this study were recruited from the rheumatology outpatient clinic with a diagnosis of SLE (according to the 1997 American College of Rheumatology). Outpatients who consented to participate and met the following criteria were included in the study: 1) 3 or more months when diagnosed with SLE; 2) 18 years old or older. Exclusion criteria were as follows: 1) patients with SLE combined with other diseases that significantly affected the HRQoL, such as severe heart, liver, kidney, blood system diseases, chronic obstructive pulmonary disease, and malignant tumors (according to medical records); 2) people with severe cognitive impairment or mental illness, and unable to cooperate (according to medical records).
Measurement of Basic Information and Clinical Symptoms
Demographic data including age (years), age when diagnosed with SLE (years), gender, level of education (middle school or below, high school, bachelor’s degree or above), income/family (whether the income can meet the family expenses). SLE symptoms from the Screening Questionnaire for Detection of SLE (SQD). SQD was a 10-item screening questionnaire with a sensitivity of 92.8% and specificity of 76.4% based on the American Rheumatism Association preliminary criteria for SLE (“Yes” answer = 1 point, “No” answer = 0 point).13
Anxiety and Depression
Anxiety and depression symptoms were assessed with the Hospital Anxiety and Depression Scale (HADS), consisting of 14 items and two subscales: Hospital Anxiety and Depression Scale Anxiety subscale (HADS-A) (7 items) and Hospital Anxiety and Depression Scale Depression subscale (HADS-D) (7 items).14 Each item uses a 4-point Likert scale (0 = not at all, to 3 = most of the time) and the total subscale score ranges from 0 to 21, with higher scores indicating more anxiety or depression. Scores on each subscale can be interpreted in ranges: normal (<8 points), suggestive of presence of either anxiety or depression (≥8 and <11 points), and probable presence of mood disorder (≥11 points).15 The Chinese version of HADS was applied in the study due to its reliability and effectiveness.16 And internal consistency of our study was good (Cronbach’s alphas was 0.82). The questionnaire was given to each patient, asking them to answer each question according to the way they had felt the last 7 days including the day of the application of the instrument.
Sleep Quality
The Pittsburgh Sleep Quality Index (PSQI) is a 19-item, self-report questionnaire that measures sleep quality and degree of sleep difficulties over the past month,17 it contains seven component scores: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleep medications, and daytime dysfunction. These are summed to yield a total PSQI score with a range of 0–21, higher scores indicate poorer sleep quality. The internal consistency was good based on a review (Cronbach’s alphas ranged from 0.70 to 0.83) and the Chinese version of PSQI has been widely used and well validated.18–20 Our study’s Cronbach’s alphas was 0.80. In our study, a PSQI score of <7 and ≥7 was defined as “good sleep quality” and “poor sleep quality,” respectively.21
Quality of Life
General health questionnaire measurement had its own defects. Such as the 36-item Short-Form Health Survey (SF-36) and Health Assessment Questionnaire. The former missed several issues of importance to patients with SLE, the latter was criticized for its non-specificity.22 Chinese version of the Systemic Lupus Erythematosus-Specific Quality of Life Questionnaire (SLEQOL-C) is a reliable assessment of health-related quality of life (HRQoL) in Chinese SLE patients. The SLEQOL-C assesses HRQoL in the past week and comprises 40 items that fall into six domains, including physical functioning (6 items), activities (9 items), symptoms (8 items), treatment (4 items), mood (4 items), and self-image (9 items). The participants were asked to rate each item on a 7-point scale ranging from 1 to 7 (including “not difficult at all”, to “extremely difficult”; “not at all troubled” to “extremely troubled”, and “not at all often” to “extremely often”). The total score, which is the sum of the 40 item scores, ranges from 40 to 280. The higher the SLEQOL-C scores, the worse the self-perceived HRQoL.23 The internal consistency of our study was good (Cronbach’s alphas was 0.85).
Statistical Analysis
The continuous variables were expressed as mean (standard deviation) if they were normal distribution, the Kolmogorov–Smirnov test was employed to determine normality. Frequency and percentages were used for categorical variables. The associations between anxiety, depression, sleep quality, HRQoL and other variables were determined using Spearman’s ρ or Pearson’s Product Moment correlation. Variables found to have significant correlation with HRQoL were then included in a multiple linear regression model. Variables with statistical significance in correlation analysis were taken as independent variables and HRQoL as dependent variables to establish a multiple linear regression model. Findings from the multiple linear regression analyses were used to construct a structural equation model to test the interrelations between variables. Model fit was assessed with path analysis, as per convention, acceptable model fit was defined as having a nonsignificant chi-square (χ2) value, a root mean square of approximation (RMSEA) <0.05, a Tucker Lewis coefficient (TLI) of ≥0.9, and a comparative fit index (CFI) of ≥0.9.24 Structural equation modeling was used to estimate the total effect of each predictor, to establish a linear model to predict HRQoL with these interrelations accounted for.
All statistical analyses were conducted by Statistical Package for the Social Sciences version 23.0 (Chicago, SPSS Inc.) and Amos version 23.0.
Results
Participant Characteristics
A total of 580 members accepted invitations to participate in the survey. There were 513 patients meeting the target criteria. All of them completed the questionnaire (response rate of 100%). Table 1 shows the participants’ demographic and clinical characteristics. The patients had a mean age of 33.12 ± 7.96 years, and 93.76% were women and the mean age when diagnosed with SLE was 25.06 ± 8.06 years. The most common symptom in patients was a rash. In total, 195 (38.01%) participants with middle school education or below, 104 (20.27%) participants with high school education, and 214 (41.72%) participants with bachelor’s degree or above. The minority (32.75%) cannot afford their families. The average score of anxiety was 6.81 (SD 4.27, range 0–21); 124 (24.17%) participants might be at risk of having at least mild anxiety and 92 (17.93%) participants might experience severe or extremely severe anxiety. The average score of depression was 7.37 (SD 4.04, range 0–19); 139 (27.10%) of participants might be at risk of having at least mild depression and 106 (20.66%) might experience severe or extremely severe depression. The average score of sleep quality was 8.41 (SD 3.61, range 0–19), with 339 people scoring 7 or more and 174 scoring less than 7. Besides, the average score of quality of life was 168.46 (SD 72.6, range 71–454).
 |
Table 1 Demographic Characteristics of SLE Patients (n=513) |
Correlation and Multiple Linear Regression Analysis
Table 2 shows the correlation values between each variable. In our study, we found that patients with higher education (r=−0.188, P <0.01), higher income (r=−0.232, P <0.01), fewer SLE symptoms (r=0.367, P<0.01), and better sleep quality (r=0.460, P<0.01) had lower levels of anxiety. In addition, anxious and depressed SLE patients are more likely to have a lower quality of life compared with those who are not anxious or depressed (r=0.657, P<0.01; r=0.614, P<0.01). And there was a significant positive correlation (r=0.538, P<0.01) between sleep score and HRQoL score. Six variables (education, income/family, number of SLE symptoms, sleep quality, anxiety, depression) have significant correlation with HRQoL were then included in a multiple linear regression model (Table 3).
 |
Table 2 Shows the Correlation Values Between Each Variable |
 |
Table 3 Variables Associated with HRQoL in Multiple Linear Regression |
Testing and Modification of the Hypothesized Path Model on the HRQoL of SLE Patients
A path analysis model was developed to examine predictors of HRQoL for SLE patients. Some non-significant paths were progressively eliminated from the model, resulting in a reduced model with only significant paths. The final model demonstrated a good fit: χ2=5.865 (P=0.118>0.05), GFI = 0.996, AGFI = 0.974, CFI = 0.998, RMSEA = 0.043. This model explained 57.3% of the variance on HRQoL in patients with SLE and all the hypothesized paths reached significance (P<0.05). The standardized path direct coefficients for the hypothetical model are presented in Figure 1. And the direct, indirect, and total effects are displayed in Table 4.
 |
Table 4 Direct and Indirect Effect of Variables on HRQoL in Study Model |
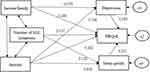 |
Figure 1 Hypothetical Causal Path Diagram for HRQoL of SLE Patients. |
We found number of SLE symptoms (β=0.137, P<0.001), income/family (β=−0.188, P<0.001), anxiety (β= 0.342, P<0.001), depression (β=0.143, P=0.004), and sleep quality (β= 0.251, P<0.001) were significant, direct predictors of HRQoL. The total direct effects on HRQoL included number of SLE symptoms (β=0.167), income (β=−0.199), anxiety (β=0.561), depression (β=0.143), sleep quality (β=0.251). Thus, anxiety has the greatest direct and total impact on HRQoL.
Discussion
Our study constructed a structural equation modeling (SEM) and used path analysis to investigate the relationships between anxiety, depression, sleep quality and HRQoL in patients with SLE. The results revealed that the fit values of the hypothetical model tested in our study were at an acceptable level. Path analysis is more superior and flexible than multiple regression analysis. In path analysis, there can be more than one dependent variable, and the variable can be either dependent or independent.12,24 As a result, path analysis can establish multiple regression models to simulate the relationship between multiple variables, so as to obtain the direct and indirect effects and causality between variables. To the best of our knowledge, our study provided the pathway analysis to assess such interaction between variables and the HRQoL among Chinese SLE patients. The model coefficients and pathways showed that a complex interaction existed between variables and HRQoL. Same as previous study, the risk and burden of SLE are high among women during their childbearing years.25 In line with what we have found, low income would reduce the quality of life of patients, as reported in the previous study.26 McCormick et al found high financial stress was an important predictor of new depression in SLE patients after controlling for disease factors and other measures.27 In the results of path analysis, income and total number of symptoms not only directly affected HRQoL but also indirectly affected HRQoL through anxiety, depression and sleep quality. While the existence of anxiety made the greatest contribution to the reduction of HRQoL, followed by depression and sleep disorder. In our research, anxiety has been revealed to have the greatest influence on declined HRQoL.28 Our results confirmed that the more symptoms of SLE, the higher the level of anxiety and depression. And the number of SLE symptoms significantly contributed to the quality of life which was consistent with previous study.29 In agreement with previous study (Ratanasiripong et al), skin manifestations (skin rash alopecia and mucosal ulcers) were the most common symptom among participants, and randomized controlled trials have shown that using cosmetics can mask rashes and improve depression, anxiety and quality of life scores.30,31 Meanwhile, patients with skin involvement also had higher likelihood of having coexisting anxiety and depression.31 Our survey showed patients with more symptoms had lower quality of life, the results were consistent with previous research, which revealed a notable relationship between quality of life and disease manifestations in SLE patients.32 The weak contribution of symptoms quantity to HRQoL in our study may be related to the short investigation period of this study and different scales for measurement of HRQoL. Previous studies have shown that the most common cause of depression and anxiety is changes in the appearance of SLE symptoms.33 In this study, it was found anxiety and depression were independent predictors of lower HRQoL in SLE patients, but little literatures have reported relation among anxiety, depression, sleep quality and HRQoL.
Furthermore, our study found, in agreement with previous studies,34,35 patients with poor sleep quality showed impaired HRQoL. Margiotta et al also explored the impact of quality of life in patients with SLE and found that patients’ quality of life could be improved by alleviating depression and fatigue.35 Fatigue in SLE patients has a strong negative effect on HRQoL in a single-center cohort study.36 Results from other studies confirmed that anxiety, depression, and sleep quality were predictors of fatigue.37–39 Azizoddin et al and Monahan et al suggested that fatigue was associated with anxiety and depression, rather than disease activity.36,40 Other studies have indicated trivial correlation between SLEQOL summary score and disease activity.23,41 However, it should be noted that anxiety, sleep quality and depression are rarely identified in early stages of disease progression. Consequently, clinicians should focus not only on clinical symptoms of SLE but also on psychological symptoms and sleep quality.
Study Limitations and Future Research
Our study contains several key limitations discussed in the following. Firstly, the data are self-reported and may be subject to reporting biases, and other than the number of symptoms, this study did not specifically assess the disease activities, comorbidities, and medication consumption, which might affect patients’ responses. These factors should be considered for future studies. Secondly, this model presented a high goodness fit and explained 57.0% of the variance of HRQoL; therefore, it is possible that the remaining 43.0% was attributed to some other factor. Lastly, as a cross-sectional study, therefore, causality is merely hypothetical. Therefore, longitudinal randomized controlled trials are needed to explore causal relationship in future research and implement intervention researches to explore the influencing factors of HRQoL in SLE.
Conclusions
This study found significant direct paths from anxiety, sleep quality, income/family, number of SLE symptoms, and depression to HRQoL in a group of SLE. Meanwhile, economic conditions and symptoms factors also need to be considered when improving HRQoL. In consequence, path analysis is an effective strategy to explore relation among anxiety, depression, sleep quality and health-related quality of life among patients with SLE.
Data Sharing Statement
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Ethical Approval and Consent to Participate
The study was conducted in accordance with the guidelines of declaration of Helsinki. This study was approved from the Biomedical Ethics Committee of Anhui Medical University (approval number: 20150111).
Acknowledgments
The authors are thankful to all the participants. Hui-Juan Chen and Hong Wang are co-first authors for this study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The foundation is supported by the National Natural Science Foundation of China (No. 81573217 and.82073652).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Schwartzman-Morris J, Putterman C. Gender differences in the pathogenesis and outcome of lupus and of lupus nephritis. Clin Dev Immunol. 2012;2012:604892. doi:10.1155/2012/604892
2. Signorini V, Elefante E, Zucchi D, Trentin F, Bortoluzzi A, Tani C. One year in review 2020: systemic lupus erythematosus. Clin Exp Rheumatol. 2020;38(4):592–601.
3. Barber MRW, Clarke AE. Socioeconomic consequences of systemic lupus erythematosus. Curr Opin Rheumatol. 2017;29(5):480–485. doi:10.1097/BOR.0000000000000416
4. Olesinska M, Saletra A. Quality of life in systemic lupus erythematosus and its measurement. Reumatologia. 2018;56(1):45–54.
5. Fung W, Lim LSH, Tomlinson G, et al. Joint trajectories of disease activity, and physical and mental health-related quality of life in an inception lupus cohort. Rheumatology. 2020;59(10):3032–3041.
6. McElhone K, Abbott J, Teh LS. A review of health related quality of life in systemic lupus erythematosus. Lupus. 2006;15(10):633–643.
7. Kasitanon N, Wangkaew S, Puntana S, et al. The reliability, validity and responsiveness of the Thai version of Systemic Lupus Erythematosus Quality of Life (SLEQOL-TH) instrument. Lupus. 2013;22(3):289–296.
8. Moustafa AT, Moazzami M, Engel L, et al. Prevalence and metric of depression and anxiety in systemic lupus erythematosus: a systematic review and meta-analysis. Semin Arthritis Rheum. 2020;50(1):84–94.
9. Bai R, Liu S, Zhao Y, et al. Depressive and anxiety disorders in systemic lupus erythematosus patients without major neuropsychiatric manifestations. J Immunol Res. 2016;2016:2829018.
10. Choi ST, Kang JI, Park IH, et al. Subscale analysis of quality of life in patients with systemic lupus erythematosus: association with depression, fatigue, disease activity and damage. Clin Exp Rheumatol. 2012;30(5):665–672.
11. Palagini L, Tani C, Mauri M, et al. Sleep disorders and systemic lupus erythematosus. Lupus. 2014;23(2):115–123. doi:10.1177/0961203313518623
12. Wolf EJ, Harrington KM, Clark SL, Miller MW. Sample size requirements for structural equation models: an evaluation of power, bias, and solution propriety. Educ Psychol Meas. 2013;76(6):913–934. doi:10.1177/0013164413495237
13. Liang MH, Meenan RF, Cathcart ES, Schur PH. A screening strategy for population studies in systemic lupus erythematosus series design. Arthritis Rheum. 1980;23(2):153–157. doi:10.1002/art.1780230204
14. Brennan C, Worrall-Davies A, McMillan D, Gilbody S, House A. The hospital anxiety and depression scale: a diagnostic meta-analysis of case-finding ability. J Psychosom Res. 2010;69(4):371–378. doi:10.1016/j.jpsychores.2010.04.006
15. Bjelland I, Dahl AA, Haug TT, Neckelmann D. The validity of the hospital anxiety and depression scale an updated literature review. J Psychosom Res. 2002;52(2):69–77. doi:10.1016/S0022-3999(01)00296-3
16. Li Q, Lin Y, Hu C, et al. The Chinese version of hospital anxiety and depression scale: psychometric properties in Chinese cancer patients and their family caregivers. Eur J Oncol Nurs. 2016;25:16–23. doi:10.1016/j.ejon.2016.09.004
17. Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. doi:10.1016/0165-1781(89)90047-4
18. Mollayeva T, Thurairajah P, Burton K, Mollayeva S, Shapiro CM, Colantonio A. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: a systematic review and meta-analysis. Sleep Med Rev. 2016;25:52–73. doi:10.1016/j.smrv.2015.01.009
19. Zheng B, Li M, Wang KL, Lv J. [Analysis of the reliability and validity of the Chinese version of Pittsburgh sleep quality index among medical college students]. Beijing Da Xue Xue Bao Yi Xue Ban. 2016;48(3):424–428. Chinese.
20. Zhu B, Xie M, Park CG, Kapella MC. Adaptation of the Pittsburgh sleep quality index in Chinese adults with type 2 diabetes. J Chin Med Assoc. 2018;81(3):242–247. doi:10.1016/j.jcma.2017.06.021
21. Dong D, Lou P, Wang J, et al. Interaction of sleep quality and anxiety on quality of life in individuals with type 2 diabetes mellitus. Health Qual Life Outcomes. 2020;18(1):150. doi:10.1186/s12955-020-01406-z
22. Radin M, El Hasbani G, Barinotti A, et al. Quality of life measures in systemic lupus erythematosus: a systematic review. Reumatismo. 2022;73(4). doi:10.4081/reumatismo.2021.1447
23. Jiang HZ, Lin ZG, Li HJ, et al. The Chinese version of the SLEQOL is a reliable assessment of health-related quality of life in Han Chinese patients with systemic lupus erythematosus. Clin Rheumatol. 2018;37(1):151–160. doi:10.1007/s10067-017-3910-4
24. Peng H, Ma X, Chen F. Examining injury severity of pedestrians in vehicle-pedestrian crashes at mid-blocks using path analysis. Int J Environ Res Public Health. 2020;17(17):6170. doi:10.3390/ijerph17176170
25. Chung MK, Park JS, Lim H, Lee CH, Lee J. Incidence and prevalence of systemic lupus erythematosus among Korean women in childbearing years: a nationwide population-based study. Lupus. 2021;30(4):674–679. doi:10.1177/0961203320984845
26. Teixeira RCA, Borba Neto EF, Christopoulos GB, Sato EI. The influence of income and formal education on damage in Brazilian patients with systemic lupus erythematosus. J Clin Rheumatol. 2017;23(5):246–251. doi:10.1097/RHU.0000000000000541
27. McCormick N, Trupin L, Yelin EH, Katz PP. Socioeconomic predictors of incident depression in systemic lupus erythematosus. Arthritis Care Res. 2018;70(1):104–113. doi:10.1002/acr.23247
28. Macedo EA, Appenzeller S, Costallat LT. Gender differences in systemic lupus erythematosus concerning anxiety, depression and quality of life. Lupus. 2016;25(12):1315–1327. doi:10.1177/0961203316638934
29. Miyawaki Y, Shimizu S, Ogawa Y, et al. Association of glucocorticoid doses and emotional health in lupus low disease activity state (LLDAS): a cross-sectional study. Arthritis Res Ther. 2021;23(1):79. doi:10.1186/s13075-021-02466-2
30. Ratanasiripong NT, Ratanasiripong P. Predictive factors of quality of life among systemic lupus erythematosus patients in Thailand: a web-based cross-sectional study. Qual Life Res. 2020;29(9):2415–2423. doi:10.1007/s11136-020-02494-6
31. Oliveira FAP, Santos F, Dias A, Neiva CLS, Telles RW, Lanna CCD. Cosmetic camouflage improves health-related quality of life in women with systemic lupus erythematosus and permanent skin damage: a controlled intervention study. Lupus. 2020;29(11):1438–1448. doi:10.1177/0961203320947802
32. Beckerman NL, Auerbach C, Blanco I. Psychosocial dimensions of SLE: implications for the health care team. J Multidiscip Healthc. 2011;4:63–72. doi:10.2147/JMDH.S19303
33. Mirbagher L, Gholamrezaei A, Hosseini N, Sayed Bonakdar Z. Sleep quality in women with systemic lupus erythematosus: contributing factors and effects on health-related quality of life. Int J Rheum Dis. 2016;19(3):305–311. doi:10.1111/1756-185X.12418
34. Moraleda V, Prados G, Martinez MP, Sanchez AI, Sabio JM, Miro E. Sleep quality, clinical and psychological manifestations in women with systemic lupus erythematosus. Int J Rheum Dis. 2017;20(10):1541–1550.
35. Margiotta DPE, Fasano S, Basta F, et al. The association between duration of remission, fatigue, depression and health-related quality of life in Italian patients with systemic lupus erythematosus. Lupus. 2019;28(14):1705–1711.
36. Azizoddin DR, Gandhi N, Weinberg S, Sengupta M, Nicassio PM, Jolly M. Fatigue in systemic lupus: the role of disease activity and its correlates. Lupus. 2019;28(2):163–173.
37. Du X, Zhao Q, Zhuang Y, Chen H, Shen B. Fatigue of systemic lupus erythematosus in China: contributors and effects on the quality of life. Patient Prefer Adherence. 2018;12:1729–1735.
38. Pinto B, Dhooria A, Grover S, Jolly M, Raj JM, Sharma A. Fatigue and its correlates in Indian patients with systemic lupus erythematosus. Clin Rheumatol. 2021;40(3):905–911.
39. Xie X, Wu D, Chen H. Prevalence and risk factors of anxiety and depression in patients with systemic lupus erythematosus in Southwest China. Rheumatol Int. 2016;36(12):1705–1710.
40. Monahan RC, Beaart-van de Voorde LJ, Eikenboom J, et al. Fatigue in patients with systemic lupus erythematosus and neuropsychiatric symptoms is associated with anxiety and depression rather than inflammatory disease activity. Lupus. 2021;30(7):1124–1132.
41. Abu-Shakra M. Quality of life, coping and depression in systemic lupus erythematosus. Isr Med Assoc J. 2016;18(3–4):144–145.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
A Cross-Sectional Study on the Relationship Between Rosacea Severity and Quality of Life or Psychological State
Yang F, Zhang Q, Song D, Liu X, Wang L, Jiang X
Clinical, Cosmetic and Investigational Dermatology 2022, 15:2807-2816
Published Date: 20 December 2022
Correlation Analysis Between Disease Activity and Anxiety, Depression, Sleep Disturbance, and Quality of Life in Patients with Inflammatory Bowel Disease
Yu R, Liu C, Zhang J, Li J, Tian S, Ding F, Liu Z, Wang T, Liu Z, Jiang C, Shi J, Wu K, Dong W
Nature and Science of Sleep 2023, 15:407-421
Published Date: 26 May 2023
Mind, Body and Machine: Preliminary Study to Explore Predictors of Treatment Response After a Sleep Robot Intervention for Adults with Insomnia
Støre SJ, Tillfors M, Wästlund E, Angelhoff C, Andersson G, Norell A
Nature and Science of Sleep 2023, 15:567-577
Published Date: 13 July 2023
Analyzing Role Overload, Mental Health, and Quality of Life Among Jordanian Female Healthcare Professionals: A Cross-Sectional Study
Alnazly EK, Allari R, Alshareef BE, Abu Al-khair F
International Journal of Women's Health 2023, 15:1917-1930
Published Date: 4 December 2023
Modulatory Effects of XIAOPI Formula on CXCL1 and Selected Outcomes in Triple-Negative Breast Cancer: A Randomized Controlled Clinical Trial
Guo L, Hong SC, Wang X, Wang SQ, Wang N, Wei XQ, Situ HL, Wang ZY
Breast Cancer: Targets and Therapy 2024, 16:289-303
Published Date: 31 May 2024
