Back to Journals » Journal of Inflammation Research » Volume 14
Undetected Jawbone Marrow Defects as Inflammatory and Degenerative Signaling Pathways: Chemokine RANTES/CCL5 as a Possible Link Between the Jawbone and Systemic Interactions?
Authors Lechner J , Schmidt M , von Baehr V, Schick F
Received 24 February 2021
Accepted for publication 7 April 2021
Published 21 April 2021 Volume 2021:14 Pages 1603—1612
DOI https://doi.org/10.2147/JIR.S307635
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Johann Lechner,1 Marlene Schmidt,2 Volker von Baehr,3 Fabian Schick4
1Head of the Clinic for Integrative Dentistry, Munich, Germany; 2Data Scientist at STEYR Motorenwerke, Steyr, Austria; 3Head of the Department of Immunology and Allergology, Institute for Medical Diagnostics, Berlin, Germany; 4Dental Surgeon, Clinic for Integrative Dentistry, Munich, Germany
Correspondence: Johann Lechner Tel +49-89-6970129
Fax +49-89-6925830
Email [email protected]
Background: Cytokines, especially chemokines, are of increasing interest in immunology. This study characterizes the little-known phenomenon of “bone marrow defects of the jawbone” (BMDJ) with known overexpression of the chemokine RANTES/CCL5 (R/C).
Purpose: Our investigation clarifies why BMDJ and the intensity of local R/C overexpression are challenging to detect, as examined in patients with seven different systemic immunological diseases. Specifically, we investigate whether R/C overexpression is specific to certain disease groups or if it represents a type of signal disruption found in all systemic immunological diseases.
Patients and Methods: In a total of 301 patients, BMDJ was surgically repaired during clinical practice to reduce “silent inflammation” associated with the presence of jaw-related pathologies. In each case of BMDJ, bone density was measured preoperatively (in Hounsfield units [HU]), while R/C expression was measured postoperatively. Each of the 301 patients suffered from allergies, atypical facial and trigeminal pain, or were diagnosed with neurodegenerative diseases, tumors, rheumatism, chronic fatigue syndrome, or parasympathetic disorders.
Results: In all BMDJ cases, strongly negative HU values indicated decreased bone density or osteolysis. Consistently, all cases of BMDJ showed elevated R/C expression. These findings were consistently observed in every disease group.
Discussion: BMDJ was confirmed in all patients, as verified by the HU measurements and laboratory results related to R/C expression. The hypothesis that a specific subset of the seven disease groups could be distinguished either based on the increased presence of BMDJ and by the overexpression of R/C could not be confirmed. A brief literature review confirms the importance of R/C in the etiology of each of the seven disease groups.
Conclusion: In this research, the crucial role played by BMDJ and the chemokine R/C in inflammatory and immune diseases is discussed for seven groups of patients. Each specific immune disease can be influenced or propelled by BMDJ-derived R/C inflammatory signaling pathways.
Keywords: bone marrow defects, jawbone, RANTES/CCL5 inflammatory signaling pathways, silent inflammation, systemic immune diseases
Background
There is no shortage of reports describing the association between circulating cytokines and disease severity. When cytokine production is dysregulated, cytokines wreak havoc by causing the immune system to turn against itself, resulting in autoimmune disease development. If the cytokine genes are not switched off, their products result in chronically activated cells within the host, which now dominate an otherwise dormant immune system.1 Cytokines are soluble factors produced by one cell. They act on another cell via the endocrine system to alter the function of the target cell ultimately. In a sense, cytokines can be thought of as “hormones” in immune and inflammatory responses. More importantly, at a molecular level, cytokines interfere with immunological signaling to a greater extent than hormones.2 The most impressive cytokines are those from chemokine families. Chemokines orchestrate cell migration from blood into the tissues.2
Despite the highly statistically significant correlations that are evident between cytokines and disease onset, one crucial lesson in cytokine biology is that a specific cytokine only plays a causal role in a given disease through the specific activation of an associated receptor. The dual functions of cytokines have emerged in recent years; one of the two identified functions is that cytokines serve as an extracellular molecule, usually referred to as a ligand, that attacks a specific receptor on the cell surface. The receptor is usually an integral membrane protein and is activated by the ligand. A juxtacrine mechanism usually makes cell-to-cell contact through the ligand.3 Many of these cytokine effects are likely to take place within the cell’s microenvironment.4 In some cases, the cytokine receptor is primarily located on one cell type responsible for a specific function.5 In other cases, the receptor can be found in nearly every cell. As a note, immunological research has made significant advances in terms of identifying these associated receptors.
In the search for potential cytokine mechanisms and chronic inflammatory connections between so-called “bone marrow defects” in the jawbone and other organs, we were able to determine the local overexpression of the chemokine RANTES/CCL5 (R/C) in old wound areas of the jawbone.6,7 An alternate consideration for R/C expression is indicated, as each cytokine binds to a specific receptor and triggers an intracellular cascade. What, then, explains the many and varied properties of a single cytokine? The key to answering this question lies within the organs or cell types on which the R/C receptors (CCR5) are located, interacting with the R/C ligand.8 CCR5 is ubiquitously expressed on numerous immune cells: predominantly on T cells, macrophages, dendritic cells, eosinophils, microglia, and a subpopulation of either breast or prostate cancer cells.9 The expression of CCR5 is selectively induced during the cancer transformation process and is not expressed in normal breast or prostate epithelial cells. Approximately 50% of human breast cancers express CCR5, primarily in cases of triple-negative breast cancer.10 In contrast, systemic levels of cytokines such as interleukin (IL)-6 or R/C levels serve as primary markers of disease severity. Our research on BMDJ suggests that persistently activated autoreactive T cells in these areas of the body, as determined by R/C overexpression, cannot switch off by themselves. These areas require surgical intervention to restore an interference-free state of R/C signaling.
R/C appears to be a suitable inflammatory marker that can be used to explore the possible links between BMDJ and systemic immunological diseases. In this research, the role of chemokine R/C in inflammatory and immune diseases is discussed.
Research Questions
The following research questions are examined in this study: To what extent the R/C signal transductions that arise from BMDJ related to specific diseases are? Are the R/C chemokine signals chronically and locally expressed from the BMDJ to such an extent that their inflammatory or degenerative effects are nonspecific and can be associated with various systemic diseases?
Patients and Methods
Patient Population
A total of 301 patients, comprising seven disease groups, were selected for this study. Each patient expressed an interest in determining whether low levels of inflammation in the jaw were present and associated with the diagnosis of a chronic immunological disorder or a painful systemic disease. The presence of BMDJ was verified preoperatively for each patient by measuring digital volume tomography (DVT)-Hounsfield units (HU). Postoperatively, the preoperative diagnosis, along with the local R/C expression profile, was further identified to determine whether it was specific to BMDJ. We thus compared the preoperative DVD-HU values in 301 patients with the postoperative laboratory results related to R/C expression in the corresponding jaw areas.
The present study of 301 patients was carried out as a retrospective case-control study and was classified as such by the forensically accredited institute, IMD-Berlin, according to DIN EN 15198/DIN EN 17025. The study was conducted in accordance with the Declaration of Helsinki. All patients provided their written informed consent (as outlined in the PLOS consent form) to participate in this study. The study presented here is patient-centered; the samples and data were obtained directly during routine clinical practice and normal medical care of the patients and evaluated retrospectively. Institutional approval was not required to publish the case details. Medication used to alleviate any sensitivities was not stopped. The use of medications to treat systemic diseases was not considered an exclusion criterion. The exclusion criteria included the use of cortisone and bisphosphonates due to their effects on bone metabolism.
The study cohort of 301 patients with suspected BMDJ comprised the following seven disease groups, as diagnosed by a specialist: allergies (including food allergies, chronic rhinitis, and weakened immune systems; n=29); atypical facial and trigeminal pain (n=47); neurodegenerative diseases (including migraine, tinnitus, multiple sclerosis, and amyotrophic lateral sclerosis; n=57); tumors (breast, prostate, pancreatic, and colon cancers; n=16); rheumatism (fibromyalgia and Lyme diseases; n=51); chronic fatigue syndrome (n=56); and parasympathetic disorders (ie, disorders related to blood pressure, dizziness, and anxiety; n=49). The patients’ mean age was 54.05 years (range: 23–75 years). The ratio of women to men was 89:225. All data were collected during normal oral surgery. All patients sought to determine the cause of their respective immune system disorders, which may have taken on the form of a “silent inflammation,” possibly induced by BMDJ. Following the clinically necessary excision of BMDJ, each abnormal tissue sample was analyzed to determine its chemokine R/C content.
Preoperative Measurement of Bone Density with OPG
A two-dimensional (2D) orthopantomogram (OPG) was carried out for each patient. Given the known limitations associated with OPG measurements concerning BMDJ, these results were not included in the analysis.11,12
Preoperative Measurement of Bone Density with DVT-HU
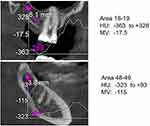
In each of the 301 patients, bone density was determined using DVT. Modern X-ray methods, which include DVTs and cone-beam computed tomograms (CBCT), enable the clinician to perform a three-dimensional (3D) assessment of the jawbone once the X-ray attenuation coefficients have been determined and as expressed in HU. In X-ray technology, the Hounsfield scale (HU) is generally recognized as a scientific assessment tool for bone density.13 HU are used in computed tomography (CT) to describe and display X–ray attenuation in grayscale images. These values are assigned to physical structures and tissues. The HU scale starts at −1000 for the attenuation coefficient of air, while it is −120 for fat, ranges from +300 to +400 for healthy cancellous bone, and from +1800 to +1900 for cortical bone. Water has a value of 0 HU.
Thus far, the measurement of bone quality and the assessment of the degree of mineralization by HU were only made possible using 3D representations of the skull, as obtained through CT. However, CT is associated with relatively high levels of radiation exposure. Methods that can determine HU attenuation coefficients have recently become available,13 where actual HU values can be derived from DVTs.14 In a prior study, SimpPlant software (3D Diagnostix, Boston, MA, USA) measurements of BMDJ, as taken from the posterior mandible, indicated that the mean CT score was 669.6 HU.14 Additional investigations classified cancellous jawbone density into five categories, with the worst jawbone density falling below 150 HU (class 5). The HU values determined in our study (range: +150 to −680) thus indicate the presence of osteolysis of the jawbone in class 5 cases.14–16
We have a DVT from Orangedental (PaX-i3D Duo), which is a 3D multi-X–ray device. It has the software required to evaluate the available HU to measure bone density. According to DIN 6868-57, viewing monitors with a contrast setting of >40:1, a brightness of at least 120 cd/m2, and a DICOM characteristic are needed. These were combined with the Orangedental PaX-i3D Duo 3D multi-X-ray device used in this validation study. The device showed the mean value of the freely selected section measured; the maximum and minimum values were presented as a curve (Figure 1).
Postoperative Measurement of R/C Expression in BMJ Areas
The challenge with areas of osteolytic and osteodestructive BMDJs is the postoperative detection of local “silent inflammation.” In previous publications, we demonstrated overexpression of the chemokine R/C that was specific to BMDJ.17,18
Postoperative Multiplex Analysis of the Cytokine Expression from BMDJ Areas
The necrotic tissue samples with a volume up to 0.5 cm3 were stored in a dry, sterile, 2 mL collecting vial (Sarstedt, Nümbrecht, Germany), closed airtight, and frozen at–20°C. In the laboratory, the samples were homogenized using mechanic force in 200 µL of cold protease inhibitor buffer (Complete Mini Protease Inhibitor Cocktail; Roche Diagnostics GmbH, Penzberg, Germany). The homogenate was centrifuged for 15 minutes at 13,400 rpm. Afterward, the supernatant was collected and centrifuged for another 25 minutes at 13,400 rpm. In each supernatant of the tissue homogenate, we measured R/C, fibroblast growth factor (FGF)-2, IL-1 receptor antagonist (IL-1ra), IL-6, IL-8, monocyte chemotactic protein-1 (MCP1), and tumor necrosis factor-alpha (TNF-a). The measurement was performed using the Human Cytokine/Chemokine Panel I (MPXHCYTO-60K; Millipore GmbH, Schwalbach, Germany), according to the manufacturer’s instructions, and analyzed using the Luminex® 200™ with xPonent® software (Luminex, Austin, TX, USA). To measure R/C in the serum, samples were pre-diluted to 1:100 in sample buffer according to the manufacturer’s instructions.
Patients without BMD and R/C Expression in the Jaw
To show that R/C overexpression was not present in the healthy jawbone, we examined bone cores that were obtained during implant placement. This healthy control group consisted of 19 patients with no BMD findings. The age range of this healthy control group was 38–71 years (mean: 54 years), with a ratio of females to males of 11/8. The mean R/C value was 149.9 pg/mL (SD ±127 pg/mL). Comparable values for healthy controls with normal jawbone tissues were not available in the literature.6
Why is R/C Expression Used as a Parameter for BMDJ?
Our earlier investigations showed that R/C expression is highly appropriate as an index of chronic inflammation when investigating the jawbone.6,7,12,17 As an example, we compared a group of 21 patients with chronic fatigue syndrome (CFS) to explore their inflammatory profile,20 as well as a group of 48 tumor cases to identify the extent of BMDJ.19 The tumor cases were divided as follows: 62% breast cancer, 15% ovarian cancer, 13% prostate cancer, and 10% melanoma. (Figure 2) The green line in shows a standard R/C value of 149.9 pg/mL (SD ±127 pg/mL).
Morphology of Fatty Degenerative Osteonecrotic BMDJ Samples
In the last few years, osteopathias of the jawbone are subject to different definitions and classifications. Concerning the conspicuous morphology of BMDJ we proposed the term “fatty–degenerative osteonecrosis/osteolysis of jawbone” (FDOJ) for this cavitational osteonecrosis,6,7,17–20 reflecting the clinical state of focal bone marrow defects (Figure 3). A total of 301 samples of fatty-degenerative osteonecrosis of the jawbone (FDOJ), as determined by the morphological analysis of the intraoperatively exposed bone marrow defects (BMDJ) (Figure 3), were examined. All 301 BMDJ/FDOJ samples were identified preoperatively with DVT-HU and postoperatively with R/C determination. Due to the softening of the spongy bone, the marrow space can be easily vacuumed and curetted. There is a complete absence of trabecular cancellous bone structures. Figure 3 shows an intraoperative tissue sample with the predominantly fatty transformation of the cancellous bone. The consistency of BMDJ/FDOJ indicates the presence of metabolic disorders and local hypoxic ischemia. Fatty-degenerative conditions prevent even slight autonomic tendencies from healing.
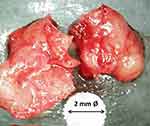 |
Figure 3 Typical samples of BMDJ/FDOJ with complete dissolution of the bone trabecula. Below the sample, the head of a ceramic drill with a 2 mm diameter is shown to provide a size comparison. |
Results
Statistical Analysis from a Group of 301 Patients
Statistical analysis was carried out with the statistical software, R (version 3.5.1). To compare the measurement distribution per disease group, the data distributions were descriptively illustrated using scatterplots.
Comparison of Local DVT-HU Values and R/C Overexpression in 301 Cases
The comparison of the DVT-HU and R/C expression profile values for all 301 patients with one of the seven selected systemic diseases and preoperative bone density measurement is shown in Tables 1 and 2. The medium value (MV) of the DVT-HU measurements of –362.9 (>300) suggests that chronic BMDJ/FDOJ is evident in the examined jawbone areas in each patient. The corresponding R/C values obtained for the postoperatively examined BMDJ/FDOJ samples showed an MV of 2968 pg/mL, suggesting the presence of extensive, chronic, inflammatory signaling from the operated jaw areas (standard value: 149.9 pg/mL).
 |
Table 1 Statistical Analysis Results of the DVT-HU Values in Each Group |
 |
Table 2 Statistical Analysis Results of the R/C Expression Levels in Each Group |
The statistical analysis results of the DVT-HU values are shown in Table 1. The statistical analysis results of the R/C expression levels are shown in Table 2.
Breakdown of Local DVT-HU Values and R/C Overexpression According to Disease Group
Table 3 compares the DVT–HU values and R/C expression levels in 301 patients with various systemic diseases who had undergone surgery to address the BMDJ/FDOJ areas.
 |
Table 3 This Table Shows the Mean Values of DVT-HU and R/C Expression for Focal BMDJ/FDOJ for Each Chronic Disease Group |
The corresponding boxplots, represented in Figures 4 and 5, show the distribution of the HU and R/C values for the study cohort of 301 patients per chronic disease group. Accordingly, the decreases in HU values are evident in the areas of BMDJ/FDOJ, with values falling well below the minimum value of 300 for healthy cancellous bone (as indicated by the green line).14–16 Figure 5 shows that the same patients also had significantly overexpressed R/C signaling compared to the common value of 149.9 pg/mL for healthy cancellous bone (also shown with a green line). Figure 4 illustrates how, regardless of disease group, negative DVT-HU values (median:–440 HU; black horizontal bars in Figure 4) indicate lower bone density in all patients. Figure 5 shows that osteolytic degeneration with pronounced R/C overexpression occurs in the corresponding marrow region. Figures 4 and 5 both show a normal distribution for the 301 HU and R/C values that were obtained; further, increasing Gamma or Poisson distribution for the R/C values is also evident.
 |
Figure 5 R/C signaling is overexpressed for all chronic disease groups compared to the common value of 149,9 pg/mL for healthy cancellous jawbone (shown by the green line). |
Discussion
Is there a relationship or correlation between the presence of chronic diseases and negative DVT-HU values, providing a reliable indicator of existing BMDJ, as determined by R/C overexpression? What is the interaction between R/C signaling pathways and systemic diseases? As all patients in each disease group demonstrated R/C overexpression in the BMDJ/FDOJ area, we can assume there may be a causal connection between these diseases and R/C expression. This R/C expression is unspecific, as it shows a possible connection with all seven disease symptoms. Our data could not determine the presence of a specific disease. The system networks of the chemokine R/C are presented in detail in the literature. Given the multifactorial relationship that R/C expression shares with other diseases, our findings confirm this lack of specificity.
Our team carried out a short review of the existing scientific publications indexed in PubMed to verify our working hypothesis. We utilized the following search terms: “RANTES CCL5 AND Allergy”, which yielded 582 hits in PubMed. The following quote from the literature confirmed our working hypothesis:
… We compared patients with chronic spontaneous urticaria (CSU), who had a significantly higher concentration of CCL5/RANTES when compared to the NHS (p<0.0001), but not of CXCL8/IL-8. (the quote was translated)21
Using the search terms “RANTES CCL5 AND Trigeminal Pain”, it was noted how there were 14 papers related to neuralgic pain. Another quote reinforced our working hypothesis:
Cytokines are now considered to be the pain mediators in neurovascular inflammation. Furthermore, cytokines may be a cause of the migraine pain: in fact, high levels of chemokines could stimulate the activation of trigeminal nerves ….22
Entering “RANTES CCL5 AND Neurodegenerative Diseases” resulted in 54 hits in PubMed. The following quote also supported our working hypothesis:
The findings of the meta-analysis demonstrated higher peripheral concentrations of IL-6, tumor necrosis factor, IL-1β, IL-2, IL-10, C-reactive protein, and RANTES in patients with PD, strengthening the clinical evidence that an inflammatory response accompanies PD ….23
Entering “RANTES CCL5 AND Tumors” yielded 1758 hits from the PubMed search, with the following quotation identified, which again supports our working hypothesis:
The CCR5 and the CCL5 ligand have been detected in some hematological malignancies, lymphomas, and a great number of solid tumors, but extensive studies on the role of the CCL5/CCR axis were performed only in a limited number of cancers.24
Using the terms “RANTES CCL5 AND Rheumatoid Arthritis” resulted in 10 hits in PubMed, by confirming our working hypothesis with the following quotation:
The present study indicated that the … understanding of molecular mechanisms underlying the development of RA, such as C-C motif chemokine 5 (CCL5), might have a negative impact in the development of RA ….25
Entering “RANTES and CFS” (chronic fatigue syndrome)/neuroinflammation resulted in 94 hits. The following quote confirmed our working hypothesis: “The protein levels of the chemokine CCL5/RANTES were remarkably increased in the astrocytes of rat injured spinal cord … ”.26 Finally, entering “RANTES CCL5 AND Blood Pressure/Anxiety” resulted in 51 hits in PubMed, respectively. The following quote was obtained, which further reinforced our working hypothesis:
As powerful drivers of diapedesis, the chemokines CCL2 and CCL5 have long been implicated in hypertension, but experimental data highlight divergent, context-specific effects of these chemokines on blood pressure and tissue injury.27
Summary
Our data showed several connections that have, until now, gone unidentified. The evaluated BMDJ/FDOJ pathology represents a chronic, osteoimmunological disturbance, which can be seen as an added burden in many immune and inflammatory diseases.
The evaluation of the 301 patients in our study that were grouped into one of seven different disease categories supported the conclusion that BMDJ/FDOJ is not only present in specific clinical cases. Instead, local proinflammatory R/C chemokine expression seems to be a common underlying factor in many immunologically related conditions, including systemic diseases.28,29
R/C expression appears to be an indicator for local BMDJ/FDOJ. Simultaneously, it may also trigger systemic diseases to develop in the immune system, which can take on the form of “silent inflammation.” The seldom noticed and largely ignored BMDJ/FDOJ should be given greater attention in medicine and dentistry, as their presence may represent a possible aggravating factor in systemic immunological diseases. Further research is needed to validate our findings.
Abbreviations
BMDJ, bone marrow defect in jawbone; CBVT, cone-beam computed tomography; DVT, digital volume tomography; DVT-HU, digital volume tomography-Hounsfield units; FDOJ, fatty-degenerative osteolysis/osteonecrosis of the jawbone; HU, Hounsfield units; OPG, orthopantomogram; R/C, RANTES/CCL5; 2D, two dimensional; 3D, three dimensional.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Raman D, Baugher PJ, Thu YM, Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007;256(2):137–165. doi:10.1016/j.canlet.2007.05.013
2. Dinarello CA. Historical review of cytokines. Eur J Immunol. 2007;37(Suppl 1):S34–S45. doi:10.1002/eji.200737772
3. Kaplanski G, Farnarier C, Kaplanski S, et al. Interleukin-1 induces interleukin-8 from endothelial cells by a juxtacrine mechanism. Blood. 1994;84(12):4242–4248. doi:10.1182/blood.V84.12.4242.bloodjournal84124242
4. Apte RN, Douvdevani A, Zoller M, et al. Cytokine-induced tumorigenicity: endogenous interleukin-1α expressed by fibrosarcoma cells confers reduced tumorigenicity. Immunol Lett. 1993;39(1):45–52. doi:10.1016/0165-2478(93)90163-V
5. Schmitz J, Owyang A, Oldham E, et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity. 2005;23(5):479–490. doi:10.1016/j.immuni.2005.09.015
6. Lechner J, von Baehr V. RANTES and fibroblast growth factor 2 in jawbone cavitations: triggers for systemic disease? Int J Gen Med. 2013;6:277–290. doi:10.2147/IJGM.S43852
7. Lechner J, Schulz T, von Baehr V. Immunohistological staining of unknown chemokine RANTES/CCL5 expression in jawbone marrow defects—osteoimmunology and disruption of bone remodeling in clinical case studies targeting on predictive preventive personalized medicine. EPMA J. 2019;10(4):1–14. doi:10.1007/s13167-019-00182-1
8. Samson M, Labbe O, Mollereau C, Vassart G, Parmentier M. Molecular cloning and functional expression of a new human CC-chemokine receptor gene. Biochemistry. 1996;35(11):3362–3367. doi:10.1021/bi952950g
9. Sicoli D, Jiao X, Ju X, et al. CCR5 receptor antagonists block metastasis to bone of v-Src oncogene-transformed metastatic prostate cancer cell lines. Cancer Res. 2014;74(23):7103–7114. doi:10.1158/0008-5472.CAN-14-0612
10. Velasco-Velázquez M, Xolalpa W, Pestell RG. CCL5/CCR5 in breast cancer. Expert Opin Ther Targets. 2014;18(11):1265–1275. doi:10.1517/14728222.2014.949238
11. Segall RO, Del Rio CE. Cavitational bone defect: a diagnostic challenge. J Endourol. 1991;17(8):396. doi:10.1016/S0099-2399(06)81993-X
12. Lechner J. Validation of dental X-ray by cytokine RANTES– comparison of X-ray findings with cytokine overexpression in jawbone. Clin Cosmet Investig Dent. 2014;6:71–79. doi:10.2147/CCIDE.S69807
13. Norton MR, Gamble C. Bone classification: an objective scale of bone density using the computerized tomography scan. Clin Oral Implants Res. 2001;12(1):79–84. doi:10.1034/j.1600-0501.2001.012001079.x
14. Misch CE. Bone density: a key determinant for clinical success. In: Misch CE, editor. Contemporary Implant Dentistry.
15. Mah P, Reeves TE, McDavid WD. Deriving Hounsfield units using grey levels in cone-beam computed tomography. Dentomaxillofac Radiol. 2010;39(6):323–335. doi:10.1259/dmfr/19603304
16. Lee S, Gantes B, Riggs M, Crigger M. Bone density assessments of dental implant sites: 3. Bone quality evaluation during osteotomy and implant placement. Int J Oral Maxillofac Implants. 2007;22(2):208–212.
17. Lechner J, von Baehr V. Chemokine RANTES/CCL5 as an unknown link between wound healing in the jawbone and systemic disease: is prediction and tailored treatments in the horizon? EPMA J. 2015;6(1):10. doi:10.1186/s13167-015-0032-4
18. Lechner J, Rudi T, von Baehr V. Osteoimmunology of tumor necrosis factor-alpha, IL-6, and RANTES/CCL5: a review of known and poorly understood inflammatory patterns in osteonecrosis. Clin Cosmet Investig Dent. 2018;10:251–262. doi:10.2147/CCIDE.S184498
19. Lechner J, von Baehr V. Hyperactivated signaling pathways of chemokine RANTES/CCL5 in osteopathies of jawbone in breast cancer patients—case report and research. Breast Cancer (Auckl). 2014;8:89–96. doi:10.4137/BCBCR.S15119
20. Lechner J, Huesker K, von Baehr V. Impact of RANTES from jawbone on chronic fatigue syndrome. J Biol Regul Homeost Agents. 2017;31(2):321–327.
21. Puxeddu I, Panza F, Pratesi F, et al. CCL5/RANTES, sVCAM-1, and sICAM-1 in chronic spontaneous urticaria. Int Arch Allergy Immunol. 2013;162(4):330–334. doi:10.1159/000354922
22. Bruno PP, Carpino F, Carpino G, Zicari A. An overview on immune system and migraine. Eur Rev Med Pharmacol Sci. 2007;11(4):245–248.
23. Qin XY, Zhang SP, Cao C, Loh YP, Cheng Y. Aberrations in peripheral inflammatory cytokine levels in Parkinson disease: a systematic review and meta-analysis. JAMA Neurol. 2016;73(11):1316–1324. doi:10.1001/jamaneurol.2016.2742
24. Aldinucci D, Colombatti A. The inflammatory chemokine CCL5 and cancer progression. Mediators Inflamm. 2014;2014:292376. doi:10.1155/2014/292376
25. Huang Y, Zheng S, Wang R, Tang C, Zhu J, Li J. CCL5 and related genes might be the potential diagnostic biomarkers for the therapeutic strategies of rheumatoid arthritis. Clin Rheumatol. 2019;38(9):2629–2635. doi:10.1007/s10067-019-04533-1
26. Zhou Y, Guo W, Zhu Z, et al. Macrophage migration inhibitory factor facilitates production of CCL5 in astrocytes following rat spinal cord injury. J Neuroinflammation. 2018;15(1):253. doi:10.1186/s12974-018-1297-z
27. Rudemiller NP, Crowley SD. The role of chemokines in hypertension and consequent target organ damage. Pharmacol Res. 2017;119:404–411. doi:10.1016/j.phrs.2017.02.026
28. von Luettichau I, Nelson PJ, Pattison JM, et al. RANTES chemokine expression in diseased and normal human tissues. Cytokine. 1996;8(1):89–98. doi:10.1006/cyto.1996.0012
29. Tsukishiro S, Suzumori N, Nishikawa H, Arakawa A, Suzumori K. Elevated serum RANTES levels in patients with ovarian cancer correlate with the extent of the disorder. Gynecol Oncol. 2006;102(3):542–545. doi:10.1016/j.ygyno.2006.01.029
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.