Back to Journals » ImmunoTargets and Therapy » Volume 9
Therapeutic Vaccines for HPV-Associated Malignancies
Authors Smalley Rumfield C ![]() , Roller N, Pellom ST
, Roller N, Pellom ST ![]() , Schlom J
, Schlom J ![]() , Jochems C
, Jochems C ![]()
Received 22 July 2020
Accepted for publication 3 September 2020
Published 7 October 2020 Volume 2020:9 Pages 167—200
DOI https://doi.org/10.2147/ITT.S273327
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Michael Shurin
Claire Smalley Rumfield, Nicholas Roller, Samuel Troy Pellom, Jeffrey Schlom,* Caroline Jochems*
Laboratory of Tumor Immunology and Biology, Center for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, MD, USA
*These authors contributed equally to this work
Correspondence: Jeffrey Schlom
Laboratory of Tumor Immunology and Biology, Center for Cancer Research, National Cancer Institute, 10 Center Drive, Room 8B09, Bethesda, MD 20892, USA
Tel +1 240-858-3463
Fax +1 240-541-4558
Email [email protected]
Abstract: Human papillomavirus (HPV)-related malignancies are responsible for almost all cases of cervical cancer in women, and over 50% of all cases of head and neck carcinoma. Worldwide, HPV-positive malignancies account for 4.5% of the global cancer burden, or over 600,000 cases per year. HPV infection is a pressing public health issue, as more than 80% of all individuals have been exposed to HPV by age 50, representing an important target for vaccine development to reduce the incidence of cancer and the economic cost of HPV-related health issues. The approval of Gardasil® as a prophylactic vaccine for high-risk HPV 16 and 18 and low-risk HPV6 and 11 for people aged 11– 26 in 2006, and of Cervarix® in 2009, revolutionized the field and has since reduced HPV infection in young populations. Unfortunately, prophylactic vaccination does not induce immunity in those with established HPV infections or HPV-induced neoplasms, and there are currently no therapeutic HPV vaccines approved by the US Food and Drug Administration. This comprehensive review will detail the progress made in the development of therapeutic vaccines against high-risk HPV types, and potential combinations with other immunotherapeutic agents for more efficient and rational designs of combination treatments for HPV-associated malignancies.
Keywords: human papillomavirus, HPV, therapeutic vaccine, cervical cancer, head and neck squamous cell carcinoma, HNSCC, combination immunotherapy
Introduction
Human papillomavirus (HPV)-associated malignancies make up about 4.5% of all cancers, afflicting more than 600,000 people worldwide each year.1 HPV-associated cancers include carcinomas of the cervix (99.7% HPV-associated, the fourth most common cancer in women worldwide2), and squamous cell carcinomas of the oropharynx, anus, rectum, penis, vagina, and vulva.
HPV is a small double-stranded DNA-virus; there are over 200 known types, most of which confer a low risk of cancer development.3,4 HPV6 and 11 are two low-risk types (lrHPV) that cause genital warts but are not carcinogenic. The high-risk HPV (hrHPV) types include HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59.
All types of HPV encode “early proteins” (E-proteins: E1, E2, E6, E7), and “late proteins” (L-proteins: L1, L2). Upon HPV infection, HPV virions will bind to the basal cell heparin sulfate proteoglycan using L1 and L2 proteins.5 Once internalized, it is thought that E1 and E2 are responsible for amplification of the viral episome, and E2 also regulates viral transcription.5 The E6 and E7 proteins are responsible for driving cell proliferation. If the infection persists uncontrolled by the host’s immune system, the hrHPV viral genome remains in the cell as an episome and will result in benign and precancerous lesions. Integration of the episome into the host genome results in the development of cervical carcinoma and cervical intraepithelial neoplasia (CIN) grades 1–3.6 Integration of the viral genome, together with dysregulation of early protein 2 (E2), which regulates the oncoproteins E6 and E7, results in the overexpression of E6 and E7. Both of these proteins drive uncontrolled cell growth.7 E6 interacts with and degrades p53 and other host cell proteins,8 while E7 interacts with and degrades Rb protein,9 both of which act as repressors of cellular proliferation.10 In addition to interfering with the p53 and Rb proteins, E6 and E7 also interfere with mammalian target of rapamycin (mTOR) signaling and senescence in the infected cells.11,12
CIN grade 1 is considered a low-grade squamous intraepithelial lesion (LSIL), and the rate of spontaneous regression is 70–80%.13 CIN grade 2 and CIN grade 3 are considered high-grade squamous intraepithelial lesions (HSIL). CIN 2 will occasionally regress, with a spontaneous regression rate of 15–23% within several years.14,15 CIN 3 is much less likely to spontaneously regress, and if untreated, 30%16 will progress to carcinoma.
For invasive cervical cancer, HPV16 is the most prevalent type (approximately 60%), HPV18 is the second (15%), and HPV45 is the third most common type.17 The use of prophylactic vaccines is predicted to help decrease these numbers in the future since the vaccines are very efficient at preventing persistent infection and the development of neoplasia. It has been estimated that the currently available nonavalent prophylactic HPV vaccine could prevent 90% of cervical cancer cases and 50% of all other HPV-related cancers (vulvar, penile, vaginal, anal, oropharyngeal, oral cavity, and laryngeal).18 This vaccine includes coverage against HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58. Prophylactic vaccines (Gardasil® (Merck, Kenilworth, NJ, USA) and Cervarix® (GlaxoSmithKline, Brentford, UK)) are subunit virus-like particles (VLPs) that act by stimulating strong neutralizing antibody responses to the capsid protein L1 on the surface of the HPV virus particle. Antibodies binding to the virus hinder it from infecting the epithelial cells, thus preventing infection.19 It has been shown that high antibody titers result in an immunoglobulin-coated capsid, which prevents the virus particle from binding to heparin sulfate proteoglycans on the basal cells, the first step of infection. This results in the antibody-covered virus being cleared by neutrophils.20,21 In the presence of low antibody titers, the virus particles are only partially hindered from binding the basal cells, and the main mechanism of action is instead caused by the prevention of the capsid interacting with the second L1 receptor on the epithelial cell surface. This results in the virus being lost from the tissue.20 However, these vaccines do not clear already established infections and do not mount the cellular CD4+ and CD8+ T-cell-based immune responses needed to combat precancerous or cancerous lesions. Due to sub-optimal prophylactic HPV vaccination rates worldwide, HPV infections and subsequent development of HPV-associated malignancies will continue to be a public health issue in the coming decades. Therefore, the development of therapeutic HPV vaccines and other cancer therapies represents a pressing public health concern.
One unique feature of HPV-associated malignancies is that the E6 and E7 proteins are constitutively expressed at high levels in the tumor cells and are not present anywhere else in the human body.22 Furthermore, since they are necessary for the transformation of malignant cells and drive the malignant cell phenotype,23 it is not likely that antigen loss would be a viable tumor escape mechanism. This makes them an ideal target for therapeutic vaccines, which are designed to generate a specific antitumor response targeting cells expressing the antigens while minimizing the risk of accidentally attacking healthy tissues. The immune responses to these proteins have been well characterized,24 and most therapeutic vaccines to date have used E6, E7, or a combination of both as the target antigens. Indeed, a healthy immune system will mount an immune response against these proteins. One recent study showed that women with abnormal pap smears who displayed HPV16 E6‒specific CD4 and CD8 T-cell responses had a favorable clinical trend associated with the regression of lesions, compared with women who did not have HPV antigen-specific T-cell responses.25,26 HPV16 E7‒specific responses were significantly associated with CD4, but not CD8, T-cell responses in this study.
Several different approaches for vaccine therapy will be discussed in this report, with examples of vaccines at different stages of development, as well as potential combinations of vaccines with other immunotherapy modalities. Immune checkpoint inhibitors can potentiate an existing immune response, while other agents can help increase an immune response by decreasing immune-suppressive elements in the tumor microenvironment (TME). Both of these mechanisms may be useful to potentiate the efficacy of a therapeutic vaccine. Current clinical trials for therapeutic HPV vaccines were queried at the NIH ClinicalTrials.gov database utilizing “HPV Vaccine” as the search string, yielding 63 studies that involve therapeutic HPV vaccines as monotherapies or in combination with other immunotherapeutics. Terminated and withdrawn clinical studies are excluded in this report, and the results are summarized in Tables 1–8. Currently, there is no therapeutic HPV vaccine approved by the US Food and Drug Administration (FDA).
 |
Table 1 Peptide-Based Vaccines |
 |
Table 2 Protein-Based Vaccines |
 |
Table 3 Viral-Vectored Vaccines |
 |
Table 4 Bacterial-Vectored Vaccines |
 |
Table 5 Cell-Based Vaccines |
 |  |  |
Table 6 DNA and RNA Vaccines |
 |
Table 7 Multi-Platform Vaccines |
 |  |  |  |
Table 8 Combination Trials |
Peptide-Based Vaccines
The advantages of peptide vaccines are that they are typically safe and tolerable for all populations and have the additional benefit of being inexpensive and easy to produce.27 Current clinical trials employing therapeutic peptide vaccines for HPV are summarized in Table 1. Potential disadvantages of peptide-based vaccines are that they often need to be combined with immunogenic adjuvants, or contain agonist epitopes, to elicit a stronger immune response.28 Short peptide-based vaccines do not require antigen processing and can bind directly to human leukocyte antigen (HLA) class I, which induces CD8+ T-cell responses, but does not stimulate CD4+ T-cell responses to maintain a long-lived effector response.29 In addition, short peptide-based (8–10 mer) vaccines have the limitation of being MHC-specific and need to be matched with the patient’s MHC type.28 One strategy to conquer this issue is to generate longer peptides that cover several potential epitopes specific for different MHCs, which allows for antigen processing by antigen-presenting cells (APCs), stimulating longer lived CD4+ and CD8+ responses.29
Synthetic Long Peptide-Based Vaccines
Such synthetic long peptides (SLP) of the HPV16 E6 and E7 oncoproteins were evaluated in a Phase I trial in women with end-stage cervical cancer.30 The vaccine consisted of a mix of nine HPV16 E6 and four HPV16 E7 synthetic long peptides, covering the entire sequences of E6 and E7, in incomplete Freund’s adjuvant (MontanideTM ISA-51), and was found to be well tolerated and resulted in a strong HPV-specific T-cell response. The same vaccine, ISA101, was also evaluated in women with HPV16-positive grade 3 vulvar intraepithelial neoplasia (VIN).31 Patients were vaccinated three or four times, and at the 12-month follow-up, 15/19 (79%) of patients had clinical responses; 9/19 had a complete response, and this response was maintained at 24 months. Antigen-specific T-cell responses were seen in all patients, and patients who had a complete response at 3 months displayed stronger and broader CD4 and CD8 T-cell responses than patients who did not attain a complete response.31 A Phase II trial of ISA101 also showed it to be well tolerated and capable of inducing a broad interferon gamma (IFNγ)‒associated T-cell response in women with advanced or recurrent HPV16-associated gynecological carcinomas, but no tumor regression was seen in this advanced setting.32 The ISA101 vaccine has therefore now been investigated in combination immunotherapy (see the section on combination therapies below).33 An ongoing clinical trial is evaluating ISA101 in anal intraepithelial neoplasia (AIN) in HIV-positive men (NCT01923116).
A second therapeutic vaccine consisting of four synthetic peptides is PepCan, in which the HPV peptides are mixed with Candin® (Candida albicans skin test reagent) as an adjuvant.34,35 A Phase I trial showed the vaccine to be safe in patients with CIN 2/3.34,35 The histological regression rate was 45% overall. Immune responses to HPV16 E6 were seen in 61% of patients and significantly increased systemic levels of TH1 cells were observed. Moreover, in 12 patients where HPV16 was detected before vaccination, viral load was significantly decreased in nine patients and undetectable in three patients after treatment.35 PepCan is currently being investigated in two Phase I/II trials (NCT03821272, NCT02481414). A separate mix of four peptides mixed with Candin® has also been investigated in a Phase I trial (NCT01653249). Vaccination with the combination of the HPV peptides and Candin® induced the maturation of Langerhans cells, and increased T-cell proliferation in humans,36 with the maturation attributed to the HPV peptides and the proliferation attributed to the Candin®. Additionally, cytokine profiles of Langerhans cells showed upregulated IL-12p40 mRNA, and increased IL-12p70 in the supernatant, consistent with the induction of a proinflammatory immune response.
A vaccine called Hespecta, or ISA201 (HPV E-Six Peptide Conjugated To AMPLIVANT®, ISA Pharmaceuticals B.V., Leiden, The Netherlands), consisting of two HPV16 E6 SLPs conjugated to a synthetic TLR2 ligand (Amplivant®) is also under investigation.37 Each peptide covers an immunodominant region of the HPV16 E6 protein and contains multiple T helper and cytotoxic lymphocyte epitopes. Preclinical studies have shown that the addition of the TLR2 adjuvant to the SLPs induces stronger dendritic cell (DC) maturation, in vivo T-cell priming, and anti-tumor immunity than SLPs alone or with the addition of a Pam3CSK4 adjuvant.38 A Phase I clinical trial to assess the safety of Hespecta was expected to be completed by December 2017, but there are no current updates on clinicaltrials.gov (NCT02821494).
Multiple other SLP vaccines are being developed preclinically, often with novel adjuvant strategies to enhance immunogenicity. Some adjuvant strategies that have been combined with SLP are CpG and nanoparticles. A novel synthetic long peptide vaccine adjuvanted with CpG is currently being investigated preclinically. SLP-CpG39 consists of a synthetic long peptide from HPV16 E7 with a centrally located MHC I epitope, adjuvanted with the TLR9 agonist CpG formulated in a squalene-based oil-in-water emulsion. TLR3, TLR4, and TLR7/8 agonists were also tested, but the TLR9 CpG agonist induced the most robust CD8 T-cell responses and inhibited tumor growth in the TC-1 murine tumor model.39 Another SLP vaccine, NP-E7Lp, consisting of an SLP from HPV16 E7 conjugated to ultra-small nanoparticles, showed significant increases in effector T cells upon injection, and an increased CD8+ T cell to regulatory T cell (Treg) ratio. In vivo, vaccination with NP-E7LP resulted in TC-1 tumor regression with complete responses.40
Short Peptide-Based Vaccines
In terms of vaccines that are based on shorter peptide sequences, PDS0101 is a short peptide-based, non-MHC-restricted vaccine adjuvanted with R-DOTAP, a liposomal carrier that activates TLR7.41,42 PDS0101 was found to be safe and tolerable in a Phase I dose-escalation trial (NCT02065973). Vaccination resulted in the regression of CIN in all patients in a non-MHC-restricted manner.43 Several Phase II trials have been initiated for the combination of PDS0101 with other immunomodulatory therapeutics, such as a Phase II trial in combination with pembrolizumab in patients with HPV16+ recurrent or metastatic head and neck squamous cell carcinoma (HNSCC) (NCT04260126), and another Phase II trial in combination with NHS-IL12 (EMD Serono, Billerica, MA, USA) and bintrafusp alfa (EMD Serono and Pfizer, New York, NY, USA), started in June 2020 at the National Cancer Institute (NCT04287868). This trial was initiated based on a preclinical study.44
A second short peptide-based vaccine, DepoVax adjuvant emulsified with HPV16 E7 peptide (DPX-E7), is a novel vaccine that is also being developed preclinically and clinically.45 Tumor-bearing mice immunized with DPX-E7 displayed fewer Treg and myeloid-derived suppressor cells (MDSC) in the tumor, and significantly lower tumor burden compared to controls.45 DPX-E7 is currently in a Phase Ib/II clinical trial for safety and efficacy (NCT02865135). The DepoVax platform is a modified water-free version of the previously described VacciMax platform, which also showed promising results with the rejection of large HPV16-expressing TC-1/A2 tumors.46
Therapeutic vaccines have also included other immune-modulating peptides in addition to HPV-peptides, such as human leukocyte antigen (HLA)-I‒ and HLA-II‒restricted melanoma antigen E (MAGE-A3) peptides or HIV peptides. A pilot study using MAGE-A3 or HPV16 peptides linked to a peptide sequence from HIV-TAT, the Trojan vaccine, supplemented with MontanideTM and granulocyte-macrophage colony-stimulating factor (GM-CSF), was shown to be safe and immunogenic.47 A Phase I dose escalation study (NCT00257738) in HNSCC patients showed no dose-limiting toxicity (DLT), and T-cell and antibody responses to HPV were seen in patients who received four vaccinations.48
Other therapeutic vaccines target HPV-positive malignancies, but they do not contain the classic HPV E6 or E7 antigens. A Phase I/IIa study (NCT01462838) uses the p16 37–63 peptide, from the cyclin-dependent kinase inhibitor p16(INK4a), adjuvanted with MontanideTM.49 P16(INK4a) is highly upregulated in HPV-associated malignancies. Patients with confirmed overexpression of p16(INK4a) in their HPV-positive malignancies were given four vaccinations over 6 months. There were no severe toxicities, and at the end of the trial, nine patients had stable disease, and five patients developed progressive disease. Another Phase I study (NCT02526316) combines this peptide vaccine with chemotherapy in order to modulate the effect of the vaccine.
Finally, a short peptide vaccine adjuvanted with very small size proteoliposomes (VSSP) has also been tested in a small number of humans for safety,50 although this clinical trial is unregistered at clinicaltrials.gov. CIGB-228 is an HLA-A2-restricted HPV16 E7 peptide, which is a known cytotoxic T lymphocyte (CTL) epitope, combined with VSSP, that produces regression of TC-1 tumors, and protects them from re-challenge due to the production of HPV16 E7-specific memory T-cell responses. CIGB-228 was tested in seven HLA-A2 positive patients who had HPV16-positive CIN 2/3 and was administered once weekly subcutaneously. There were no serious adverse events (AE), and at the end of the study 57.1% of patients had complete regression and 14.3% of patients had histological downgrading. All patients also showed an increase in IFNγ-producing cells in peripheral blood mononuclear cells (PBMC).50
The development of short peptide-based vaccines will become easier in the future due to artificial intelligence (AI) and advancement in algorithms that can better predict T-cell epitopes51 for improved immunogenicity for multiple hrHPV types,52 as well as for many HLA types,53 which will help overcome HLA-restriction.
Protein-Based Vaccines
Protein-based vaccines are processed by antigen-presenting cells (APCs), resulting in presentation of antigens via HLA class I and class II for the generation of CD8+ and CD4+ T-cell responses without type restriction. Potential advantages of protein-based vaccines are that they are typically safe and tolerable, including for those individuals who are immunocompromised.54 However, a potential disadvantage of protein-based vaccines is that they can be poorly immunogenic for the generation of CD8+ T-cell responses and therefore may require immunogenic adjuvants to augment the efficacy and to avoid major histocompatibility complex (MHC) II processing for antibody-based immunity.54 Clinical studies employing protein-based vaccines are listed in Table 2.
Tissue antigen-cervical intraepithelial neoplasia (TA-CIN) is a fusion protein that consists of HPV16 proteins L2, E6, and E7.55 A Phase I study was recently completed in CIN for safety and efficacy, as measured by increases in antigen-specific responses56 (NCT02405221). A Phase II trial conducted in the UK in VIN grade 2/3 showed significant T-cell infiltration and regression of lesions in the majority of patients after administration of the vaccine adjuvanted with imiquimod.57
TVGV1 is a fusion protein vaccine candidate that uses a Pseudomonas aeruginosa exotoxin fusion with HPV16 E7, and a KDEL endoplasmic reticulum retention signal, adjuvanted with CpG ODN or GPI-0100.58 TVGV1 with both adjuvants was able to induce multifunctional T-cell responses that showed efficacy against the C3.43 in vivo tumor model (HPV16-transformed B6 tumor).58 There is an ongoing Phase IIA clinical trial in women with high-grade HPV cervical infection, but the status of this trial is unclear on clinicaltrials.gov (NCT02576561).
GTL001 (ProCervix) is a combination of HPV16 and HPV18 E7 proteins fused to CyaA, a Bordetella pertussis adenylate cyclase that has had its toxic components removed, and adjuvanted with imiquimod cream, a common treatment for HPV-positive genital warts.59 GTL001 was evaluated in a Phase II study that enrolled 233 HPV16 and HPV18 positive patients with both normal and abnormal cervical cytology (NCT01957878), and in a second trial, where patients received one dose of vaccine or placebo adjuvanted with imiquimod. Viral clearance rates were found to be the same between the vaccine and placebo groups 2 years later, so the study was terminated (NCT02689726). Further development of GTL001 was stopped, and a second-generation vaccine, GTL002, has recently been in preclinical testing for in vivo efficacy in generating antigen-specific T-cell responses as well as regression in the TC-1 model.60
SGN-00101 (also known as HSP-E7) is based on the fusion of HPV16 E7 with recombinant heat shock protein 65 (HSP65) from Mycobacterium bovis.61 Clinical responses to this vaccine have been seen in AIN,62 CIN3,63 and cervical HSIL,64 with increases in HPV-specific CTLs as well as regression of HSIL. Two Phase II studies have recently been completed (NCT00054041, NCT00091130).
Other protein vaccines are being investigated preclinically, including novel fusion proteins or recombinant lipidated proteins. The fusion protein LALFE7,65 now called CIGB550-E7, has been explored preclinically for its use in mediating anti-tumor responses in mice bearing TC-1 tumors.66 CIGB550-E7 is a cell-penetrating peptide linked to an HPV16 E7 mutein, adjuvanted with VSSP. Vaccination with this CIGB550-E7 fusion protein in mice generated both anti-tumor responses as well as cell-mediated immune responses. The recombinant lipidated protein rE6mE7m67 activates TLR2 and stimulates and upregulates the costimulatory molecules CD40 and CD80 on bone marrow‒derived dendritic cells. In vivo, rlipo-rE6mE7m can activate CTL and inhibit TC-1 tumor growth.
Viral Vectors
Viral vectors, including both DNA and RNA viruses, are some of the most well-tested antigen delivery systems to induce an immune response.68 Viral vectors directly infect host cells, and induce presentation of class-restricted antigens on the cell surface. They can be engineered to express any antigen of interest.69 Potential advantages of these vectors are that they are highly immunogenic and produce rapid antibody and CD8 T-cell responses to antigens present in the vector.68 Potential disadvantages of this platform include the development of neutralizing antibody responses to the vectors, requiring alternative prime-boost strategies. Clinical trials for therapeutic HPV viral-vectored vaccines are summarized in Table 3.
DNA Virus-Based Viral Vectors
Some of the most widely used viral vectors are the vaccinia virus vectors, which are stable vectors capable of holding large amounts of transgenic DNA. Attenuated poxviruses have been used in vaccine regimens for the eradication of smallpox, and thus have a long history of safety in humans. Individuals immunized against smallpox, however, may have neutralizing antibodies against poxviral vectors.70,71 Tipapkinogen Sovacivec (TS) vaccine is an attenuated and replication-deficient modified vaccinia Ankara (MVA) vector, with inserted genes that code for human IL-2, HPV16 E6, and HPV16 E7. The Phase II trial (NCT01022346) in CIN 2/3 with a 2½-year follow-up showed reversion of CIN 2/3 in vaccinated patients regardless of hrHPV type.72
TG4001 is another MVA-vectored HPV16 E6, E7, and IL-2 expressing vaccine that has shown positive results in Phase Ib/II. It was shown to be safe and resulted in regression of CIN in patients receiving the vaccine.73 TG4001 is currently being evaluated in combination with avelumab (anti-programmed cell death protein-1 ligand (PD-L1) antibody, Merck KGaA, Darmstadt, Germany, and Pfizer) in a Phase Ib/II trial in HPV16-positive recurrent or metastatic cancers including oropharyngeal HNSCC (NCT03260023). Current data from the Phase Ib portion of this trial have shown a partial response in 3/9 patients with oropharyngeal, anal, cervical, and vaginal refractory or metastatic cancer, with no DLTs or serious AEs.74 TA-HPV is a live recombinant MVA vector that expresses HPV16/18 E6 and E7 proteins, which has been evaluated in patients (NCT00002916) and is currently in combination trials with DNA vaccines. It was shown to be safe and immunogenic, and generated HPV-specific CTL responses in a non-HLA–restricted manner in four patients, and HPV-specific serological responses in eight other patients.71
Human adenoviruses, another widely used viral vector, have been used for gene therapy and can transduce large amounts of foreign DNA. They have tropism for a variety of cell types and can transduce both quiescent and actively dividing cells, making them an ideal vector for therapeutic vaccines.75 Immunity to human adenoviruses is common, as they are a widely circulating subset of human viruses. Immunity to Ad5, one of the most commonly used therapeutic adenoviruses, approaches 60% in North America and Europe, and up to 90% in sub-Saharan Africa.68 Thus, rare human adenoviruses such as Ad26 and Ad35 have recently been investigated as potential vectors for HPV antigens, encoding E2, E6, and E7 fusion proteins for HPV16 and HPV18 positive malignancies.76 This vaccine showed efficacy in the murine TC-1 model and elicited robust T-cell immunity. An additional recent paper described the use of an intramuscular prime and an intravaginal boost regimen using adenovirus types 26 and 35 expressing a fusion of HPV16 E6 and E7 oncoproteins.77 The authors found induction of HPV-specific CD8 T cells producing IFNγ and tumor necrosis factor (TNF)α in the cervicovaginal tract of treated mice and concluded that this diverse prime-boost regimen is a promising strategy for persistent HPV infection and CIN.
In order to take advantage of the safety of both human adenoviruses and MVA vectored viruses and a heterologous prime-boost strategy, a trial using a combination of the two viruses has been initiated (NCT03610581). This trial will be investigating a combination of priming Ad26 vector expressing HPV16 or HPV18, and a boost of MVA expressing HPV16/18. Indeed, this heterologous prime-boost of Ad26 and MVA has recently been used to elicit anti-Ebola glycoprotein responses, and was well tolerated78 (NCT02376426).
Non-human primate adenoviruses are an attractive alternative to human adenovirus vectors, as there is no pre-existing immunity in human populations, and they retain close homology with human adenoviruses. Currently, there are two non-human primate vectors in development. One is a chimpanzee adenovirus vector, which was built with a synthetic gene designed by selecting conserved regions from each of six early proteins to represent five hrHPV genotypes.79 In preclinical studies, this gene was delivered by three different methods in prime-boost regimens: plasmid DNA, chimpanzee adenovirus (ChAdOx1), and MVA vectors. The combination of ChAdOx1 and MVA vectors led to the strongest and most durable HPV-specific T-cell responses. Vaccine-induced T cells were polyfunctional and trafficked to the cervix following administration.79 A clinical observational study (16/SW/0331) in humans showed antigen-specific responses by IFNγ ELISpot in women who had been vaccinated and had current or past hrHPV infections. Another non-human primate vector is a novel gorilla adenovirus currently being explored for multiple therapeutic modalities including infectious disease and cancer.80 Preclinical studies have used gorilla adenovirus vectors for the treatment of malaria and respiratory syncytial virus (RSV),81,82 as well as for HPV-positive malignancies in preclinical models (unpublished data). A Phase I clinical trial in HNSCC is planned at the National Cancer Institute (NCT04432597).
RNA Virus-Based Viral Vectors
RNA virus-based vectors have been emerging for therapeutic use and are typically alphaviruses or arenaviruses. RNA replicon vaccines insert RNA sequences encoding target antigens into non-replicating viral vectors, which can be used with multiple administrations for sustained antigen expression without risk of cellular transformation or chromosomal integration.83 Compared to DNA viruses, RNA viral vectors are relatively labor-intensive and unstable, and have dose-limiting toxicities.83 Additionally, human safety data for these vectors are not well established. Only one RNA viral vector, Semliki Forest virus (SFV) replicons encoding E6 and E7, known as Vvax001, is entering a Phase I trial for safety and efficacy in humans (NCT03141463), having induced HPV-specific cytotoxic T-cell responses and reduced tumor burden in mice.84,85
Additional RNA viral vectors are currently being investigated in animal models. An arenavirus-based vector, HB-201, is a lymphocytic choriomeningitis virus (LCMV)‒based vector that increased HPV-specific CTL and cleared TC-1 tumors in vivo.86 HB-201 is currently in a Phase I/II trial (NCT04180215), using HB-201 alone or in combination with nivolumab. The Venezuelan equine encephalitis virus (VEE)–based viral vector, E7-VRP, eliminated established C3 tumors in the majority of mice, and protected mice from tumor re-challenge.87 Another group developing VEE-based vaccines also showed promising results in C3 and TC-1 tumors in mice.88 The Sindbis virus–based vector, VP22-E7, has been used in preclinical studies with similar effects, resulting in tumor regression and generation of HPV-specific T-cell responses.89
Bacterial Vectors
Bacterial vectors have been explored as potential live vectors because of their ability to produce robust innate and adaptive immune responses.90 Bacterial vectors act similarly to viral vehicles in that they can be engineered to express the antigen of interest, therefore generating immune responses against the specific target.69 Potential advantages are that bacteria are “natural” adjuvants due to their wide range of pathogen-associated molecular pattern molecules (PAMPs) and damage-associated molecular pattern molecules (DAMPs), which activate toll-like receptors (TLRs) 2, 4, 5, 7, 8, and 9 and other innate immune mechanisms.91 Adaptive immune responses induced by bacterial vectors are equally powerful, due to the host of inflammatory cytokines bacterial infections produce. Potential disadvantages of this platform include the potential for toxicity due to the robust immune response. Current clinical trials employing bacterial vectors are summarized in Table 4.
Listeria-Based Vectors
Listeria monocytogenes, in particular, has been extensively developed into a potential therapeutic vaccine. Listeria is a facultatively intracellular Gram-negative bacteria, and upon infection of the host, induces phagocytosis in responding macrophages. Once engulfed into the phagolysosome, Listeria uses the protein listeriolysin-O (LLO) to escape the phagosome and live freely in the cytosol of host cells.92–94 Because of its intracellular niche, Listeria can activate both CD8+ and CD4+ T cells through the MHC I and MHC II pathways. Listeria induces significant increases in the production of IFNγ, IFNα, IFNβ, and a variety of chemokines, and is a potent activator of TLRs 2 and 5, resulting in further cytokine and chemokine production to control infection. Due to the pro-inflammatory properties and direct antigen processing inherent in Listeria infections, it has been explored as a therapeutic vector for multiple cancer types, including colon, prostate, and breast (NCT03265080, NCT02386501, NCT02325557). More recently, Listeria has been used as a therapeutic vaccine vector for HPV-positive malignancies. The most developed Listeria monocytogenes bacterial vectored HPV vaccine is ADXS11-001.95 ADXS11-001 is composed of Listeria monocytogenes with an HPV16 E7 protein fused to LLO, or Lm-LLO-E7. Preclinically, ADXS11-001 resulted in the regression of TC-1 tumors in C57BL/6 mouse models and induced memory as well as antigen-specific T cells. ADXS11-001 first moved into clinical trials for cervical cancer in 2009, showing an acceptable safety profile, and inducing mixed antigen-specific responses.95 Adverse events were reported, including pyrexia and fatigue. A Phase II study showed increased survival in advanced cervical cancer patients compared to historical controls,96 with a median overall survival (OS) of 8.3 months vs. 4.7 months. This study also included an ADXS11-001 plus cisplatin arm and reported a median OS of 8.8 months.96 Currently, a Phase III trial is ongoing for advanced cervical cancer (NCT02653604). Patients with cervical carcinoma will be given an infusion of ADXS11-001 every 3 weeks for three doses, then every 8 weeks for five doses. The results are expected in October 2024. Additional Phase I/II trials are also ongoing for a variety of indications, including oropharyngeal carcinoma, anal carcinoma, and persistent or recurrent cervical cancer (NCT02002182, NCT02399813, NCT01266460). Preliminary data indicate expanded T-cell receptor (TCR) clones post-vaccination with ADXS11-001 in patients with oropharyngeal cancer,97 and additional preliminary data indicate that ADXS11-001 can be administered with standard chemotherapy for anal cancer.98 One clinical trial for ADXS11-001 was terminated when a patient developed systemic listeriosis;99 however, this is the only known case found in the use of this agent.
Lactobacillus-Based Vectors
Other bacteria investigated as live vectors include the Lactobacillus species, including L. lactis, L. casei, and L. plantarum. These bacteria have been used for 30 years for therapeutic heterologous gene expression. They are administered non-invasively, typically orally or intranasally, and are considered relatively safe due to their lack of endotoxin. A Phase I/IIa dose-escalation study has been conducted using L. casei expressing HPV16 E7 (GLBL101c) in 10 patients with CIN3.100 No patient experienced AEs, and 70% of the participants receiving the optimized dose had their CIN3 downgraded to CIN2. The presence of cervical E7-specific lymphocytes directly correlated to pathological downgrade.
Lactobacillus lactis is an additional lactobacterium vector being explored preclinically and in early clinical studies. An engineered L. lactis expressing HPV16 E6 was administered orally to TC-1‒bearing C57BL/6 mice, resulting in reduced tumor burden as well as increased survival compared to controls.101 This oral vaccine (NZ8123-HPV-16-optiE6) was moved to a Phase I study with 46 healthy volunteers for dose-escalation, tolerability, and safety.102 No significant AEs were reported, and a dose-dependence was observed for humoral and persistent cell-mediated responses. Lactobacillus casei has been used for early in vivo work using L. casei expressing HPV16 E7, and another strain of L. casei expressing HPV16 E6.103,104 Immunization with L. casei expressing E6 or E7 led to an increase in E6- or E7-specific IFNγ positive T cells, and a decrease in TC-1 tumor burden in C57BL/6 mice. Other groups have seen similar results.105,106 In addition, L. plantarum expressing HPV16 E7 has been used similarly, yielding serum antibodies and antigen-specific T cells.107
Cell-Based Vaccines
A wide variety of cell-based vaccines are being explored as options for advanced HPV-associated malignancies in clinical trials, summarized in Table 5. This platform involves the patient’s APCs being directly loaded with HPV antigens and infused back into the patient. A potential advantage of this platform is that antigen-specific cells are introduced directly to the patient, eliminating the trial and error associated with generating antigen-specific cells within the tumor microenvironment via vaccine. This approach, however, may be hampered by HLA restriction or by long amounts of time necessary to generate personalized cell-based therapies, as well as prohibitive costs for large-scale treatment of patients.
Several of these studies consist of PBMC transplantation plus HPV peptide (NCT00003977, NCT00019110). Study NCT00019110 focused on harvesting PBMC from patients with advanced or recurrent cervical, vaginal, anal, or oropharyngeal HPV-positive cancer, treating the PBMC with granulocyte-macrophage colony-stimulating factor (GM-CSF), then pulsing the PBMC with HPV16 E6 and E7 proteins. Study NCT00003977 uses a similar methodology in patients with advanced cervical cancer and pulses the PBMC with lipidated HPV16 E7. DC-based therapies currently in clinical trials are being used for recurrent cervical cancer (NCT00155766) and for CIN 1/2 (NCT03870113). In study NCT03870113, autologous DC are loaded with HPV16/18 E6/E7 peptides and are injected back into the patient’s adjacent lymph node six times per week. Data from these studies have not yet been released.
BVAC-C consists of B cells and monocytes utilized as APCs, loaded with HPV16/18 E6/E7 peptides, and transfused back into patients with recurring or metastatic cervical cancer (NCT02866006). Data from the Phase I trial108 showed that BVAC injection resulted in mild AEs, and no DLTs in patients with platinum-resistant recurrent cervical cancer. The overall response rate was 11%, and the median progression-free survival was 6.8 months. Patients also exhibited increased activation of natural killer T (NKT) cells, NK cells, and HPV-specific T cells post-vaccination.108
DNA-Based Vaccines
DNA-based vaccines are a rapidly expanding area of vaccine research because they are safe and tolerable for all patient populations and easy to produce, and multiple plasmids encoding different antigens can be added without compromising safety or efficacy.109–112 DNA-based vectors act as shuttle systems to deliver and express antigens directly into target cells.69 The addition of a variety of sequences and codon optimization within the DNA shuttle helps increase transcription efficiency, expression of the antigen of interest, and targeting to the endoplasmic reticulum for downstream generation of humoral and adaptive immune responses.69 The disadvantages of DNA vaccines, however, are low transfection efficiency and immunogenicity; this modality requires specialized equipment for vaccination and additional adjuvants for improved immune responses.113 DNA vaccines can be administered by subdermal injection, but this delivers subpar uptake by dermal DC, stimulation of which is required for efficient antigen presentation and enhanced immune responses. Multiple studies are ongoing using DNA vaccines in the therapeutic treatment of HPV-positive malignancies. The clinical trials employing DNA and RNA vaccines are summarized in Table 6.
HPV DNA-Based Vaccines
The DNA-based vaccine VGX3100 is undergoing a variety of clinical trials and trial formats, as both a monotherapy and in combination with various TME-modulating immunotherapeutics. VGX-3100 is a DNA vaccine containing a mixture of two plasmids that encode optimized consensus E6 and E7 genes of HPV16 and 18.114 In the Phase I trial in women with CIN 2/3, the vaccine was given three times intramuscularly, followed by electroporation.114,115 VGX-3100 was shown to be safe, tolerable, and to induce HPV-specific CD8 T cells expressing granzyme B and perforin and exhibiting full cytolytic functionality. The randomized double-blind placebo-controlled Phase IIb trial in patients with CIN 2/3 showed efficacy against HPV16/18‒induced CIN.116 VGX-3100 was administered to 125 patients, and 42 patients were given placebo. Histopathological regression was seen in 49.5% of vaccine-treated patients, compared to 30.6% of controls. Furthermore, when 13/18 patients from the Phase I trial were given an additional boost of VGX-3100 vaccine after 9 months’ follow-up on the original trial, both cellular and humoral immune responses were augmented, including IFNγ, TNFα, CD8 T-cell activation and lytic proteins.115 TCR sequencing also showed localization of HPV-specific T-cell clones to the cervical mucosa, which may suggest the mechanism of lesion regression and HPV16 and 18 elimination observed in the clinical trials. Currently, VGX-3100 is in a Phase III clinical trial (REVEAL 1, NCT03185013) in 198 patients with confirmed HPV16/18 positive CIN 2/3. VGX-3100 is also being evaluated in multiple additional clinical trials for other HPV-positive malignancies, including in HIV-positive patients with high-grade anal lesions (NCT03603808), CIN grade 2 or 3 (NCT01304524), cervical HSIL (NCT03721978), and a Phase II trial in vulvar HSIL (NCT03180684). INO-3112 (now MEDI0457) has also been developed as a combination of VGX-3100 and INO9012, which is a plasmid encoding human IL-12 to enhance the pro-inflammatory response to the HPV antigens encoded in VGX-3100.117 The Phase I trial showed safety and elevated antigen-specific T-cell activity in 18/21 evaluable patients. The increased T-cell activity was observed out to 1 year post-therapy. It is currently being investigated in clinical trials in cervical cancer (NCT02172911) and HNSCC (NCT02163057), as well as in combination with the checkpoint inhibitor durvalumab (NCT03162224, NCT03439085).
GX-188e is a DNA vaccine containing plasmid DNA encoding E6 and E7 proteins of HPV16 and HPV18.118 In a Phase I study, women with CIN3 were immunized with GX-188e. In seven of nine patients, there was complete regression and viral clearance by 36 weeks post-immunization.116 A Phase II study in a larger population with HPV16/18 positive CIN3 was conducted to test the efficacy by histopathological results of cervical biopsy and to determine the optimal dose. They found that 52% of the 72 patients enrolled had histopathologic regression of CIN3 by 20 weeks after the first injection, and 67% of the patients showed regression by 36 weeks after the first injection.108 An additional Phase II trial is registered online (NCT02596243), but the status is unknown.
HPV DNA- and Immunogenic Protein-Based DNA Vaccines
VB10.16 (Vaccibody AS, Oslo, Norway) is another novel DNA vaccine that encodes the HPV16 E6/E7 protein, a dimerization entity, and a protein that specifically binds to APCs. An exploratory open-label Phase I/IIa trial (NCT02529930) was presented at the 2019 American Association for Cancer Research annual meeting,119 and showed strongly encouraging safety, tolerability, and immunogenicity results. The study also found upregulation of PD-L1 expression after therapy, which suggests that a combination with checkpoint inhibitors may be beneficial.
Several DNA vaccines in clinical trials are based on variations of the plasmid pNGVLa encoding sequences for HPV16 E7. Two studies have been previously completed: pNGVL4a-CRT/E7, the pNGVL4a plasmid expressing HPV16 E7 linked to calreticulin (CRT), was administered to patients with HPV16-positive CIN 2/3 three times over 8 weeks, and histologic regression to CIN1 occurred in 30% of patients (NCT00988559). Sixty-nine percent of the patients experienced minor AEs associated with vaccination.120 pNGVL4a-Sig/E7(detox)Hsp70, the pNGVL4a plasmid expressing HPV16 E7 linked to SigE7(detox)-heat shock protein 70, was administered three times to patients with HPV16 CIN 2/3, and patients were assessed at week 15 (NCT00121173). Increases in HPV E7-specific T cells were minor, though complete regression of CIN occurred in 33% of patients.121 A Phase I study using pNGVLa expressing HPV16 E6, E7, and L2 proteins linked to CRT has been registered but is not yet recruiting (NCT04131413). These vaccines are also currently being investigated in combination with protein-based therapeutic HPV vaccines as well (see the multi-platform vaccine section).
Another DNA vaccine (SP-SA-E7-1BBL) encoding SA-4-1BBL fused to HPV16 E7 antigen has shown promising results in preclinical studies.122 SA-4-1BBL is an oligomeric form of the ligand 4–1BB receptor of the TNF superfamily, which has been shown to have pro-inflammatory effects when it is engaged. The fusion of SA-4-1BBL to HPV16 E7, administered via gene gun, showed increased anti-tumor efficacy in the TC-1 model compared to controls, and also increased IFNγ-producing E7-specific T cells.
RNA-Based Vaccines
RNA-based vaccines have been pioneered in other malignancies, but few are available for HPV-positive malignancies. The synthetic mRNA technology is new, but recent studies have shown that it is relatively safe due to its non-integrating nature.123 Potential advantages of this technology are that encoded antigens are delivered in a non-HLA-restricted manner, synthetic mRNA is inexpensive and fast to produce, and mRNA is rapidly degraded and cleared. Additionally, mRNA is a natural TLR7/8 ligand.123 Potential disadvantages are that since this technology is new, delivery systems in vivo are still being tested and optimized. Additionally, there is potential for mRNA vaccines to cause toxicity due to the inherent inflammatory activity of mRNA.124 RNA-based vaccines are often paired with other agents, like liposomes, for increased stabilization and additional adjuvant effects, and are able to transit directly into the cytosol to the cell translation machinery that creates the antigen it encodes.69,125 The clinical trials employing RNA-based vaccines are summarized in Table 6.
HPV16 RNA-LPX is a novel synthetic RNA-based vaccine for HPV-positive malignancies. Preclinical studies in mouse models of this mRNA encapsulated in RNA-lipoplex showed that it was selectively taken up by DC in lymphoid compartments.126 Vaccination resulted in complete regression of two HPV-positive murine tumor models (TC-1 and C3) and protection from re-challenge, and showed a combinatorial effect when administered with a checkpoint inhibitor.127 The Phase I HARE-40 trial (NCT03418480) is evaluating the HPV16 mRNA LPX vaccine with and without anti-CD40.
An E7-Trimix RNA vaccine is currently being investigated preclinically.128 The vaccine consists of an mRNA-based vaccine encoding for CD40L, constitutively active TLR4 (caTLR4), and CD70 (Trimix), which was administered together with mRNA encoding HPV16 E7. When injected in the subiliac lymph nodes of C57BL6 mice bearing TC-1 tumors, it decreased tumor burden and increased CD8 T-cell infiltration.
Multi-Platform Vaccines
Other vaccines include fusions of two diverse vaccine platforms, such as utilizing both viral-vectored vaccines and peptide-based vaccines, as well as an endogenously engineered exosome-based vaccine. Multi-platform approaches combine the benefits of diverse avenues of immunization while overcoming some of the hurdles that individual platforms may present when used alone. Accordingly, multiple ongoing clinical trials are using a DNA vaccine prime of HPV16 E7, and an HPV peptide vaccine boost (NCT00788164, NCT03913117, NCT03911076). Preclinical studies for pNGVL4a-sig/E7(detox)/HSP70 DNA HPV vaccine with TA-HPV boost, a vaccinia vectored HPV16 E6/E7, showed potent antigen-specific T-cell responses.120,129,130 Another DNA vaccine, pNGVL4aCRTE6E7L2, with TA-CIN boost, a fusion protein of HPV16 E6, E7, and L2 linked together (a vaccine combination known as PVX-6), showed similar antigen-specific responses. Other heterologous prime-boost strategies include a viral vector prime with a protein vaccine boost, or vice versa. TA-CIN, a protein-based vector, was boosted with TA-HPV, a vaccinia-based vector in patients with AIN;131,132 however, the levels of antigen-specific T cells were not significantly increased compared to TA-HPV alone. In another study, TA-HPV was injected first, then boosted with TA-CIN in patients with VIN.56 Patients showed increased HPV-specific T cells; however, there was no correlation between these responses and clinical regression of VIN. Clinical trials for multi-platform vaccine strategies are listed in Table 7.
Finally, a new type of vaccine is being tested for its potential as an HPV therapeutic. This endogenously engineered exosome-based vaccine is administered by intramuscular inoculation, and consists of a DNA vector expressing Nef fused to HPV16 E7,133 which results in a continuous source of endogenously engineered exosomes. These exosomes are engineered to deliver HPV16 E7 protein upon fusion with Nef exosome-anchoring protein and elicit a strong HPV16 E7 T-cell response.134 This platform resulted in potent antitumor T-cell responses and a reduction in tumor burden in the TC-1 murine model. To our knowledge, this vaccine has not yet been in clinical trials.
Combination Therapies
Rationale for Combining HPV Therapeutic Vaccines with Other Treatment Modalities
Therapeutic vaccines against HPV-associated malignancies commonly target the oncoproteins E6 and E7 to elicit a T-cell response against these proteins. To effectively create a response, the vaccine must deliver the antigens to APCs and activate an HPV antigen-specific response in either CD8+ T cells and/or CD4+ T cells. While there are many different methods for the delivery of antigens to APCs, the response is dependent upon the quality of the antigen-specific T cells produced. If the delivery of the vaccine to the APCs is successful, there are still several factors present either on the tumor itself or in the TME that can act to attenuate the anti-tumor response. Overcoming inhibitory signals in the TME is important to create a robust anti-tumor response, and the combination of therapeutic vaccines with other treatment modalities may augment the efficacy of therapeutic vaccination (Figure 1). Ongoing combination trials are listed in Table 8.
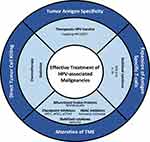 |
Figure 1 Current and potential immunotherapeutic combinations for the treatment of HPV-associated malignancies. |
Therapeutic HPV Vaccines in Combination with PD-1/PD-L1 Axis Inhibitors
Multiple preclinical studies have been published that investigate the effect of combining checkpoint inhibitors with HPV therapeutic vaccination. One of the major inhibitory pathways T cells encounter in the TME is the programmed cell death protein-1 (PD-1)/programmed cell death protein-1 ligand (PD-L1) axis. PD-1 is expressed on the surface of T cells and when ligated by PD-L1 results in an immunosuppressive response leading to reduced activity of those T cells. Multiple monoclonal antibodies (mAb) have been developed and FDA-approved for therapeutic targeting of the PD-1/PD-L1 axis in cancer.
Additionally, HPV E6/7 expression has been correlated with PD-L1 expression. Overexpression of E7 in transfected PC3 (prostate cancer, HPVNEG) cells led to a corresponding overexpression of PD-L1. Conversely, when HPV E7 expression was silenced in the CaSki (cervical cancer, HPV16+) cell line, PD-L1 expression was reduced.135 Increased expression of PD-L1 has been correlated with immune escape and evasion of immunosurveillance,136 and increased expression of PD-L1 in HNSCC has been found to independently correlate with decreased OS.137 Several preclinical studies have evaluated the effect of combining therapeutic HPV vaccination with anti-PD-L1: Treatment of mice harboring PD-1/PD-L1 checkpoint resistant C3 and TC-1 tumors with RNA-LPX E7 vaccination plus anti-PD-L1 led to complete remission in 10 of 15 mice and improved survival by 40% compared to RNA-LPX E7 monotherapy.127 Another study used a DC-targeting fusion protein containing HPV16 E7 in combination with anti-PD-L1 therapy. It showed similar results with a significant decrease in tumor volume and increased survival, with 20% of mice surviving at least 120 days.138 These preclinical studies have provided a strong rationale for combining anti-PD-L1 and anti-PD-1 therapies with HPV therapeutic vaccines.
Clinical Studies
The ISA101 vaccine was recently evaluated in a Phase II study in combination with the anti-PD1 checkpoint inhibitor nivolumab (Bristol-Myers Squibb Co., New York, NY, USA) (NCT02426892).33 The overall response rate was 33%, with a median duration of response of 10.3 months. The ISA101 vaccine alone had shown promising results in CIN but failed to induce responses in patients with advanced cervical cancer. Similarly, nivolumab alone had previously shown a response rate of only 20% in a similar patient population. Recent data from a clinical trial (NCT03444376) using GX188E and pembrolizumab (anti-PD1, Merck) also showed increases in efficacy using the combination versus the checkpoint inhibitor alone.139 There are multiple clinical trials investigating the combination of vaccine and checkpoint inhibition of the PD-1/PD-L1 axis (NCT03439085, NCT03946358, NCT03618953, NCT04001413, NCT04084951, NCT04260126, NCT02291055, NCT03260023, NTC03669718, NCT04369937).
Anecdotally, one patient (out of 22), who was enrolled on a Phase Ib/II trial investigating the efficacy of MEDI0457, a DNA vaccine targeting HPV16/18 E6/E7 with an IL-12 encoding plasmid (NCT02163057), had a complete response (CR) by radiography, with robust induction of antigen-specific PD-1+ CD8+ T cells following four cycles of MEDI0457 treatment. After the patient progressed, nivolumab was added to the therapy, which led to a rapid and durable CR.117 This suggests that before nivolumab treatment the antigen-specific T cells may have been inhibited by the PD-1/PD-L1 axis, and the checkpoint inhibitor allowed these T cells to efficiently eliminate the tumor.
Therapeutic HPV Vaccines in Combination with Other Checkpoint Inhibitors
Another major inhibitory pathway for T cells is caused by ligation of CTLA-4 with B7. This ligation prevents CD28 from interacting with B7, thereby disrupting costimulation. CTLA-4 is upregulated on the plasma membrane of T cells after their activation.136 A counterpart to the co-inhibitory nature of CTLA-4 is 4–1BB, a costimulatory receptor that can activate both T cells and APCs. 4–1BB is a tumor necrosis factor receptor (TNFR) involved in survival signaling in T cells, and activation of the 4–1BB pathway has been shown to promote the production of the inflammatory cytokines IL-12 and IL-6.140
Much like with PD-L1 expression, CTLA-4 expression has been linked with HPV E7 expression. Transfection of the keratinocyte cell line HaCaT with HPV11 E7 resulted in increased CTLA-4 expression in microarray, RT-PCR, and Western blot analyses.141 These results were also reproduced in both SiHa and HeLa cells overexpressing both HPV16 E7 and HPV18 E7. The mechanism of action for E7 control of CTLA-4 expression was determined to be through the reduction of JHDM1B, a histone demethylase that led to reduced H3K36me2 enrichment in the CTLA-4 promoter. Targeting of CTLA-4 in HPV+ tumors may provide additional benefit when combined with HPV therapeutic vaccination.
In addition to classical T-cell activation, a novel subset of highly cytotoxic CD4+ and CD8+ T cells, named ThEO and TcEO, respectively, has been shown to be induced by 4–1BB stimulation.142 A study by Bartkowiak et al combined an E6/E7 peptide vaccine with either 4–1BB agonist or anti-CTLA-4. In the combination of vaccine plus anti-CTLA-4, the TC-1 tumors in two of 10 C57BL/6 mice regressed, but the response was not durable. However, the combination treatment of vaccine plus 4–1BB agonist resulted in tumor regression in all mice, with complete regression in five of eight mice. This anti-tumor effect was driven by an increase in CD8 T cells compared to Tregs (17:1 CD8 to Treg ratio for 4–1BB agonist treatment versus 6.5:1 with vaccine alone).143 Additionally, a conjugated E7 vaccine plus 4–1BBL was able to improve the 90-day survival rate by 50% in mice bearing TC-1 tumors after a single treatment.144 The conjugation of biotinylated E7 to streptavidin-4-1BBL (SA-4-1BBL) also significantly increased the intratumoral CD8+ T effector to Treg ratio, and near-complete eradication of lung tumors was seen in the TC-1 lung metastasis model. The addition of a TLR4 agonist, monophosphoryl lipid A (MPL), produced an even stronger 90-day survival benefit in the TC-1 model. All mice receiving E7 protein plus SA-4-1BBL/MPL achieved complete eradication of tumors during the 90-day watch period after a single treatment.145 Treatment was well tolerated with no increase in autoimmunity, tested by antibodies to single-stranded (ss) DNA, changes in liver enzymes (ALT and AST), or kidney function (BUN and creatinine).
Anti-4-1BB mAb treatment in combination with recombinant IL-2 (rIL-2) and pE7 DNA vaccine resulted in a cure rate of 67% in mice bearing TC-1 tumors compared to 27% in mice treated with control antibody plus rIL-2 and pE7 DNA vaccine.146 The triple combination significantly increased antigen-specific CTL lytic activity and IFNγ production in isolated CD8+ spleen cells. An ongoing clinical trial is investigating the effects of combining ISA101b with utomilumab, which binds to 4–1BB, in advanced oropharyngeal cancer patients (NCT03258008).
Therapeutic HPV Vaccines in Combination with HDAC Inhibitors
The HPV oncoprotein E6 is a known epigenetic regulator through its actions either directly or indirectly on histone acetyltransferases (HATs).147 Thus, targeting HATs through histone deacetylase inhibitors (HDACi) may prove to be a useful therapeutic strategy for HPV-associated malignancies. Vorinostat (Merck KGaA), an FDA-approved pan-HDACi, has been shown to inhibit HPV18 DNA amplification by up to 98.7% in a dose-dependent manner.148 The mechanism of action was determined to be through both the inhibition of S-phase entry, as measured by BrdU incorporation, and by increased Bim expression resulting in increased apoptosis. Increased Bim expression was driven by decreasing EZH2 expression. HDAC-3/4 are also known regulators of Bim expression in a non-EZH2‒dependent manner.149 Classically, HPV infection also results in reduced p53 protein via increased ubiquitination and subsequent degradation.150 Independent of HPV, HDACi have also been shown to increase MHC class I and II, and CD40 expression, and to enhance DNA vaccination immune responses.151,152 HDACs have also been shown to downregulate p53 function and lead to reduced potential to activate the BAX promotor.153 A combination of vaccine plus HDACi could therefore potentially have a strong synergistic effect by targeting multiple pathways in HPV-positive malignancies.
AR-42 (Arno Therapeutics, Flemington, NJ, USA), another pan-HDACi, was shown to significantly increase survival and reduce tumor growth in mice bearing TC-1 tumors when combined with an HPV E7 DNA vaccine.154 Mice treated with the combination of vaccine plus AR-42 had increased CD8+ E7-specific T cells in PBMC (17.78%) compared to DNA vaccine alone (7.89%) and AR-42 alone (0.83%). This result was mirrored in the spleen with more IFNγ+ E7-specific T cells in the combination-treated mice compared to the control-treated mice. In vitro, AR-42–treated TC-1 cells were lysed at a greater rate by CD8+ HPV16 E7-specific T cells and had increased MHC-I expression. Taken together, these studies provide a strong justification for combining therapeutic HPV vaccination with HDAC inhibitors.
Therapeutic HPV Vaccines in Combination with Other Therapeutic Modalities
The combination of a therapeutic vaccine with multiple immunotherapeutic agents is currently being investigated in a trial (NCT04287868) with PDS0101, bintrafusp alfa (EMD Serono and Pfizer), and NHS-IL12 (EMD Serono). This Phase II trial is based on a preclinical study showing additive anti-tumor efficacy when the vaccine was combined with the tumor targeting immunocytokine NHS-IL12 and the dual anti-PD-L1 and TGFβ-trapping agent bintrafusp alfa.44
Current standard of care (carboplatin, paclitaxel, with or without bevacizumab) was combined with ISA101/ISA101b in patients with advanced or recurrent cervical cancer (NCT02128126). Patients were given three doses of vaccine 2 weeks after each dose of standard chemotherapy, and T-cell responses were assessed by IFNγ ELISpot.155 A strong correlation was found between the strength of the immune response and overall survival, highlighting the importance of combining therapeutic HPV vaccines with standard of care, at minimum, to increase survival in patient populations with advanced malignancies.
A recently published study investigated the safety and tolerability of fimaporfin in combination with HPV E7 peptides in healthy volunteers (NCT02947854). Fimaporfin (TPCS2a, PCI Biotech, Oslo, Norway) is a photosensitizer drug being designed to enhance the effects of other drugs in a site-specific, light-directed manner. The combination includes Hiltonol® (Oncovir, Inc., Washington, DC), which is a TLR3 agonist poly-ICLC. The clinical trial found the combination to be safe, and it enhanced the T-cell response to HPV antigens.156 A recently completed Phase I study (NCT02526316) combined the peptide vaccine P6_37-63 with chemotherapy in order to modulate the effect of the vaccine. The results have not yet been presented.
Conclusions
The increase in prophylactic HPV vaccination in young populations will likely decrease the incidence and frequency of HPV-associated malignancies in future decades, especially in HPV16 and HPV18, which were the first types of hrHPV targeted for prophylactic vaccination. Ongoing observational clinical trials are underway to get a more complete picture of how prophylactic HPV vaccination against HPV16/18 using Gardasil® (Merck) and Cervarix® (GlaxoSmithKline) (both directed against hrHPV 16/18) is changing the landscape of HPV types currently causing infection and malignancies (NCT02937155). Prophylactic vaccination across certain populations in developed countries, however, has been uneven due to resistance to vaccination. As of 2016, 49.5% of female and 37.5% of male adolescents aged 13–17 were up to date with the HPV vaccination series.157 Additionally, access to prophylactic vaccination is unequal in developing countries due to prohibitive cost and distribution difficulties. Thus, there will still be a need for therapeutic HPV vaccination as the burden of HPV malignancies will remain high for years to come. Furthermore, recent studies have found HPV expression in non-small cell lung cancer (NSCLC), and the virus appears to play a carcinogenic role for this disease.158 HPV infection was also shown to correlate with higher PD-L1 expression and better clinical response to immune checkpoint inhibition in a metastatic lung adenocarcinoma study presented at the 2020 American Society of Clinical Oncology annual meeting.159 Further clinical studies are needed to evaluate if therapeutic HPV vaccines may be of additional benefit for this patient population.
Therapeutic vaccines for HPV-associated malignancies are a rapidly evolving field with many candidates in clinical trials, and additional candidates in promising preclinical studies. Apparent across the wide variety of ongoing clinical trials is the abundance of treatment methods, doses, types of malignancy being treated, and outcomes used to measure the efficacy of each treatment, thus making comparisons between treatment modalities difficult. Any treatment that is approved for use would be a great advancement to the field; however, more research must be done on potential biomarkers for additional standards by which to measure the outcome. Additional shortcomings such as anti-vector immunity, HLA-restriction of peptides, ease of production, and ease of delivery are all inherent in one or more platforms. The variety of platforms, however, remains important for multiple options for patient care, as each case is treated individually. The development of multiple platforms for simultaneous use is important for addressing the limitations of the respective platforms; for example, peptide-based vaccines can be used as a boost for viral-based vaccines to avoid anti-vector immunity (NCT03911076).
Furthermore, utilizing combination strategies for advanced cases of HPV-positive malignancy has resulted in the proliferation of clinical trials using multiple immunotherapeutics in the past several years. Many clinical trials are now exploring the contribution of checkpoint inhibitor therapy, and the combination of therapeutic HPV vaccine and anti-PD1 therapy has so far been shown to be a potent combination.33 In this study, the overall response rate was 33%, and the median duration of response was 10.3 months. ISA101 alone showed promise for CIN, but did not induce any responses in patients with advanced cervical cancer. Similarly, nivolumab alone was previously shown to have a response rate of only 20% in a similar patient population.33 Other combinations of vaccines and checkpoint inhibitors have shown similar results, such as recent data from the combination of GX-188e and pembrolizumab.139 Thus, combination therapy may address some of the potential shortcomings of therapeutic vaccines, by decreasing the inhibition of antigen-specific T cells, modulating the immunosuppressive TME, and increasing pro-inflammatory cytokines provided by immunocytokines.
Abbreviations
AE, adverse event; AI, artificial intelligence; AIN, anal intraepithelial neoplasia; APC, antigen-presenting cell; CIN, cervical intraepithelial neoplasia; CR, complete response; CRT, calreticulin; CTL, cytotoxic T lymphocytes; DAMP, damage-associated molecular pattern molecules; DC, dendritic cells; DLT, dose-limiting toxicity; E2, early protein 2; FDA, US Food and Drug Administration; GM-CSF, granulocyte-macrophage colony-stimulating factor; HAT, histone acetyltransferases; HDACi, histone deacetylase inhibitor; HLA, human leukocyte antigen; HNSCC, head and neck squamous cell carcinoma; HPV, human papillomavirus; hrHPV, high-risk HPV; HSIL, high-grade squamous intraepithelial lesion; IFN, interferon; IL, interleukin; lrHPV, low-risk HPV; LCMV, lymphocytic choriomeningitis virus; LLO, listeriolysin-O; LSIL, low-grade squamous intraepithelial lesion; mAb, monoclonal antibodies; MAGE-A3, melanoma antigen E; MDSC, myeloid-derived suppressor cells; MHC, major histocompatibility complex; MPL, monophosphoryl lipid A; MVA, modified vaccinia Ankara; NK, natural killer; NSCLC, non-small cell lung cancer; OS, overall survival; PAMP, pathogen-associated molecular pattern molecules; PBMC, peripheral blood mononuclear cells; PD-1, programmed cell death protein-1; PD-L1, programmed cell death protein-1 ligand; r, recombinant; RSV, respiratory syncytial virus; RT-PCR, reverse transcription polymerase chain reaction; SA-4-1BBL, streptavidin-4-1BBL; SFV, Semliki Forest virus; SLP, synthetic long peptides; ss, single-stranded; TA-CIN, tissue antigen-cervical intraepithelial neoplasia; TCR, T-cell receptor; TLR, toll-like receptors; TME, tumor microenvironment; TNF, tumor necrosis factor; TNFR, tumor necrosis factor receptor; Treg, regulatory T cells; TS, Tipapkinogen Sovacivec; VEE, Venezuelan equine encephalitis virus; VEGF, vascular endothelial growth factor; VIN, vulvar intraepithelial neoplasia.
Acknowledgments
The authors thank Debra Weingarten for her editorial assistance in the preparation of this manuscript.
Funding
This study was supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, National Institutes of Health.
Disclosure
The authors report no conflicts of interest in this work.
References
1. de Martel C, Plummer M, Vignat J, Franceschi S. Worldwide burden of cancer attributable to HPV by site, country and HPV type. Int J Cancer. 2017;141(4):664–670. doi:10.1002/ijc.30716
2. Arbyn M, Weiderpass E, Bruni L, et al. Estimates of incidence and mortality of cervical cancer in 2018: a worldwide analysis. Lancet Glob Health. 2020;8(2):
3. Egawa N, Egawa K, Griffin H, Doorbar J. Human papillomaviruses; epithelial tropisms, and the development of neoplasia. Viruses. 2015;7(7):3863–3890. doi:10.3390/v7072802
4. Munoz N, Bosch FX, de Sanjose S, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med. 2003;348(6):518–527. doi:10.1056/NEJMoa021641
5. Doorbar J, Quint W, Banks L, et al. The biology and life-cycle of human papillomaviruses. Vaccine. 2012;30(Suppl 5):F55–F70. doi:10.1016/j.vaccine.2012.06.083
6. Jing Y, Wang T, Chen Z, et al. Phylogeny and polymorphism in the long control regions E6, E7, and L1 of HPV Type 56 in women from southwest China. Mol Med Rep. 2018;17(5):7131–7141. doi:10.3892/mmr.2018.8743
7. Lehoux M, D’Abramo CM, Archambault J. Molecular mechanisms of human papillomavirus-induced carcinogenesis. Public Health Genomics. 2009;12(5–6):268–280. doi:10.1159/000214918
8. White EA, Kramer RE, Tan MJ, Hayes SD, Harper JW, Howley PM. Comprehensive analysis of host cellular interactions with human papillomavirus E6 proteins identifies new E6 binding partners and reflects viral diversity. J Virol. 2012;86(24):13174–13186. doi:10.1128/jvi.02172-12
9. Zhang B, Chen W, The RA. E7 proteins of low- and high-risk human papillomaviruses share the ability to target the pRB family member p130 for degradation. Proc Natl Acad Sci U S A. 2006;103(2):437–442. doi:10.1073/pnas.0510012103
10. Almeida AM, Queiroz JA, Sousa F, Sousa Â. Cervical cancer and HPV infection: ongoing therapeutic research to counteract the action of E6 and E7 oncoproteins. Drug Discov Today. 2019;24(10):2044–2057. doi:10.1016/j.drudis.2019.07.011
11. Hoppe-Seyler K, Bossler F, Braun JA, Herrmann AL, Hoppe-Seyler F. The HPV E6/E7 oncogenes: key factors for viral carcinogenesis and therapeutic targets. Trends Microbiol. 2018;26(2):158–168. doi:10.1016/j.tim.2017.07.007
12. Taghizadeh E, Jahangiri S, Rostami D, et al. Roles of E6 and E7 human papillomavirus proteins in molecular pathogenesis of cervical cancer. Curr Protein Pept Sci. 2019;20(9):926–934. doi:10.2174/1389203720666190618101441
13. Insinga RP, Dasbach EJ, Elbasha EH. Epidemiologic natural history and clinical management of human papillomavirus (HPV) disease: a critical and systematic review of the literature in the development of an HPV dynamic transmission model. BMC Infect Dis. 2009;9:119. doi:10.1186/1471-2334-9-119
14. Moscicki AB, Ma Y, Wibbelsman C, et al. Rate of and risks for regression of cervical intraepithelial neoplasia 2 in adolescents and young women. Obstet Gynecol. 2010;116(6):1373–1380. doi:10.1097/AOG.0b013e3181fe777f
15. Rositch AF, Burke AE, Viscidi RP, Silver MI, Chang K, Gravitt PE. Contributions of recent and past sexual partnerships on incident human papillomavirus detection: acquisition and reactivation in older women. Cancer Res. 2012;72(23):6183–6190. doi:10.1158/0008-5472.can-12-2635
16. McCredie MR, Sharples KJ, Paul C, et al. Natural history of cervical neoplasia and risk of invasive cancer in women with cervical intraepithelial neoplasia 3: a retrospective cohort study. Lancet Oncol. 2008;9(5):425–434. doi:10.1016/s1470-2045(08)70103-7
17. Guan P, Howell-Jones R, Li N, et al. Human papillomavirus types in 115,789 HPV-positive women: a meta-analysis from cervical infection to cancer. Int J Cancer. 2012;131(10):2349–2359. doi:10.1002/ijc.27485
18. de Sanjosé S, Brotons M, Pavón M. The natural history of human papillomavirus infection. Best Pract Res Clin Obstet Gynaecol. 2018;47:2–13. doi:47doi:10.1016/j.bpobgyn.2017.08.015
19. Villa LL, Ault KA, Giuliano AR, et al. Immunologic responses following administration of a vaccine targeting human papillomavirus types 6, 11, 16, and 18. Vaccine. 2006;24(27–28):5571–5583. doi:10.1016/j.vaccine.2006.04.068
20. Day PM, Kines RC, Thompson CD, et al. In vivo mechanisms of vaccine-induced protection against HPV infection. Cell Host Microbe. 2010;8(3):260–270. doi:10.1016/j.chom.2010.08.003
21. Schiller J, Lowy D. Explanations for the high potency of HPV prophylactic vaccines. Vaccine. 2018;36(32Pt A):4768–4773. doi:10.1016/j.vaccine.2017.12.079
22. NSL Y-T, Ito Y, Jha S. High-risk human papillomaviral oncogenes E6 and E7 target key cellular pathways to achieve oncogenesis. Int J Mol Sci. 2018;19(6). doi:10.3390/ijms19061706
23. Araldi RP, Assaf SMR, Carvalho R, et al. Papillomaviruses: a systematic review. Genet Mol Biol. 2017;40:1–21.
24. Singh KP, Verma N, Akhoon BA, et al. Sequence-based approach for rapid identification of cross-clade CD8+ T-cell vaccine candidates from all high-risk HPV strains. 3 Biotech. 2016;6(1):39. doi:10.1007/s13205-015-0352-z
25. Nakagawa M, Gupta SK, Coleman HN, Sellers MA, Banken JA, Greenfield WW. A favorable clinical trend is associated with CD8 T-cell immune responses to the human papillomavirus type 16 e6 antigens in women being studied for abnormal pap smear results. J Low Genit Tract Dis. 2010;14(2):124–129. doi:10.1097/LGT.0b013e3181c6f01e
26. Kim KH, Greenfield WW, Cannon MJ, Coleman HN, Spencer HJ, Nakagawa M. CD4+ T-cell response against human papillomavirus type 16 E6 protein is associated with a favorable clinical trend. Cancer Immunol Immunother. 2012;61(1):63–70. doi:10.1007/s00262-011-1092-5
27. Hung CF, Wu TC, Monie A, Roden R. Antigen-specific immunotherapy of cervical and ovarian cancer. Immunol Rev. 2008;222:43–69. doi:10.1007/s00262-012-1259-8
28. van der Burg SH, Bijker MS, Welters MJ, et al. Improved peptide vaccine strategies, creating synthetic artificial infections to maximize immune efficacy. Adv Drug Deliv Rev. 2006;58(8):916–930. doi:10.1016/j.addr.2005.11.003
29. Melief CJM, van Hall T, Arens R, Ossendorp F, van der Burg SH. Therapeutic cancer vaccines. J Clin Invest. 2015;125(9):3401–3412. doi:10.1172/JCI80009
30. Kenter GG, Welters MJ, Valentijn AR, et al. Phase I immunotherapeutic trial with long peptides spanning the E6 and E7 sequences of high-risk human papillomavirus 16 in end-stage cervical cancer patients shows low toxicity and robust immunogenicity. Clin Cancer Res. 2008;14(1):169–177. doi:10.1158/1078-0432.ccr-07-1881
31. Kenter GG, Welters MJ, Valentijn AR, et al. Vaccination against HPV-16 oncoproteins for vulvar intraepithelial neoplasia. N Engl J Med. 2009;361(19):1838–1847. doi:10.1056/NEJMoa0810097
32. van Poelgeest MI, Welters MJ, van Esch EM, et al. HPV16 synthetic long peptide (HPV16-SLP) vaccination therapy of patients with advanced or recurrent HPV16-induced gynecological carcinoma, a phase II trial. J Transl Med. 2013;11:88. doi:10.1186/1479-5876-11-88
33. Massarelli E, William W, Johnson F, et al. Combining immune checkpoint blockade and tumor-specific vaccine for patients with incurable human papillomavirus 16-related cancer: a phase 2 clinical trial. JAMA Oncol. 2019;5(1):67–73. doi:10.1001/jamaoncol.2018.4051
34. Greenfield WW, Stratton SL, Myrick RS, et al. A phase I dose-escalation clinical trial of a peptide-based human papillomavirus therapeutic vaccine with candida skin test reagent as a novel vaccine adjuvant for treating women with biopsy-proven cervical intraepithelial neoplasia 2/3. Oncoimmunology. 2015;10(4):e1031439. doi:10.1080/2162402x.2015.1031439
35. Coleman HN, Greenfield WW, Stratton SL, et al. Human papillomavirus type 16 viral load is decreased following a therapeutic vaccination. Cancer Immunol Immunother. 2016;65(5):563–573. doi:10.1007/s00262-016-1821-x
36. Wang X, Coleman HN, Nagarajan U, Spencer HJ, Nakagawa M. Candida skin test reagent as a novel adjuvant for a human papillomavirus peptide-based therapeutic vaccine. Vaccine. 2013;31(49):5806–5813. doi:10.1016/j.vaccine.2013.10.014
37. Slingerland M, Speetjens F, Welters M, et al. A phase I study in patients with a human papillomavirus type 16 positive oropharyngeal tumor treated with second generation synthetic long peptide vaccine conjugated to a defined adjuvant. J Clin Oncol. 2016;34(15_suppl):TPS3113–TPS3113. doi:10.1200/JCO.2016.34.15_suppl.TPS3113
38. Zom GG, Willems M, Khan S, et al. Novel TLR2-binding adjuvant induces enhanced T cell responses and tumor eradication. J Immunother Cancer. 2018;6(1):146. doi:10.1186/s40425-018-0455-2
39. Maynard SK, Marshall JD, MacGill RS, et al. Vaccination with synthetic long peptide formulated with CpG in an oil-in-water emulsion induces robust E7-specific CD8 T cell responses and TC-1 tumor eradication. BMC Cancer. 2019;19(1):540. doi:10.1186/s12885-019-5725-y
40. Galliverti G, Tichet M, Domingos-Pereira S, et al. Nanoparticle conjugation of human papillomavirus 16 E7-long peptides enhances therapeutic vaccine efficacy against solid tumors in mice. Cancer Immunol Res. 2018;6(11):1301–1313. doi:10.1158/2326-6066.cir-18-0166
41. Gandhapudi SK, Ward M, Bush JPC, Bedu-Addo F, Conn G, Woodward JG. Antigen priming with enantiospecific cationic lipid nanoparticles induces potent antitumor CTL responses through novel induction of a type I IFN response. J Immunol. 2019;202(12):3524–3536. doi:10.4049/jimmunol.1801634
42. Vasievich EA, Chen W, Huang L. Enantiospecific adjuvant activity of cationic lipid DOTAP in cancer vaccine. Cancer Immunol Immunother. 2011;60(5):629–638. doi:10.1007/s00262-011-0970-1
43. Wood L, Edwards L, Ferris D, et al. A novel enantio-specific cationic lipid R-DOTAP + HPV16 E6 & E7 antigens induces potent antigen-specific CD8+ T cell responses in-vivo in subjects with CIN and high-risk human papillomavirus infection.
44. Smalley Rumfield C, Pellom ST, Morillon YM
45. Karkada M, Quinton T, Blackman R, Mansour M. Tumor inhibition by DepoVax-based cancer vaccine is accompanied by reduced regulatory/suppressor cell proliferation and tumor infiltration. ISRN Oncol. 2013;2013:753427. doi:10.1155/2013/753427
46. Daftarian PM, Mansour M, Pohajdak B, et al. Rejection of large HPV-16 expressing tumors in aged mice by a single immunization of VacciMax® encapsulated CTL/T helper peptides. J Transl Med. 2007;5:26. doi:10.1186/1479-5876-1185-1126
47. Voskens CJ, Sewell D, Hertzano R, et al. Induction of MAGE-A3 and HPV-16 immunity by trojan vaccines in patients with head and neck carcinoma. Head Neck. 2012;34(12):1734–1746. doi:10.1002/hed.22004
48. Zandberg DP, Rollins S, Goloubeva O, et al. A phase I dose escalation trial of MAGE-A3- and HPV16-specific peptide immunomodulatory vaccines in patients with recurrent/metastatic (RM) squamous cell carcinoma of the head and neck (SCCHN). Cancer Immunol Immunother. 2015;64(3):367–379. doi:10.1007/s00262-014-1640-x
49. Reuschenbach M, Pauligk C, Karbach J, et al. A Phase 1/2a study to test the safety and immunogenicity of a p16(INK4a) peptide vaccine in patients with advanced human papillomavirus-associated cancers. Cancer. 2016;122(9):1425–1433. doi:10.1002/cncr.29925
50. Solares AM, Baladron I, Ramos T, et al. Safety and immunogenicity of a human papillomavirus peptide vaccine (CIGB-228) in women with high-grade cervical intraepithelial neoplasia: first-in-human, proof-of-concept trial. ISRN Obstet Gynecol. 2011;2011:292951. doi:10.5402/2011/292951
51. Jabbar B, Rafique S, Salo-Ahen OMH, et al. Antigenic peptide prediction from E6 and E7 oncoproteins of HPV types 16 and 18 for therapeutic vaccine design using immunoinformatics and MD simulation analysis. Front Immunol. 2018;9:3000. doi:10.3389/fimmu.2018.03000.eCollection02018
52. Namvar A, Panahi HA, Agi E, Bolhassani A. Development of HPV(16,18,31,45) E5 and E7 peptides-based vaccines predicted by immunoinformatics tools. Biotechnol Lett. 2020;42(3):403–418. doi:10.3389/fimmu.2018.03000
53. Khan A, Junaid M, Kaushik AC, et al. Computational identification, characterization and validation of potential antigenic peptide vaccines from hrHPVs E6 proteins using immunoinformatics and computational systems biology approaches. PLoS One. 2018;13(5):e0196484. doi:10.1007/s10529-020-02792-6
54. Su JH, Wu A, Scotney E, et al. Immunotherapy for cervical cancer: research status and clinical potential. BioDrugs. 2010;24(2):109–129. doi:10.2165/11532810-000000000-00000
55. van der Burg SH, Kwappenberg KM, O’Neill T, et al. Pre-clinical safety and efficacy of TA-CIN, a recombinant HPV16 L2E6E7 fusion protein vaccine, in homologous and heterologous prime-boost regimens. Vaccine. 2001;19(27):3652–3660. doi:10.1016/s0264-410x(01)00086-x
56. Davidson EJ, Faulkner RL, Sehr P, et al. Effect of TA-CIN (HPV 16 L2E6E7) booster immunisation in vulval intraepithelial neoplasia patients previously vaccinated with TA-HPV (vaccinia virus encoding HPV 16/18 E6E7). Vaccine. 2004;22(21–22):2722–2729. doi:10.1016/j.vaccine.2004.01.049
57. Daayana S, Elkord E, Winters U, et al. Phase II trial of imiquimod and HPV therapeutic vaccination in patients with vulval intraepithelial neoplasia. Br J Cancer. 2010;102(7):1129–1136. doi:10.1038/sj.bjc.6605611
58. Da Silva DM, Skeate JG, Chavez-Juan E, et al. Therapeutic efficacy of a human papillomavirus type 16 E7 bacterial exotoxin fusion protein adjuvanted with CpG or GPI-0100 in a preclinical mouse model for HPV-associated disease. Vaccine. 2019;37(22):2915–2924. doi:10.1016/j.vaccine.2019.04.043
59. Esquerré M, Bouillette-Marussig M, Goubier A, et al. GTL001, a bivalent therapeutic vaccine against human papillomavirus 16 and 18, induces antigen-specific CD8+ T cell responses leading to tumor regression. PLoS One. 2017;12(3):e0174038. doi:10.1371/journal.pone.0174038
60. Esquerré M, Misseri Y, Totain E. Preclinical proof of concept of GTL002, a multivalent candidate for the immunotherapy of human papilloma virus HPV16/18/45/31/33/52-infected women. Eurogin. 2016;123.
61. Chu NR, Wu HB, Wu TC, Boux LJ, Mizzen LA, Siegel MI. Immunotherapy of a human papillomavirus type 16 E7-expressing tumor by administration of fusion protein comprised of mycobacterium bovis BCG Hsp65 and HPV16 E7. Cell Stress Chaperones. 2000;5(5):401–405. doi:10.1379/1466-1268(2000)005<0401:ioahpt>2.0.co;2
62. Palefsky JM, Berry JM, Jay N, et al. A trial of SGN-00101 (HspE7) to treat high-grade anal intraepithelial neoplasia in HIV-positive individuals. AIDS. 2006;20(8):1151–1155. doi:10.1097/01.aids.0000226955.02719.26
63. Einstein MH, Kadish AS, Burk RD, et al. Heat shock fusion protein-based immunotherapy for treatment of cervical intraepithelial neoplasia III. Gynecol Oncol. 2007;106(3):453–460. doi:10.1016/j.ygyno.2007.04.038
64. Roman LD, Wilczynski S, Muderspach LI, et al. A phase II study of Hsp-7 (SGN-00101) in women with high-grade cervical intraepithelial neoplasia. Gynecol Oncol. 2007;106(3):558–566. doi:10.1016/j.ygyno.2007.05.038
65. Granadillo M, Vallespi MG, Batte A, et al. A novel fusion protein-based vaccine comprising a cell penetrating and immunostimulatory peptide linked to human papillomavirus (HPV) type 16 E7 antigen generates potent immunologic and anti-tumor responses in mice. Vaccine. 2011;29(5):920–930. doi:10.1016/j.vaccine.2010.11.083
66. Granadillo M, Batte A, Alfonso AB, et al. Impact on antitumor response using a new adjuvant preparation as a component of a human papillomavirus type 16 therapeutic vaccine candidate. Vaccine. 2019;37(30):3957–3960. doi:10.1016/j.vaccine.2019.05.060
67. Sher YP, Lee C, Liu SY, et al. A therapeutic vaccine targeting HPV E6/E7 with intrinsic toll-like receptor 2 agonist activity induces antitumor immunity. Am J Cancer Res. 2018;8(12):2528–2537. doi:10.1371/journal.pone.0196484
68. Ewer KJ, Lambe T, Rollier CS, et al. Viral vectors as vaccine platforms: from immunogenicity to impact TriVax-HPV: an improved peptide-based therapeutic vaccination strategy against human papillomavirus-induced cancers. Curr Opin Immunol. 2016;41(8):47–54. doi:10.1016/j.coi.2016.05.014
69. Guo C, Manjili MH, Subjeck JR, Sarkar D, Fisher PB, Wang X-Y. Therapeutic cancer vaccines: past, present, and future. Adv Cancer Res. 2013;119:421–475. doi:10.1016/b978-0-12-407190-2.00007-1
70. Borysiewicz LK, Fiander A, Nimako M, et al. A recombinant vaccinia virus encoding human papillomavirus types 16 and 18, E6 and E7 proteins as immunotherapy for cervical cancer. Lancet. 1996;347(9014):1523–1527. doi:10.1016/s0140-6736(96)90674-1
71. Kaufmann AM, Stern PL, Rankin EM, et al. Safety and immunogenicity of TA-HPV, a recombinant vaccinia virus expressing modified human papillomavirus (HPV)-16 and HPV-18 E6 and E7 genes, in women with progressive cervical cancer. Clin Cancer Res. 2002;8(12):3676–3685.
72. DM H, P N, G D, et al. The efficacy and safety of tipapkinogen sovacivec therapeutic HPV vaccine in cervical intraepithelial neoplasia grades 2 and 3: randomized controlled phase II trial with 2.5 years of follow-up. Gynecol Oncol. 2019;153(3). doi::10.1016/j.ygyno.2019.03.250
73. Brun JL, Dalstein V, Leveque J, et al. Regression of High-Grade Cervical Intraepithelial Neoplasia with TG4001 Targeted Immunotherapy. Vol. 204. United States: © 2011 Mosby, Inc; 2011.
74. Le Tourneau C, Delord J, Cassier P, et al. Phase Ib/II trial of TG4001 (tipapkinogene sovacivec), a therapeutic HPV-vaccine, and avelumab in patients with recurrent/metastatic (r/m) HPV-16+ cancers (poster).
75. Gabitzsch ES, Tsang KY, Palena C, et al. The generation and analyses of a novel combination of recombinant adenovirus vaccines targeting three tumor antigens as an immunotherapeutic. Oncotarget. 2015;6(31):31344–31359. doi:10.18632/oncotarget.5181
76. Khan S, Oosterhuis K, Wunderlich K, et al. Development of a replication-deficient adenoviral vector-based vaccine candidate for the interception of HPV16- and HPV18-induced infections and disease. Int J Cancer. 2017;141(2):393–404. doi:10.1002/ijc.30679
77. Çuburu N, Khan S, Thompson CD, et al. Adenovirus vector-based prime-boost vaccination via heterologous routes induces cervicovaginal CD8+ T cell responses against HPV16 oncoproteins. Int J Cancer. 2018;142(7):1467–1479. doi:1410.1002/ijc.31166
78. Mutua G, Anzala O, Luhn K, et al. Safety and immunogenicity of a 2-dose heterologous vaccine regimen with Ad26.ZEBOV and MVA-BN-filo ebola vaccines: 12-month data from a phase 1 randomized clinical trial in Nairobi, Kenya. J Infect Dis. 2019;220(1):57–67. doi:10.1093/infdis/jiz071
79. Hancock G, Blight J, Lopez-Camacho C, et al. A multi-genotype therapeutic human papillomavirus vaccine elicits potent T cell responses to conserved regions of early proteins. Sci Rep. 2019;9(1):18713. doi:10.5402/2011/292951
80. Duncan M, Cranfield M, Torano H, et al. Adenoviruses isolated from wild gorillas are closely related to human species C viruses. Virology. 2013;444(1–2):119–123. doi:10.1016/j.virol.2013.05.041
81. Limbach K, Stefaniak M, Chen P, et al. New gorilla adenovirus vaccine vectors induce potent immune responses and protection in a mouse malaria model. Malar J. 2017;16(1). doi:10.1186/s12936-017-1911-z
82. Johnson T, Rangel D, Graham B, Brough D, Gall J. Genetic vaccine for respiratory syncytial virus provides protection without disease potentiation. Mol Ther. 2014;22(1):196–205. doi:10.1038/mt.2013.142
83. Lundstrom K. Replicon RNA viral vectors as vaccines. Vaccines. 2016;4(4). doi:10.3390/vaccines4040039
84. Daemen T, Riezebos-Brilman A, Regts J, Dontje B, van der Zee A, Wilschut J. Superior therapeutic efficacy of alphavirus-mediated immunization against human papilloma virus type 16 antigens in a murine tumour model: effects of the route of immunization. Antivir Ther. 2004;9(5):733–742.
85. Daemen T, Riezebos-Brilman A, Bungener L, Regts J, Dontje B, Wilschut J. Eradication of established HPV16-transformed tumours after immunisation with recombinant semliki forest virus expressing a fusion protein of E6 and E7. Vaccine. 2003;21(11–12):1082–1088. doi:10.1016/s0264-410x(02)00558-3
86. Schmidt S, ElGazzar A, Bonilla WV, et al. B131: live-attenuated LCMV-based vector for active immunotherapy of HPV16+ cancer.
87. Velders MP, McElhiney S, Cassetti MC, et al. Eradication of established tumors by vaccination with venezuelan equine encephalitis virus replicon particles delivering human papillomavirus 16 E7 RNA. Cancer Res. 2001;61(21):7861–7867.
88. Cassetti MC, McElhiney SP, Shahabi V, et al. Antitumor efficacy of venezuelan equine encephalitis virus replicon particles encoding mutated HPV16 E6 and E7 genes. Vaccine. 2004;22(3–4):520–527. doi:10.1016/j.vaccine.2003.07.003
89. Cheng WF, Hung CF, Hsu KF, et al. Cancer immunotherapy using Sindbis virus replicon particles encoding a VP22-antigen fusion. Hum Gene Ther. 2002;13(4):553–568. doi:10.1089/10430340252809847
90. Dietrich G, Spreng S, Favre D, Viret JF, Guzman CA. Live attenuated bacteria as vectors to deliver plasmid DNA vaccines. Curr Opin Mol Ther. 2003;5(1):10–19.
91. Janeway CA
92. Cossart P, Pizarro-Cerdá J, Lecuit M. Invasion of mammalian cells by Listeria monocytogenes: functional mimicry to subvert cellular functions. Trends Cell Biol. 2003;13(1):23–31. doi:10.1016/s0962-8924(02)00006-5
93. Schnupf P, Portnoy DA. Listeriolysin O: a phagosome-specific lysin. Microbes Infect. 2007;9(10):1176–1187. doi:10.1016/j.micinf.2007.05.005
94. Mollenkopf H, Dietrich G, Kaufmann SH. Intracellular bacteria as targets and carriers for vaccination. Biol Chem. 2001;382(4):521–532. doi:10.1515/bc.2001.066
95. Maciag PC, Radulovic S, Rothman J. The first clinical use of a live-attenuated Listeria monocytogenes vaccine: a phase I safety study of Lm-LLO-E7 in patients with advanced carcinoma of the cervix. Vaccine. 2009;27(30):3975–3983. doi:10.1016/j.vaccine.2009.04.041
96. Basu P, Mehta A, Jain M, et al. A randomized phase 2 study of ADXS11-001 Listeria monocytogenes-Listeriolysin O immunotherapy with or without cisplatin in treatment of advanced cervical cancer. Int J Gynecol Cancer. 2018;28(4):764–772. doi:10.1097/igc.0000000000001235
97. Krupar R, Pathak R, Imai N, et al. Changes in local and peripheral T cell diversity after HPV E7 antigen-expressing Listeria-based immunotherapy (ADXS11-001) prior to robotic surgery for HPV-positive oropharyngeal cancer (abstr 5639).
98. Safran H, Leonard KL, Perez K, et al. Tolerability of ADXS11-001 Lm-LLO Listeria-based immunotherapy with mitomycin, fluorouracil, and radiation for anal cancer. Int J Radiat Oncol Biol Phys. 2018;100(5):1175–1178. doi:10.1016/j.ijrobp.2018.01.004
99. Sacco JJ, Evans M, Harrington KJ, et al. Systemic listeriosis following vaccination with the attenuated Listeria monocytogenes therapeutic vaccine, ADXS11-001. Hum Vaccin Immunother. 2016;12(4):1085–1086. doi:10.1080/21645515.2015.1121338
100. Kawana K, Adachi K, Kojima S, et al. Oral vaccination against HPV E7 for treatment of cervical intraepithelial neoplasia grade 3 (CIN3) elicits E7-specific mucosal immunity in the cervix of CIN3 patients. Vaccine. 2014;32(47):6233–6239. doi:10.1016/j.vaccine.2014.09.020
101. Taghinezhad SS, Mohseni AH, Keyvani H, Razavilar V. Protection against human papillomavirus type 16-induced tumors in C57BL/6 mice by mucosal vaccination with Lactococcus lactis NZ9000 expressing E6 oncoprotein. Microb Pathog. 2019;126:149–156. doi:10.1016/j.micpath.2018.10.043
102. Taghinezhad SS, Mohseni AH, Keyvani H, Razavi MR. Phase 1 safety and immunogenicity trial of recombinant lactococcus lactis expressing human papillomavirus type 16 E6 oncoprotein vaccine. Mol Ther Methods Clin Dev. 2019;15:40–51. doi:10.1016/j.omtm.2019.08.005
103. Poo H, Pyo HM, Lee TY, et al. Oral administration of human papillomavirus type 16 E7 displayed on Lactobacillus casei induces E7-specific antitumor effects in C57/BL6 mice. Int J Cancer. 2006;119(7):1702–1709. doi:10.1002/ijc.22035
104. Lee TY, Kim YH, Lee KS, et al. Human papillomavirus type 16 E6-specific antitumor immunity is induced by oral administration of HPV16 E6-expressing Lactobacillus casei in C57BL/6 mice. Cancer Immunol Immunother. 2010;59(11):1727–1737. doi:10.1007/s00262-010-0903-4
105. Adachi K, Kawana K, Yokoyama T, et al. Oral immunization with a Lactobacillus casei vaccine expressing human papillomavirus (HPV) type 16 E7 is an effective strategy to induce mucosal cytotoxic lymphocytes against HPV16 E7. Vaccine. 2010;28(16):2810–2817. doi:10.1016/j.vaccine.2010.02.005
106. Ribelles P, Benbouziane B, Langella P, Suárez JE, Bermúdez-Humarán LG. Protection against human papillomavirus type 16-induced tumors in mice using non-genetically modified lactic acid bacteria displaying E7 antigen at its surface. Appl Microbiol Biotechnol. 2013;97(3):1231–1239. doi:10.1007/s00253-012-4575-1
107. Cortes-Perez NG, Azevedo V, Alcocer-González JM, et al. Cell-surface display of E7 antigen from human papillomavirus type-16 in Lactococcus lactis and in Lactobacillus plantarum using a new cell-wall anchor from lactobacilli. J Drug Target. 2005;13(2):89–98. doi:10.1080/10611860400024219
108. Choi YJ, Hur SY, Kim TJ, et al. A phase II, prospective, randomized, multicenter, open-label study of GX-188E, an HPV DNA vaccine, in patients with cervical intraepithelial neoplasia 3. Clin Cancer Res. 2020;26(7):1616–1623. doi:10.1158/1078-0432.ccr-19-1513
109. Gurunathan S, Klinman DM, Seder RA. DNA vaccines: immunology, application, and optimization*. Annu Rev Immunol. 2000;18:927–974. doi:10.1146/annurev.immunol.18.1.927
110. Donnelly JJ, Ulmer JB, Liu MA. DNA vaccines. Dev Biol Stand. 1998;95:43–53.
111. Ledwith BJ, Manam S, Troilo PJ, et al. Plasmid DNA vaccines: investigation of integration into host cellular DNA following intramuscular injection in mice. Intervirology. 2000;43(4–6):258–272. doi:10.1159/000053993
112. Barber GN. Cytoplasmic DNA innate immune pathways. Immunol Rev. 2011;243(1):99–108. doi:10.1111/j.1600-065X.2011.01051.x
113. Jorritsma SHT, Gowans EJ, Grubor-Bauk B, Wijesundara DK. Delivery methods to increase cellular uptake and immunogenicity of DNA vaccines. Vaccine. 2016;34(46):5488–5494. doi:10.1016/j.vaccine.2016.09.062
114. Bagarazzi ML, Yan J, Morrow MP, et al. Immunotherapy against HPV16/18 generates potent TH1 and cytotoxic cellular immune responses. Sci Transl Med. 2012;4(155):155ra138. doi:10.1126/scitranslmed.3004414
115. Morrow MP, Kraynyak KA, Sylvester AJ, et al. Augmentation of cellular and humoral immune responses to HPV16 and HPV18 E6 and E7 antigens by VGX-3100. Mol Ther Oncolytics. 2016;3:16025. doi:10.1038/mto.2016.25
116. Trimble CL, Morrow MP, Kraynyak KA, et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: a randomised, double-blind, placebo-controlled phase 2b trial. Lancet. 2015;386(10008):2078–2088. doi:10.1016/s0140-6736(15)00239-1
117. Aggarwal C, Cohen RB, Morrow MP, et al. Immunotherapy targeting HPV16/18 generates potent immune responses in HPV-associated head and neck cancer. Clin Cancer Res. 2019;25(1):110–124. doi:10.1158/1078-0432.ccr-18-1763
118. Kim TJ, Jin HT, Hur SY, et al. Clearance of persistent HPV infection and cervical lesion by therapeutic DNA vaccine in CIN3 patients. Nat Commun. 2014;5:5317. doi:10.1038/ncomms6317
119. Hillemanns P, Petry KU, Woelber L, et al. CT209: safety, efficacy and immunogenicity of VB10.16, a therapeutic DNA vaccine targeting human papillomavirus (HPV) 16 E6 and E7 proteins for high grade cervical intraepithelial neoplasia (CIN 2/3): 6-month data from an exploratory open-label phase I/2a trial.
120. Alvarez RD, Huh WK, Bae S, et al. A pilot study of pNGVL4a-CRT/E7(detox) for the treatment of patients with HPV16+ cervical intraepithelial neoplasia 2/3 (CIN2/3). Gynecol Oncol. 2016;140(2):245–252. doi:10.1016/j.ygyno.2015.11.026
121. Trimble CL, Peng S, Kos F, et al. A phase I trial of a human papillomavirus DNA vaccine for HPV16+ cervical intraepithelial neoplasia 2/3. Clin Cancer Res. 2009;15(1):361–367. doi:10.1158/1078-0432.ccr-08-1725
122. Garza-Morales R, Perez-Trujillo JJ, Martinez-Jaramillo E, et al. A DNA vaccine encoding SA-4-1BBL fused to HPV-16 E7 antigen has prophylactic and therapeutic efficacy in a cervical cancer mouse model. Cancers. 2019;11(1):96. doi:10.3390/cancers11010096
123. Diken M, Kranz LM, Kreiter S, Sahin U. mRNA: a versatile molecule for cancer vaccines. Curr Issues Mol Biol. 2017;22:113–128. doi:10.21775/cimb.022.113
124. Liu M. A comparison of plasmid DNA and mRNA as vaccine technologies. Vaccines. 2019;7:2. doi:10.3390/vaccines7020037
125. Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines — a new era in vaccinology. Nat Rev Drug Discov. 2018;17(4):261–279. doi:10.1038/nrd.2017.243
126. Kranz LM, Diken M, Haas H, et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534(7607):396–401. doi:10.1038/nature18300
127. Grunwitz C, Salomon N, Vascotto F, et al. HPV16 RNA-LPX vaccine mediates complete regression of aggressively growing HPV-positive mouse tumors and establishes protective T cell memory. Oncoimmunology. 2019;8(9):e1629259. doi:10.1080/2162402x.2019.1629259
128. Bialkowski L, van Weijnen A, Van der Jeught K, et al. Intralymphatic mRNA vaccine induces CD8 T-cell responses that inhibit the growth of mucosally located tumours. Sci Rep. 2016;6:22509. doi:10.1038/srep22509
129. Maldonado L, Teague JE, Morrow MP, et al. Intramuscular therapeutic vaccination targeting HPV16 induces T cell responses that localize in mucosal lesions. Sci Transl Med. 2014;6(221):221ra213. doi:10.1126/scitranslmed.3007323
130. Sun YY, Peng S, Han L, et al. Local HPV recombinant vaccinia boost following priming with an HPV DNA vaccine enhances local HPV-specific CD8+ T-cell-mediated tumor control in the genital tract. Clin Cancer Res. 2016;22(3):657–669. doi:10.1158/1078-0432.ccr-15-0234
131. Smyth LJ, Van Poelgeest MI, Davidson EJ, et al. Immunological responses in women with human papillomavirus type 16 (HPV-16)-associated anogenital intraepithelial neoplasia induced by heterologous prime-boost HPV-16 oncogene vaccination. Clin Cancer Res. 2004;10(9):2954–2961. doi:10.1158/1078-0432.ccr-03-0703
132. Fiander AN, Tristram AJ, Davidson EJ, et al. Prime-boost vaccination strategy in women with high-grade, noncervical anogenital intraepithelial neoplasia: clinical results from a multicenter phase II trial. Int J Gynecol Cancer. 2006;16(3):1075–1081. doi:10.1111/j.1525-1438.2006.00598.x
133. Di Bonito P, Accardi L, Galati L, Ferrantelli F, Federico M. Anti-cancer vaccine for HPV-associated neoplasms: focus on a therapeutic HPV vaccine based on a novel tumor antigen delivery method using endogenously engineered exosomes. Cancers. 2019;11(2). doi:10.3390/cancers11020138
134. Di Bonito P, Chiozzini C, Arenaccio C, et al. Antitumor HPV E7-specific CTL activity elicited by in vivo engineered exosomes produced through DNA inoculation. Int J Nanomedicine. 2017;12:4579–4591. doi:10.2147/ijn.s131309
135. Liu C, Lu J, Tian H, et al. Increased expression of PD-L1 by the human papillomavirus 16 E7 oncoprotein inhibits anticancer immunity. Mol Med Rep. 2017;15(3):1063–1070. doi:10.3892/mmr.2017.6102
136. Postow MA, Callahan MK, Wolchok JD. Immune checkpoint blockade in cancer therapy. J Clin Oncol. 2015;33(17):1974–1982. doi:10.1200/jco.2014.59.4358
137. Müller T, Braun M, Dietrich D, et al. PD-L1: a novel prognostic biomarker in head and neck squamous cell carcinoma. Oncotarget. 2017;8(32):52889–52900. doi:10.18632/oncotarget.17547
138. Liu Z, Zhou H, Wang W, Fu YX, Zhu M. A novel dendritic cell targeting HPV16 E7 synthetic vaccine in combination with PD-L1 blockade elicits therapeutic antitumor immunity in mice. Oncoimmunology. 2016;5(6):e1147641. doi:10.1080/2162402x.2016.1147641
139. Hur S, Park JS, Kim Y-M, et al. Efficacy and safety results of pembrolizumab combined with GX-188E, a therapeutic DNA vaccine administration in patients with HPV 16- and/or 18- positive advanced cervical cancer: phase II interim analysis results.
140. Wang C, Lin GH, McPherson AJ, Watts TH. Immune regulation by 4-1BB and 4-1BBL: complexities and challenges. Immunol Rev. 2009;229(1):192–215. doi:10.1111/j.1600-065X.2009.00765.x
141. Zhou Q, Chen L, Song Y, et al. Induction of co-inhibitory molecule CTLA-4 by human papillomavirus E7 protein through downregulation of histone methyltransferase JHDM1B expression. Virology. 2019;538:111–118. doi:10.1016/j.virol.2019.10.001
142. Curran MA, Geiger TL, Montalvo W, et al. Systemic 4-1BB activation induces a novel T cell phenotype driven by high expression of Eomesodermin. J Exp Med. 2013;210(4):743–755. doi:10.1084/jem.20121190
143. Bartkowiak T, Singh S, Yang G, et al. Unique potential of 4-1BB agonist antibody to promote durable regression of HPV+ tumors when combined with an E6/E7 peptide vaccine. Proc Natl Acad Sci U S A. 2015;112(38):E5290–E5299. doi:10.1073/pnas.1514418112
144. Sharma RK, Schabowsky RH, Srivastava AK, et al. 4-1BB ligand as an effective multifunctional immunomodulator and antigen delivery vehicle for the development of therapeutic cancer vaccines. Cancer Res. 2010;70(10):3945–3954. doi:10.1158/0008-5472.can-09-4480
145. Srivastava AK, Yolcu ES, Dinc G, Sharma RK, Shirwan H. SA-4-1BBL/MPL as a novel immune adjuvant platform to combat cancer. OncoImmunology. 2016;5(1):e1064580. doi:10.1080/2162402x.2015.1064580
146. Kim H, Kwon B, Sin JI. Combined stimulation of IL-2 and 4-1BB receptors augments the antitumor activity of E7 DNA vaccines by increasing Ag-specific CTL responses. PLoS One. 2013;8(12):e83765. doi:10.1371/journal.pone.0083765
147. Zhang Y, Dakic A, Chen R, Dai Y, Schlegel R, Direct LX. HPV E6/Myc interactions induce histone modifications, Pol II phosphorylation, and hTERT promoter activation. Oncotarget. 2017;8(56):96323–96339. doi:10.18632/oncotarget.22036
148. Banerjee NS, Moore DW, Broker TR, Chow LT. Vorinostat, a pan-HDAC inhibitor, abrogates productive HPV-18 DNA amplification. Proc Natl Acad Sci U S A. 2018;115(47):E11138–E11147. doi:10.1073/pnas.1801156115
149. Tanimoto A, Takeuchi S, Arai S, et al. Histone deacetylase 3 inhibition overcomes BIM deletion polymorphism-mediated osimertinib resistance in EGFR-mutant lung cancer. Clin Cancer Res. 2017;23(12):3139–3149. doi:10.1158/1078-0432.ccr-16-2271
150. Thomas M, Pim D, Banks L. The role of the E6-p53 interaction in the molecular pathogenesis of HPV. Oncogene. 1999;18(53):7690–7700. doi:10.1038/sj.onc.1202953
151. Magner WJ, Kazim AL, Stewart C, et al. Activation of MHC class I, II, and CD40 gene expression by histone deacetylase inhibitors. J Immunol. 2000;165(12):7017–7024. doi:10.4049/jimmunol.165.12.7017
152. Vanniasinkam T, Ertl H, Tang Q. Trichostatin-A enhances adaptive immune responses to DNA vaccination. J Clin Virol. 2006;36(4):292–297. doi:10.1016/j.jcv.2006.04.009
153. Juan LJ, Shia WJ, Chen MH, et al. Histone deacetylases specifically down-regulate p53-dependent gene activation. J Biol Chem. 2000;275(27):20436–20443. doi:10.1074/jbc.M000202200
154. Lee SY, Huang Z, Kang TH, et al. Histone deacetylase inhibitor AR-42 enhances E7-specific CD8+ T cell-mediated antitumor immunity induced by therapeutic HPV DNA vaccination. J Mol Med (Berl). 2013;91(10):1221–1231. doi:10.1007/s00109-013-1054-9
155. Gerritsen WR, Melief CJ, Welters MJ, et al. Association of T cell responses after vaccination with HPV16 long peptides for late stage cervical cancer with prolonged survival. J Clin Oncol. 2017;35(15_suppl):5525. doi:10.1200/JCO.2017.35.15_suppl.5525
156. Selbo PK, Janetzki S, Welters MJP, et al. Phase I clinical study for validation of fimaporfin-based photochemical internalisation: a novel technology for enhancing cellular immune responses important for therapeutic effect of peptide-and protein-based vaccines. Poster 109P presented at: European Society for Medical Oncology (ESMO) 2019; September 27-October 1, 2019; Barcelona, Spain. Ann Oncol. 2019;30(Supp11):xi40–xi41. doi:10.1093/annonc/mdz451.018
157. Walker TY, Elam-Evans LD, Singleton JA, et al. National, regional, state, and selected local area vaccination coverage among adolescents aged 13–17 years – United States, 2016. MMWR Morb Mortal Wkly Rep. 2017;66(33):874–882. doi:10.15585/mmwr.mm6633a2
158. Hasegawa Y, Ando M, Kubo A, et al. Human papilloma virus in non-small cell lung cancer in never smokers: a systematic review of the literature. Lung Cancer. 2014;83(1):8–13. doi:10.1016/j.lungcan.2013.10.002
159. Rojas Puentes LL, Ruiz-Patiño A, Mayorga Gonzalez DC, et al. Human papillomavirus infection in metastatic lung adenocarcinoma is associated with better outcomes with immune checkpoint inhibitor treatment. J Clin Oncol. 2020;38(15_suppl):e21569–e21569. doi:10.1200/JCO.2020.38.15_suppl.e21569
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
