Back to Journals » Cancer Management and Research » Volume 15
The Treatment of Diffuse Large B-Cell Lymphoma (Triple Expression) Involving the Breast, Spleen, and Bone in a Male Patient with Viral Hepatitis B: A Rare Case Report
Authors Zhou W , Gao Q , Wang L, Li W, He C, Li Y, Feng L, Liu W, Liu L, Wang Y
Received 17 July 2023
Accepted for publication 30 August 2023
Published 6 September 2023 Volume 2023:15 Pages 983—988
DOI https://doi.org/10.2147/CMAR.S427217
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Harikrishna Nakshatri
Weiling Zhou,1 Qian Gao,1 Lianjing Wang,2 Weijing Li,2 Cuiying He,2 Yanting Li,3 Lei Feng,1 Wei Liu,2 Lihong Liu,2 Yuan Wang1
1Department of Endocrine and Metabolic Diseases, The Fourth Hospital of Hebei Medical University, Shijiazhuang, HeBei Province, 050000, People’s Republic of China; 2Department of Hematology, The Fourth Hospital of Hebei Medical University, Shijiazhuang, HeBei Province, 050000, People’s Republic of China; 3Department of Glandular Surgery, The Second Hospital of Hebei Medical University, Shijiazhuang, HeBei Province, 050000, People’s Republic of China
Correspondence: Lihong Liu, Department of Hematology, The Fourth Hospital of Hebei Medical University, Shijiazhuang, HeBei Province, 050000, People’s Republic of China, Email [email protected] Yuan Wang, Department of Endocrine and Metabolic Diseases, The Fourth Hospital of Hebei Medical University, Shijiazhuang, HeBei Province, 050000, People’s Republic of China, Email [email protected]
Background: Diffuse large B-cell lymphoma (DLBCL) involving the breast, spleen, and bone in a male patient with hepatitis B virus (HBV) infection is extremely rare in clinical practice.
Case Presentation: We report a case of DLBCL involving the breast, spleen, and bone (triple expression of Bcl-2+, Bcl-6+, and 70% positive C-mcy) in a male patient with HBV admitted to our hospital. The patient was treated with EPOCH× 4, lenalidomide+EPOCH× 2 chemotherapy, intermittent methotrexate intrathecal injections to prevent central invasion, and autologous stem cell transplantation (ASCT). The patient is currently in complete remission, and the follow-up time was 43 months.
Conclusion: A patient with DLBCL involving the breast, spleen, and bone can be treated with a combination of multiple regimens. If the patient’s economic conditions permit it, ASCT can be considered.
Keywords: breast, viral hepatitis B, diffuse large B-cell lymphoma, triple expression, autologous stem cell transplantation
Introduction
The prevalence of any breast malignancy is very low in the male population, and male breast diffuse large B-cell lymphoma (DLBCL) is even rarer. Male breast cancer accounts for about 1% of all breast malignancies.1 Moreover, primary DLBCL of the breast accounts for only about 0.5% of breast tumors;2–4 this may be attributed to the fact that breast tissue contains fewer lymph nodes. Correspondingly, there is no effective unified treatment program.
As far as we know, there have been no large-scale reports and few reports about male breast DLBCL, which have mostly been reported as individual cases. Only a few male breast DLBCL cases can be found worldwide. We report the case of a male patient with chronic hepatitis B virus (HBV) infection complicated with DLBCL (triple expression: Bcl-2+, Bcl-6+, and 70% positive C-myc, involving the breast, spleen, and bone).
Case Presentation
In June 2019, a 45-year-old man found a mass about the size of an egg in the left breast without obvious pain. He had been diagnosed with HBV 20 years previously. The patient’s disease course and treatment history is summarized in Figure 1. Ultrasonography indicated the following: an extremely hypoechoic nodule with a size of 3.3×2.9 × 1.2cm was observed in the subcutaneous fat layer in the outer upper quadrant of the left breast, showing a large lobulated regular shape with a clear boundary, an uneven internal echo, an enhanced peripheral soft tissue echo, and no abnormal axillary lymph nodes (Figure 2). In accordance with the examination results, a breast biopsy was performed. The pathology indicated a malignant tumor, and immunohistochemistry was recommended to exclude lymphoma (Figure 3). The immunohistochemistry results indicated the following: CD20+, Ki-67 (80% positive cells), Bcl-2+, Bcl-6+, C-myc (70% positive), CD3−, AE1/AE3−, CD2−, CD30−, CD1−, CD5−, and MUM1−. The in situ hybridization results were EBER−, and the puncture tissue was considered to be non-Hodgkin’s lymphoma, which is consistent with DLBCL. Further gene fluorescence in situ hybridization (FISH) detection showed no recombination of Bcl-2, Bcl-6, and C-myc. During the first admission, there were no obvious abnormalities in the blood routine, coagulation function, β2 microglobulin, sedimentation rate, liver and kidney function and bone marrow aspiration. Lactate dehydrogenase (LDH) was 400 U/L↑ and hepatitis B DNA quantitative was 5.27*105 IU/mL↑. PET/CT (Figure 4) showed soft tissue nodules in the upper quadrant of the left breast, abnormal high glucose metabolism in the corresponding parts on PET (SUVmax 9.9), multiple circular low density in the spleen (SUVmax 15.8), mass soft tissue density in the right posterior chest wall, and the cortex of the adjacent posterior ribs of the right 10th was blurred, seemingly discontinuous. Uneven density in the medullary cavity (SUVmax 15.1) with the possibility of multiple site invasion of lymphoma, was considered high. The patient had a history of chronic viral hepatitis B and no history of other chronic diseases or malignant tumors. Due to active virus replication, the patient was treated with EPOCH (vindesine 1 mg + pirirubicin hydrochloride 19.4 mg + etoposide 0.097 g ×4d + cyclophosphamide 1.45 g × 1d) chemotherapy based on liver protection and antiviral replication. He was admitted to hospital for the second time in September 2019, when a gene mutation test showed CD70 mutation, indicating a poor prognosis, indicating first-line autotransplantation; lenalidomide + EPOCH chemotherapy was given to the patient. Considering the high risk of central invasion of the patient’s primary disease, a large methotrexate dose was given to prevent central invasion after chemotherapy. Fully explain Autologous stem cell transplantation (ASCT) related side effects to patients and their families, including high-dose chemotherapy may cause damage to important organs, gastrointestinal reactions, bone marrow suppression, bleeding, infection, hemolytic reaction, etc., and do a good job in psychological counseling and emotional stability of patients. ASCT carried out successful under full assessment and preparation. Although various complications such as bone marrow suppression and infection occurred in this patient, he was obviously alleviated after symptomatic treatment. After chemotherapy with EPOCH×4, lenalidomide + EPOCH×2, intermittent methotrexate intrathecal injections to prevent central invasion and the ASCT, the patient was in complete remission. The latest review was in March 2023 (Figure 5), when the breast ultrasound showed no obvious space occupation, PET/CT showed small flakes with slightly low density in the splenic parenchyma, the margin was not clear, and no obvious abnormal glucose hypermetabolism was observed on PET, which was considered to be the change after the lymphoma treatment, the disappearance of the breast and bone tumors. The patient and their families were satisfied with our overall treatment.
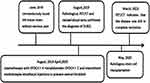 |
Figure 1 Summary of disease course and treatment situation. |
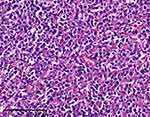 |
Figure 3 HE staining showed diffuse, relatively uniform, medium sized cells with obvious atypia, considering to be diffuse large B-cell lymphoma. The ruler in indicates 20 μm. |
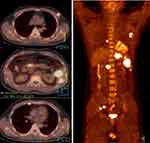 |
Figure 4 PET/CT showed diffuse large B-cell lymphoma involving breast, spleen, and bone. |
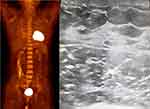 |
Figure 5 The ultrasound and PET/CT indicated complete remission of the DLBCL. |
DLBCL of the breast is relatively rare, and DLBCL of the male breast is even rarer. Only one case was found in lymphoma patients in our hospital over the past 15 years.
Discussion
DLBCL is made up of mature B-cells and is one of the most common types of non-Hodgkin’s lymphoma (NHL). DLBCL is a potentially curable disease that usually responds to systemic chemotherapy and synchronous or adjuvant radiotherapy.5 Many patients are misdiagnosed with breast cancer and receive surgical treatment. However, one study showed that surgical treatment did not improve patient survival.2 Our rare case was a male patient with hepatitis B involving breast, spleen, and bone DLBCL. We treated the patient with EPOCH×4, lenalidomide +EPOCH×2 chemotherapy and ASCT. The patient is in complete remission and has been followed up for 43 months.
ASCT refers to the harvesting of autologous stem cells into foreign blood and the transplantation of the purified stem cells back into the body. ASCT has shown strong advantages in the treatment of recurrent lymphoma or refractory lymphoma and could improve the survival time of patients.6–8 Due to the small number of cases, there is no exact and effective treatment plan for breast DLBCL at present. This case achieved an ideal therapeutic effect through chemotherapy and ASCT, which could provide a reference for the formulation of future treatment plans.
HBV, tuberculosis, and acquired immune deficiency syndrome are the three most common infectious diseases in the world. In 2015, there were 257 million chronic HBV carriers worldwide, of whom 887,000 died from HBV-related complications, mainly cirrhosis and hepatocellular carcinoma.9 A large Italian study showed an increased risk of DLBCL in HBsAg-positive patients (hazard ratio 1.74; 95% CI 1.45–2.09).10 However, the mechanism by which HBV may induce NHL is not clear and may be related to chronic B-cell activation that promotes DNA damage and transformation into NHL,11 explaining the clinical existence of many hepatitis patients with lymphoma. In addition, the presence of immune and inflammatory cells helps regulate tumor growth and invasion in hematologic malignancies and DLBCL. DLBCL cells interact with mature stromal cells and blood vessels to confer tumor protection and suppress the immune response while providing nutrients and oxygen supply.12 Therefore, tumor microenvironment analysis is an important aspect of assessing DLBCL progression.
Limitations
This is a single case report, and further studies are needed to determine the generalizability of the treatment approach used in this case.
Conclusion
In this case, a male patient with DLBCL involving the breast, spleen, and bone, who was treated with a combination of multiple regimens, showed a good response without signs of recurrence after 43 months of follow-up. Therefore, if the patient’s economic conditions are relatively good, this treatment plan combined with ASCT can be considered. As this is only an individual case, more cases are needed to verify this result.
Ethics Statement
The study was reviewed and approved by the Ethic Committee of the The Fourth Hospital of Hebei Medical University (approval number, 2022KY384).
Informed Consent Statement
Informed consent was obtained from the patient involved in this study.
Author Contributions
All authors made a significant contribution to the work reported, whether in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This research was supported by the Department of Science and Technology of Hebei Province of China (No. 20221232, No. 20230803 and No. 20221266).
Disclosure
The authors disclose no conflicts of interest related to this work.
References
1. Abdelwahab Yousef AJ. Male breast cancer: epidemiology and risk factors. Semin Oncol. 2017;44(4):267–272. doi:10.1053/j.seminoncol.2017.11.002
2. Zhou W, Yu G, Liu L, et al. Primary diffuse large B-cell lymphoma of the breast: a retrospective study of outcomes and insulin resistance. Saudi Med J. 2023;44(1):38–44. doi:10.15537/smj.2023.44.1.20220677
3. Wiseman C, Liao KT. Primary lymphoma of the breast. Cancer. 1972;29(6):1705–1712. doi:10.1002/1097-0142(197206)29:6<1705::AID-CNCR2820290640>3.0.CO;2-I
4. Sabaté JM, Gómez A, Torrubia S, et al. Lymphoma of the breast: clinical and radiologic features with pathologic correlation in 28 patients. Breast J. 2002;8(5):294–304. doi:10.1046/j.1524-4741.2002.08509.x
5. Cancer stat-facts non-Hodgkin’s lymphoma; 2020. Available from: https://seer.cancer.gov/statfacts/html/nhl.html.
6. Cortez AJ, Dulley FL, Saboya R, et al. Autologous hematopoietic stem cell transplantation in classical Hodgkin’s lymphoma. Rev Bras Hematol Hemoter. 2011;33(1):10–14. doi:10.5581/1516-8484.20110007
7. Sun L, Li S, El-Jawahri A, et al. Autologous stem cell transplantation in elderly lymphoma patients in their 70s: outcomes and analysis. Oncologist. 2018;23(5):624–630. doi:10.1634/theoncologist.2017-0499
8. Tardy MP, Gastaud L, Ojeda-Uribe M, et al. Z-BeEAM (Ibritumomab tiuxetan, Bendamustine, Etoposide, Cytarabine, Melphalan) before autologous stem cell transplantation is safe and efficient for refractory large B-cell lymphoma. Exp Hematol Oncol. 2015;4(1):18. doi:10.1186/s40164-015-0013-2
9. World Health Organization. Global hepatitis report, 2017. World Health Organization; 2017. Available from: https://www.afro.who.int/publications/global-hepatitis-report-2017.
10. Engels EA, Cho ER, Jee SH. Hepatitis B virus infection and risk of non-Hodgkin lymphoma in South Korea: a cohort study. Lancet Oncol. 2010;11(9):827–834. doi:10.1016/S1470-2045(10)70167-4
11. Marcucci F, Mele A. Hepatitis viruses and non-Hodgkin lymphoma: epidemiology, mechanisms of tumorigenesis, and therapeutic opportunities. Blood. 2011;117(6):1792–1798. doi:10.1182/blood-2010-06-275818
12. Solimando AG, Annese T, Tamma R, et al. New insights into diffuse large B-cell lymphoma pathobiology. Cancers. 2020;12(7):1869. doi:10.3390/cancers12071869
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

