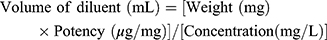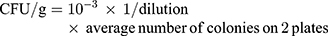Back to Journals » International Journal of Nanomedicine » Volume 15
The Role of Silver Nanoparticles in a Treatment Approach for Multidrug-Resistant Salmonella Species Isolates
Authors Farouk MM , El-Molla A, Salib FA , Soliman YA , Shaalan M
Received 7 July 2020
Accepted for publication 31 August 2020
Published 23 September 2020 Volume 2020:15 Pages 6993—7011
DOI https://doi.org/10.2147/IJN.S270204
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Anderson Oliveira Lobo
Manar M Farouk,1 Amal El-Molla,1 Fayez A Salib,1 Yousef A Soliman,2 Mohamed Shaalan3
1Department of Internal Medicine and Infectious Diseases, Faculty of Veterinary Medicine, Cairo University, Giza 12211, Egypt; 2Central Laboratory for Evaluation of Veterinary Biologics (CLEVB), Cairo, Egypt; 3Department of Pathology, Faculty of Veterinary Medicine, Cairo University, Giza 12211, Egypt
Correspondence: Manar M Farouk
Department of Internal Medicine and Infectious Diseases, Faculty of Veterinary Medicine, Cairo University, Giza 12211, Egypt
Tel +02 01067903499
Email [email protected]
Purpose: The main objective of this study is to investigate the antibacterial activity of silver nanoparticles (AgNPs) against multidrug-resistant Salmonella isolates recovered from diarrheic sheep and goats
Methods: This study used chemical reduction synthesis of AgNPs to evaluate their antimicrobial effects by estimation of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) for each isolate using the microplate dilution method and tetrazolium salt reduction test to detect the viability percentage. In vivo treatment efficacy was assessed in mice by determining the viable count of Salmonella Enteritidis recovered from feces and by hematologic, biochemical and histopathologic examinations to confirm that use of AgNPs has no toxic or pathologic effects and to evaluate its ability in tissue regeneration following treatment.
Results: All recovered strains were identified as MDR with a prevalence of 4% and 3.6% in sheep and goats, respectively. The results of TEM, DLS, Zeta potential, and FTIR revealed typical characteristics of the synthesized AgNPs. Silver nanoparticles showed antibacterial activity against all recovered strains with MIC of ≤ 0.02– 0.313 μg/mL (mean average 0.085± 0.126 μg/mL) and MBC of 0.078– 1.250 μg/mL (average 0.508± 0.315 μg/mL). In vivo efficacy of AgNPs was observed by a reduction in the number of viable S. Enteritidis recovered from feces in an S. Enteritidis infected mouse model, with complete shedding stopping between treatment days 4 and 6. Hematologic, serum biochemical, and histopathologic analyses proved the ability of AgNPs to suppress inflammatory reaction caused by S. Enteritidis infection.
Conclusion: The study proved the effective ability of AgNPs to fight MDR Salmonella spp. in vitro and in vivo without adverse effects.
Keywords: multidrug resistance, silver nanoparticles, Salmonella, mice, minimum inhibitory concentration, minimum bactericidal concentration
Introduction
Sheep and goats are well known for their potential as livestock to transform feed into beneficial products such as fiber, milk, and meat.1 Salmonellosis is an infectious disease in animals that causes severe losses of livestock and serious public health problems worldwide.2 Salmonella is a key etiologic agent of infectious diarrhea and a major zoonotic pathogen globally.3 Antimicrobial agents are valuable tools for infectious diseases’ treatment and keeping animals healthy and productive. Simultaneously, antimicrobial-resistant pathogens pose serious and costly problems for animal health.4 The main reason for development of multidrug resistance among Salmonella spp. in humans is the misuse of antibiotics resulting from a lack of uniform policy and disregard for hospital infection control standards.5 This issue has sparked vigorous debate on the possibility that the use of antibiotics in animal feed will lead to a similar crisis;6 for example, antibiotic use as a growth stimulator is usually thought to promote the emergence and distribution of antibiotic-resistant strains in animal farms.6,7 However, veterinary data on the prevalence of antimicrobial resistance in animal pathogens are scarce, especially in developing countries, such as Egypt, where this study was conducted.8 Because of the threat of Salmonella spp. infections in Egypt, it is important to study the resistance mechanisms to antimicrobials in Salmonella spp. and their effect on livestock.9
Infectious diseases represent one of the biggest health problems worldwide. Although, numerous antibiotics are available for commercial use, they mostly lack potency against newly evolved multidrug-resistant (MDR) pathogens, which gives rise to massive doses of antibiotics and the need to continuously develop new antibiotics. Recently the potency of metallic nanoparticles has been proven to control MDR pathogens, both independently and jointly with existing available antibiotics.10 Specifically, the promising properties of silver nanoparticles (AgNPs) have been shown to be suitable for multiple biomedical purposes, including their impressive ability for the formulation of novel antimicrobials.11 Recent studies regarding the antibacterial activities of AgNPs for MDR ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter sp.) showed impressive results for four of six pathogens in this group of bacteria.12 Nanosilver is one of the most commonly used nanomaterials, and its particle size ranges from 1–100 nm.13,14 The antimicrobial properties of silver (Ag+) have been well recognized for decades.15 Silver nanoparticles (AgNPs) have received considerable attention due to their biological, physical, and chemical properties based on their catalytic activity and bactericidal effects.16 One of the strengths of AgNPs versus traditional antibiotics is their interaction with various bacterial cellular reactions such that resistance to them is becoming less prevalent.17 The exact antibacterial modes of action for AgNPs are unclear.18 Nevertheless, various researchers have proposed several modes of action,17,19 including interference with the bacterial cell wall, production of reactive oxygen species, generation of Ag+ ions, and interaction with DNA.20 The key antibacterial mechanisms of AgNPs are either by liberation of Ag+ ions or by the intracellular deposition of nanoparticles.21 Moreover, the bactericidal activity is primarily due to inhibition of the synthesis of nucleic acid, protein, and the cell wall, as well as microbial cell disruption.22–24 Despite the steadily increasing presence of Ag-containing products in the market22 and extensive reports on the antimicrobial activity of AgNPs,25 insufficient data are currently available about the use of AgNPs as in vivo therapeutic agents.26 Different treatment methods have been proposed to eradicate salmonellosis; however, a critical consideration is that the use of antibiotics poses a risk of serious and costly animal health disorders and also leads to the rapid development of resistance. The emergence of antibiotic microbial resistance is one of the major challenges today, and there is an increasingly urgent need to promote safe approaches for the containment of MDR salmonellosis, such as the investigation of antimicrobial alternatives. According to the best of our knowledge, most of the previous studies did not investigate the antibacterial effect of AgNPs against the antibiotic-resistant Salmonella strains clinically isolated from small ruminants, especially the serovar Salmonella Enteritidis. Therefore, this study aims to determine the occurrence of MDR salmonellosis in diarrheic goats and sheep and to demonstrate antimicrobial efficacy of synthesized AgNPs both in vivo and in vitro for salmonellosis control. The present study was also designed to synthesize AgNPs through chemical co-reduction method with trisodium citrate and sodium borohydride. The synthesized AgNPs was coated with PVP and carrying a negative charge. AgNPs were characterized by transmission electron microscopy (TEM), DLS and zeta potential analysis, and Fourier transform infrared spectroscopy (FTIR) analysis.
Materials and Methods
Salmonella Species Isolation and Identification
A total number of 325 and 193 fecal swabs were collected from diarrheic sheep and goats, respectively, from different localities in Giza Governorate, Egypt. The swabs were microbiologically examined for Salmonella spp. isolation following ISO-6579 standards27 and biochemically identified according to Quin et al.28 Then the isolates were serotyped following the Kauffman-White scheme using a commercially available antisera kit (SIFIN),29 as mentioned in Supplementary Appendix 1.
Molecular Confirmation of Salmonella Species Isolates
To extract DNA from the bacterial isolates, the boiling method by Wani et al30 was used, and molecular verification was achieved by Salmonella spp. invA gene amplification.31 The primer sequence and thermal profile used is listed in Table 1.
 |
Table 1 Oligonucleotide Primer Sequence and Thermal Profile for invA Gene |
Antimicrobial Susceptibility Test
The minimum inhibitory concentration (MIC) values for 10 antimicrobials were evaluated using broth microdilution assay in 96 well microtiter plates according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines.32 Briefly, 100 μL of sterile Mueller Hinton broth was introduced into each well of a 96-well microtiter plate. Two rows were used as controls, a sterility control row containing 200 μL of sterile Mueller Hinton broth was used as a sterility indicator, and a culture control row containing 100 μL of sterile Mueller Hinton broth and 100 μL of subculture broth was used to verify the broth’s ability to support microbial growth. To determine MIC for antimicrobial agents, 100 μL of working solution of each agent at a specified concentration was introduced into the top row of the 96-well microtiter plate, and double-fold serial dilutions were performed according to the specified test range for each agent. Thereafter, 100 μL of subculture broth adjusted to the 0.5 McFarland standard was introduced to all wells except in the sterility control row, and all antimicrobials were tested in triplicate to ensure reproducibility of results. After confirming reproducibility, each plate was sealed with a sterile adhesive film and incubated at 37°C for 24 hours. Results were recorded when there was sufficient turbidity in the culture control row and no growth or turbidity in the sterility control row. The growth in each well was compared with that in the culture control, and the MIC was determined as the lowest concentration of the antimicrobial that completely inhibits growth. The results were interpreted according to Clinical and Laboratory Standards Institute (CLSI) and EUCAST guidelines.33,34 The stock solution for each powdered antimicrobial agent was prepared, and the amount of weighed antimicrobial powder and the volume of diluent were calculated from the following equation:
The weighed antimicrobial powders were dissolved in 1 mL of dimethyl sulfoxide (DMSO) solvent and diluted to the final stock concentration with sterile distilled water. Working solutions were prepared according to the test range for each antimicrobial, which was chosen according to World Health Organization guidelines,35 as listed in Table 2.
 |
Table 2 Details of the Tested Antimicrobials and Their Antimicrobial Phenotypic Sensitivity Patterns, Results |
Multiple Antimicrobial Resistance Index (MAR) Determination
The multiple antimicrobial resistance index (MAR) of each isolate was estimated using the Paul et al36 formula, in which the number of antimicrobials to which isolates were resistant was divided by the total number (10) of antimicrobials to which the test isolate has been evaluated for susceptibility. The interpretation of MAR results was performed according to Christopher et al37 as follows: isolates with an index <0.3 are considered to have narrow drug resistance (NDR); ≥0.3–0.7 are MDR, 0.8–0.9 are extensively drug-resistant (XDR); and pan drug resistant (PDR) isolates are resistant to all agents.
Synthesis of Silver Nanoparticles (AgNPs)
AgNPs synthesis was applied through a chemical reduction method according to El Mahdy et al.38 Briefly, a reducing solution of sodium borohydride (NaBH4) (research Lab®) was prepared by dissolving 1.89 g of NaBH4 in 50 mL distilled water and then refrigerated. The Ag source solution was prepared by dissolving 0.272 g of silver nitrate (AgNO3) in 344 mL of distilled water, followed by stirring for 15 minutes with a magnetic stirrer. In 48 mL of distilled water, a stabilizing mixture of 0.504 g of polyvinyl pyrrolidone (PVP) (CDH®) and 2.912 g of trisodium citrate (ADWIC®) were dissolved, stirred for 15 minutes, and mixed with the prepared AgNO3 solution. Then, 8 mL of the fresh, cold-reducing NaBH4 solution was added to the mixture followed by stirring for 30 minutes. After that step, the reduction reaction was investigated for when the color of solution changed from colorless into a dark yellow or brown color. Finally, the prepared stock solution was kept in a dark glass bottle away from direct sunlight at 4°C. The yield of each step in the synthesis process is mentioned in Supplementary Appendix 2.
Silver Nanoparticles Characterization
Determination of the morphology and size of the integrated nanoparticles was made by using transmission electron microscopy (TEM) (JEM-1400-JEOL), which operated at an accelerating voltage of 80 kV. Briefly, one drop of AgNPs solution was deposited on the carbon-coated grid and left to evaporate at room temperature, thus forming a monolayer. The particle size was estimated using Image SP_ Viewer software. In addition, distribution of particle size and zeta potential analysis were measured at 25°C using a nano-zetasizer (Zetasizer Nano ZS, Malvern Paralytical) where deionized water was used as the dispersant. Briefly, the sample was diluted 1:10 and then exposed to ultrasonication for 5 minutes for estimation of particle size distribution and it was diluted 1:1000 and three measurements were conducted for zeta potential analysis. Fourier transform infrared spectroscopy (FTIR) analysis was performed by FTIR spectroscopy (Hitachi Ltd., Tokyo, Japan) and recorded AgNPs in the frequency range of 4000–400 cm−1 to identify the different functional groups in molecules responsible for the silver ion reduction and PVP-capping of AgNPs.
Determination of Silver Nanoparticles Concentration
An advanced microwave digestion system was used for digestion of the AgNPs sample, and the Ag+ concentration was determined by using elemental concentrations of the nanoparticle solution through inductively coupled plasma atomic emission spectroscopy (ICP-AES) (Themo Sci, Model: iCAP6000 series) in which argon gas was used for excitation of the element atom and the blank value for the element was deduced from the sample value.
In vitro Antimicrobial Activity Assay
Bacterial Growth Inhibition
One purified colony per each isolate was inoculated in brain heart infusion broth (Oxoid) and incubated at 37°C for 4 hours in a shaker incubator (225 rpm). Bacterial culture dilutions at a certain concentration of 1×106 colony forming unit (CFU)/mL in brain heart infusion broth were prepared and confirmed by plate counting. One milliliter each of AgNPs solution and each diluted broth of the Salmonella spp. isolate diluted broth were mixed to obtain the final inoculum containing 5×105 CFU/mL. In parallel, a control negative tube for each isolate was obtained by a mixture preparation containing the same volume of sterile distilled water and broth containing the bacteria, followed by overnight incubation of tubes at conditions just described. Finally, 0.1 mL from each tube was plated on the Mueller Hinton agar (MHA) plate (Oxoid) and incubated overnight in an ordinary incubator at 37°C, followed by colony counting according to Bresee et al.39
MIC
Serial microdilution assays were conducted to quantify the MIC values for AgNPs that inhibited bacterial growth, based on the method by Shaalan et al40 with some modifications. Briefly, 100 μL of sterile Mueller-Hinton broth was introduced into each well of a 96-well microtiter plate, except for one well that was left as a blank according to microplate reader software instructions. Moreover, three rows from each plate were used as controls, as follows: one negative control row of AgNPs at starting concentration 5 μg/mL was used as blank values, which were deduced from the test values; one positive control row of ciprofloxacin at starting concentration of 0.1 mg/mL was included due to its antibacterial activity; and one culture control was included to verify that the broth was capable of supporting microbial growth. To test the antimicrobial properties of the AgNPs sample, 100 μL of AgNPs at a starting concentration of 5 mg/L were individually transferred into the top row of the micro-titer plate and double-fold serial dilutions were performed; all samples were tested in triplicate to ensure accurate and reproducible results. Before the addition of bacteria to the microtiter plates, all cultures used were subcultured into the suitable broth and diluted until just turbid (0.5 McFarland standard) to ensure an approximate concentration of 1.5×108 CFU/mL. Hereafter 100 µL of subculture was added to all 96 wells, except in the negative control row, for which 100 µL of sterile Mueller-Hinton broth was added. Each plate was subsequently sealed with a sterile adhesive-sealing film to prevent evaporation of the test sample and incubated at 37°C for 24 hours.
Tetrazolium Salt Reduction Assay
Various colorimetric assays quantify the survival of cells through reducing salts of tetrazolium into formazan pigments, whereas the color change from colorless to pink can be quantified with a spectrophotometer.19 After incubation of microplates, 40 µL of indicator solution at a concentration of 5 mg/mL was added to each well of the micro-titer plates and incubated at 37°C for 15 minutes in a shaking incubator (150 rpm). Once an observable color change was observed within the culture control column after completion of the incubation period, the microplate plate reader (BioTek ELX-800) was used to measure the optical density of each plate at 570 nm. As defined by Shekar et al,41 MIC was the minimum concentration of AgNPs that inhibited 20% of the test microorganisms’ growth. The percentage of bacterial inhibition by AgNPs concentration was computed using the following equation for optical density (OD) after blank correction:
Minimum Bactericidal Concentration (MBC)
After measurement of ODs, 100 µL from each well was plated on an MHA plate (Oxoid) and incubated overnight in an ordinary incubator at 37°C to determine the least concentration that has no obvious growth (MBC) after 20 hours of incubation.
In vivo Antimicrobial Activity Assay
Experimental Design
A total of 35 male mice of 25±3 g body weight were divided into five groups, as listed in Tables 3 and 4, and kept in plastic cages (7 mice/cage) in a well-ventilated room at ambient room temperature and humidity with natural light. Mice were acclimated for 1 week before starting the experiment and were reared with free access to commercial pelleted feed (21% protein) and water. After the acclimatization period, mice were immunosuppressed by intraperitoneal injection with a dose of 30 mg/kg/24 hours of cyclophosphamide (Endoxan®) for 3 successive days.42 On the last day of immunosuppression, all mice were fasted overnight followed by oral administration of 0.1 mL of 5% solution of sodium bicarbonate (NaHCO3) to counteract the stomach pH immediately before oral infection.43 Infection with 1 mL of normal saline solution containing 1.5×108 CFU/mL of the isolated Salmonella Enteritidis strain (isolate No.1) given by oral gavage to all infected groups; the other non-infected groups received normal saline only to exclude the stress factor. For verification of infection occurrence, the feces bacterial load of mice was estimated on the last day before and 2 days after infection; the onset of infection showed a gradual rise in bacterial load over the 2 days. The treatment regime was administered for 1 week with 10 μg/Kg body weight daily of AgNPs and 4 mg/Kg body weight daily of cefotaxime to the corresponding treated groups, in combination with administration of saline to other non-treated groups. Cefotaxime has been chosen for positive control group because the third-generation of cephalosporins, including cefotaxime, are successfully used in clinical practice as a therapeutic approach to salmonellosis.44 As well as the phenotypic sensitivity pattern of the used S. Enteritidis strain against cefotaxime was evaluated using the disc diffusion method according to the standards of CLSI,33 where the zone of inhibition equalled 30 mm which was interpreted according to CLSI,33 and it was identified as sensitive to cefotaxime (Farouk, unpublished work). All procedures for use of mice model in this research complied with ethics regulations of Institutional Animal Care and Use Committee, Faculty of Veterinary Medicine-Cairo University (FVMCU), and received the approval number: VetCU20022020121.
 |
Table 3 Overall Mean of Selected Hematological and Serum Biochemical Parameters Following Treatment of Mice in the First Four Groups and Statistical Comparison Between Them |
 |
Table 4 Overall Mean of Selected Hematological and Serum Biochemical Parameters Following Treatment of Mice in the Three Infected Groups and Statistical Comparison Between Them |
Bacteriologic Analyses
Fecal samples were collected from all groups at the end of the acclimatization period and subjected to routine bacteriologic examination to confirm that mice were free of Salmonella spp. The Salmonella spp. were counted per gram of feces in all infected groups every day after beginning the treatment protocol. For the feces collected, 1 g was dissolved in 1 mL sterile normal saline, and then 100 μL aliquots from each fecal suspension were serially diluted (10-fold) in sterile saline. Viable counts were determined by plating 10 μL from each dilution on duplicate xylose lysine deoxycholate agar plates that were later incubated at 37°C overnight. The standard colonies were identified as well as counted on the plates. Moreover, the actual count was calculated using the following equation.
Hematologic and Biochemical Parameters Analyses
Before dissection, mice were anesthetized using chloroform vapor after the treatment regime ended. Blood samples were obtained into two separate plain and ethylenediaminetetraacetic acid (EDTA)-containing tubes by cardiac puncture.45 The blood in the EDTA tube was used to estimate parameters of hematology by an automated veterinary hematology analyzer. The serum separation was performed through centrifugation of a plain vacutainer at 3000 xg for 10 minutes and kept at −20°C till estimation of serum albumin, alanine aminotransferase (ALT), urea, and creatinine using dedicated test kits (Spectrum Diagnostics) according to manufacturer instructions.
Histopathologic Examination
After treatment completion and blood collection, all mice were euthanized and dissected. Liver and intestine were removed carefully and fixed in formalin saline 10% solution. Cut sections of liver and intestine with 5 μm thickness were prepared and stained with hematoxylin and eosin dye, then microscopically inspected and photographed.46
Statistical Analysis
All quantitative data obtained from the in vitro study were presented as mean±standard deviation, as shown in Supplementary Tables 1A–E). For the in vivo study, statistical analysis was performed with SAS® version 9.4. A statistical significance was regarded at P<0.05. Statistical significance between the first four groups was determined using two-way ANOVA and one-way ANOVA between the three infected groups. All statistical analysis processes are illustrated in Supplementary Appendix 3.
Results
Isolation of Salmonella spp. From Diarrheic Sheep and Goats
Of the examined 325 and 193 fecal samples from diarrheic sheep and goats, respectively, Salmonella spp. was isolated from 13/325 (4%) and 7/193 (3.6%) samples, respectively (Table 5). All Salmonella spp. isolates were serotyped, as shown in Table 5. The identified isolates were as follows: nine Salmonella Mississippi, two Salmonella Durham, one Salmonella Ferruch, and one Salmonella ParatyphiA for sheep; two Salmonella Enteritidis, one Salmonella Allerton, one Salmonella Bonariensis, one Salmonella Kottbus, one Salmonella Mississippi, and one Salmonella Stanleyville for goats. In addition, agarose gel electrophoresis of polymerase chain reaction products revealed a specific amplicon size of 284 bp in all isolates, as shown in Figure 1. All recovered isolates complied with the typical biochemical identity of Salmonella spp., as shown in Supplementary Appendix 1.
 |
Table 5 Isolation Rate and Serotyping of Salmonella Species Isolates |
Antimicrobial Phenotypic Resistance Pattern and MAR Indexing
All isolates were resistant to colistin, doxycycline, and oxytetracycline, and all were sensitive to amikacin and enrofloxacin, and, moreover, showed variable patterns to the other five antimicrobials (Table 2). The resistance rates were 95%, 70%, 60%, 15%, and 5% for sulfamethoxazole-trimethoprim, amoxicillin, ciprofloxacin, florfenicol, and gentamycin, respectively. All 20 isolates had an index range of 0.4–0.7 (Table 6), which was identified as MDR Salmonella spp.
 |
Table 6 MDR Index and Overall Antimicrobial Activity of the Synthesized Silver-Nanoparticles on Tested MDR Salmonella Species Strains (MIC, MBC, and Inhibition % at MIC) |
Characterization and Concentration of AgNPs
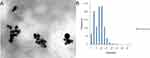
Imaging on TEM showed spherically shaped AgNPs (Figure 2A) with a diameter mean of 6.8±2.28 nm (range 2.95–12.2 nm), the highest frequency of sizes was noted at the size of 10 nm, as shown in the histogram (Figure 2B), the average particle size distribution of the prepared AgNPs by volume is shown in Figure 3A, and the highest peak of size distribution was observed at 24.77 nm. Zeta potential analysis results showed that AgNPs were carrying a negative charge of −37.3±5.7 mV (Figure 3B), the FT-IR spectrum confirmed AgNPs synthesis and stability due to the reduction and capping by PVP and showed seven peaks (Figure 4) at the following wave numbers: 3463, 2852, 2072, 1638, 1391, 1294, and 550 cm−1. Moreover, measurement by ICP-AES demonstrated 5.096 μg/mL of silver ion concentration.
 |
Figure 4 FTIR spectra of PVP capped AgNPs in the frequency range of 4000–400 cm−1. |
In vitro Antibacterial Activity of AgNPs
Bacterial Growth Inhibition
The synthesized AgNPs exhibited growth inhibiting activity against all 20 tested strains of MDR Salmonella spp., and the bacterial count was less than the initial count before incubation (<5×105 CFU/mL) for all tested isolates.
MIC and Viability % of Salmonella spp
The MICs of the synthesized AgNPs ranged from ≤0.002–0.313 μg/mL, and the mean average was 0.085±0.126 μg/mL (Table 6). The AgNPs concentrations ranged from 5 μg/mL to 0.002 μg/mL, which were assessed for their efficacy in the present study, as detailed in Tables 7–9.
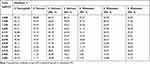 |
Table 7 Percentage of Bacterial Inhibition of S. ParatyphiA, S. Ferruch, S. Durham (No. 1 and 2) and S. Mississippi (No. 1-3) Isolates at Different Serial Dilutions of Silver Nanoparticles |
 |
Table 8 Percentage of Bacterial Inhibition of S. Mississippi (No. 4-9) and S. Stanleyville Isolates at Different Serial Dilutions of Silver Nanoparticles |
MBC
The initial MBCs were recorded as the lowest AgNPs concentration to show no change in color when the color of the culture control row matches that of the pH endpoint control (Figure 5). Final readings were recorded after incubation of inoculated MHA plates, which was determined by no obvious growth of the corresponding dilution comparison with obvious growth of culture control inoculated plates. The MBCs ranged from 0.078–1.250 μg/mL with an average mean of 0.508±0.315 μg/mL (Table 6).
In vivo Antibacterial Activity
Treatment Efficacy
The experimental infection of mice was confirmed by noticing a steady increase in the viable count of Salmonella Enteritidis in feces within 2 days of infection. Oral administration of AgNPs was found to induce noticeable decreases in the count of viable Salmonella Enteritidis retrieved from feces, as shown in Figure 6, where the numbers of viable S. Enteritidis were 4.4×104, 2.16×104, and 1.507×104 CFU/g feces in the first, second, and third day of treatment, respectively. Oral treatment with cefotaxime resulted in quite a decrease in the viable count in comparison to the AgNPs treated group, where the viable S. Enteritidis counts were 6.9×104, 3.6×104, and 3.4×104 CFU/g feces in the first, second, and third day of treatment, respectively. Salmonella Enteritidis shedding in feces stopped during days 4 and 6 of the treatment protocol in both the AgNPs and cefotaxime treated groups. Also, the viable counts of S. Enteritidis decreased in the feces of the other infected control group, and shedding stopped 2 days later in comparison with the other two treated groups.
 |
Figure 6 Effects of treatment with AgNPs and cefotaxime on fecal shedding of Salmonella Enteritidis (CFU/g) in comparison to the infected and non-treated group. |
Effect of the Synthesized AgNPs on Hemato-Biochemical Parameters
In comparison between the first four groups, as shown in Table 3, a significant difference was evident in some parameters, including band neutrophils count, mean corpuscular hemoglobin concentration (MCHC), serum albumin, and ALT. Moreover, there was no evidence of significance for other parameters. Regarding band neutrophils count, there was a significant increase in the infected and non-treated group compared with the other three groups, which did not differ from each other due to significant effects of infection (F1,15=4.61, P=0.0485), treatment (F1,15=5.90, P=0.0281), and infection-treatment interaction (F1,15=5.90, P=0.0281). In the case of MCHC, serum albumin, and ALT parameters, there was a significant decrease in albumin and an increase in MCHC and ALT in both infected groups versus the non-infected, and no evidence of difference between both the infected groups and both the non-infected groups, however, differences were noted due to the infection effect for albumin (F1,13=5.93, P=0.0301), ALT (F1,13=5.31, P=0.0383), and MCHC (F1,15=5.35, P=0.0353). In comparing the three infected groups, however, the only significant evidence was in band neutrophils count, and the three groups were completely different from each other, as shown in Table 4. The count was the highest in the infected and nontreated group, followed by the count of the infected and cefotaxime-treated group, then the infected and AgNPs-treated group (F2,11=4.91, P=0.0299).
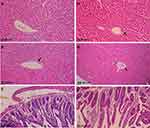
Effect of the Synthesized AgNPs on the Histology of Liver and Intestine
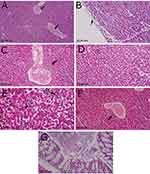
Histopathologic lesions were mainly observed in the negative control group of S. Enteritidis-infected and non-treated mice. The inflammatory and degenerative lesions were predominant in liver, including congestion of hepatic blood vessels, acute inflammatory response with infiltration of mononuclear cells, dilatation of the hepatic sinusoids, perivascular cuffing with inflammatory cells, and subcapsular edema (Figure 7A–F). The intestine samples showed necrobiotic changes and acute inflammatory reaction (Figure 7G). In contrast, the infected and AgNPs-treated group expressed a mild inflammatory reaction that was less than that for the non-treated groups and maintained of the integrity and structure of hepatocytes, whereas the intestine showed a normal histologic pattern (Figure 8A–C). A similar normal histological pattern with mild pathological changes was noted in the AgNPs-treated non-infected group (Figure 8D–F).
Discussion
Recently the epidemiology of Salmonella spp. and recently evolved resistant strains have caused concern in both human and veterinary medicine because of their zoonotic impact. Although the existence and spread of MDR strains in seemingly healthy slaughtered sheep and goats have previously been recorded,47 there is less information about its prevalence in diseased sheep and goats. In the current study of diarrheic sheep and goats all within the Giza Governorate in Egypt, Salmonella spp. were isolated in 4% (13/325) of fecal samples in sheep and from 3.6% (7/193) of the sample in goats (Table 5). Nearly similar isolation rates were reported in other studies of diarrheic lambs, sheep, and goats: 3.6% in diarrheic lambs,9 4.5% among diarrheic sheep,48 and 5.26% in both diarrheic sheep and goats.49
The concern of antibiotic resistance bacteria in Africa has been gaining particular interest throughout the previous decade. Furthermore, very little has been understood concerning the significant extent of the threat, because monitoring for antibiotics resistance to date has been only implemented within a limited number of countries.50 In the current study, all recovered isolates were detected as MDR based on their MAR range of >0.3 to <0.8. However, these isolates showed different phenotypic resistant patterns to the 10 tested antimicrobial agents. The most evident pattern of complete resistance (100%) was seen for colistin, doxycycline, and oxytetracycline, followed by 95%, 70%, and 60% for sulfamethoxazole–trimethoprim, amoxicillin, and ciprofloxacin, respectively. Moreover, the most effective agents were amikacin, enrofloxacin, gentamicin, and florfenicol (Table 2).
The emergence of such MDR serovars acts as a key factor in therapeutic failure for treatment of infections in both animals and humans,51 constitutes a great challenge for this pathogen control, and advocates for the need to investigate antimicrobial alternatives as a method of control. Owing to their antimicrobial activity, metallic nanoparticles may serve as potential alternatives for some traditional antibiotics.40 This study integrated AgNPs through a chemical reduction reaction. For this integration, AgNO3, NaBH4, and PVP were used as a source for Ag+, a reducing agent, and stabilizing agent to prevent agglomeration of particles, respectively.52 Trisodium citrate acted simultaneously as both stabilizing and reducing agent, Furthermore, the use of PVP and sodium citrate improved the stability of newer nanoparticles.53
In the current study, AgNPs were integrated through chemical reduction reaction to clarify its ability to overcome all clinically recovered MDR Salmonella spp. isolates and it showed efficient antimicrobial activities against all of them. Furthermore, it showed efficient inhibitory activities on all isolates ranging from 84.81–99.92% at concentration 5 μg/mL, MIC range (≤0.002–0.313 μg/mL) with an average mean of 0.085±0.126 μg/mL, and MBC range (0.078–1.250 μg/mL) with an average of 0.508±0.315 μg/mL. These findings are compatible with results of a previous study54 that reported the antibacterial activity of AgNPs against Salmonella Typhimurium. The MBC/MIC (Table 6) ratio of AgNPs for different isolates showed variant bacteriostatic to bactericidal effects of AgNPs, which may be justified by the different resistance patterns of the tested isolates. The bactericidal effect of AgNPs is attributed to the reaction between the charges of bacterial cell wall and metal ions.55 Moreover, the main factors affecting bactericidal properties are stability, size, surface area, and shape of AgNPs.56 The FTIR spectrum reveals the stability of the synthesized AgNPs where the silver ion reduction and PVP-capping of AgNPs occur simultaneously; the FT-IR spectrum shows strong bonding at peak 1 at wave number 3463 cm−1, which corresponds to the N-H stretches with the amine group, and peak 2 at 2852 cm−1, which corresponds to C–H stretches with the alkenes group. A strong absorption peak at 1638 cm−1 is corresponding to carbonyl group stretching of PVP, indicating the PVP capping for AgNPs which prevents its reaggregation and agglomeration. The two peaks at 1391 and 1294 cm−1 are reported for bond vibrations of N–H–O complex and NO3 group, respectively. The last band at 550 cm−1 corresponds to carbonyl stretch.57 The synthesized AgNPs in this study are spherical in shape and this finding may be attributed to its wider antibacterial activity in inhibiting all isolates58 and they are carrying a negative charge of −37.3±5.7 mV, which indicates good stability of the synthesized particles.59 The negative charge of AgNPs is attributed to the interaction between the PVP and metallic nanoparticles surface.60 Moreover, the bactericidal efficacy of AgNPs against gram-negative bacteria depends on AgNPs concentration.58 It was observed that the percent of inhibition and bactericidal efficacy of synthesized AgNPs were in a direct proportional relationship with AgNPs concentration (Tables 7–9), as shown in a previous report by Swarnavalli et al,58 which noted that the higher antibacterial effects were observed with increasing the AgNPs concentration. Furthermore, the bactericidal effects of AgNPs increased by decreasing its size.58,61 The size range of synthesized AgNPs is 2.95–12.2 nm, based on TEM measurements, while the DLS analysis showed an average of particle size distribution (at 24.77 nm). DLS measurements are affected by dispersants, which results in higher size values than obtained by TEM analyses,62 which is also similar to the previous findings58,61 and is considered a key factor responsible for the observed antimicrobial activity. This finding can be justified as follows: the maximum contact area, surface area-to-volume ratio, and AgNPs relative concentration will be increased when AgNPs size decreases.58,61 This relationship can be the result of the powerful penetration of the nanoparticles and a wide area of interaction between the nanoparticles and microorganisms.58,61 The previous in vivo studies did not declare the phenotypic resistance characteristics of the challenge strain used, so in the present study in vivo antimicrobial activity of integrated AgNPs was visualized against MDR S. Enteritidis, which was recovered from a diarrheic goat with MAR value of 0.5. It was obvious that this strain was pathogenic to mice based on histopathologic and hemato-biochemical evidence in infected mice, as well as a marked decrease of the number of viable S. Enteritidis isolates recovered from feces of the treated groups, with shedding stopped completely between days 4 to 6 of treatment. In addition, the viable count gradually decreased during the treatment period, and there was a difference in cefotaxime-treated and AgNPs-treated groups, which are findings compatible with those of Abd-Elhakeem et al.26 Also, the therapeutic dose of AgNPs was less than that of the cefotaxime dose, and the efficacy of AgNPs versus cefotaxime was greater in decreasing bacterial count. Therefore, our study spotlights the in vivo efficacy of AgNPs on MDR S. Enteritidis strain in comparison to cefotaxime use as the conventional antibiotic.
Hematologic, biochemical, and histopathologic examinations in different groups were used to ascertain whether AgNPs had no toxic or pathologic effect in the AgNPs control group and to evaluate its ability in the tissue healing process following treatment in the test group. Regarding the hematologic findings, as shown in Table 3, there was a significant increase in band neutrophils count in the infected and non-treated group versus the other three groups due to the significant effects of infection, treatment, and infection–treatment interaction. These findings may be attributed to infection that resulted in an increased band neutrophils count. Treatment decreased band neutrophils count, and, with regard to the treatment effect as shown in Table 4, the count was significantly different between three groups and the count was higher in infected and non-treated group than in both other groups. Also, it was observed that count decreased in both treated groups compared with the non-treated group. However, the treatment effect of AgNPs was more evident in decreasing the count than in the cefotaxime-treated group. Furthermore, with regard to the infection effect, there was a significant decrease in albumin and an increase in MCHC and ALT in both infected groups versus the other non-infected groups, and no evidence of difference between both infected groups and between both non-infected groups. The increase of liver enzyme ALT and decrease in albumin is supported by previous studies, which reported that Salmonella spp. infection results in hepatic granulomas or paratyphoid nodules, causing release of liver enzymes in serum and decreasing its ability to produce albumin.63 In addition, previous studies found that the significant difference in the increased band neutrophils count in the non-treated versus treated groups may be attributed to the contributions of neutrophils in the effort for salmonellosis control.64,65 The response to strong inflammatory stimulus leads to an increased demand for neutrophils production and thus results in left shift neutrophilia because of the liberation of immature band neutrophils into circulation.66 Although the significant decrease in neutrophils may be attributed to the ability of mice to respond to treatment and the efficacy of AgNPs and cefotaxime in eliminating infection and decreasing inflammatory reactions, the effect of AgNPs is more evident than that for cefotaxime. So the hematobiochemical findings provided evidence for the clinical pathologic effect of the challenge of the S. Enteritidis strain in infected groups and the ability of AgNPs to suppress the developed inflammatory reaction. Moreover, there was no evidence of significant adverse toxic or pathologic effects of AgNPs on these test subjects, especially liver and kidney function tests which act as valuable indicators for toxicity; liver and kidneys are responsible for detoxification and excretion.
Mice are considered an ideal model for studying Salmonella spp. pathogenesis because they are a natural host. However, the limitation in this model is the absence of diarrhea in mice after oral infection. Oral streptomycin was administered before experimental infection with S. Typhimurium to create the ideal clinical signs of diarrhea.67 Nevertheless, because the aim was to evaluate the therapeutic effect of AgNPs, the use of streptomycin may have altered the results. Alternatively, a 3-day regime of I/P cyclophosphamide can be used as an immunosuppressive agent before oral infection.42 The pathologies induced by the infection of S. Enteritidis are mainly inflammatory, with the inflammatory findings in liver similar to those in previous studies on mice,42,68 including congestion, edema, and inflammatory cells infiltrations. The necrotic changes in intestine are in line with Lee et al.69 The findings of this current study showed that in the groups treated with AgNPs, the severity of inflammatory response is greatly reduced. This finding is attributed partially to killing activity of AgNPs toward gram-negative microorganisms.70–72 The other action of AgNPs is an anti-inflammatory effect,73 which helps reduce the severity of inflammation induced by salmonellosis. Finally, the in vivo study declared the safety and potency of the integrated AgNPs in the treatment of MDR salmonellosis so it can serve as an alternative antimicrobial, especially at the occurrence of antibiotic-resistant bacterial infections.74
Conclusion
This study focused on MDR Salmonella spp. in diarrheal sheep and goats in Egypt for which conventional antimicrobial therapy may fail to provide a response, the ability of AgNPs in their inhibition of Salmonella spp. both in vitro and in vivo without adverse effects, and an estimation of MIC to determine an effective concentration for when a commercial product can be prepared. The study findings support that AgNPs may be used as a therapeutic agent. However, further studies are needed to develop and design a safe and effective delivery system of AgNPs for small ruminants.
Abbreviations
Ag+ ions, silver ions; AgNPs, silver nanoparticles; ALT, alanine aminotransferase; CFU, colony forming unit; ICP-AES, inductively coupled plasma atomic emission spectroscopy; MAR, multiple antimicrobial resistance; MBC, minimum bactericidal concentration, MCHC, mean corpuscular hemoglobin concentration; MDR, multidrug resistant; MHA, Mueller-Hinton agar; MIC, minimum inhibitory concentration; OD, optical density; PVP, polyvinyl pyrrolidone; TEM, transmission electron microscope.
Data Sharing Statement
All data used to generate these results is available in the main text.
Ethics Approval and Consent to Participate
The study was approved by Ethics of Institutional Animal Care and Use Committee, Faculty of Veterinary Medicine-Cairo University (FVMCU) and received the approval number: VetCU20022020121.
Acknowledgments
The authors would like to thank Dr. Hisham A. Abdelrahman for sharing his expertise in performing statistical analysis of this study and we gratefully thank Dr. Fatma A. Fadl for her help in biochemical examinations.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
1. Wilsmore T. Diseases of Small Ruminants in Ethiopia, the Veterinary Epidemiology and Economics Research Unit (VEERU) School of Agriculture Policy and Development. UK: The University of read; 2006:6–7.
2. Montagne M, Martel A, Le Moual H. Characterization of the catalytic activities of the PhoQ histidine protein kinase of Salmonella enterica serovar Typhimurium. J Bacteriol. 2001;183:1787–1791. doi:10.1128/JB.183.5.1787-1791.2001
3. Hoelzer K, Soyer Y, Rodriguez L, et al. The prevalence of multidrug resistance is higher among bovine than human Salmonella enteric serotype Newport, Typhimurium, and 4, 5, 12: i:- isolates in the United States but differs by serotype and geographic region. Appl Environ Microbiol. 2010;76:5947–5959. doi:10.1128/AEM.00377-10
4. Van den Bogaard AE, Stobberingh EE. Antibiotic usage in animals: impact on bacterial resistance and public health. Drugs. 1999;85:589–607. doi:10.2165/00003495-199958040-00002
5. Sivakumar T, Avinash Saravanavel N, Prabhu D, et al. Characterization of multidrug resistant patterns of Salmonella sp. World J Med Sci. 2012;7:64–67.
6. Marshall BM, Levy SB. Food animals and antimicrobials: impact on human health. Clin Microbiol Rev. 2011;24:718–733.
7. Yap MN. The double life of antibiotics. Mo Med. 2013;110:320–324.
8. Ban F. Food safety of animal origin and surveillance for drug residues in animal products. Henan Anim Sci Vet Med. 2001;22:5–7.
9. Abd El-Twab AA, El-Hofy IF, Amira MR. Molecular characterization of quinolones and β-lactams resistant Salmonella serovars determinants in diarrheic calves, lambs and goats-kids in the Middle of Nile Delta, Egypt. Benha Vet Med J. 2016;30:171–182. doi:10.21608/bvmj.2016.31361
10. Priyanka S, Abhroop G, Santosh P, et al. Antimicrobial effects of biogenic nanoparticles. J Nanomater. 2018;8:1–19.
11. Burdusel CA, Gherasim O, Grumezescu MA, Mogoantă L, Ficai A, Andronescu E. Biomedical applications of silver nanoparticles: an up-to-date overview. Nanomaterials. 2018;8:681. doi:10.3390/nano8090681
12. Porcaro F, Carlini L, Ugolini A, et al. Synthesis and structural characterization of silver nanoparticles stabilized with 3-mercapto-1-propansulfonate and 1-thioglucose mixed thiols for antibacterial applications. Materials. 2016;9:1028. doi:10.3390/ma9121028
13. Kim JS, Kuk E, Yu KN, et al. Antimicrobial effects of silver nanoparticles. Nanomedecine: NBM. 2007;3:95–101. doi:10.1016/j.nano.2006.12.001
14. Suresh AK, Pelletier DA, Wang W, et al. Cytotoxicity induced by engineered silver nanocrystallites is dependent on surface coatings and cell types. Langmuir. 2012;28:2727–2735. doi:10.1021/la2042058
15. Durán N, Marcato PD, Conti RD, Alves OL, Costa FTM, Brocchi M. Potential use of silver nanoparticles on pathogenic bacteria, their toxicity and possible mechanisms of action. J Braz Chem Soc. 2010;21:949–959. doi:10.1590/S0103-50532010000600002
16. Han DW, Woo YI, Lee MH, et al. In-vivo and in-vitro biocompatibility evaluations of silver nanoparticles with antimicrobial activity. J Nanosci Nanotechnol. 2012;12:5205–5209. doi:10.1166/jnn.2012.6367
17. Knetsch ML, Koole LH. New strategies in the development of antimicrobial coatings: the example of increasing usage of silver and silver nanoparticles. Polymers. 2011;3:340–366.
18. Mosselhy AD, Abd El-Aziz M, Hanna M, et al. Comparative synthesis and antimicrobial action of silver nanoparticles and silver nitrate. J Nanopart Res. 2015;17:473. doi:10.1007/s11051-015-3279-8
19. Seil JT, Webster TJ. Antimicrobial applications of nanotechnology: methods and literature. Int J Nanomedicine. 2012;7:2767–2781.
20. Swain P, Nayak SK, Sasmal A, et al. Antimicrobial activity of metal based nanoparticles against microbes associated with diseases in aquaculture. World J Microbiol Biotechnol. 2014;30:2491–2502.
21. Hoseinnejad M, Jafari SM, Katouzian I. Inorganic and metal nanoparticles and their antimicrobial activity in food packaging applications. Crit Rev Microbiol. 2018;44:161–181. doi:10.1080/1040841X.2017.1332001
22. Jain J, Arora S, Rajwade JM, et al. Silver nanoparticles in therapeutics: development of an antimicrobial gel formulation for topical use. Mol Pharm. 2009;6:1388–1401. doi:10.1021/mp900056g
23. Gopinath PM, Narchonai G, Dhanasekaran D, Ranjani A, Thajuddin N. Mycosynthesis, characterization and antibacterial properties of AgNPs against multidrug resistant (MDR) bacterial pathogens of female infertility cases. Asian J Pharm Sci. 2015;10:138–145. doi:10.1016/j.ajps.2014.08.007
24. Sanyasi S, Majhi RK, Kumar S, et al. Polysaccharide-capped silver nanoparticles inhibit biofilm formation and eliminate multi-drug-resistant bacteria by disrupting bacterial cytoskeleton with reduced cytotoxicity towards mammalian cells. Sci Rep. 2016;6:24929. doi:10.1038/srep24929
25. Grigor’eva A, Saranina I, Tikunova N, et al. Fine mechanisms of the interaction of silver nanoparticles with the cells of Salmonella typhimurium and Staphylococcus aureus. Biometals. 2013;26:479–488. doi:10.1007/s10534-013-9633-3
26. Abd-Elhakeem AM, Ingy B, Amira R. Efficacy of silver nanoparticles as antimicrobial agent against Salmonella infection and accompanied biochemical, immunological and histopathological changes in rats. Egy J Pure &appl Sci. 2016;54:13–19.
27. International Organization for Standardization −6579. Microbiology of Food and Animal Feeding Stuff-Horizontal Method for the Detection of Salmonella. Geneva: ISO; 1998.
28. Quinn PJ, Markey BK, Carter ME, et al. Veterinary Microbiology and Microbial Diseases. Great Britain by HPG. Bodmin, Cornwall, UK: Books Ltd; 2002:114–118.
29. Grimont PA, Weill FX. WHO Collaborating Centre for Reference and Research on Salmonella “Antigenic Formulae of the Salmonella Serovars”.
30. Wani SA, Bhat MA, Samanta I, et al. Isolation and characterization of Shiga toxin-producing Escherichia coli (STEC) and Enteropathogenic Escherichia coli (EPEC) from calves and lambs with diarrhea in India. Lett Appl Microbiol. 2003;37:121–126. doi:10.1046/j.1472-765X.2003.01364.x
31. Rahn K, De Grandis SA, Clarke RC, et al. Amplification of an invA gene sequence of Salmonella Typhimurium by polymerase chain reaction as a specific method of detection of Salmonella. Mol Cell Probes. 1992;6:271–279. doi:10.1016/0890-8508(92)90002-F
32. EUCAST. European Committee For Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin Microbiol Infect. 2003;9(1–7).
33. CLSI. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Fourth Informational Supplement. CLSI document M100-S24. Wayne, PA: Clinical and Laboratory Standards Institute; 2014.
34. European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 7.1. 2017. Available from: http://www.eucast.org.
35. Global Salm-Surv. A Global Salmonella Surveillance and Laboratory Support Project of the World Health Organization. Laboratory Protocols Level 1 Training Course: Isolation of Salmonella.
36. Paul S, Bezbaruah RL, Roy MK, et al. Multiple antibiotic resistance (MAR) index and its reversion in Pseudomonas aeruginosa. Letters Appl Microbiol. 1997;24:169–171. doi:10.1046/j.1472-765X.1997.00364.x
37. Christopher AF, Hora S, Ali Z. Investigation of plasmid profile, antibiotic susceptibility pattern multiple antibiotic resistance index calculation of Escherichia coli isolates obtained from different human clinical specimens at tertiary care hospital in Bareilly-India. Ann Trop Med PH. 2013;6:285–289.
38. El Mahdy MM, Eldin TAS, Aly HS, et al. Evaluation of hepatotoxic and genotoxic potential of silver nanoparticles in albino rats. Exp Toxicol Pathol. 2015;67:21–29. doi:10.1016/j.etp.2014.09.005
39. Bresee J, Bond CM, Worthington RJ, et al. Nanoscale structure activity relationships, mode of action, and biocompatibility of gold nanoparticle antibiotics. J Am Chem Soc. 2014;136:5295–5300. doi:10.1021/ja408505n
40. Shaalan IM, El‑Mahdy MM, Theiner S, et al. In vitro assessment of the antimicrobial activity of silver and zinc oxide nanoparticles against fish pathogens. Acta Vet Scand. 2017;59:49. doi:10.1186/s13028-017-0317-9
41. Shekar BRC, Nagarajappa R, Jain R, et al. Minimum inhibitory concentration of the plant extracts’ combinations against dental caries and plaque microorganisms: an in vitro study. J Indian Assoc Public Health Dent. 2016;14:456–462. doi:10.4103/2319-5932.195842
42. Tala SD, Gasting D, Fodouop CPS, et al. In vivo anti-salmonella activity of aqueous extract of Euphorbia prostrata Aiton (Euphorbiaceae) and its toxicological evaluation. Asian Pac J Trop Biomed. 2015;5:310–318. doi:10.1016/S2221-1691(15)30350-6
43. Srinivasan A, Foley J, Ravindran R, et al. Low-dose salmonella infection evades activation of flagellin-specific CD4 T cells. J Immunol. 2004;173:4091–4099. doi:10.4049/jimmunol.173.6.4091
44. Ekinci B, Coban AY, Birinci A, et al. In vitro effects of cefotaxime and ceftriaxone on Salmonella typhi within human monocyte-derived macrophages. Clin Microbiol Infect. 2002;8:810–813. doi:10.1046/j.1469-0691.2002.00457.x
45. Gatsing D, Aliyu R, Kuiate JR, et al. Toxicological evaluation of the aqueous extract of Allium sativum bulbs on laboratory mice and rats. Cameroon J Exp Biol. 2005;1:39–45.
46. Gamble M. The hematoxylins and eosin. Bancroft JD, Gamble M, editors. Theory and Practice of Histological Techniques.
47. Molla W, Molla B, Alemayehu D, et al. Occurrence and antimicrobial resistance of Salmonella serovars in apparently healthy slaughtered sheep and goats of central Ethiopia. Trop Anim Health Prod. 2006;38:455–462. doi:10.1007/s11250-006-4325-4
48. Erhoma AA. Bacteriological studies on salmonellosis in sheep. M.V.Sc. thesis (Bacteriology). Faculty of veterinary medicine, Cairo university; 2007.
49. Nasr M, Hammouda HA, Bakeer NM, et al. Epidemiological, clinical and bacteriological studies on bacterial lamb enteritis at Behera Province, Egypt. Alex J Vet Sci. 2014;43:8–16. doi:10.5455/ajvs.163829
50. World Health Organization. Antimicrobial resistance: global report on surveillance. Geneva; 2014. Available from: https://www.who.int/drugresistance/documents/surveillancereport/en/.
51. Kimang’a AN. A situational analysis of antimicrobial drug resistance in Africa: are we losing the battle? Ethiop J Health Sci. 2012;22:135–143.
52. Wang H, Qiao X, Chen J, Wang X, Ding S. Mechanisms of PVP in the preparation of silver nanoparticles. Mater Chem Phys. 2005;94:449–453. doi:10.1016/j.matchemphys.2005.05.005
53. Cao VD, Tran NQ, Nguyen TPP. Synergistic efect of citrate dispersant and capping polymers on controlling size growth of ultrafne copper nano‑particles. J Exp Nanosci. 2015;10:576–587. doi:10.1080/17458080.2013.848298
54. Omara TS, Zwrah FM, Samy AA. Minimum bactericidal concentration of chemically synthesized silver nanoparticles against pathogenic Salmonella and Shigella strains isolated from layer poultry farms. J App Pharm. 2017;7:214–221.
55. Prema P, Thangapandiyan S, Immanuel G. CMC stabilized nano silver synthesis, characterization and its antibacterial and synergistic effect with broad spectrum antibiotics. Carbohydr Polym. 2017;158:141. doi:10.1016/j.carbpol.2016.11.083
56. Su Y, Zhao L, Meng F, et al. Silver nanoparticles decorated lipase-sensitive polyurethane micelles for on-demand release of silver nanoparticles. Colloids Surf B Biointerfaces. 2017;152:238–244. doi:10.1016/j.colsurfb.2017.01.036
57. Kumar M, Devi P, Kumar A. Structural analysis of PVP capped silver nanoparticles synthesized at room temperature for optical, electrical and gas sensing properties. J Mater Sci Mater Electron. 2017;28:5014–5020. doi:10.1007/s10854-016-6157-y
58. Swarnavalli GCJ, Dinakaran S, Raman N, et al. Bio inspired synthesis of monodispersed silver nanoparticles using Sapindus emarginatus pericarp extract –Study of antibacterial efficacy. J Saudi Chem Soc. 2017;21:172–179. doi:10.1016/j.jscs.2015.03.004
59. Saeb AT, Alshammari AS, Al-Brahim H, Al-Rubeaan KA. Production of silver nanoparticles with strong and stable antimicrobial activity against highly pathogenic and multidrug resistant bacteria. Sci World J. 2014;2014:704708. doi:10.1155/2014/704708
60. Haesuwannakij S, Kimura T, Furutani Y, et al. The impact of the polymer chain length on the catalytic activity of poly (N-vinyl-2-pyrrolidone)-supported gold nanoclusters. Sci Rep. 2017;7:1–8. doi:10.1038/s41598-017-10165-9
61. Agnihotri S, Mukherji S, Mukherji S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. RSC Adv. 2014;4:3974. doi:10.1039/C3RA44507K
62. Souza TG, Ciminelli VS, Mohallem NDS. A comparison of TEM and DLS methods to characterize size distribution of ceramic nanoparticles. J Phys Conf Ser. 2016;733:6–11. doi:10.1088/1742-6596/733/1/012039
63. Hasbun HJ, Osorio R, Hasbun A. Hepatic dysfunction in typhoid fever during pregnancy. Infect Dis Obstet Gynecol. 2006;2006:64828.
64. Conlan JW. Critical roles of neutrophils in host defense against experimental systemic infections of mice by Listeria monocytogenes, Salmonella typhimurium, and Yersinia enterocolitica. Infect Immun. 1997;65:630–635. doi:10.1128/IAI.65.2.630-635.1997
65. Vassiloyanakopoulos AP, Okamoto S, Fierer J. The crucial role of polymorphonuclear leukocytes in resistance to Salmonella Dublin infections in genetically susceptible and resistant mice. Proc Natl Acad Sci. 1998;95:7676–7681. doi:10.1073/pnas.95.13.7676
66. Rosenfeld JA, Dial MS. Abnormalities in the Red and White Blood Cell Populations; Clinical Pathology for the Veterinary Team.
67. Palmer DA, Slaush MJ. Special collection advancing knowledge of biological mechanisms of the superorganism for next generation (NextGen) microbial dose-response modeling: salmonellosis 3. Mechanisms of Salmonella pathogenesis in animal models. Hum Ecol Risk Assess. 2017;23:1877–1892. doi:10.1080/10807039.2017.1353903
68. Kalia P, Kumar NR, Harjai K. The therapeutic potential of propolis against damage caused by Salmonella enterica serovar Typhimurium in mice liver: a biochemical and histological study. ARCH BIOL SCI. 2015;67:807–816. doi:10.2298/ABS141105040K
69. Lee M, Kwon AH, Kwon YD, et al. Antibacterial activity of medicinal herb extracts against Salmonella. Int J Food Microbiol. 2006;111:270–275. doi:10.1016/j.ijfoodmicro.2006.06.004
70. Losasso C, Belluco S, Cibin V, et al. Antibacterial activity of silver nanoparticles: sensitivity of different Salmonella serovars. Front Microbiol. 2014;5:227. doi:10.3389/fmicb.2014.00227
71. Seong M, Lee GD. Silver nanoparticles against Salmonella enterica serotype typhimurium: role of inner membrane dysfunction. Curr Microbiol. 2017;74:661–670. doi:10.1007/s00284-017-1235-9
72. Sondi I, Salopek-Sondi B. Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J Colloid Interface Sci. 2004;275:177–182. doi:10.1016/j.jcis.2004.02.012
73. Wong YKK, Cheung FOS, Huang L, et al. Further evidence of the anti-inflammatory effects of silver nanoparticles. Chem Med Chem. 2009;4:1129–1135. doi:10.1002/cmdc.200900049
74. Shaalan M, Sellyei B, El-Matbouli M, et al. Efficacy of silver nanoparticles to control flavobacteriosis caused by Flavobacterium johnsoniae in common carp Cyprinus carpio. Dis Aquat Organ. 2020;137:175–183. doi:10.3354/dao03439
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.