Back to Journals » Cancer Management and Research » Volume 12
The miRNA, miR-125b, Inhibited Invasion and Metastasis of Gastric-Cancer Cells by Triggering the STAT3 Signaling Pathway
Authors Xu X, Dang Z, Zhang J, Feng Y, Wei Z
Received 23 April 2020
Accepted for publication 3 August 2020
Published 17 September 2020 Volume 2020:12 Pages 8569—8580
DOI https://doi.org/10.2147/CMAR.S259513
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Harikrishna Nakshatri
Xiangqian Xu,1,2,* Zhongqin Dang,2,* Junping Zhang,3 Yingpu Feng,4 Zheng Wei1,3
1Gastroenterology Department, Henan University of Chinese Medicine, Zhengzhou, Henan, 450000, People’s Republic of China; 2Gastroenterology Department, The Second Affiliated Hospital of Henan University of Traditional Chinese Medicine, Zhengzhou, Henan, 450002, People’s Republic of China; 3Oncology Department, Henan Academy Institute of Traditional Chinese Medicine, Zhengzhou, Henan, 450000, People’s Republic of China; 4Oncology Department, Henan Province People’s Hospital, Zhengzhou, Henan, 450003, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yingpu Feng Email [email protected]
Zheng Wei Email [email protected]
Objective: To investigate the function and the mechanism of miR-125b in the invasion and metastasis of gastric cancer and provide experimental basis for finding and developing new therapeutic strategies for gastric cancer.
Methods: The difference of miR-125b expression in gastric cancer tissues and adjacent tissues was detected by qRT-PCR. The same test was performed in different gastric cancer cell lines. The effect of miR-125b on SGC-7901 and BGC-823 gastric cancer cell viability was examined using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Transwell assay was used to detect the effect of miR-125b on invasion and metastasis of gastric cancer cells. The target gene STAT3 of miR-125b was identified and validated by dual luciferase reporter assay. Western blot assay and immunofluorescence staining were used to detect the effect of miR-125b on the expression and distribution of STAT3 protein. The inhibitor and activator of STAT3 were used to confirm the effect of STAT3 on invasion and metastasis of gastric cancer cells. Peritoneal metastasis experiment and IHC were used to study the inhibitory effect of miR-125b on the metastasis of gastric cancer in vivo.
Results: The results of qRT-PCR showed that 125b expression was significantly lower in gastric cancer than in adjacent tissues, which indicated poor prognosis for gastric-cancer patients. Furthermore, two gastric-cancer cell lines, SGC-7901 and BGC-823, exhibited lower miR-125b levels than the normal cell line HEK293. After treatment with miR-125b mimics, cell proliferation was markedly inhibited. Meanwhile, the invasion and metastasis of gastric cancer cells were also inhibited after treated with miR-125b mimics. We also identified the signal transducer and activator of transcription 3 (STAT3) as a potential target of miR-125b based on patient data from The Cancer Genome Atlas (TCGA). Dual luciferase assays revealed that miR-125b directly inhibited STAT3 by binding to its 3′-untranslated region (UTR). Immunofluorescence assay showed that miR-125b could affect the subcellular distribution of STAT3. Moreover, treatment with miR-125b mimics or stattic inhibited invasion and migration in the gastric cancer cell lines, and IL-6 could reverse the inhibitory effect. Finally, nude mice xenografted with gastric-cancer cells expressing miR-125b mimics exhibited smaller tumors and lower transfer rates than mice engrafted with control group cells.
Conclusion: These data suggested that miR-125b inhibited invasion and metastasis in gastric cancer by inhibiting STAT3; therefore, miR-125b and STAT3 could be potential therapeutic targets in the treatment of gastric cancer.
Keywords: miR-125b, invasion and metastasis, gastric cancer, STAT3
Introduction
Gastric cancer is one of the most common cancers in China, especially in the country’s vast rural areas.1,2 Its incidence and mortality rates are higher than those of many other cancers.3,4 Early symptoms of gastric cancer are atypical; the disease is usually diagnosed in the middle or late stage.5,6 Invasion and metastasis in gastric cancer seriously lower the survival rate after surgery.7 Therefore, it is very important to find a molecular target to inhibit gastric-cancer invasion and metastasis. Micro-ribonucleic acids (miRNAs) are a class of endogenous noncoding small RNAs that play key roles in many physiological processes.8–10 Generally, miRNAs are 22–24 nucleotides (NT) long and complementary to the messenger RNA (mRNA) 3′-noncoding region, thereby inhibiting translation or activating transcription of target genes.11–13 Most importantly, miRNAs usually regulate various stages of tumor progression, including invasion, migration and apoptosis.14–16 The miRNA miR-125b is known to regulate malignant behaviors of cancer.17 Due to the low expression of miR-125b in gastric-cancer tissue, it is seen as a tumor suppressor. However, the specific role and mechanism of miR-125b in gastric-cancer metastasis are unclear. Therefore, we conducted both in vitro and in vivo experiments to reveal the molecular mechanism by which miR-125b inhibits gastric-cancer invasion and metastasis. This study provides the necessary experimental basis for finding a molecular target to inhibit said invasion and metastasis, as well as a new strategy for the clinical treatment of gastric cancer.
Patients and Methods
Patients and Tissue Specimens
We obtained tumor and non-tumor tissue specimens from gastric-cancer patients who were surgically treated at the hospital of Henan University of Chinese Medicine (Zhengzhou, China). No patient had a history of radiotherapy or chemotherapy before sampling, and diagnosis of gastric cancer was pathologically confirmed. The Institutional Ethics Committee of Henan University of Chinese Medicine approved this study. All patients were invited to sign informed-consent forms.
Reagents and Antibodies
We purchased fetal bovine serum (FBS) and Dulbecco’s Modified Eagle Medium (DMEM) from GIBCO BRL (Thermo Fisher Scientific, Waltham, Massachusetts, US); Nuclear Protein Extraction Kit and IL-6 were purchased from Sigma-Aldrich (St. Louis, Missouri, US); a RNA Extraction Kit from QIAGEN (Hilden, Germany); Matrigel was purchased from BD Biosciences (Franklin Lakes, NJ, USA); an Immunohistochemistry (IHC) Kit from Thermo Fisher; and methyl thiazolyl tetrazolium (MTT), a Bicinchoninic Acid (BCA) Protein Quantitative Kit and a Dual Luciferase Reporter Gene Assay Kit from Beyotime Institute of Biotechnology (Nanjing, China). Antibodies against signal transducer and activator of transcription 1 (STAT3), STAT3 inhibitor (stattic) and β-actin were purchased from Cell Signaling Technology (CST; Danvers, MA, USA); other analytical-grade chemicals were obtained from other commercial sources.
Cell Culture and Transfection
We obtained the human gastric-cancer cell lines SGC-7901 and BGC-823 from the Cell Bank of the Chinese Academy of Sciences, Shanghai Institute of Cell Biology (Shanghai, China). SGC-7901 and BGC-823 cells were cultured in DMEM containing 10% FBS and maintained at 37° C in a humidified incubator in a 5% CO2 atmosphere.
miR-125b Mimic Transfection
We seeded SGC-7901 and BGC-823 cells (1.5×105 per well) into a six-well plate and incubated them for 12 h, followed by transfection with miR-125b mimics (5′-UCCCUGAGACCCUAACUUGUGA-3′) using Lipofectamine 2000 (Beyotime) per manufacturer’s instructions. For all experiments, miR-125b mimics were synthesized by GenePharma (Shanghai, China), from which we also purchased negative controls.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
To assess the transfection efficiency of the miR-125b mimics, we performed a qRT-PCR assay. Primers were prepared and synthesized by Sangon Biotech, Ltd. (Shanghai, China). For miR-125b, the forward primer was 5′-CGAACAGAAATTGCCTGTCA-3′, while the reverse primer was 5′-ACCAAATTTCCAGGATGCAA-3′. The control was Homo sapiens RNAU6. Primers for U6 were 5′-CTCGCTTCGGCAGCACA-3′ (forward) and 5′-AACGCTTCACGAATTTGCGT-3′ (reverse). We extracted total RNA using the RNA Extraction Kit. Total RNA was used as a reverse-transcription template to obtain complementary deoxyribonucleic acid (cDNA), which we then fully mixed with the enzymes and primers. PCR conditions were as follows: 95° C for 5 min, followed by 40 cycles of 95°C for 15 s, 60°C for 60 s and 72°C for 10 s. Relative expression of miR-125b was calculated by the ΔΔCq method.
Cell Proliferation Assay
We analyzed SGC-7901 and BGC-823 cell proliferation via and methyl thiazolyl tetrazolium (MTT) assay. Briefly, cells were plated at a density of 3000 cells/well into a 96-well plate and incubated at 37° C for 12, 24, 48 and 72 h. Then, we incubated them with DMEM (containing 0.5 mg/mL MTT) for 4 h. Formazan crystals were dissolved with 150 μL dimethyl sulfoxide (DMSO). After 10 min oscillation, we measured the absorbance of each well (including blanks) at 490 nm in an automatic microplate reader (Bio-Rad, USA). The formula for the cell growth inhibition rate was as follows: Inhibition rate = (1 − absorbance of mimics group/absorbance of blank control group) × 100%.
Cell Invasion and Migration Assays
We performed cell invasion and migration assays using Transwell membranes (Corning, Inc., Corning, NY, USA) per manufacturer’s instructions. SGC-7901 and BGC-823 cells were suspended in 200 μL serum-free media and then added into the upper Boyden chamber on an uncoated or Matrigel-coated Transwell membrane; meanwhile, 600 μL culture medium containing 10% FBS was added into the lower chamber and cultured for 24 h. At the end of treatment, we removed the cells from the upper surface with cotton swabs. Cells that had invaded across the Matrigel onto the lower surface of the membrane were fixed with methanol and stained with 0.1% crystal violet. We then obtained images under a microscope (Nikon Corp., Tokyo, Japan) at 100× magnification, randomly capturing five fields per membrane. Migratory and invasive cells were counted and averaged. We performed migration assays using the same procedure, except without Matrigel-coated polycarbonate membranes. Each experiment was repeated three times.
Dual Luciferase Reporter Assay
We predicted transcription factor-binding sites in the promoter region of human miR-125b using the PicTar (https://pictar.mdc-berlin.de/) and TargetScan (http://www.targetscan.org) biological-analysis websites. The putative STAT3-binding site was 5′-CTCAGG-3′, and the mutant STAT3-binding site was 5′-GAGUCC-3′. We created the STAT3 luciferase reporter constructs by amplifying the human STAT3 mRNA 3′ untranslated-region (UTR) sequence via PCR and then cloning it into the XbaI site of the pGL3-promoter construct. Next, we inserted sequences of the human miR-125b promoter region into pGL3-basic vector (Sangon Biotech, Shanghai, China), after which we co-transfected the plasmids with Renilla luciferase expression vector (pRL-TK) and then transfected them into SGC-7901 and BGC-823 cells using Lipofectamine 2000 per manufacturers’ instructions. After transfection for 24 h, we measured luciferase activity in the samples using the Dual Luciferase Assay Kit.
Western Blot Analysis
We determined expression levels of STAT3 via Western blot. Cells or tissues were lysed with cold lysis buffer supplemented with a protease inhibitor mixture. Total protein concentration was measured by BCA assay and equalized with extraction reagent. We loaded equivalent amounts of protein, subjected them to 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel and transferred them electrophoretically onto polyvinylidene difluoride (PVDF) membranes. Next, we blocked the membranes with 5% skim milk for 2 h at room temperature (RT) and incubated them overnight at 4° C with STAT3 antibodies. Afterward, we washed them 3 times, added the secondary antibody and incubated them at RT for 2 h, after which we analyzed them using a Western blotting analysis system (Bio-Rad, USA).
Immunofluorescence (IF) Assay
We seeded BGC-823 cells into a 6-well plate with glass slides at the bottom. The cells were then fixed with 4% paraformaldehyde for 20 min, followed by permeabilization with 0.1% Triton X-100 for 3–5 min. Then, we incubated the cells with 1% bovine serum albumin (BSA) for 1 h, followed by overnight incubation with the anti-STAT3 antibody. Next, we incubated the samples with the secondary antibody (containing tetramethylrhodamine isothiocyanate [TRITC]–conjugated phalloidin) for 30–60 min. After staining with 4′,6-diamidino-2-phenylindole (DAPI) for 3–5 min, we visualized fluorescence images with a laser scanning confocal microscope (Olympus, Japan). Finally, we used Western blot analysis to confirm the distribution of STAT3 protein.
Xenograft Mouse Model
To examine the effect of miR-125b on the invasion and metastasis of tumor cells in vivo, we transfected BGC-823 with luciferase vector (GeneBiotech, Shanghai, China) and with or without miR-125b mimic treatment. We purchased 4-week-old nude mice from the Comparative Medicine Center of Soochow University (Suzhou, China). The Institutional Ethics Committee of Henan University of Chinese Medicine and The Animal Experiment Ethics Committee of Henan University of Chinese Medicine approved this study. All animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals (US National Institutes of Health, Bethesda, Maryland, US). Next, we injected 100 µL control and miR-125b mimic–transfected BGC-823 cells (1×107/mL) intraperitoneally into mice, and we recorded tumor growth and volume weekly for 3 weeks. To determine the location, growth and metastasis of tumors in vivo, we injected near-infrared fluorescence (NIRF) dye (IR-783) into mouse veins after 7 days and analyzed the fluorescent signal using an animal living imaging system (PerkinElmer, Waltham, Massachusetts, US). Rates of tumor growth and metastasis in the 2 groups of mice were then plotted and analyzed.
Immunohistochemistry (IHC)
All specimens were fixed in neutral buffered formalin and embedded in paraffin. Slides were cut at 5 um, deparaffinized in xylene and rehydrated in graded ethanol. After we blocked nonspecific binding sites by exposing them to 10% BSA in PBS for 20 min, we incubated the sections overnight at 4° C with STAT3 antibodies. Afterward, we rinsed the slides with phosphate-buffered saline (PBS) and incubated them with biotinylated immunoglobulin G (IgG) for 20 min at 37° C. Finally, the sections were slightly counterstained with hematoxylin for 30 s before coverslip mounting.
Statistical Analysis
We analyzed all data with SPSS statistical software version 16.0 (IBM Corp., Armonk, New York, US). All data are expressed as the mean ± standard deviation (SD), and P-values <0.05, <0.01 and <0.001 were established as statistically significant. Each experiment was repeated at least 3 times.
Results
miR-125b Was Downregulated in Gastric Cancer
We analyzed miRNA sequencing (miRNA-Seq) data from 30 frozen gastric-cancer samples to identify the critical miRNAs involved in this cancer. The results showed that miR-125b was significantly lower in gastric-cancer tissues than in matched adjacent normal tissues (Figure 1A). Furthermore, qRT-PCR analysis showed that miR-125b was significantly decreased in the SGC-7901 and BGC-823 gastric-cancer cell lines compared with HEK293 cells (Figure 1B).
miR-125b Inhibited the Proliferation of Gastric-Cancer Cells
To test the inhibitory effect of miR-125b on cells, we treated the cells with miR-125b mimics to generate cells with markedly enhanced miR-125b levels (Figure 2A). Moreover, we conducted a monoclonal-cell assay and MTT experiment. The results showed that cell proliferation was inhibited markedly after miR-125b was overexpressed by mimics. The number of monoclonal cells was considerably less than that in the control group (Figure 2B). After treatment with mimics, SGC-7901 and BGC-823 cell proliferation was significantly inhibited compared with the control group at 12, 24, 48 and 72 h (Figure 2C).
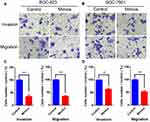
miR-125b Inhibited Invasion and Migration of Gastric-Cancer Cells in vitro
We used invasion and migration assays to test the effect of miR-125b mimics on gastric-cancer invasion and migration in vitro. Toward this end, we compared control, SGC-7901 and BGC-823 cells transfected with miR-125b mimics in Transwell invasion and migration assays 24 h after treatments. We observed that cells transfected with miR-125b-mimics showed a significant reduction in migration and invasion (Figure 3A–D). This suggested that miR-125b inhibited gastric-cancer cell invasion and metastasis.
STAT3 Was the Target of miR-125b
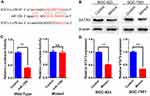
To identify the putative targets of miR-125b, we performed a TargetScan in which the 3′-UTR of STAT3 mRNA was recognized as the potential target of miR-125b (Figure 4A), and we used a dual luciferase reporter assay to validate whether STAT3 was a direct target of miR-125b. We observed that co-transfection of miR-125b mimics remarkably enhanced the luciferase activity of STAT3 3′-UTR reporter, but the luciferase activity of STAT3 reporter with mutated miR-125b binding sites remained basal (Figure 4C and D). This indicated that miR-125b specifically targeted STAT3. Furthermore, we quantified the expression of STAT3 in SGC-7901 and BGC-823 cells before and after treatment with miR-125b mimics and again observed that it was significantly lower in the mimic groups than in the control group (Figure 4B). Together, these results strongly suggested that STAT3’s promotion of invasion and metastasis in gastric cancer depended on its regulation by miR-125b.
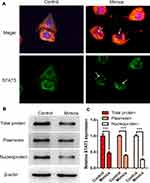
miR-125b Affected the Expression and Subcellular Distribution of STAT3
To explore the molecular mechanism by which miR-125b regulated STAT3, we performed cytoskeleton staining of BGC-823 cells with anti-STAT3 antibody and TRITC-conjugated phalloidin (Figure 5). We observed that gastric-cancer cells treated with miR-125b mimics showed low expression of STAT3 in cytoplasm and especially in nuclei (Figure 5A, arrows). Expression of STAT3 in mimic-treated cells was lower than in control cells (Figure 5A). Western blot analysis demonstrated that treatment with miR-125b mimics decreased STAT3 levels in cytoplasm and especially nuclei of BGC-823 cells (Figure 5B and C). This suggested that as a nuclear transcription factor, miR-125b could inhibit entry of STAT3 into the nucleus.
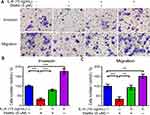
The Expression of STAT3 Directly Affects the Invasion and Metastasis of Gastric Cancer Cells
In order to further verify that miR-125b inhibits the invasion and metastasis of gastric cancer cells by regulating STAT3, we treated gastric cancer cells with STAT3 inhibitor (stattic) and activator (IL-6) respectively to detect their invasion and metastasis. The results showed that the invasion and metastasis of gastric cancer cells were significantly reduced after STAT3 was inhibited. On the contrary, after STAT3 was activated, the invasion and metastasis of gastric cancer cells were significantly enhanced, as shown in Figure 6.
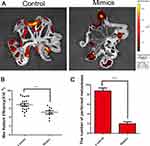
miR-125b Inhibited Metastatic Tumors on the Mesentery in vivo
To confirm the anti-tumor effects of miR-125b and observe the extent of tumor metastasis in the abdominal cavities of mice, BGC-823 cells were treated with either miR-125b mimics or control and then injected into the mice’s peritoneal cavities. We then visualized tumor sizes and distribution using the animal living imaging system (PerkinElmer, Waltham, Massachusetts, US), after which we removed the mesentery and assessed cell migration. The results indicated that peritoneal carcinomatosis was significantly inhibited in the miR-125b mimic group compared with the control group (Figure 7A). The fluorescence signal data and the number of metastases are shown in Figure 7B and C, respectively.
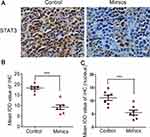
miR-125b Regulated the STAT3 Signaling Pathway in vivo
To further verify the signal transduction pathway mediating miR-125b’s anti-invasion and anti-migration effects in vivo, we prepared xenografts of mice as paraffin-embedded sections. IHC analysis revealed that miR-125b could significantly downregulate expression of STAT3 in vivo (Figure 8A and B). More importantly, it could inhibit STAT3 from entering the nucleus in vivo (Figure 8C).
Discussion
Gastric cancer is a common digestive-system tumor in China.2,3 Invasion and metastasis are the main factors suppressing the survival rate for this cancer, especially in rural areas.6,7 Therefore, it is highly important to find an effective molecular target for the treatment of gastric cancer. In recent years, the role of miRNAs in the pathogenesis of cancer has become increasingly prominent.18–20 A miRNA acts as an oncogene or tumor suppressor gene by regulating the expression of target genes.21,22 In this study, we found that miR-125b significantly inhibited gastric-cancer invasion and metastasis. Expression of miR-125b was stably inhibited in gastric-cancer tissues; therefore, we speculated that miR-125b might hinder tumor development—that is to say, it could have anti-tumor effects. We verified the above speculation preliminarily by analyzing clinical specimens of gastric cancer and finding that miR-125b was stably expressed at a low level in gastric-cancer cells. Compared with the normal cell line HEK293, miR-125b was also expressed at low levels in gastric-cancer cell lines BGC-823 and SGC-7901. To further confirm its role in gastric cancer, we treated BGC-823 and SGC-7901 cells with miR-125b-mimics to induce them to overexpress miR-125b. Cell clone formation and MTT experiments showed that miR-125b markedly inhibited the proliferation of gastric-cancer cells. In addition, a Transwell experiment showed that miR-125b could significantly reduce the number of gastric-cancer transmembrane cells. This implied that miR-125b had an obvious inhibitory effect on cell invasion and metastasis.
As we all know, STAT3 is an important transcription factor. In the physiological state, it plays a crucial role in the physiological function of normal cells.23 STAT3 overactivation induces abnormal expression of genes associated with cell proliferation, migration and apoptosis.24 Activating the STAT3 pathway promotes cell proliferation, malignant transformation, inhibition of cellular apoptosis and the invasion and metastasis of tumor cells.25 Through target gene prediction and a dual-luciferase reporter gene experiment, we determined and validated that STAT3 was the direct target gene of miR-125b. By analyzing clinical samples, we found that as a target of miR-125b, STAT3 was stably overexpressed in gastric-cancer tissues. To further reveal the molecular mechanism underlying miR-125b inhibition of tumor invasion and metastasis via the STAT3 pathway, we performed cell IF and cytoskeletal staining. The results provided new evidence that miR-125b inhibited STAT3 expression and entry into the nucleus. Taken together, these findings showed that STAT3 was indeed involved in the malignant behavior of gastric cancer (including invasion and metastasis) and that miR-125b could regulate the STAT3 pathway. Western blot experiments further confirmed the regulatory effect of miR-125b on STAT3 protein: miR-125b could downregulate STAT3 expression levels and inhibit STAT3 from entering the nucleus. Taken together, the abovementioned experimental results illustrated that miR-125b inhibited gastric-cancer cell invasion and migration by inhibiting the expression of STAT3 and its entry into the nucleus, and then inhibited cell invasion and metastasis. Our experiment with peritoneal metastases in nude mice proved the above results in vivo.
In summary, we demonstrated that miR-125b inhibited gastric-cancer cell invasion and migration by inhibiting (a) STAT3 expression and entry into the nucleus, (b) STAT3’s function as a nuclear transcription factor and (c) cancer cell invasion and metastasis. We expect miR-125b to become a new target for prognosis and treatment of gastric cancer. This study provides a potential target and experimental basis for the development of molecule-targeted drugs for gastric cancer.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Wanqing C, Zheng R, Baade PD, et al. Cancer statistics in China. CA Cancer J Clin. 2015;6:115–132.
2. Li X, Deng Y, Tang W, et al. Urban-rural disparity in cancer incidence, mortality, and survivals in Shanghai, China, during 2002 and 2015. Front Oncol. 2018;8:579. doi:10.3389/fonc.2018.00579
3. Zheng RS, Sun KX, Zhang SW, et al. Report of cancer epidemiology in China, 2015. Zhonghua Zhong Liu Za Zhi. 2019;41(1):19–28. doi:10.3760/cma.j.issn.0253-3766.2019.01.005
4. Yang Z, Zeng H, Xia R, et al. Annual cost of illness of stomach and esophageal cancer patients in urban and rural areas in China: A multi-center study. Chin J Cancer Res. 2018;30(4):439–448. doi:10.21147/j.issn.1000-9604.2018.04.07
5. Zong L, Abe M, Seto Y, et al. The challenge of screening for early gastric cancer in China. Lancet. 2016;388(10060):2606. doi:10.1016/S0140-6736(16)32226-7
6. Pasechnikov V, Chukov S, Fedorov E, et al. Gastric cancer: prevention, screening and early diagnosis. World J Gastroenterol. 2014;20(38):13842–13862. doi:10.3748/wjg.v20.i38.13842
7. Strong VE, Wu AW, Selby LV, et al. Differences in gastric cancer survival between the U.S. and China. J Surg Oncol. 2015;112(1):31–37. doi:10.1002/jso.23940
8. Roberts AP, Jopling CL. Targeting viral infection by microRNA inhibition. Genome Biol. 2010;11:201–204. doi:10.1186/gb-2010-11-1-201
9. Baumjohann D, Ansel KM. MicroRNA-mediated regulation of T helper cell differentiation and plasticity. Nat Rev Immunol. 2013;13(666678):666–678. doi:10.1038/nri3494
10. Sayed D, Abdellatif M. MicroRNAs in development and disease. Physiol Rev. 2011;91:827–887. doi:10.1152/physrev.00006.2010
11. Carrington JC, Ambros V. Role of microRNAs in plant and animal development. Science. 2003;301:336–338. doi:10.1126/science.1085242
12. Hobert O. Gene regulation by transcription factors and microRNAs. Science. 2008;319:1785–1786. doi:10.1126/science.1151651
13. Blahna MT, Hata A. Regulation of miRNA biogenesis as an integrated component of growth factor signaling. Curr Opin Cell Biol. 2013;25:233–240. doi:10.1016/j.ceb.2012.12.005
14. Di Leva G, Garofalo M, Croce CM. MicroRNAs in cancer. Annu Rev Pathol. 2014;9:287–314.
15. Chen Z, Liu X, Hu Z, et al. Identification and characterization of tumor suppressor and oncogenic miRNAs in gastric cancer. Oncol Lett. 2015;10:329–336. doi:10.3892/ol.2015.3179
16. Lovat F, Valeri N, Croce CM. MicroRNAs in the pathogenesis of cancer, Semin. Oncol. 2011;3:724–733.
17. Lu Y, Zhao X, Liu Q, et al. lncRNA MIR100HG-derived miR-100 and miR-125b mediate cetuximab resistance via Wnt/β-catenin signaling. Nat Med. 2017;23(11):1331–1341. doi:10.1038/nm.4424
18. Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017;16(3):203–222. doi:10.1038/nrd.2016.246
19. Lin S, Gregory RI. MicroRNA biogenesis pathways in cancer. Nat Rev Cancer. 2015;15(6):321–333. doi:10.1038/nrc3932
20. Ling H, Fabbri M, Calin GA. MicroRNAs and other non-coding RNAs as targets for anticancer drug development. Nat Rev Drug Discov. 2013;12(11):847–865. doi:10.1038/nrd4140
21. Di Leva G, Garofalo M, Croce CM. MicroRNAs in cancer. Annu Rev Pathol. 2014;9:287–314. doi:10.1146/annurev-pathol-012513-104715
22. Hermeking H. MicroRNAs in the p53 network: micromanagement of tumour suppression. Nat Rev Cancer. 2012;12(9):613–626. doi:10.1038/nrc3318
23. Yu H, Lee H, Herrmann A, Buettner R, Jove R. Revisiting STAT3 signalling in cancer: new and unexpected biological functions. Nat Rev Cancer. 2014;14(11):736–746. doi:10.1038/nrc3818
24. Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9(11):798–809. doi:10.1038/nrc2734
25. Khandekar MJ, Cohen P, Spiegelman BM. Molecular mechanisms of cancer development in obesity. Nat Rev Cancer. 2011;11(12):886–895. doi:10.1038/nrc3174
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.