Back to Journals » Journal of Multidisciplinary Healthcare » Volume 13
The Applicability of the Patient-Specific Functional Scale (PSFS) in Rehabilitation for Patients with Acquired Brain Injury (ABI) – A Cohort Study
Authors Evensen J , Soberg HL , Sveen U, Hestad KA , Bronken BA
Received 21 April 2020
Accepted for publication 24 August 2020
Published 9 October 2020 Volume 2020:13 Pages 1121—1132
DOI https://doi.org/10.2147/JMDH.S259151
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Janne Evensen,1 Helene Lundgaard Soberg,2,3 Unni Sveen,2,3 Knut A Hestad,4,5 Berit Arnesveen Bronken4
1Department of Physical Medicine and Rehabilitation, Innlandet Hospital Trust, Gjøvik, Norway; 2Faculty of Health Sciences, Oslo Metropolitan University, Oslo, Norway; 3Department of Physical Medicine and Rehabilitation, Oslo University Hospital, Oslo, Norway; 4Department of Health and Nursing Sciences, Faculty of Health- and Social Sciences, The Inland Norway University of Applied Sciences, Elverum, Norway; 5Department of Research, Innlandet Hospital Trust, Brumunddal, Norway
Correspondence: Janne Evensen
Department of Physical Medicine and Rehabilitation, Innlandet Hospital Trust, Gjøvik 2819, Norway
Tel +47 61 11 75 00
Email [email protected]
Aim: The primary aim of this study was to investigate the applicability of the Patient-Specific Functional Scale (PSFS) in patients with acquired brain injury (ABI) admitted to a specialized rehabilitation unit in a regional hospital. A secondary aim was to identify patient characteristics and functioning that predicted changes in the PSFS.
Patients and Methods: In a cohort study, 59 patients with ABI were assessed for the ability to complete the PSFS. A trained multidisciplinary team applied the PSFS as part of a collaborative development of rehabilitation goals. The modified Rankin Scale (mRS), the Functional Ambulation Categories (FAC), the Rivermead Behavioural Memory Test (RBMT), the Norwegian Basic Aphasia Assessment (NBAA) and the Loewenstein Occupational Therapy Cognitive Assessment (LOTCA) were used to identify characteristics of the sample. Multivariate regression analyses were performed to investigate associations between changes in the PSFS score from admission to discharge and a selected set of participant baseline characteristics and functioning.
Results: Fifty-four patients (92%) of the patients with ABI were able to complete the PSFS. The five (8%) who were unable to complete the PSFS had severe cognitive or language impairment. The PSFS score improved by a mean of 2.6 (SD 2.0) points from admission to discharge. The LOTCA score made the strongest unique contribution to explain the change in the PSFS score (beta = 0.477, p= 0.020).
Conclusion: In the present study, most patients with ABI (92%) were able to complete the PSFS. Cognitive function on admission was a predictor of improved functioning on the PSFS.
Keywords: multidisciplinary rehabilitation, patient-specific outcome measure, patient-identified goals, shared decision making
Plain Language Summary
Patient-identified goals are recommended to be documented and monitored in rehabilitation, and an outcome measure is needed for that purpose. The Patient-Specific Functional Scale (PSFS) is helpful to identify the patients` goals because the patients are involved in the identification and rating of their own goals. In the PSFS, the patients rate their problems in functioning on a scale from 0 to 10 where zero is “unable to perform the activity” and 10 is “performs the activity without difficulties or at the same level as before the illness.” The rehabilitation goals were set based on the PSFS.
The aim was to investigate the applicability of the PSFS for patients with acquired brain injury admitted to a specialized rehabilitation unit in a regional hospital. We also wanted to identify patient characteristics and functioning that may predict changes in self-reported functioning. The health professional applied the PSFS as part of a collaborative development of rehabilitation goals.
The results showed that almost all (54 of 59) patients were able to complete the PSFS. The five patients who were unable to complete the PSFS had severe cognitive or language impairment. The patients` cognitive function made the strongest contribution in explaining changes in the PSFS score. Patients with higher cognitive function improved significantly more on the PSFS. The results indicate that the PSFS may be a useful tool to document and monitor goals in rehabilitation for patients with acquired brain injury.
Introduction
Goal setting is considered a key component of rehabilitation,1 and there is growing evidence that rehabilitation is most effective when the goal-setting process is patient-centred.2,3 Evidence suggests that the use of shared decision-making within goal setting may increase confidence and a sense of ownership, thus having a positive impact on the patient’s rehabilitation process and increasing their motivation to achieve their goals.1,4
Survival rates and life expectancy have increased for individuals with acquired brain injury (ABI).5,6 Stroke survivors constitute the largest group of individuals with ABI, and approximately 60.000 live with a post stroke condition in Norway.6 Traumatic brain injury (TBI) and subarachnoid hemorrhage (SAH) also constitute a large number of the ABI group. Patients with ABI may have common challenges such as paresis, visual impairment, reduced cognitive function and language impairment. Emotional symptoms and psychosocial challenges are also common.7 These functional changes have been shown to have an impact on the functional outcomes over time.8,9 Person-centred rehabilitation performed by a multidisciplinary team is required in ABI-rehabilitation.7
The aim of rehabilitation is to ensure that people can realize their functional potential in the environments in which they live and work.10 Assessments based on outcome measures are required to detect functional change and to evaluate the benefits of rehabilitation for individuals with ABI. Patient-reported outcome measures (PROMs) are recommended to capture the patients` experience,11 most often with predefined and standardized questions. Patient-specific outcome measures are a sub-category of PROMs that do not consist of standardized questions but enable patients to identify their own problems and goals and to rate their current level of functioning.12
A clinical practice guideline containing a core set of outcome measures for adults with neurological conditions undergoing rehabilitation was developed by Moore et al.11 They emphasized that clinicians should document patient-identified goals and monitor changes using a relevant outcome measure. Patient-specific outcome measures are helpful to identify the patients' goals because the patients are actively involved in the identification and rating of their problems, whereupon individualized treatment goals can be set.12 Furthermore, patient-specific outcome measures may be more sensitive to change than measures with standardized questions.13,14 The Patient-Specific Functional Scale (PSFS) was developed for eliciting and recording the patients` functional problems.15 The patients identify important activities they have difficulties performing due to injury or illness, and they rate their current level of functioning associated with each activity. The activities identified in the PSFS are then redefined as the patients` goals for the rehabilitation. The PSFS was intended to be applicable to a large number of clinical profiles.15 The PSFS is widely used to monitor changes in patient-identified goals for individuals with musculoskeletal disorders.12,17
Regarding the measurement properties of the PSFS, Stratford et al15 found the average of the minimum clinically important difference scores for 3 activities to be 0.8 points (“small change”), 3.2 points (“medium change”), and 4.3 points (“large change”) in patients with chronic low back pain. The PSFS has been translated into Norwegian,18 and has been tested for reliability and responsiveness in patients with musculoskeletal disorders in primary care in Norway. It was found that a 2-point change in the PSFS score can be considered clinically and statistically relevant.19 Horn et al have summarized peer-reviewed literature on the reliability, validity and responsiveness of the PSFS.20 The PSFS is found to have satisfactory measurement properties in individuals with different types of musculoskeletal disorders. They emphasized that there also is potential for use of the PSFS for patients with neurological condition, and one study that investigated PSFS on patients with multiple sclerosis found satisfactory concurrent validity.21 Patient-specific measurements such as the Canadian occupational performance measure (COPM)41,42 involve the identification of activities and rating of activities on a numeric rating scale similar to the PSFS. In a review article it was found that the test-retest reliability of the COPM in patients with stroke was high (r = 0.89), and discriminant validity was confirmed.22,23 The PSFS has been used in studies as an outcome measure to evaluate the impact of rehabilitation in primary health care for individuals with neurological conditions.21,24,25 It has been claimed that disturbances of consciousness, cognition and language impairment could be an obstruction to ascertaining the patients’ goals11 and to rating their own activity problems or goals.26,27 Individuals with cognitive or language impairment have systematically been excluded from studies that used the PSFS as an outcome measure.24,25 One study included patients with different diseases affecting the peripheral or the central nervous system. The authors found that the PSFS was easy to administer and easily understood by patients. However, patients with moderate to severe cognitive or language impairments were excluded.24 A study conducted in primary care in Copenhagen, including 1092 people aged 52–97 years with various diagnoses,26 recommended the PSFS as an useful and effective measure in rehabilitation practice. None of these studies investigated whether patients with ABI suffering from different levels of language or other cognitive impairment were able to complete the PSFS, but rather emphasized that cognitive impairment could be an obstruction to the application of the PSFS. Thus, there is a paucity of knowledge regarding the applicability of the PSFS in patients with ABI suffering of different levels of language or other cognitive impairment. Moreover, specialized rehabilitation in a regional hospital provides service to patients in an earlier stage after an ABI diagnosis than rehabilitation services in primary health care. Thus, more knowledge about the applicability of the PSFS in specialized rehabilitation in a hospital is a need.
The PSFS has been applied in rehabilitation where some of the patients had an ABI diagnose, although the measurement has not been validated for this target group.20 Therefore, the first step in a validation process to investigate if the PSFS is a suitable measurement for patients with ABI is to investigate its applicability.12,28 Applicability is defined by pragmatic quality criteria allowing the use of a measurement with a given population or in a specific context. The applicability is good when the respondent and the examiner burden are minimal, score distribution is adequate and the format is compatible with the respondents` age, culture, language and abilities.29 Some of these aspects of the PSFS are known from previous research; it can be used across ages and levels of disability severity and by various professions across different levels of health services.24 It requires few resources regarding training and procurement of equipment.16 However, the applicability of the PSFS in patients with ABI often at risk of language and cognitive impairments is still unresolved.
Therefore, the primary aim of this study was to investigate the applicability of the PSFS in patients with ABI admitted to a specialized rehabilitation unit in a regional hospital by determining: (1) The proportion of patients with ABI who were able to complete the PSFS, (2) The extent to which individuals with language or other cognitive impairment, or individuals in an early phase after a brain injury were able to complete the PSFS, (3) The time used for the collaborative development of the rehabilitation goals and completion of the PSFS. A secondary aim was to identify patient characteristics and functioning that predicted changes in the PSFS.
Participants and Methods
Study Design, Participants and Setting
This was a cohort study conducted at a specialized rehabilitation unit in a regional hospital in Norway. Patients with ABI admitted from October 2016 to April 2017 for a stay longer than 10 days were asked to participate. Participants were included in the study based on the following criteria: (1) diagnosis of stroke, subarachnoid haemorrhage (SAH) or traumatic brain injury (TBI) and (2) ability to give their consent to participate. Patients were excluded if (1) they could not communicate in Norwegian language and (2) presence of a progressive neurological disease because an aggravation of the disease may affect the rehabilitation outcome.
An interdisciplinary model of rehabilitation was applied, and it comprised ten health professionals. Each patient had a primary contact person that coordinated the rehabilitation activities during the hospital stay. The primary contact was a nurse, social worker, occupational therapist or physiotherapist.
Procedures
The PSFS was applied as part of a collaborative development of rehabilitation goals by patients and the primary contact on the second day of the rehabilitation stay. The health professionals had received instruction and training before the introduction of the PSFS at the rehabilitation unit. A local written procedure based upon a Norwegian evidence based guideline regarding shared decision-making in the goal-setting process,30 was developed and made available for the health professionals. The training also included observation of a collaborative development process of rehabilitation goals and completion of the PSFS, practical exercises in term of role-play and supervision. In the local procedure, there are examples demonstrating how long-term goals can be transformed into specific and short term goals, which could be an activity in the PSFS. An example of how the PSFS was used in the collaborative development of goals in this study is provided in the appendix. The patients received a brochure about goal setting in a rehabilitation process. The health professionals assessed whether the patient was able to complete the PSFS based on collaboration between the patients` judgement and the professionals’ clinical judgement.
Measurements
The PSFS was the main outcome variable, and was applied the second day after admission to the rehabilitation unit and at discharge. The patients rated their performance on self-identified activities on a numeric rating scale from 0 to 10 where zero is “unable to perform the activity” and 10 is “performs the activity without difficulties or at the same level as before the injury/illness”. The patients selected a value that best described their current level of performance in each chosen activity. The primary contact conducted the collaborative development of the rehabilitation goals and completion of the PSFS. The time used for the collaborative development of rehabilitation goals and completion of the PSFS was recorded. On discharge, the patients who were able to complete the PSFS at admission rated the chosen activities previously identified. The patients did not see their PSFS admission scores during the discharge assessment.
Members of the multidisciplinary rehabilitation team conducted the test and measurements during the first week of the patients’ rehabilitation stay. To identify the characteristics and functioning of the patients, we utilized five clinical measurements: 1) The modified Rankin Scale (mRS), 2) the Functional Ambulation Categories (FAC), 3) the Rivermead Behavioural Memory Test (RBMT), 4) the Loewenstein Occupational Therapy Cognitive Assessment (LOTCA) and 5) the Norwegian Basic Aphasia Assessment. The measurements are described in the following. The mRS measures functional independence on a 7-point ordinal scale (0 −6). A score of 0 indicates no symptoms, a score of 5 severe disability, being bedridden and requiring constant care, and a score of 6 is being dead.31 Ambulation was measured with the FAC. It describes assistance required to walk, and has a 6-point ordinal scale (0–5). A score of 0 indicates no functional gait and a score of 5 indicates independent gait function.32 The mRS and the FAC have demonstrated excellent reliability, and validity in stroke.33,34 In addition, information about sex, age, diagnosis, length of stay and time post injury was collected from the medical records.
Occupational therapists assessed cognitive skills with the RBMT35 and the LOTCA.36 The RBMT assesses everyday memory problems for individuals with brain injury and has been translated into Norwegian.37 The instrument has 12 subtests and two scales, a profile score (0–24) and a screening score (0–12). The profile score is used in the current study. Each subtest on the profile score is scored 2 points for correct, 1 point partially correct and 0 for incorrect. The profile score is divided into four levels: 24–22 points indicate normal memory, 21–17 points indicate mild memory impairment, 16–10 points indicate moderate memory impairment and 9–0 points indicate severe memory impairment. The LOTCA (Version 2) measures a wide range of basic cognitive functions, and it was developed for individuals with ABI. It contains 26 subtests in six areas: orientation, visual perception, spatial perception, motor praxis, visuomotor organization, thinking operations and a single score for attention and concentration. The subtests are scored on a 4, 5, or 8 points ordinal scale, and all visuomotor organization subtests are timed. The total score of LOTCA is 27–119 (worst to best).38 The LOTCA has been tested for validity and reliability for patients with ABI, and excellent internal consistency and inter-rater reliability for subtests have been reported.36 The measurement has been useful in the early detection of vascular cognitive impairment,39 and it might be suitable for assessing the cognitive ability of post-stroke patients with aphasia.40
Speech pathologists assessed aphasia with the Norwegian Basic Aphasia Assessment (NBAA).41,42 The eight subtests included spontaneous speech (this score was subjective and therefore not included in the total score), auditory comprehension (71 points), repetition,39 speech production,40 reading aloud,20 reading comprehension,22 writing,10 and syntax.6 The total score (aphasia coefficient) is calculated 0–217 points (worst to best). Due to the clinical experience of a ceiling effect on NBAA, patients with mild or no aphasia were not tested. Hence, the patients in this study were categorized into the following two groups: (1) patients with aphasia tested with the NBAA or not tested with the NBAA due to major cognitive problems, (2) Patients with mild aphasia or no symptoms of aphasia (not tested with NBAA). Patients with severe cognitive impairment were not tested with the NBAA (n=2), RBMT (n=17) or the LOTCA (n=17) because it was considered unethical performing the measurements if the patients were exposed to many tasks that they could not manage.43
Analyses
Continuous data are presented by means and standard deviation (SDs), median, range and interquartile range. The categorical variables are presented as frequencies and percentages. A mean score for the PSFS ratings for the 1–5 identified activities at admission and discharge for each patient was calculated. The change in the PSFS mean score was calculated. A paired-samples t-test was conducted to test for significant changes in the PSFS score. P-values ˂ 0.05 were considered statistically significant. The time post injury was divided into three groups; (1) ≤ 9 days after the brain injury, (2) ≥ 10 and ≤ 89 days after the brain injury and (3) ≥ 90 days after the brain injury, because we wanted to investigate if the patients in an early phase after the brain injury were able to complete the PSFS. Further in the analysis, the variable was dichotomized as (1) ≤ 89 and (2) ≥ 90 days44,45 to identify patient characteristics that predicted changes in the PSFS.
Univariate regression analysis was performed to screen for predictor variables that were associated with the change in the PSFS scores. The choice of independent variables used in the univariate analyses was based on a review of the literature and clinical judgement. A multivariate regression analysis was performed using a stepwise backward elimination method to determine which of the independent variables that predicted change in functioning on the PSFS from admission to discharge. Variables with a p value < 0.2 in the univariate analyses were included. Independent variables with an inter-correlation > 0.7 (Pearson r) were not introduced into the multivariate regression model simultaneously. The analysis showed multicollinearity between the LOTCA and the RBMT (Pearson r= 0.758). The LOTCA was chosen for the multiple regression analysis because it assesses a wider range of basic cognitive functions than the RMBT that mainly assesses memory.
Baseline characteristics used in the multiple regression analysis were length of stay, time post injury ≤ 89/≥ 90 days, the LOTCA score. Age and gender were regarded as possible confounders and included in the analysis. Further regression diagnostics were performed to investigate any violation of the assumptions of normality, linearity, multicollinearity and homoscedasticity. Estimated regression coefficients from the univariate and multivariate regression analyses are reported with 95% confidence intervals and p-values. The results are presented with B, standardized beta, R2 and adjusted R2. The SPSS version 25 (Armonk, NY, 2017) was used for the statistical analysis.
Results
In all, 59 patients with ABI were included. Figure 1 shows a flowchart of the participants.
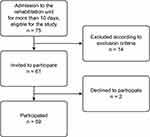 |
Figure 1 Flowchart of the participants. |
The participant baseline characteristics and functioning are reported in Table 1. Their mean age was 66 (SD=11) years, and 64% were men. Of these, 92% suffered from stroke, while the rest were diagnosed with either TBI (3%) or SAH (5%). More than half of the patients (58%) had their brain injury ≥ 10 and ≤ 89 days before the admission to the rehabilitation unit. Only 15% of them had brain injury ≤ 9 days before the admission.
 |
Table 1 Patients’ Characteristics |
The results from the test and measurements are reported in Table 2. Seventeen patients had aphasia. Of these, 15 were tested with NBAA, whereas two patients were not tested because of severe cognitive impairment. Forty-one patients were tested with the LOTCA, with a mean score of 104 points (SD 12.0). Forty-one patients (69%) were tested with RBMT with a mean score of 17.3 (SD 5.4).
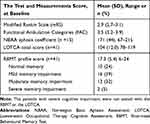 |
Table 2 The Test and Measurements Score |
Fifty-four patients (92%) were able to complete the PSFS. The five patients (8%) who were unable to complete the PSFS identified at least one activity, but they were unable to rate the activities. The patients identified 184 activities with a median of four activities (range 1–5). The characteristics of the patients who completed the PSFS (n=54) and the patients who were unable to complete PSFS (n=5) are presented in Table 3. Fourteen of 17 patients with aphasia were able to complete the PSFS. The lowest score in NBAA for a patient who completed the PSFS was114 of 217 points. Two patients with severe memory impairment and 13 with moderate memory impairment on the RBMT were able to complete the PSFS. Twenty patients with a LOTCA total score below 104 were able to complete the PSFS. The lowest score of a patient completing the PSFS in cognitive functioning on the LOTCA was 78 of 119 points. Nine patients were admitted to the rehabilitation unit during the first 9 days after their brain injury, and all of them were able to complete the PSFS.
 |
Table 3 The Characteristics of the Patients Who Were Able and Unable to Complete the PSFS. Relative Frequencies (%) Based on the 59 Patients |
In total, five patients were unable to complete the PSFS. Three of them had aphasia, two with an aphasia coefficient on the NBAA on 67 and 199 points, respectively. The third with aphasia was not tested with the NBAA due to severe cognitive impairment. Four patients were not tested by the RBMT because of severe cognitive impairment, and one had severe memory impairment. None of these five patients was tested with the LOTCA because of severe cognitive impairment. Mean time used for the collaborative development of rehabilitation goals and completion of the PSFS was 28 (SD 11.0, range 7–75) minutes.
Figure 2 shows the PSFS mean score and SD at admission and at discharge. The PSFS score improved significantly, with a mean change of 2.6 (SD 2.0) points from admission to discharge (p<0.001). Most of the 54 patients (93%) improved on the PSFS, with a mean score of 6.6 (SD 1.9) points at discharge compared to 4.0 (SD 2.2) points at admission.
 |
Figure 2 The PSFS mean score (and SD) at admission and discharge. |
The patient characteristics at baseline that predicted changes in the PSFS from admission to discharge are reported in Table 4. Univariate analyses showed that length of stay, time post injury (≤ 89 or ≥ 90 days), the LOTCA score and the RBMT score were predictors of changes in the PSFS score from admission to discharge with p-values ˂ 0.2. Only for RMBT and LOTCA, the associations were statistically significant.
 |
Table 4 Univariate Regression Results of the Independent Variables Associated with Changes in the PSFS Score from Admission to Discharge |
Results from the multivariate regression model are reported in Table 5. Length of stay and LOTCA total score were significant predictors of change in the PSFS score. The LOTCA made the strongest unique contribution in explaining the variance in the change in functioning on the PSFS with a standardized beta of 0.477, p= 0.020. A ten points increase in cognitive function on the LOTCA gave a 0.85 point higher change score on the PSFS. The contribution of length of stay was also significant (p= 0.041). For every 10 days elongation at the rehabilitation unit, a 0.68 points higher change in functioning on the PSFS was observed. The model explained 24.9% (adjusted R2 = 0. 249) of the change in PSFS score and accounted for a significant amount of the variance in the change in PSFS score from admission to discharge (F=5.413, p= 0.003).
 |
Table 5 Results from the Multivariate Regression Model Testing Factors Predictors of Change in the PSFS Score from Admission to Discharge |
Discussion
The primary aim of this study was to investigate the clinical applicability of PSFS in individuals with ABI. The study shows that 54 (92%) patients with ABI in this sample were able to complete the PSFS, and that the five (8%) who were unable to complete had severe cognitive or language impairment. The patients who failed to complete the PSFS identified at least one activity, but were unable to rate the activity. Furthermore, all of the patients admitted to the rehabilitation unit during the first 9 days after their brain injury were able to complete the PSFS. The mean time spent on the collaborative development of the rehabilitation goals and completion of the PSFS was 28 mins. The improved functioning on the PSFS was between a small and medium change15 of 2.6 (SD 2.0) points from admission to discharge. Cognitive functioning assessed by the LOTCA made the strongest unique contribution to explaining the change in the PSFS score from admission to discharge.
The number of studies that have examined the clinical applicability of the PSFS is limited. To reflect on the clinical applicability of the PSFS, the results in this study will be compared with studies using other patient-specific measurements in goal-setting processes, such as the Canadian occupational performance measure (COPM),46,47 the self-identified goals assessment (SIGA),27 the goal-attainment scale (GAS)48 and the patient-specific complaints (PSC) questionnaire.49 These measurements involve the identification of activities and rating of activities on a numeric rating scale similar to the PSFS.
The results in the present study indicate that patients with different levels of cognitive as well as language impairment may benefit from using the PSFS as a tool to identify personal goals in rehabilitation. Previous studies have not recommended the PSFS for individuals with cognitive or language impairment or for those in the early phase after their brain injury.16 Individuals with cognitive or language impairment have been systematically excluded from previous studies that used the PSFS as an outcome measure.24,25 However, one study where patients with moderate to severe cognitive or language impairments were excluded found that the PSFS was easily understood by those with mild aphasia and mild cognitive impairment.24 Some of the patients in the present study were able to identify activities and rate them despite moderate or even severe cognitive and language impairment. The procedure-incorporating collaborative development of the rehabilitation goals and the completion of the PSFS may have contributed to the high number of patients who were able to complete the PSFS in the present study. This number might have been lower if the patients did not collaborate with a professional; hence, the collaboration contributes to the applicability of PSFS in patients with ABI.
The patients who were unable to complete the PSFS were not able to rate the activities on a scale from 0 to 10. Difficulties with rating have been documented in other studies as well.12,26,27 The literature also suggests that identifying activities can be challenging.26,47 Patients undergoing rehabilitation may have wishes or expectations other than being able to perform activities better. Patients’ goals may involve, for example, pain relief, changing of lifestyle, gaining of knowledge and disease management.50 What is of importance to a patient can be difficult to formulate and score in the PSFS. In the primary care study in Copenhagen, the PSFS was introduced for patients receiving rehabilitation services. One conclusion was that the health professionals should be trained in using the PSFS as part of the guidance of the patient to identify goals that can be rated, without overruling the patients’ own expressed values and goals.26 In the current study, the health professionals received instruction, practical training and a written procedure before the study started, which could have contributed to the high percentage of patients able to complete the PSFS (92%).
Most patients with aphasia were able to complete the PSFS in the present study. Individuals with aphasia have been shown wanting to participate in goal setting, and they are able to express their own goals for rehabilitation given sufficient facilitation for communication.51 In the collaborative development of rehabilitation goals for patients with aphasia, it may be of value for the health professionals to have competence in the use of “Supported Conversation for Adults with Aphasia” (SCA).52 Also, a family member53 or a speech pathologist may be of great help in the collaborative goal setting and completion of the PSFS in this patient group. In the present study, the health professionals were instructed in principles for SCA and they had experience regarding communicating with patients suffering from aphasia. Examples of principles are to give the patients time to answer, speak in short sentence and using key words, pictures or drawings. When the patients had severe aphasia, the speech therapist attended the collaborative goal-setting conversation.
In the present study, the nine patients admitted to the rehabilitation unit during the nine first days after their ABI were able to complete the PSFS. In this phase, the patients may not be aware of their own problems and may not fully understand the consequences of the injury.47 Hence, in a future study investigating the validity and reliability of the PSFS for patients with ABI, focus should also be on patients in an early phase after the brain injury. The injury itself can also lead to reduced self-awareness.54 During the rehabilitation stay, patients may become more aware of their problems. Therefore, some might rate themselves with a discharge score lower than or the same as the admission rating, despite improved functioning. This possibility highlights that in some cases, self-rating tools such as the PSFS may not be sensitive to positive change; however, assessment performed by clinicians or other collateral reports do indicate change.48 A prerequisite for the use of the PSFS is that the patients must be aware of a problem as a consequence of the injury.26 A benefit of using the PSFS is that through discussion and negotiation with the patient, the health professional could obtain useful information about the patients` awareness of their own problems. Hence, the PSFS could be a tool to increase the patients´ self-awareness.49
The time spent on the collaborative development of goals and the completion of the PSFS ranged from seven to 75 mins with a mean time on 28 mins. The database of rehabilitation measures states that it takes 4 mins to fill out the PSFS,16 but this is without the collaborative goal-setting process. In this study, the PSFS was applied as part of a collaborative development of rehabilitation goals, like the literature recommend.4 The time spent on administration of other patient-specific measures in goal setting as COPM and GAS, ranged from 15 to 50 mins.12 Stevens et al12 argue that the time was worthwhile as the goal-setting process also fostered teamwork and a client-centered approach. The same arguments are pertinent in this study. In addition, patients with ABI often need more time to be able to express themselves and their concerns. This supports the use of the mean time of 28 mins as acceptable on this key component of rehabilitation.
Concerning the changes in the PSFS score, the results in the present study showed a change of 2.6 points in the mean score from admission to discharge. In a study, where the impact of community rehabilitation service for individuals with neurological conditions was evaluated, the PSFS score improved by 3 points after 3 months of rehabilitation.24 However, the duration of the rehabilitation period was longer compared to the present study (mean 22 days), and the time post injury was probably longer than in the current study, since it was in a community setting. It is known that the reactive neurobiological recovery of patients with ABI is most significant during the first 3 months after injury onset.44,55 The PSFS change in the current study (mean 2.6 points) can be considered relatively large within the limited time period of the inpatient rehabilitation stay (mean 22 days). In an early phase after the brain injury, some of the change in the PSFS may be explained by reactive neurobiological recovery. In a systematic review20, the minimal clinically important difference (MCID) on the PSFS in a range of conditions was found to be between 2 and 3 points. PSFS responsiveness has yet not been tested in individuals with ABI; hence, future research should test the PSFS for reliability and responsiveness for these patients.
The LOTCA made the strongest unique contribution in explaining the change in PSFS from admission to discharge. One implication of this finding could be that patients with cognitive impairment show less progress when assessed by the PSFS. Previous studies have supported the use of cognitive assessment of patients with stroke to assist in the prediction of outcomes.9,56 In particular, impairment of executive function, attention and processing speed have been found to predict dependence in activities of daily living (ADLs) and home life.8,57 The LOTCA measures a wide range of basic cognitive functions, containing orientation, visual perception, spatial perception, motor praxis, visuomotor organization, thinking operations, attention and concentration. However, the results of the present study do not tell us which forms of cognitive impairment that predicted outcome. To the best of our knowledge, no study on cognitive impairment as a predictor for patient-specific outcome measures has been published. Patient-specific outcome measures such as the PSFS can identify unique problems for each patient that are not assessed by other standardized outcome measures. Therefore, it should be considered an important outcome measure. Future research should investigate different types of cognitive impairments and other basic characteristics as predictors for change in the PSFS.
Limitations and Strengths of the Current Study
The small sample size of this study limits the generalizability of the findings.
Concerning the measurements, LOTCA is validated for patients with ABI,36 but there is not an official and approved Norwegian version. However, the version used in the current study is widely used in rehabilitation after ABI in Norwegian hospitals, and occupational therapists have experienced that the LOTCA is a useful broad screening assessment instrument of cognitive function in patients with stroke and TBI in Norway.58 The LOTCA has been found to be better customized for this target group than the Mini-Mental State Examination (MMSE) and the functional independence measure (FIM) cognitive subscale in predicting functional change after stroke rehabilitation.39,59
Patient-specific measures offer the advantage of identifying the salient issues for each patient and are more likely to focus on only the relevant areas for the patients. One disadvantage that has been put forward is that without standardization of the items, the scales do not measure exactly the same in each patient. In this situation, the numeric score may not convey a common meaning, and the value of analysing the data statistically and calculating parameters such as means and correlations can be questionable.60 However, the PSFS has been found to be an appropriate measure for statistical comparisons in clinical research for patients with musculoskeletal diseases. One study supported the concurrent, convergent, and discriminant validities, scale consistency, distribution, and responsiveness of the PSFS for both between-group discrimination and assessment of change over time in group-level data.61
Conclusion
In the present study, 54 out of 59 patients with ABI (92%) were able to complete the PSFS. All patients managed to identify activities that they had difficulties performing, but 5 patients (8%) were unable to rate the activities due to severe cognitive or language impairments. Cognitive function on admission was found as a predictor of improved functioning, assessed by the PSFS. The results from this study indicate that professionals should not predetermine whether a patient is able to complete the PSFS but introduce the PSFS as part of the goal-setting process for patients with ABI. Further research on a larger scale is required to draw conclusions on the clinical applicability of the PSFS for individuals with ABI.
Ethics
All participants received information about the study and provided written informed consent in accordance with the Declaration of Helsinki. The study was initially considered as a quality improvement project by the Norwegian Regional Committee for Medical Research Ethics in 2016 (2016/1048 A) and was approved by the Data Protection Officer (approval no. 2016/92). Later the study was included as a sub-study of a larger research project, which required a new application for approval. The study was approved by the Regional Committees for Medical and Health Research Ethics in 2020 (reference number 71,114 (0302/2020)).
Acknowledgments
The authors would like to thank the staff at the Rehabilitation unit, where the study was conducted, for their support.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Norwegian Directorate of Health (grant number: 15/4855-11). The writing of this article was funded by Innlandet Hospital Trust (grant number: 150386).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Levack WM, Weatherall M, Hay-Smith EJ, Dean SG, McPherson K, Siegert RJ. Goal setting and strategies to enhance goal pursuit for adults with acquired disability participating in rehabilitation. Cochrane Database Syst Rev. 2015;(7):Cd009727.
2. Plewnia A, Bengel J, Korner M. Patient-centeredness and its impact on patient satisfaction and treatment outcomes in medical rehabilitation. Patient Educ Couns. 2016;99(12):2063–2070. doi:10.1016/j.pec.2016.07.018
3. Sugavanam T, Mead G, Bulley C, Donaghy M, van Wijck F. The effects and experiences of goal setting in stroke rehabilitation - a systematic review. Disabil Rehabil. 2013;35(3):177–190. doi:10.3109/09638288.2012.690501
4. Rose A, Rosewilliam S, Soundy A. Shared decision making within goal setting in rehabilitation settings: a systematic review. Patient Educ Couns. 2017;100(1):65–75. doi:10.1016/j.pec.2016.07.030
5. Ellekjær H, Selmer R. Hjerneslag – like mange rammes, men prognosen er bedre. Tidsskr Nor. 2007;127:740–743.
6. Akerkar R, Bakken IJ. Hjerte- Og Karregisteret. Oslo: Folkehelseinstituttet; 2018.
7. Grabljevec K, Singh R, Denes Z, et al. Evidence based position paper on physical and rehabilitation medicine professional practice for adults with acquired brain injury. The European PRM position (UEMS PRM Section). Eur J Phys Rehabil Med. 2018.
8. Whyte E, Skidmore E, Aizenstein H, Ricker J, Butters M. Cognitive impairment in acquired brain injury: a predictor of rehabilitation outcomes and an opportunity for novel interventions. PM R. 2011;3(6 Suppl 1):S45–51. doi:10.1016/j.pmrj.2011.05.007
9. Wagle J, Farner L, Flekkoy K, et al. Early post-stroke cognition in stroke rehabilitation patients predicts functional outcome at 13 months. Dement Geriatr Cogn Disord. 2011;31(5):379–387. doi:10.1159/000328970
10. World Health Organization. Rehabilitation in Health Systems. Geneva: World Health Organization; 2017.
11. Moore JL, Potter K, Blankshain K, Kaplan SL, O’Dwyer LC, Sullivan JE. A core set of outcome measures for adults with neurologic conditions undergoing rehabilitation: a clinical practice guideline. J Neurol Phys Ther. 2018;42(3):174–220. doi:10.1097/NPT.0000000000000229
12. Stevens A, Beurskens A, Koke A, van der Weijden T. The use of patient-specific measurement instruments in the process of goal-setting: a systematic review of available instruments and their feasibility. Clin Rehabil. 2013;27(11):1005–1019. doi:10.1177/0269215513490178
13. Hall AM, Maher CG, Latimer J, Ferreira ML, Costa LO. The patient-specific functional scale is more responsive than the Roland Morris disability questionnaire when activity limitation is low. Eur Spine J. 2011;20(1):79–86. doi:10.1007/s00586-010-1521-8
14. Pengel LH, Refshauge KM, Maher CG. Responsiveness of pain, disability, and physical impairment outcomes in patients with low back pain. Spine. 2004;29(8):879–883. doi:10.1097/00007632-200404150-00011
15. Stratford P, Gill C, Westaway M. Assessing disability and change on individual patients: a report of a patient specific measure. Physiother Can. 1995;47(4):258–263. doi:10.3138/ptc.47.4.258
16. Shirley Ryan Ability Lab. Rehabilitation measures database; 2013. Available from: https://www.sralab.org/rehabilitation-measures/database.
17. Kjeken I. The BRIDGE rehabilitation trial (BRIDGE) clinical trials.gov. U.S. National Library of Medicine; 2017. Available from: https://clinicaltrials.gov/ct2/show/NCT03102814?term=patient±specific±functional±scale&rank=5.
18. Grotle M, Kjeken I. Patient-Specific Functional Scale, Norwegian Translation. 1996.
19. Moseng T, Tveter AT, Holm I. Pasient-Spesifikk Funksjons Skala - Et nyttig verktøy for fysioterapeuter i primærhelsetjenesten. Nor J Physiother. 2013;(2).
20. Horn KK, Jennings S, Richardson G, Vliet DV, Hefford C, Abbott JH. The patient-specific functional scale: psychometrics, clinimetrics, and application as a clinical outcome measure. J Orthop Sports Phys Ther. 2012;42(1):30–42. doi:10.2519/jospt.2012.3727
21. Hammer A, Nilsagard Y, Forsberg A, Pepa H, Skargren E, Oberg B. Evaluation of therapeutic riding (Sweden)/hippotherapy (United States). A single-subject experimental design study replicated in eleven patients with multiple sclerosis. Physiother Theory Pract. 2005;21(1):51–77. doi:10.1080/09593980590911525
22. Cup EH, Scholte Op Reimer WJM, Thijssen MCE, van Kuyk-minis MAH. Reliability and validity of the Canadian occupational performance measure in stroke patients. Clin Rehabil. 2003;17(4):402–409. doi:10.1191/0269215503cr635oa
23. Yang SY, Lin CY, Lee YC, Chang JH. The Canadian occupational performance measure for patients with stroke: a systematic review. J Phys Ther Sci. 2017;29(3):548–555. doi:10.1589/jpts.29.548
24. Barker RN, Sealey CJ, Polley ML, Mervin MC, Comans T. Impact of a person-centred community rehabilitation service on outcomes for individuals with a neurological condition. Disabil Rehabil. 2017;39(11):1136–1142. doi:10.1080/09638288.2016.1185803
25. Dean SG, Poltawski L, Forster A, et al. Community-based rehabilitation training after stroke: results of a pilot randomised controlled trial (ReTrain) investigating acceptability and feasibility. BMJ Open. 2018;8(2):e018409. doi:10.1136/bmjopen-2017-018409
26. Schnohr CW, Damkjær L, Bjørner JB. Oversættelse og validering af en dansk udgave af PSFS (Patient Specifik Funktionel Skala) – et nyttigt værktøj for terapeuter. Fysioterapeuten. 2016;01.
27. Melville LL, Baltic TA, Bettcher TW, Nelson DL. Patients’ perspectives on the self-identified goals assessment. Am J Occup Ther. 2002;56(6):650–659. doi:10.5014/ajot.56.6.650
28. de Vet HC, Knol DL, Terwee CB, Mokkink LB. Measurement in Medicine: A Practical Guide. Cambridge: Cambridge University Press; 2010.
29. Auger C, Demers L, Swaine B. Making sense of pragmatic criteria for the selection of geriatric rehabilitation measurement tools. Arch Gerontol Geriatr. 2006;43(1):65–83. doi:10.1016/j.archger.2005.09.004
30. Evensen J, Skøien R, Sareneva E, et al. Rehabilitering. Samvalg I Målavklaringsprosessen; Kunnskapsbasert Fagprosedyre. Innlandet Hospital Trust, editor. Helsebiblioteket; 2015.
31. Rankin J. Cerebral vascular accidents in patients over the age of 60. III. Diagnosis and treatment. Scott Med J. 1957;2(6):254–268.
32. Holden MK, Gill KM, Magliozzi MR, Nathan J, Piehl-Baker L. Clinical gait assessment in the neurologically impaired. Phys Ther. 1984;64(1):35–40.
33. Mehrholz J, Wagner K, Rutte K, Meissner D, Pohl M. Predictive validity and responsiveness of the functional ambulation category in hemiparetic patients after stroke. Arch Phys Med Rehabil. 2007;88(10):1314–1319. doi:10.1016/j.apmr.2007.06.764
34. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007;38(3):1091–1096. doi:10.1161/01.STR.0000258355.23810.c6
35. Wilson B, Cockburn J, Baddeley A, Hiorns R. The development and validation of a test battery for detecting and monitoring everyday memory problems. J Clin Exp Neuropsychol. 1989;12(11(6)):855–870. doi:10.1080/01688638908400940
36. Katz N, Itzkovich M, Averbuch S, Elazar B. Loewenstein occupational therapy cognitive assessment (LOTCA) battery for brain-injured patients: reliability and validity. Am J Occup Ther. 1989;43(3):184–192. doi:10.5014/ajot.43.3.184
37. Wilson BA, Cockburn J, Baddeley AD. The rivermead behavioural memory test: håndbok - voksne. Namsos. 1997;32.
38. Katz N, Itzkovich M, Averbuch S. The loewenstein occupational therapy cognitive assessment. Arch Phys Med Rehabil. 2002;83(8):1179. doi:10.1016/S0003-9993(02)70008-7
39. Wang SY, Gong ZK, Sen J, Han L, Zhang M, Chen W. The usefulness of the loewenstein occupational therapy cognition assessment in evaluating cognitive function in patients with stroke. Eur Rev Med Pharmacol Sci. 2014;18(23):3665–3672.
40. Yu ZZ, Jiang SJ, Li J, et al. Clinical application of loewenstein occupational therapy cognitive assessment battery-second edition in evaluating of cognitive function of Chinese patients with post-stroke aphasia. Chin Med Sci J. 2013;28(3):167–171.
41. Sundet K, Finset A, Reinvang I. Neuropsychological predictors in stroke rehabilitation. J Clin Exp Neuropsychol. 1988;10(4):363–379. doi:10.1080/01688638808408245
42. Reinvang I. Aphasia and Brain Organization. New York, Boston, MA: Springer, Plenum Press; 1985.
43. Krohne K, Slettebo A, Bergland A. Cognitive screening tests as experienced by older hospitalised patients: a qualitative study. Scand J Caring Sci. 2011;25(4):679–687. doi:10.1111/j.1471-6712.2011.00878.x
44. Hebert D, Lindsay MP, McIntyre A, et al. Canadian stroke best practice recommendations: stroke rehabilitation practice guidelines, update 2015. Int J Stroke. 2016;11(4):459–484. doi:10.1177/1747493016643553
45. Duncan PW, Lai SM, Keighley J. Defining post-stroke recovery: implications for design and interpretation of drug trials. Neuropharmacology. 2000;39(5):835–841. doi:10.1016/S0028-3908(00)00003-4
46. Tuntland H, Aaslund MK, Langeland E, Espehaug B, Kjeken I. Psychometric properties of the Canadian occupational performance measure in home-dwelling older adults. J Multidiscip Healthc. 2016;9:411–423. doi:10.2147/JMDH.S113727
47. Wressle E, Marcusson J, Henriksson C. Clinical utility of the Canadian occupational performance measure–Swedish version. Can J Occup Ther. 2002;69(1):40–48. doi:10.1177/000841740206900104
48. Doig E, Fleming J, Kuipers P, Cornwell PL. Clinical utility of the combined use of the Canadian occupational performance measure and goal attainment scaling. Am J Occup Ther. 2010;64(6):904–914.
49. Stevens A, Moser A, Koke A, van der Weijden T, Beurskens A. The patient’s perspective of the feasibility of a patient-specific instrument in physiotherapy goal setting: a qualitative study. Patient Prefer Adherence. 2016;10:425–434.
50. Berdal G, Sand-Svartrud AL, Bo I, et al. Aiming for a healthier life: a qualitative content analysis of rehabilitation goals in patients with rheumatic diseases. Disabil Rehabil. 2018;40(7):765–778. doi:10.1080/09638288.2016.1275043
51. Berg K, Askim T, Balandin S, Armstrong E, Rise MB. Experiences of participation in goal setting for people with stroke-induced aphasia in Norway. A qualitative study. Disabil Rehabil. 2017;39(11):1122–1130. doi:10.1080/09638288.2016.1185167
52. Kagan A, Black SE, Duchan FJ, Simmons-Mackie N, Square P. Training volunteers as conversation partners using “supported conversation for adults with aphasia” (SCA): a controlled trial. J Speech Lang Hear Res. 2001;44(3):624–638.
53. Levack WM, Siegert RJ, Dean SG, McPherson KM. Goal planning for adults with acquired brain injury: how clinicians talk about involving family. Brain Inj. 2009;23(3):192–202. doi:10.1080/02699050802695582
54. Sinanović O. Neuropsychology of acute stroke. Psychiatr Danub. 2010;22(2):278–281.
55. Winters C, Kwakkel G, Eeh VW, Nijland RHM, Veerbeek JM, Meskers CGM. Moving stroke rehabilitation forward: the need to change research. NeuroRehabilitation. 2018;43(1):19–30. doi:10.3233/NRE-172393
56. Larson EB, Kirschner K, Bode RK, Heinemann AW, Clorfene J, Goodman R. Brief cognitive assessment and prediction of functional outcome in stroke. Top Stroke Rehabil. 2003;9(4):10–21. doi:10.1310/84YN-Y640-8UEQ-PDNV
57. Park SH, Sohn MK, Jee S, Yang SS. The characteristics of cognitive impairment and their effects on functional outcome after inpatient rehabilitation in subacute stroke patients. Ann Rehabil Med. 2017;41(5):734–742. doi:10.5535/arm.2017.41.5.734
58. Leknes R, Grue R, Næss T, Lillefjell M. Erfaringer med bruk av kartleggingsverktøyet LOTCA. Ergoterapeuten. 2016;59(6):36–45.
59. Zwecker M, Levenkrohn S, Fleisig Y, Zeilig G, Ohry A, Adunsky A. Mini-mental state examination, cognitive FIM instrument, and the loewenstein occupational therapy cognitive assessment: relation to functional outcome of stroke patients. Arch Phys Med Rehabil. 2002;83(3):342–345. doi:10.1053/apmr.2002.29641
60. Jolles BM, Buchbinder R, Beaton DE. A study compared nine patient-specific indices for musculoskeletal disorders. J Clin Epidemiol. 2005;58(8):791–801. doi:10.1016/j.jclinepi.2005.01.012
61. Abbott JH, Schmitt JS. The patient-specific functional scale was valid for group-level change comparisons and between-group discrimination. J Clin Epidemiol. 2014;67(6):681–688. doi:10.1016/j.jclinepi.2013.11.002
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
