Back to Journals » OncoTargets and Therapy » Volume 14
TEAD4 as a Prognostic Marker Promotes Cell Migration and Invasion of Urinary Bladder Cancer via EMT
Authors Huang Z , Yan Y, Tang P, Cai J, Cao X, Wang Z, Zhang F, Shen B
Received 9 November 2020
Accepted for publication 7 January 2021
Published 10 February 2021 Volume 2021:14 Pages 937—949
DOI https://doi.org/10.2147/OTT.S290425
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Alberto Bongiovanni
Zhengnan Huang,1,* Yilin Yan,1,* Pengfei Tang,2 Jinming Cai,1 Xiangqian Cao,1 Zeyi Wang,2 Fang Zhang,1 Bing Shen1,2
1Department of Urology, Shanghai General Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, 200080, People’s Republic of China; 2Department of Urology, Shanghai General Hospital Affiliated to Nanjing Medical University, Shanghai, 200080, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Bing Shen; Fang Zhang
Department of Urology, Shanghai General Hospital, Shanghai Jiaotong University School of Medicine, No. 100 Haining Road, Hongkou District, Shanghai, 200080, People’s Republic of China
Tel +86-021-5213-0011-361; +86-21-63240090-2514
Email [email protected]; [email protected]
Purpose: As a member of TEA Domain Transcription Factors (TEADs), TEAD4 was found to be upregulated in urinary bladder cancer (UBC). This study focused on investigating the clinical value and potential functions of TEAD4 in UBC.
Materials and Methods: Patients’ samples, TCGA-BLCA and multiple GEO datasets were applied to explore the expression pattern of TEAD4 in UBC. Cox regression and Kaplan–Meier survival analyses were carried out to evaluate the prognostic significance of TEAD4 in UBC. Wound healing and transwell assays were performed to explore the biological functions of TEAD4 in UBC cells.
Results: The results of TCGA-BLCA, GEO datasets, Western blotting and immunohistochemistry staining (IHC) indicated that TEAD4 was strikingly elevated in UBC tissues as compared to their normal counterparts, and upregulation of TEAD4 was significantly correlated with clinical stage, pathological grade and poor clinical outcome. Functional studies demonstrated that TEAD4 knockdown suppressed cell migration and invasion by reducing the expression of epithelial-mesenchymal transition (EMT) related markers and transcription regulators.
Conclusion: Our results suggest that TEAD4 may serve as a novel prognostic biomarker and a promising therapeutic target for UBC, and act as a pro-tumorigenic gene to promote cell migration and invasion by inducing EMT.
Keywords: TEAD4, urinary bladder cancer, prognostic biomarker, migration and invasion, EMT
Introduction
Urinary bladder cancer (UBC) is one of the most common malignant tumors in the urinary system, with the characteristics of easy metastasis and high recurrence.1,2 Although various advancements in surgical treatments and medical therapies, approximately half of patients with UBC develop metastasis or recurrence within 2 years of diagnosis.3 Major prognostic factors include advanced TNM stage, pathological grade and cervical lymph node metastasis.4 Therefore, apart from elucidating the metastasis mechanisms of UBC, it is imperative to identify new prognostic biomarkers for early detection and keep watch over treatment effectiveness in UBC patients.
Previous studies have recognized the Hippo signaling pathway as a crucial and indispensable regulator in multiple biological processes, including organ size control, cell fate determination, cell proliferation and tumor suppression.5–7 By repressing YAP and TAZ transcription co-activators, the Hippo pathway exerts its important regulatory roles in response to multiple intracellular and extracellular signals.8,9 Once the hippo signaling is dysregulated, YAP and TAZ will be activated and accumulate in the nucleus, and subsequently drive gene transcription mainly through forming complexes with TEA domain transcription factors (TEAD1–4).10–13 The family members contain a TEA domain for the binding with DNA elements and a transactivation domain for the interaction with transcription coactivators which were required for activation of TEADs as transcription factors.14,15 Increasing evidence supported that TEAD transcription factors not only participate in a variety of biological process, but also play essential roles in tumor initiation and progression.16–18 TEADs had been reported to promote cancer development and progression by binding with or without YAP/TAZ in multiple solid tumors, including cholangiocarcinoma,19 colorectal cancer,20,21 breast22 and gastric cancers.23 These data intrigued us to examine the expression pattern of TEADs in UBC and to explore whether they are correlated with UBC.
Through the analysis of TEADs expression between UBC samples and their normal counterparts in TCGA-BLCA and GSE13507 datasets, we found that TEAD4 was the only upregulated gene among four TEAD family members. We also investigated the correlations of its overexpression with clinicopathological parameters and prognosis. Further functional studies were performed to examine whether TEAD4 overexpression was required for cell aggressiveness.
Materials and Methods
Gene Expression Profiling
The RNA sequencing data was downloaded from the Cancer Genome Atlas (TCGA) dataset. The mRNA expression level of TEAD4 was conversed with Log (FPKM). Other public microarray data including GSE13507, GSE31684, GSE32584, GSE32894 and GSE48075 were downloaded from the Gene Expression Omnibus (GEO) dataset. Clinical data on the UBC specimen downloaded from TCGA and GEO datasets is listed in Supplementary Table 1.
Patients and Tissue Specimens
Sixteen pairs of fresh UBC tissues and adjacent nontumorous tissues were acquired from the patients at the time of surgery from the Shanghai General Hospital. A cohort of 50 paraffin-embedded UBC tissues and relevant clinical information were also collected. All patients gave informed consent, and the study protocol acquired official approval by the ethics committees of the Shanghai General Hospital. Clinical data on the UBC specimen used is listed in Supplementary Tables 2 and 3.
Cell Culture
The normal urothelial cell (SV-HUC-1) and UBC cell lines (J82, T24, 5637, SW780, UMUC-3 and SCaBER) were bought from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). All cells were cultured in RPMI 1640 medium (Hyclone) supplemented with 10% FBS at 37°C with 5% CO2.
TEAD4 Knockdown
The siRNA sequences including TEAD4 siRNA-1 (5′-GAA CGU CCC AUG AUG UGA ATT-3′, 5′-UUC ACA UCA UGG GAC GUU CTT-3′), TEAD4 siRNA-2 (5′-GAC AGA GUA UGC UCG CUA UTT-3′,5′-AUA GCG AGC AUA CUC UGU CTT-3′) and negative control siRNA (5′-UUC UCC GAA CGU GUC ACG UTT-3′, 5′-ACG UGA CAC GUU CGG AGA ATT-3′) were bought from GenePharma (Shanghai, China). Transfection of siRNA was carried out with Lipofectamine 3000 (Thermo Fisher Scientific).
Quantitative Real-Time PCR
TRIzol reagent (TaKaRa) was used to isolate the total RNA of cultured cells. Prime-Script RT-PCR kit (TaKaRa) was applied to transcribe RNA into cDNA. The mRNA expression levels of genes were examined using SYBR Green in an ABI 7500 StepOne Plus Real Time PCR instrument (Applied Biosystem). Each reaction was performed in triplicate. The specific primer sequences used are listed in Table 1.
 |
Table 1 List of Primer Sets for qRT-PCR |
Western Blotting
RIPA buffer with phosphatase and protease inhibitor was used to extract total protein in tissues or cells. Proteins were separated by SDS-PAGE and transferred onto the PVDF membrane. After blocking in 5% non-fat milk with PBST, primary antibodies against TEAD4 (GTX108750, GeneTex), E-cadherin (20,874-1-AP, Proteintech), N-cadherin (22,018-1-AP, Proteintech), Fibronectin (sc-8422, Santa Cruz), Twist1/2 (GTX127310, GeneTex) or GAPDH (sc-32,233, Santa Cruz) were used. The membranes were then incubated in the horseradish peroxidase (HRP)-conjugated secondary antibodies after washing in PBST three times. ECL substrate kit (Tanon Science & Technology, Shanghai, China) was used to visualize the immunoreactive bands on the blots.
Immunohistochemistry (IHC)
The paraffin-embedded sections were prepared for IHC staining according to the standard procedures. Briefly, after antigen retrieval, sections were incubated with primary anti-TEAD4 antibody (GTX108750, GeneTex). DAB visualization kit (DAB-0031, MaixinBio, China) was applied, followed by the procedures described previously.24
Wound Healing
Cells were seeded in 6-well plates until confluent and then a 100 μL pipette tip was used for wound scraping on the cell monolayer and cultured in serum-free medium. Cells were photographed after 0, 24, and 48 h to estimate the wound closure.
Transwell Migration and Invasion Assay
In vitro transwell with or without Matrigel matrix was applied to estimate the migration and invasion abilities of UBC cells. For invasion assay, the mixture of Matrigel and PBS (1:8) was added to upper chambers and then incubated at 37°C. After the glue solidified, transfected cells in 100 μL serum-free medium were then seeded into the upper chambers allowed to migrate or invade, and the bottom well was added with the culture medium plus 15% FBS. After 24 h, transwells were stained by 0.2% crystal violet. The migrated or invaded cells were photographed and counted in 3 casually random fields.
Gene Set Enrichment Analysis (GSEA)
The UBC samples from TCGA-BLCA and GSE32894 were grouped as high TEAD4 expression group and low TEAD4 expression group based on the median transcriptional expression of TEAD4. GSEA software was applied to compare the expression profiles between these two groups. A nominal P-value <0.05 and FDR <0.25 were considered to a statistically significant gene set.
Statistical Analysis
All graphs were plotted and analyzed with GraphPad Prism 8.0 software. Between-groups comparisons were analyzed using Student’s t-tests. A Chi-square test was applied to evaluate the correlation between TEAD4 expression level and clinicopathological features. The overall survival (OS) was estimated by Kaplan–Meier (Log rank test) method. Pearson correlation was applied to measure the strength of correlation between TEAD4 and EMT-related genes. P>0.05 was considered not statistically significant, and the following denotations were used: *P<0.05, **P<0.01 and ***P <0.001.
Results
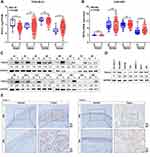
TEAD4 is Highly Upregulated in UBC
To explore the distinct prognostic and potential therapeutic value of four TEAD family members in UBC patients, we first surveyed the expression of four TEADs between UBC tumor tissues and adjacent normal tissues in TCGA-BLCA and GSE13507 datasets. As shown in Figure 1A and B, TEAD4 was the only one TEAD family member that was strikingly elevated in UBC tissues as compared to their normal counterparts both in TCGA-BLCA (P<0.01, Figure 1A) and GSE13507 (P<0.001, Figure 1B) datasets. To confirm the aberrant TEAD4 expression in UBC from the datasets, Western blotting was performed in 16 paired UBC tissues and their adjacent noncancerous tissues. Upregulation of TEAD4 protein level was observed in UBC samples (Figure 1C). Consistently, TEAD4 protein expression level was higher in most UBC cell lines as compared to non-malignant normal urothelial cell SV-HUC-1 (Figure 1D). Then, we further examined the expression level of TEAD4 in UBC tissues by immunohistochemical (IHC) staining. The results showed that TEAD4-positive staining was barely observed in normal urothelial epithelium, while strong TEAD4-positive nuclear staining was detected in cancer cells from UBC samples (Figure 1E). Collectively, we concluded that TEAD4 expression was significantly increased in UBC.
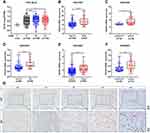
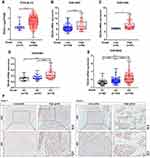
Increased Expression of TEAD4 is Associated with Clinical Stage and Pathological Grade
To investigate the association between TEAD4 expression level and clinicopathological characteristics, multiple datasets with clinical stage and pathological grade information were enrolled. In TCGA-BLCA dataset, TEAD4 mRNA expression level was notably increased in Stage III or Stage IV as compared to Stage II (P<0.05, Figure 2A). In GEO datasets, the mRNA expression of TEAD4 was higher in advanced stages (T2–4) than that in low stages (Tis+T1 or Ta+T1) (P<0.01, Figure 2B–F). Consistently, our IHC data showed the representative images of TEAD4 expression in Ta and T1–4 stages patients, the protein level of TEAD4 was higher in advanced stages (Figure 2G). In addition, overexpressed TEAD4 mRNA expression in high-grade patients was observed (P<0.05, Figure 3A–E). In line with this, IHC data also showed that more intense positive nuclear TEAD4 staining was detected in high-grade patients as compared to low-grade patients (Figure 3F). Further analyses revealed that TEAD4 protein level was remarkably correlated with T stage (P=0.002, Table 2) and tumor grade (P<0.001, Table 2). Moreover, univariate Cox regression analysis revealed that T stage, grade, M stage and TEAD4 expression were significantly associated with patient prognosis (Table 3). However, as shown in Supplementary Figure 1, there were no significant associations between TEAD4 mRNA expression and M stage in TCGA-BLCA and most GEO datasets.
 |
Table 2 The Association Between TEAD4 Protein Levels and Clinicopathological Features of UBC Patients (n=50) |
 |
Table 3 Univariate and Multivariate Analyses of Clinicopathological Features and Survival Time of UBC Patients (n=50) |
Overexpression of TEAD4 is Significantly Related to Unfavorable Prognosis in Patients with UBC
To better investigate the clinical value of TEAD4 in UBC, Kaplan–Meier survival analysis was applied to assess the correlation between TEAD4 expression and clinical outcome. The results showed that patients with high TEAD4 mRNA level had shorter overall survival time in all 5 datasets (P<0.05, Figure 4A–E). Consistently, the same conclusion was drawn from our IHC cohort (n=50), higher TEAD4 protein level heralded worse prognosis (P=0.0148, Figure 4F). Taken together, these data revealed a trend toward high level of TEAD4 and poor patient prognosis.
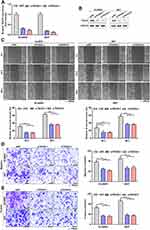
TEAD4 Knockdown Inhibits UBC Cells Migration and Invasion
The above results strongly supported the view that TEAD4 may have a tumor-promoting effect in UBC. We then focused on its potential oncogenic roles by conducting siRNA-mediated loss-of-function approach. We selected SCaBER and 5637 cells for knockdown experiments for the relatively higher endogenous TEAD4 in these cell lines (Figure 1D). TEAD4 knockdown was confirmed by qRT-PCR and Western blotting after 2 independent TEAD4 siRNAs were transfected into SCaBER and 5637 cells (Figure 5A and B). We then explored the effects of TEAD4 on migration and invasion abilities of UBC cells using wound healing and in vitro transwell with or without Matrigel matrix layer assays. Wound healing demonstrated that significant impairment in wound closure at different time points in TEAD4-silenced SCaBER and 5637 cells (Figure 5C). In line with this, depletion of TEAD4 in SCaBER and 5637 notably suppressed cell migration and invasive abilities (Figure 5D and E). Therefore, these functional data indicated that TEAD4 greatly contributed to metastatic ability of UBC cells by promoting cell migration and invasion.
TEAD4 Induces UBC Cells Epithelial-Mesenchymal Transition
Epithelial-mesenchymal transition (EMT) is well known to play an essential role in malignant tumor progression, which endows tumor cells with increased metastatic potential.25,26 Therefore, we speculated that TEAD4 overexpression may be able to induce EMT in UBC cells. To confirm this hypothesis, GSEA was carried out to analyze the differences of EMT gene sets in transcriptome levels between high and low TEAD4. As a result, high mRNA expression of TEAD4 was significantly associated with genes related to EMT both in TCGA-BLCA (Figure 6A) and GSE32894 (Figure 6B) datasets. We then analyzed the correlation between TEAD4 and various EMT-related markers as well as transcription regulators in TCGA-BLCA and GSE32894 datasets. TEAD4 expression level showed a close correlation with EMT-related genes (Figure 6C and D). We next examined the mRNA expression level of these genes in SCaBER and 5637 cells under the treatment of siRNAs to TEAD4. The results showed that the expression of E-cadherin was upregulated, whereas the levels of N-cadherin and FN1 were downregulated, once TEAD4 was depleted (Figure 6E and F). Consistent with these findings, two master EMT-related transcription factors-TWIST1 and TWIST2, but not another four EMT-related transcription factors (SNAI1/2 and ZEB1/2), were also significantly decreased in both SCaBER and 5637 cells (Figure 6E and F). Western blotting further confirmed the elevation of E-cadherin and the reductions of N-cadherin, Fibronectin and Twist1/2 upon TEAD4 depletion (Figure 6G). Collectively, these data suggested that TEAD4 overexpression was required for EMT in UBC cells.
Discussion
As the pivotal mediators in Hippo signaling pathway, TEAD family proteins have been reported to be involved in the initiation and progression of diverse cancers. Researches on this area mainly aimed at TEAD1 and TEAD4. The expression level of TEAD1 was reported to be elevated in the prostate cancer27 and medulloblastomas,28 which led to the malignant progression of the tumors. Overexpression of TEAD4 in carcinomas was widespread, including colorectal cancer,20,21 oral squamous cell carcinomas,29 lung adenocarcinoma,30 and head neck squamous cell carcinoma.31 Intriguingly, TEAD1 and TEAD4 were found to be abundantly expressed gastric cancer, which together resulted in the tumor aggressiveness.23 As TEADs play such critical roles in cancer development, it thus gives us a hint that suppression of TEAD family might provide a promising strategy for future cancer intervention and therapies. In the present study, we found that TEAD4, but not other TEAD family members, was remarkably upregulated at mRNA level in UBC tissues as compared to adjacent normal tissues in two public datasets. Its overexpression was mainly in the nuclei of UBC cells determined by immunohistochemistry. Western blotting from UBC cell lines and tumor tissues also demonstrated aberrant protein upregulation of TEAD4. However, the exact molecular mechanisms underlying its overexpression await further exploration.
In addition, bioinformatics analyses from TCGA-BLCA and GEO datasets revealed strong associations between TEAD4 mRNA expression level and clinical stage, pathological grade as well as clinical outcome. Consistently, our data from UBC samples also demonstrated that TEAD4 overexpression was strongly associated with advanced clinical stage, high pathological grade and unfavorable prognosis. Furthermore, univariate Cox regression analysis revealed that TEAD4 expression, as well as T stage, grade, M stage, were closely associated with patient prognosis. However, we failed to reveal significant associations between TEAD4 mRNA level and M stage in TCGA-BLCA and most GEO datasets. We reasoned that it is conceivable due to heterogeneity of UBC. Therefore, larger number of patients with UBC from multiple centers is required to collect for further accurate analysis. Recent study revealed that metformin suppressed UBC cell progression by inhibiting the AMPK/Yap1/TEAD4/CCNE1/2 axis, which provided novel molecular therapeutic targets involved with TEAD4 for UBC.32 In gastric tumorigenesis, miR-4269 inhibited the oncogenic role of TEAD4.23 Similarly, pharmacologic inhibition of TEAD4 inhibited glucocorticoid receptor-induced breast cancer chemoresistance.33 The above data indicated that TEAD4 was a promising therapeutic target in malignant tumor.
TEAD4 overexpression in UBC suggested that TEAD4 might function as an oncogene and its upregulation might lead to the development and progression of UBC. To confirm our hypothesis, functional experiments were carried out. The results demonstrated that TEAD4 depletion suppressed the UBC cell migration and invasion abilities. To elucidate the underlying mechanisms of TEAD4-mediated carcinogenic effects, we hypothesized that TEAD4 facilitated the UBC development via inducing EMT, which is a cellular programme that is crucial for embryogenesis, wound healing and malignant progression.25,26 EMT is triggered by several extracellular signals, including components of the extracellular matrix and growth factors, and is mediated by the activation of EMT transcription factors such as Snail1/2, Slug, Twist1/2, and ZEB1/2. Accumulating evidence reveals that EMT is the main driving force of cancer progression from onset to metastasis, and plays a key role in inducing cancer progression, metastasis and drug resistance.34–36 GSEA and Pearson correlation analyses demonstrated that TEAD4 was significantly associated with genes related to EMT in two public datasets. Consistently, our data also validated that TEAD4 induced EMT in UBC as evidenced by the EMT markers and transcription regulators expression changes. In our previous study, we revealed that honokiol was able to inhibit the UBC cells EMT through repressing the expression of Twist1.37 Apart from this, Foxo3a was also demonstrated to negatively regulate Twist1 and thereby suppressed urothelial cancer invasiveness.38 Here, we provided a potential positive upstream regulator of Twist, namely TEAD4, which might modulate the process of EMT. However, the specific mechanism contributing to the TEAD4-induced EMT in UBC remains unclear and needs to further exploration.
To sum up, our data revealed that TEAD4 was upregulated in UBC and identified it as a new prognostic biomarker and therapeutic target, as well as a pro-tumorigenic gene for UBC development and progression.
Conclusion
In summary, our findings revealed that TEAD4 was highly elevated in UBC tissues compared with adjacent nontumorous counterpart, and its aberrant upregulation was significantly associated with advanced clinical stage, high pathological grade and unfavorable prognosis of UBC. Subsequently, knockdown of TEAD4 could substantially inhibit UBC cell migration and invasion via suppressing EMT. Collectively, our results indicated that TEAD4 may serve as a novel prognostic biomarker and a promising therapeutic target for UBC.
Acknowledgments
This work was supported by the National Natural Science Foundation (82072821), Shanghai Songjiang Municipal Science and Technology Commission Natural Science Foundation (17SJKJGG10) and Shanghai Specialized Research Fund for Integrated Chinese and Western Medicine in General Hospitals (ZHYY-ZXYJHZX-1-201705).
Disclosure
The authors declare that they have no competing interests.
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. doi:10.3322/caac.21590
2. Knowles MA, Hurst CD. Molecular biology of bladder cancer: new insights into pathogenesis and clinical diversity. Nat Rev Cancer. 2015;15(1):25–41. doi:10.1038/nrc3817
3. Sternberg CN, Bellmunt J, Sonpavde G, et al. ICUD-EAU International consultation on bladder cancer 2012: chemotherapy for urothelial carcinoma-neoadjuvant and adjuvant settings. Eur Urol. 2013;63(1):58–66. doi:10.1016/j.eururo.2012.08.010
4. Kluth LA, Black PC, Bochner BH, et al. Prognostic and prediction tools in bladder cancer: a comprehensive review of the literature. Eur Urol. 2015;68(2):238–253. doi:10.1016/j.eururo.2015.01.032
5. Dey A, Varelas X, Guan KL. Targeting the Hippo pathway in cancer, fibrosis, wound healing and regenerative medicine. Nat Rev Drug Discov. 2020;19(7):480–494. doi:10.1038/s41573-020-0070-z
6. Yu FX, Zhao B, Guan KL. Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell. 2015;163(4):811–828. doi:10.1016/j.cell.2015.10.044
7. Zheng Y, Pan D. The hippo signaling pathway in development and disease. Dev Cell. 2019;50(3):264–282.
8. Calses PC, Crawford JJ, Lill JR, Dey A. Hippo pathway in cancer: aberrant regulation and therapeutic opportunities. Trends Cancer. 2019;5(5):297–307. doi:10.1016/j.trecan.2019.04.001
9. Ma S, Meng Z, Chen R, Guan KL. The hippo pathway: biology and pathophysiology. Annu Rev Biochem. 2019;88:577–604. doi:10.1146/annurev-biochem-013118-111829
10. Zanconato F, Cordenonsi M, Piccolo S. YAP/TAZ at the roots of cancer. Cancer Cell. 2016;29(6):783–803. doi:10.1016/j.ccell.2016.05.005
11. Koo JH, Guan KL. Interplay between YAP/TAZ and metabolism. Cell Metab. 2018;28(2):196–206. doi:10.1016/j.cmet.2018.07.010
12. Pobbati AV, Hong W. A combat with the YAP/TAZ-TEAD oncoproteins for cancer therapy. Theranostics. 2020;10(8):3622–3635. doi:10.7150/thno.40889
13. Lin KC, Park HW, Guan KL. Regulation of the hippo pathway transcription factor TEAD. Trends Biochem Sci. 2017;42(11):862–872. doi:10.1016/j.tibs.2017.09.003
14. Holden JK, Cunningham CN. Targeting the hippo pathway and cancer through the TEAD family of transcription factors. Cancers. 2018;10:3. doi:10.3390/cancers10030081
15. Gibault F, Sturbaut M, Bailly F, Melnyk P, Cotelle P. Targeting Transcriptional Enhanced Associate Domains (TEADs). J Med Chem. 2018;61(12):5057–5072. doi:10.1021/acs.jmedchem.7b00879
16. Huh HD, Kim DH, Jeong HS, Park HW. Regulation of TEAD transcription factors in cancer biology. Cells. 2019;8:6. doi:10.3390/cells8060600
17. Zhou Y, Huang T, Cheng AS, Yu J, Kang W, To KF. The TEAD family and its oncogenic role in promoting tumorigenesis. Int J Mol Sci. 2016;17:1.
18. Pobbati AV, Hong W. Emerging roles of TEAD transcription factors and its coactivators in cancers. Cancer Biol Ther. 2013;14(5):390–398. doi:10.4161/cbt.23788
19. Marti P, Stein C, Blumer T, et al. YAP promotes proliferation, chemoresistance, and angiogenesis in human cholangiocarcinoma through TEAD transcription factors. Hepatology. 2015;62(5):1497–1510. doi:10.1002/hep.27992
20. Liu Y, Wang G, Yang Y, et al. Increased TEAD4 expression and nuclear localization in colorectal cancer promote epithelial-mesenchymal transition and metastasis in a YAP-independent manner. Oncogene. 2016;35(21):2789–2800. doi:10.1038/onc.2015.342
21. Tang J-Y, Yu C-Y, Bao Y-J, et al. TEAD4 promotes colorectal tumorigenesis via transcriptionally targeting YAP1. Cell Cycle. 2018;17(1):102–109. doi:10.1080/15384101.2017.1403687
22. Lamar JM, Stern P, Liu H, Schindler JW, Jiang ZG, Hynes RO. The Hippo pathway target, YAP, promotes metastasis through its TEAD-interaction domain. Proc Natl Acad Sci U S A. 2012;109(37):E2441–2450. doi:10.1073/pnas.1212021109
23. Zhou Y, Huang T, Zhang J, et al. TEAD1/4 exerts oncogenic role and is negatively regulated by miR-4269 in gastric tumorigenesis. Oncogene. 2017;36(47):6518–6530. doi:10.1038/onc.2017.257
24. Yan YL, Huang ZN, Zhu Z, et al. Downregulation of TET1 promotes bladder cancer cell proliferation and invasion by reducing DNA hydroxymethylation of AJAP1. Front Oncol. 2020;10:667. doi:10.3389/fonc.2020.00667
25. Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20(2):69–84. doi:10.1038/s41580-018-0080-4
26. Nieto MA, Huang RY, Jackson RA, Thiery JP. Emt: 2016. Cell. 2016;166(1):21–45.
27. Knight JF, Shepherd CJ, Rizzo S, et al. TEAD1 and c-Cbl are novel prostate basal cell markers that correlate with poor clinical outcome in prostate cancer. Br J Cancer. 2008;99(11):1849–1858. doi:10.1038/sj.bjc.6604774
28. Takeuchi S, Kasamatsu A, Yamatoji M, et al. TEAD4-YAP interaction regulates tumoral growth by controlling cell-cycle arrest at the G1 phase. Biochem Biophys Res Commun. 2017;486(2):385–390. doi:10.1016/j.bbrc.2017.03.050
29. Fernandez LA, Northcott PA, Dalton J, et al. YAP1 is amplified and up-regulated in hedgehog-associated medulloblastomas and mediates Sonic hedgehog-driven neural precursor proliferation. Genes Dev. 2009;23(23):2729–2741. doi:10.1101/gad.1824509
30. Gu C, Huang Z, Chen X, et al. TEAD4 promotes tumor development in patients with lung adenocarcinoma via ERK signaling pathway. Biochimica et Biophysica Acta. 2020;1866:12. doi:10.1016/j.bbadis.2020.165921
31. Zhang W, Li J, Wu Y, et al. TEAD4 overexpression promotes epithelial-mesenchymal transition and associates with aggressiveness and adverse prognosis in head neck squamous cell carcinoma. Cancer Cell Int. 2018;18:178. doi:10.1186/s12935-018-0675-z
32. Wu Y, Zheng Q, Li Y, et al. Metformin targets a YAP1-TEAD4 complex via AMPKalpha to regulate CCNE1/2 in bladder cancer cells. J Exp Clin Cancer Res. 2019;38(1):376. doi:10.1186/s13046-019-1346-1
33. He L, Yuan L, Sun Y, et al. Glucocorticoid receptor signaling activates TEAD4 to promote breast cancer progression. Cancer Res. 2019;79(17):4399–4411. doi:10.1158/0008-5472.CAN-19-0012
34. De Craene B, Berx G. Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer. 2013;13(2):97–110. doi:10.1038/nrc3447
35. Yeung KT, Yang J. Epithelial-mesenchymal transition in tumor metastasis. Mol Oncol. 2017;11(1):28–39. doi:10.1002/1878-0261.12017
36. Mittal V. Epithelial mesenchymal transition in tumor metastasis. Annu Rev Pathol. 2018;13:395–412. doi:10.1146/annurev-pathol-020117-043854
37. Shen L, Zhang F, Huang R, Yan J, Shen B. Honokiol inhibits bladder cancer cell invasion through repressing SRC-3 expression and epithelial-mesenchymal transition. Oncol Lett. 2017;14(4):4294–4300. doi:10.3892/ol.2017.6665
38. Shiota M, Song Y, Yokomizo A, et al. Foxo3a suppression of urothelial cancer invasiveness through Twist1, Y-box-binding protein 1, and E-cadherin regulation. Clin Cancer Res. 2010;16(23):5654–5663. doi:10.1158/1078-0432.CCR-10-0376
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.