Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 14
Short-Term Changes in Metabolically Healthy Overweight/Obesity Status Impact the Susceptibility to Type 2 Diabetes in Chinese Adults
Authors Min J , Hu X, Zhang J , Zeng T , Wang Y, Tian S, Liu G, Zhong X, Qiu K, Peng M, Chen L
Received 30 March 2021
Accepted for publication 27 May 2021
Published 9 June 2021 Volume 2021:14 Pages 2561—2571
DOI https://doi.org/10.2147/DMSO.S313475
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Antonio Brunetti
Jie Min, Xiang Hu, Jiaoyue Zhang, Tianshu Zeng, Ying Wang, Shenghua Tian, Geng Liu, Xueyu Zhong, Kangli Qiu, Miaomiao Peng, Lulu Chen
Department of Endocrinology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, People’s Republic of China
Correspondence: Lulu Chen
Department of Endocrinology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, 1277 Jiefang Avenue, Wuhan, Hubei, 430022, People’s Republic of China
Tel +86 27 8572 6082
Fax +86 27 8535 6365
Email [email protected]
Purpose: Changes in transition from metabolically healthy overweight/obesity (MHO) to metabolically unhealthy overweight/obesity (MUO) are associated with the risk for cardiometabolic complications. This study aims to investigate the effects of short-term dynamic changes in body mass index (BMI) and metabolic status on the risk of type 2 diabetes (T2D) and to identify biological predictors for the MHO-to-MUO transition.
Patients and Methods: A total of 4604 subjects from the REACTION study were included for a 3-year follow-up. Subjects were categorized based on their BMI and metabolic syndrome status. Overweight/obesity was defined as BMI ≥ 24 kg/m2. Metabolically healthy was defined as having two or fewer of the metabolic syndrome components proposed by the Chinese Diabetes Society. Thus, subjects were divided into four groups: metabolically healthy normal weight (MHNW), MHO, metabolically unhealthy normal weight (MUNW), and MUO.
Results: Compared with MHNW, MHO was not predisposed to an increased risk for T2D (OR 1.08, 95% CI 0.64– 1.83, P = 0.762). However, a 3-year transition probability of 20.6% was identified for subjects who shifted from MHO to MUO; this conversion increased the risk of T2D by 3-fold (OR 3.04, 95% CI 1.21– 7.68, P = 0.018). The fatty liver index independently predicted the MHO-to-MUO transition with an OR 3.14 (95% CI 1.56– 7.46, P = 0.002) when comparing the fourth quartile to the first quartile.
Conclusion: This study reveals that metabolic changes affect the short-term susceptibility to T2D in the overweight/obese Chinese population, and the fatty liver index is an efficient clinical parameter for identifying those with a metabolic deterioration risk.
Keywords: overweight, obesity, metabolic syndrome, type 2 diabetes
Introduction
Overweight and obesity, which affect more than one third of the world’s population,1 are associated with the development of type 2 diabetes (T2D), cardiovascular disease, and many other chronic diseases.2 However, research remains to be done regarding the varying levels of exposure to cardiometabolic complications in the obese population. One of the specific interests explored in this study is a subgroup of overweight/obese subjects who experience a low risk for cardiometabolic abnormalities; this subgroup is defined as metabolically healthy overweight/obesity (MHO).3
Little consensus has been made on the association between MHO and the incidence rate of T2D in the past decades. Some studies have found that MHO is a safe phenotype as possessing no significant relevance in the risk for developing T2D than that of healthy lean subjects4,5 while other studies have reported a low but positive risk.6,7 Despite the inconsistent findings regarding the association between metabolic health and the risk for diabetes, the unstable nature of MHO may deteriorate to metabolically unhealthy overweight/obesity (MUO) overtime, and ultimately increase the risk for the development of diabetes.8–10 The Nurses’ Health Study delineated a decline of healthy metabolic profile with increased age after a 30-year follow-up and suggested an early intervention to impede the transition from healthy to unhealthy metabolic status in both overweight and obese populations.9 Thus, it is urgent to investigate the short-term MHO-to-MUO transition impact on the T2D risk and identify the predictors for this imminent transition. However, most studies address the long-term effects of MHO phenotype on diabetic risk without considering body mass index (BMI) across metabolic changes and its predictors, especially in Chinese adults.5,11–14
In this study, we aim to 1) examine the association of BMI-metabolic health categories and the risk for the development of T2D over a 3-year follow-up period by using data from the Risk Evaluation of cAncers in Chinese diabeTic Individuals (REACTION) study;15 2) investigate the effects of dynamic metabolic health status changes in the MHO phenotype on the risk of T2D; 3) explore the clinical parameters predicting the changes from metabolically healthy to unhealthy state of MHO individuals during this period.
Patients and Methods
Study Population
The population studied was derived from the REACTION study in which investigators studied the association between glucose metabolism and risk for cancer in the Chinese population.15 The cohort profile being studied here is a subgroup of REACTION subjects, which were followed up for a 3-year period. 10,420 subjects residing in rural areas of Yichang city, Hubei province ranging from ages 35 to 91 years were enrolled in this study from October 2011 to January 2012. 6243 subjects completed the follow-up examination between September 2014 and October 2015. Baseline exclusionary criteria included subjects diagnosed with any of the following diseases: T2D, cardiovascular disease (CVD), stroke, pancreas disease, kidney disease, and tumor. Additionally, baseline BMI was evaluated for exclusionary criteria. Ultimately, 4604 subjects participated in the final analysis. This study adheres to the Principles of the Declaration of Helsinki and the study protocol was approved by the Ethics Committee of Tongji Medical College of Huazhong University of Science and Technology. All subjects were provided a verbal explanation of the study protocol and were also provided written details regarding their voluntary participation in the study; all subjects provided written informed consent.
Measurement
Well-trained specialists collected the anthropometric data. Weight was measured to the nearest 0.1 kg with subjects wearing indoor clothes but no shoes. Height was measured to the nearest 0.1 cm without shoes. While wearing minimal clothing, waist and hip circumferences were measured to the nearest 0.1 cm. BMI was calculated as weight in kilograms divided by the square of the height in meters. Blood pressure was conducted two to three times, with each measurement taken after the subject was sedentary for a duration of 5 minutes, and the mean blood pressure was calculated for analysis. The International Physical Activity Questionnaire (IPAQ), a short list of questions used to determine activity level, was conducted to evaluate the physical activity of subjects.
The details of biochemical measurements are described in the REACTION study.15 All subjects fasted for a minimum of 8 hours prior to sample collection. During the 75-g oral glucose tolerance test (OGTT), a 2-hour blood sample was obtained for 2-hour plasma glucose (2hPG). HOMA-IR was calculated for the evaluation of insulin resistance.16 To represent ectopic fat distribution, we calculated fatty liver index (FLI)17 using the formula: FLI = (e 0.953 × loge (triglycerides) + 0.139 × BMI + 0.718 × loge (GGT) + 0.053 × waist circumference-15.745)/(1 + e 0.953 × loge (triglycerides) + 0.139 × BMI + 0.718*loge (GGT) + 0.053 × waist circumference-15.745) × 100.
Definition of Metabolically Healthy, Overweight, and Obesity
Metabolism status was determined by five components of metabolic syndrome proposed by the Chinese Diabetes Society18 as follows: 1) waist circumference ≥90 cm for men and ≥85cm for women; 2) fasting plasma glucose (FPG) ≥6.1 mmol/L or OGTT 2hPG ≥7.8 mmol/L, or confirmed diabetes that was under treatment; 3) blood pressure ≥130/85 mmHg, or diagnosed hypertension and on antihypertensive therapy; 4) fasting TG ≥ 1.70 mmol/L; 5) fasting HDL‐C <1.04 mmol/L. Metabolically healthy was defined as having two or fewer of these components. Metabolically unhealthy was defined as the presence of three or more of the five components of metabolic syndrome.
Obesity was determined based on the BMI criteria. Normal weight was defined as 18.5 ≤ BMI < 24 kg/m2; overweight was defined as 24 ≤ BMI < 28 kg/m2; obesity was defined as BMI ≥ 28 kg/m2 according to the Chinese guidelines on the prevention and control of obesity.19 Underweight individuals, which were defined as subjects with a BMI < 18.5 kg/m2, were excluded.
Subjects were divided into four groups: 1) metabolically healthy normal weight (MHNW); 2) metabolically healthy overweight/obesity (MHO); 3) metabolically unhealthy normal weight (MUNW); 4) metabolically unhealthy overweight/obesity (MUO).
Definition of Type 2 Diabetes
Type 2 diabetes was defined using the criteria proposed by the Chinese Diabetes Society18 in which a FPG ≥ 7.0 mmol/L or 2hPG ≥11.1 mmol/L determines diabetes. We excluded other types of diabetes via medical history, family history, and physical examination.
Statistical Analysis
Continuous variables are expressed in mean ± SD or median (interquartile range 25–75%) while categorical variables are presented in frequency and percentages. The Student’s t-test, χ2 test, or ANOVA were used to compare the characteristics of this cohort classified by their metabolic health and BMI categories. Logistic regression analysis was used to estimate the odds ratio (OR) and their 95% confidence interval (CI) of incident T2D during the 3-year follow-up. Three multivariate-adjusted models were fitted: Model 1 was adjusted for age and sex; Model 2 was Model 1 plus the adjustment for smoking, physical activity, sitting MET, and family history (diabetes, CVD, and stroke); Model 3 was Model 2 plus the adjustment for FPG, BMI, and waist circumference.20 First, we analyzed the risk for developing T2D in the cohort by the BMI-metabolic status using MHNW phenotype as the reference category. We further analyzed the risk for T2D considering the transition from MHO to MUO during the 3-year follow-up period using the stable MHNW subgroup as the reference category. To evaluate the probability of transition in the context of BMI-metabolic status within this follow-up period, we used a multiple regression model treating the baseline phenotype as the independent variable and age as a covariate.7 Finally, we conducted multivariate logistic regression analysis with backward stepwise selection to determine the independent association between clinical and biochemical parameters and the MHO-to-MUO transition. Incidence MUO or non-MUO transition from baseline-MHO were treated as dependent variables. Sex, age, smoking, physical activity, sitting MET, BMI, waist circumference, systolic blood pressure (SBP), diastolic blood pressure (DBP), FPG, 2hPG, TG, HDL, LDL, HOMA-IR, and FLI were considered as variables initially. We considered a P value <0.05 to be statistically significant. All the analyses above were conducted using SPSS Statistics (version 23).
Results
Baseline Characteristics
Data from 4604 subjects (1554 men and 3050 women) with a mean age of 55.36 years for men and 53.35 years for women were analyzed. The anthropometric data, family history, and biochemical measurements classified by BMI-metabolic health status are summarized in Table 1.
 |
Table 1 Baseline Characteristics of the Cohort |
At baseline, 1239 (26.9%) subjects were categorized as MHO, constituting 68.2% in the overweight/obese population and 32.2% in the metabolically healthy population. MHO subjects were characterized as being younger in age and having lower past smoking rates in comparison to other groups. Although both metabolically healthy, the MHO group yielded relatively worse 2hPG, blood pressure, and lipid profile and were more insulin resistant (HOMA-IR: 1.88 ± 0.87) compared with metabolically healthy normal weight (MHNW) subjects (HOMA-IR: 1.34 ± 0.82), P < 0.05 for all the variables mentioned above according to post hoc analysis; their FPG levels were comparable. FLI levels showed that the liver fat content in MHO (29.54 ± 16.95) was 3-fold higher than those in MHNW (10.51 ± 8.84). When compared with MUO, the MHO group displayed a better metabolic condition with half the FLI level. In another hand, although MUNW subjects were within normal BMI range, they presented higher levels of waist and hip circumference, and were more insulin resistant than MHNW subjects according to post hoc analysis. There were no significant differences between the four groups considering the characteristics of marital status, education, alcohol consumption history, physical activity, sedentary status, family histories of diabetes, CVD, and stroke (P ≥ 0.05).
Risk of Incident T2D for the Cohort After 3-Year Follow-Up
Table 2 shows the distribution of subjects among the 4 groups MHNW, MHO, MUNW and MUO; and the risk of incident for T2D stratified by baseline metabolic health status and BMI. The overall cumulative incidence of T2D (4.20%) for the MHO group was one-third of those in the MUO group (14.73%) and was comparable to MHNW. Compared with MHNW, MHO subjects were not at an increased risk for the incident of T2D (OR 1.08, 95% CI 0.64–1.83, P = 0.762), MUO subjects had a 3-fold higher risk (OR 3.02, 95% CI 1.63–5.60, P < 0.001) when adjusted for age, sex, smoking, physical activity, sedentary time, FPG, BMI, waist circumference, and family history of diabetes, CVD, and stroke.
 |
Table 2 Risk of Incident T2D by Categories of BMI and Metabolic Status |
Changes in BMI Across Metabolic Status in the Cohort and Their Risk for Incident T2D
Table 3 shows the 3-year transition probabilities categorized by BMI-metabolic status. Four out of five subjects in the MHNW group maintained their status of MHNW (stable MHNW), while only 2.3% of them converted to MUO (MHNW-to-MUO) during the 3-year follow-up. By comparison, two thirds of the baseline MHO subjects remained stable (stable MHO), and 20.6% of them transitioned to MUO (MHO-to-MUO). On the other hand, MUO also presented an unstable trait with 34.7% shifted to MHO (MUO-to-MHO).
 |
Table 3 Transition Probabilities of BMI Cross Metabolic Status Changes from Baseline to Follow-Up |
Compared with the MHO subgroup, at baseline, the MHO-to-MUO subgroup presented an elevated level of systolic blood pressure, TG, LDL, and more insulin resistance but had equivalent FPG levels (Supplement Table S1). In addition, the FLI level in the MHO-to-MUO subgroup was almost 1.5-fold higher than that of the stable MHO subgroup (Supplement Table S1).
Table 4 shows adjusted ORs using multiple logistic analysis for incident T2D when considering the metabolic status changes in overweight/obese subjects in a 3-year follow-up. Compared with stable MHNW (MHNW-to-MHNW), subjects in the stable MHO subgroup (MHO-to-MHO) did not burden an increased T2D risk (OR 1.02, 95% CI 0.45–2.33 P = 0.959) after adjusting variables. However, when metabolic health deteriorated (MHO-to-MUO), these subjects had a 3-fold increased risk for T2D (OR 3.04, 95% CI, 1.21–7.68, P = 0.018). To further investigate how metabolic improvement affected the risk for T2D in the short term, we did the same analysis in the baseline MUO subjects. Compared with stable MHNW subgroup, stable MUO subjects (MUO-to-MUO) suffered an 8-fold higher risk for T2D (OR 8.18, 95% CI 3.53–18.99, P < 0.001), but the subjects with metabolic improvement (MUO-to-MHO) showed insignificant change in the risk for T2D (OR 0.97, 95% CI 0.31–3.03, P = 0.961). We also compared the MHO-to-MUO with stable MHO subgroups and compared MUO-to-MHO with stable MUO subgroups (Supplement Table S2), reconfirming that metabolic health shielded the overweight or obese subjects from increased diabetic risk even though their BMI remained consistent in this short-term follow-up.
 |
Table 4 Risk for Incident T2DM According to the BMI-Metabolic Status Changes in 3-Year Follow-Up |
Independent Predictors for the Transition from MHO to MUO Phenotype
To determine which parameters independently predict the metabolically healthy to unhealthy transition in the overweight/obese population (MHO-to-MUO) during a 3-year follow-up, we generated a model using backward stepwise logistic regression (Table 5). Elevated levels of metabolic components such as waist circumference, systolic blood pressure, FPG, and TG, along with the female sex were predictors for transitioning from MHO to MUO. Moreover, increased FLI level independently predicted this transition with an OR of 3.14 (95% CI 1.56–7.46, P = 0.002) when comparing the fourth quartile with the first quartile.
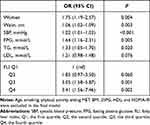 |
Table 5 Multivariate Logistic Regression Analysis for the Transition from Metabolically Healthy to Unhealthy in Overweight/Obese Population |
Discussion
In this study we investigated the association of BMI categories in the context of dynamical metabolic status changes and the risk for T2D. Our study found that metabolically healthy overweight/obese (MHO) subjects constituted for one-fourth of the rural Chinese adult population and were not predisposed to an increased risk for T2D than the lean healthy population (MHNW) within the 3-year follow-up. However, the MHO phenotype was unstable, and one-fifth of them progressed to a metabolically unhealthy state (MUO), consequently increasing their risk for T2D. The FLI was an independent clinical parameter for predicting this metabolic transition from MHO to MUO in the short-term of 3 years.
A limited number of studies have evaluated the stability of BMI-metabolic status changes and their effects on the development of T2D. In general, the transition probability from metabolically healthy to unhealthy in obese individuals (MHO-to-MUO) increases over time. For instance, the Framingham Heart Study reported a 4-year transition probability of 40%,7 and the Nurses’ Health Study disclosed a 20-year conversion rate of 84% in women.9 In agreement with these studies, our results show a 20.6% transition probability for an even shorter prediction period (3 years) than the aforementioned studies. Besides, MUO individuals also have the chance of reverting to MHO. Based on the instability of metabolism status, we indicated the effects of metabolic changes on the risk for T2D in the obese population beyond weight loss. On the one hand, deterioration of metabolic status facilitates the development of T2D in obese individuals. We observed that overweight/obese subjects who developed a metabolically unhealthy status (MHO-to-MUO) had a 3-fold increased risk for incident T2D than the stable MHNW subgroup. By contrast, those who maintained a healthy metabolism status did not suffer the relevant risk even though they remained in the same BMI category. These results were consistent with another Chinese study that recorded a 3.5-fold increased risk for T2D in MHO-to-MUO transition within a longer follow-up (6 years).11 This is also supported by studies that targeted other cardiovascular outcomes.21 On the other hand, improvement of metabolic status lowers the risk for developing diabetes in obese individuals. Compared to the obese subjects who maintained a status of metabolically unhealthy (stable MUO) and underwent a high diabetic risk during follow-up, those who restored metabolic health (MUO-to-MHO) were shielded from significant increased risk. Our study reveals the unstable attributes of metabolic status in the obese population and its effects on T2D development. More importantly, we indicate the short-term harmful consequences of transitioning from MHO to MUO, emphasizing the importance of timely identification of the subset of MHO individuals who carry the risk for progression to an unhealthy metabolic state.
The most interesting finding of our study is that the FLI independently predicts a short-term MHO-to-MUO transition. The FLI, which is based on metabolic-related parameters and reflects the ectopic fat accumulation content in the liver, has been shown to predict fatty liver17 and is helpful in identifying nonalcoholic fatty liver disease (NAFLD) in the Chinese population.22 We illustrated that healthy overweight/obese subjects had only half the level of FLI than that of unhealthy subjects. Moreover, the higher the FLI level within these healthy overweight/obese subjects, the higher the risk of progression to unhealthy status. These results verified the phenotypic traits of lower ectopic fat deposition (visceral, liver, and skeletal muscle) in MHO subjects versus in MUO subjects. MHO subjects have the expansion capacity of subcutaneous adipose tissue (especially in the lower body), decreasing the ectopic fat accumulation and protecting against insulin resistance.3,23,24 Ectopic fat deposition is positively associated with a decline in insulin sensitivity, an increase in adipocytokine level and proinflammatory activity in obese individuals.25 Less ectopic fat accumulation is one explanation for the relatively low risk for T2D in MHO individuals, which is also in line with the adipose tissue expandability hypothesis that links obesity to T2D.26 Impressively, MUNW, which is characterized by lower leg fat mass and a 3-fold higher risk for cardiovascular events in comparison to that of MHNW, is considered a lipodystrophy-like phenotype in this context.27 Fatty liver confirmed by abdominal ultrasonography in one study has also been shown as a risk factor for progression from MHNW to metabolic abnormal phenotype.28 Along with these findings, waist circumference, which provides better characteristic information for the visceral fat accumulation than BMI,29 has been shown to more strongly associated with the risk of incident T2D than the latter in Chinese population.30,31 In our study, increased waist circumference is also a predictor for the MHO-to-MUO transition. Our results support this biological mechanism underlying MHO and, for the first time, show that the FLI is an independent clinical predictor for the short-term MHO-to-MUO transition, implicating that the relatively higher ectopic fat deposition in MHO predispose subjects to an unhealthy metabolic status, which increase the susceptibility to cardiometabolic complications.
Both intrinsic and extrinsic factors that drive an unhealthy response to the excess fat accumulation could be implicated in the mechanism underly MHO-to-MUO transition. Apart from genetic predisposition, poor dietary habits (eg, increase total fat intake), less physical activity, disrupted circadian rhythm, and psychological unhealthy have also considered taking charge of this metabolic shift, which provide us the intervention targets for lifestyle and medical therapy.32,33 Although we did not confirm the relationship between diet or exercise and the transition probability from MHO to MUO, other studies found that lifestyle interventions significantly improve metabolic conditions without weight loss, partly due to their anti-inflammation function. Polyphenols, which have been found beneficial for cardiovascular risk factors in T2D patients (the TOSCA.IT study),34 could also extend the lifespan with improved plasma lipid levels and endotoxemia and decreased immune cell infiltration to the adipose tissue in obese mice, which was independent of body weight.35,36 Further studies on the prevention factors of MHO-to-MUO transition are needed.
Whether a healthy metabolic profile protects obese individuals from the incidence of diabetes has been debated for decades. In our 3-year follow-up, there was no significant difference in the risk for development of T2D in the MHO group compared with MHNW, while the MUO subjects had a 3-fold higher risk. Our results were consistent with other studies which found that individuals with MHO phenotype might be protected or at a lower risk for obese-related metabolic complications, such as T2D.4,5,37 However, other studies have yielded different results which support that an abnormal BMI predisposes individuals to an elevated risk for T2D, independently of metabolic status.6,38 One explanation for this divergence may be due to the different duration of the follow-up periods. As we discussed before, subjects with MHO-to-MUO transition were more prone to cardiometabolic complications,8 and cardiometabolic complications were also age-related abnormalities. The longer the follow-up, the more susceptible to these chronic diseases. The other interpretation for results diversity ascribes to the discorded definitions of metabolic health. When other studies using metabolic syndrome defined by NCEP ATP-III or using insulin sensitivity to determine the metabolically healthy status,39 we preferred the definition of metabolic syndrome prescribed by the Chinese Diabetes Society, which adheres to NCEP ATP-III but is modified to fit the Chinese population.18,40 Although all the metabolic health standards conclude the critical feature of MHO, which is the lack of insulin resistance,41 no unified metabolic health criteria result in the diverse association between MHO and cardiometabolic diseases. Hence, the standardized MHO criteria are needed.
One limitation of our study was that we did not employ imaging technologies to evaluate body fat composition, such as Dual-energy X-ray absorptiometry (DEXA) scan and computed tomography (CT) scan. CT scans may further verify our hypothesis that relatively higher ectopic fat deposition predisposes MHO individuals to an unstable, unhealthy metabolic condition in the Chinese population. It may also help to assess the efficacy of FLI in predicting the MHO-to-MUO transition. Additionally, we could also compare FLI with other clinical parameters that have also been shown to anticipate this shift.42–44
Identifying the MHO phenotype provides an insight into exploring preventative tools and treatment strategies in the obese population. First, not all obese individuals are the same in terms of risk for diabetes and other cardiovascular diseases. Categorizing obesity by the metabolic condition will reduce the adverse medical outcomes and socioeconomic bearings. Although MHO is not as naïve as we previously thought (such as when transitioning to the unhealthy status), it still presents a broad subgroup of the obese population with less risk for developing cardiometabolic complications.20,45 According to a large meta-analysis that included 40 population-based studies worldwide, approximately 35% of all the obese individuals were metabolically healthy despite the variety of definitions.46 This result implicates that one-third of obese people could benefit from less aggressive anti-obese strategies (such as bariatric surgery), although they still need to be treated. Second, identifying MHO phenotype affords a substitute destination for anti-obese therapies. Obesity is a chronic relapsing disease in which long-term weight loss is hard to maintain due to complicated compensatory mechanisms.47 Matthias et al suggested that in addition to weight loss, sustaining a metabolically healthy status could be an alternative target for obese individuals when considering cardiometabolic complications.41 In the specific obesity treatment program, individuals undergo metabolic health status changes accompanied by a series of weight loss and regain.3 Aiming to reach a status of metabolic health could lower the risk for cardiometabolic complications and encourage obese individuals to continue anti-obese treatments when they fail weight loss attempts or go through weight regain. As we discussed before, diet interventions such as polyphenol-rich food supplements could be additional to anti-obese treatments to improve metabolic health in obese people. Undoubtedly, we should be aware that obesity also increases the risk for other diseases such as sleep apnea syndrome, asthma, depression, and cancer.48 Whether MHO could protect obese individuals from these diseases besides cardiometabolic complications requires further study beyond our discussion.
Conclusions
In conclusion, metabolic health shelters overweight or obese individuals with low T2D risk, but the dynamic change from metabolically healthy to unhealthy status increases this risk. Increased liver fat accumulation could predispose MHO individuals to a status of unhealthy metabolism, and FLI independently predicts this short-term healthy-to-unhealthy transition.
Abbreviations
2hPG, 2-hour Plasma Glucose; BMI, Body Mass Index; CVD, Cardiovascular Disease; CI, Confidence Interval; CT, Computed Tomography; DBP, Diastolic Blood Pressure; DEXA, Dual-Energy X-ray Absorptiometry; FLI, Fatty Liver Index; FPG, Fasting Plasma Glucose; HOMA-IR, Homeostatic Model Assessment for Insulin Resistance; IPAQ, International Physical Activity Questionnaire; MET, Metabolic Equivalent of Task; MHNW, Metabolically Healthy Normal Weight; MHO, Metabolically Healthy Overweight/obesity; MUNW, Metabolically Unhealthy Normal Weight; MUO, Metabolically Unhealthy Overweight/obesity; NAFLD, Nonalcoholic Fatty Liver Disease; OGTT, Oral Glucose Tolerance Test; OR, Odds Ratio; PA, physical activity.; REACTION study, Risk Evaluation of cAncers in Chinese diabeTic Individuals study; SBP, Systolic Blood Pressure; T2D, Type 2 Diabetes; WHR, Waist-Hip Ratio.
Acknowledgments
We would like to thank all the investigators from Endocrinology Department, Union Hospital, Tongji Medical College, Huazhong University of Science and from Technology and Yiling Hospital of Yichang, and all the participants who took part in this study. This study was supported by grants from the National Natural Science Foundation of China (81471069, 81770843 and 81800762) and the National Key R&D Program of China Grants 2016YFC0901200 and 2016 YFC0901203 from the Ministry of Science and Technology.
Disclosure
Dr Xiang Hu reports grants from NSFC, during the conduct of the study. The authors report no other conflicts of interest in this work.
References
1. Ng M, Fleming T, Robinson M, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9945):766–781.
2. Afshin A, Forouzanfar MH, Reitsma MB, et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017;377(1):13–27.
3. Bluher M. Metabolically healthy obesity. Endocr Rev. 2020;41:3.
4. Meigs JB, Wilson PW, Fox CS, et al. Body mass index, metabolic syndrome, and risk of type 2 diabetes or cardiovascular disease. J Clin Endocrinol Metab. 2006;91(8):2906–2912.
5. Luo D, Liu F, Li X, et al. Comparison of the effect of ‘metabolically healthy but obese’ and ‘metabolically abnormal but not obese’ phenotypes on development of diabetes and cardiovascular disease in Chinese. Endocrine. 2015;49(1):130–138.
6. Hinnouho GM, Czernichow S, Dugravot A, et al. Metabolically healthy obesity and the risk of cardiovascular disease and type 2 diabetes: the Whitehall II cohort study. Eur Heart J. 2015;36(9):551–559.
7. Echouffo-Tcheugui JB, Short MI, Xanthakis V, et al. Natural history of obesity subphenotypes: dynamic changes over two decades and prognosis in the Framingham heart study. J Clin Endocrinol Metab. 2019;104(3):738–752.
8. Navarro-Gonzalez D, Sanchez-Inigo L, Fernandez-Montero A, Pastrana-Delgado J, Alfredo Martinez J. Are all metabolically healthy individuals with obesity at the same risk of diabetes onset? Obesity (Silver Spring). Dec. 2016;24(12):2615–2623.
9. Eckel N, Li Y, Kuxhaus O, Stefan N, Hu FB, Schulze MB. Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease risk across BMI categories in 90 257 women (the Nurses’ Health Study): 30 year follow-up from a prospective cohort study. Lancet Diabetes Endocrinol. 2018;6(9):714–724.
10. Gao M, Lv J, Yu C, et al. Metabolically healthy obesity, transition to unhealthy metabolic status, and vascular disease in Chinese adults: a cohort study. PLoS Med. 2020;17(10):e1003351.
11. Wang B, Zhang M, Wang S, et al. Dynamic status of metabolically healthy overweight/obesity and metabolically unhealthy and normal weight and the risk of type 2 diabetes mellitus: a cohort study of a rural adult Chinese population. Obes Res Clin Pract. 2018;12(1):61–71.
12. Xu R, Gao X, Wan Y, Fan Z. Association of metabolically healthy overweight phenotype with abnormalities of glucose levels and blood pressure among Chinese adults. JAMA Netw Open. 2019;2(10):e1914025.
13. Liu X, Zhang J, Wu J, et al. The impact of BMI categories on metabolic abnormality development in Chinese adults who are metabolically healthy: a 7-year prospective study. Diabetes Metab Syndr Obes. 2020;13:819–834.
14. Wei Y, Wang J, Han X, et al. Metabolically healthy obesity increased diabetes incidence in a middle-aged and elderly Chinese population. Diabetes Metab Res Rev. 2020;36(1):e3202.
15. Bi Y, Lu J, Wang W, et al. Cohort profile: risk evaluation of cancers in Chinese diabetic individuals: a longitudinal (REACTION) study. J Diabetes. 2014;6(2):147–157.
16. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004;27(6):1487–1495.
17. Bedogni G, Bellentani S, Miglioli L, et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33.
18. Jia W, Weng J, Zhu D, et al. Standards of medical care for type 2 diabetes in China 2019. Diabetes Metab Res Rev. 2019;35(6):e3158.
19. Chen C, Lu FC. Department of Disease Control Ministry of Health PRC. The guidelines for prevention and control of overweight and obesity in Chinese adults. Biomed Environ Sci. 2004;17:
20. Bell JA, Kivimaki M, Hamer M. Metabolically healthy obesity and risk of incident type 2 diabetes: a meta-analysis of prospective cohort studies. Obes Rev. 2014;15(6):504–515.
21. Kaur A, Johnston DG, Godsland IF. Does metabolic health in overweight and obesity persist? - Individual variation and cardiovascular mortality over two decades. Eur J Endocrinol. 2016;175(2):133–143.
22. Huang X, Xu M, Chen Y, et al. Validation of the fatty liver index for nonalcoholic fatty liver disease in middle-aged and elderly Chinese. Medicine (Baltimore). 2015;94(40):e1682.
23. Despres JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881–887.
24. Magkos F. Metabolically healthy obesity: what’s in a name?. Am J Clin Nutr. 2019;110(3):533–539.
25. Amato MC, Giordano C, Galia M, et al. Visceral Adiposity Index: a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33(4):920–922.
26. Virtue S, Vidal-Puig A. Adipose tissue expandability, lipotoxicity and the Metabolic Syndrome – an allostatic perspective. Biochim Biophys Acta. 2010;1801(3):338–349.
27. Stefan N, Schick F, Haring HU. Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab. 2017;26(2):292–300.
28. Hashimoto Y, Hamaguchi M, Fukuda T, Ohbora A, Kojima T, Fukui M. Fatty liver as a risk factor for progression from metabolically healthy to metabolically abnormal in non-overweight individuals. Endocrine. 2017;57(1):89–97.
29. Despres JP. Body fat distribution and risk of cardiovascular disease: an update. Circulation. 2012;126(10):1301–1313.
30. Hou X, Chen S, Hu G, et al. Stronger associations of waist circumference and waist-to-height ratio with diabetes than BMI in Chinese adults. Diabetes Res Clin Pract. 2019;147:9–18.
31. Fan Y, Wang R, Ding L, et al. Waist circumference and its changes are more strongly associated with the risk of Type 2 diabetes than body mass index and changes in body weight in Chinese adults. J Nutr. 2020;150(5):1259–1265.
32. Kruger HS, Ricci C, Pieters M, et al. Lifestyle factors associated with the transition from healthy to unhealthy adiposity among black South African adults over 10 years. Nutr Metab Cardiovasc Dis. 2021;S0939-4753(21.
33. Dagpo TD, Nolan CJ, Delghingaro-Augusto V. Exploring therapeutic targets to reverse or prevent the transition from metabolically healthy to unhealthy obesity. Cells. 2020;9:7.
34. Vitale M, Vaccaro O, Masulli M, et al. Polyphenol intake and cardiovascular risk factors in a population with type 2 diabetes: the TOSCA.IT study. Clin Nutr. 2017;36(6):1686–1692.
35. Baur JA, Pearson KJ, Price NL, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444(7117):337–342.
36. Aires V, Labbe J, Deckert V, et al. Healthy adiposity and extended lifespan in obese mice fed a diet supplemented with a polyphenol-rich plant extract. Sci Rep. 2019;9(1):9134.
37. McLaughlin T, Abbasi F, Lamendola C, Reaven G. Heterogeneity in the prevalence of risk factors for cardiovascular disease and type 2 diabetes mellitus in obese individuals: effect of differences in insulin sensitivity. Arch Intern Med. 2007;167(7):642–648.
38. Twig G, Afek A, Derazne E, et al. Diabetes risk among overweight and obese metabolically healthy young adults. Diabetes Care. 2014;37(11):2989–2995.
39. Schulze MB. Metabolic health in normal-weight and obese individuals. Diabetologia. 2019;62(4):558–566.
40. Huang PL. A comprehensive definition for metabolic syndrome. Dis Model Mech. 2009;2(5–6):231–237.
41. Stefan N, Haring HU, Schulze MB. Metabolically healthy obesity: the low-hanging fruit in obesity treatment?. Lancet Diabetes Endocrinol. 2018;6(3):249–258.
42. Gilardini L, Zambon A, Soranna D, Croci M, Invitti C. Predictors of the transition from metabolically healthy obesity to unhealthy obesity. Eat Weight Disord. 2018;23(6):739–744.
43. Zhao L, Ni Y, Ma X, et al. A panel of free fatty acid ratios to predict the development of metabolic abnormalities in healthy obese individuals. Sci Rep. 2016;6:28418.
44. Achilike I, Hazuda HP, Fowler SP, Aung K, Lorenzo C. Predicting the development of the metabolically healthy obese phenotype. Int J Obes (Lond). 2015;39(2):228–234.
45. Lotta LA, Abbasi A, Sharp SJ, et al. Definitions of metabolic health and risk of future Type 2 Diabetes in BMI categories: a systematic review and network meta-analysis. Diabetes Care. 2015;38(11):2177–2187.
46. Lin H, Zhang L, Zheng R, Zheng Y. The prevalence, metabolic risk and effects of lifestyle intervention for metabolically healthy obesity: a systematic review and meta-analysis: a PRISMA-compliant article. Medicine (Baltimore). 2017;96(47):e8838.
47. Bray GA, Kim KK, Wilding JPH, World Obesity F. Obesity: a chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes Rev. 2017;18(7):715–723.
48. Bray GA. Medical consequences of obesity. J Clin Endocrinol Metab. 2004;89(6):2583–2589.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
