Back to Journals » International Journal of General Medicine » Volume 14
Serum Klotho Levels Contribute to the Prevention of Disease Progression
Authors Nakanishi K , Nishida M, Taneike M, Yamamoto R, Moriyama T, Yamauchi-Takihara K
Received 10 November 2020
Accepted for publication 31 December 2020
Published 22 January 2021 Volume 2021:14 Pages 229—236
DOI https://doi.org/10.2147/IJGM.S291437
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Kaori Nakanishi, Makoto Nishida, Manabu Taneike, Ryohei Yamamoto, Toshiki Moriyama, Keiko Yamauchi-Takihara
Health Care Division, Health and Counseling Center, Osaka University, Osaka, Japan
Correspondence: Keiko Yamauchi-Takihara
Health Care Division, Health and Counseling Center, Osaka University, 1-17 Machikaneyama, Toyonaka, Osaka 560-0043, Japan
Tel +81-6-6850-6012
Fax +81-6-6850-6040
Email [email protected]
Background: Assessing the progression of a disorder from its pre-clinical state is important in the prevention of various diseases. In the present study, we evaluated the role of serum levels of αKlotho (αKl) in the progression of several pre-clinical disorders.
Methods: This cohort study included 80 males who underwent their annual health checkup during the entry period between April 2005 and March 2008. Physical and biochemical parameters were obtained from all subjects. The associations of baseline serum levels of soluble αKl (sαKl) with the progression of the disorders were assessed in the study.
Results: Baseline serum levels of sαKl were significantly lower in subjects developing a high fasting plasma glucose (FPG) level than in subjects not developing a high FPG level. Logistic multivariable analysis showed that baseline serum levels of sαKl and FPG levels significantly associated with a high FPG level progression. It is suggested that low sαKl levels are associated with the progression of hyperglycemia. Evaluation of serum levels of sαKl in subjects with multiple disorders revealed that those with more pre-clinical disorders progression tended to show lower sαKl levels.
Conclusion: A decrease in serum levels of sαKl could be associated with the progression of pre-clinical disorders.
Keywords: αKlotho, metabolic disorder, hyperglycemia, hypertriglyceridemia, hypertension
Introduction
It is well-known that the pre-clinical state of disorders, including obesity, hypertension, dyslipidemia, hyperglycemia, and hyperuricemia, is a serious risk factor for developing various diseases, such as cardiovascular disease, stroke, and type 2 diabetes mellitus. Moreover, as represented by the metabolic syndrome, cluster of these disorders would be a more serious risk for these diseases than a single disorder.1–3 Recently, several studies reported the relationship between these diseases and Klotho (Kl).4–6
Kl was originally discovered as an anti-aging gene, Klotho. Klotho mutant mice have short life spans and exhibit multiple aging phenotypes, including skin atrophy, ectopic calcification, osteoporosis, atherosclerosis, and pulmonary emphysema. The gene encodes a single-pass transmembrane protein αKl, which is primarily expressed in the distal tubule of kidneys, parathyroid gland, and choroid plexus.7,8 αKl regulates mineral metabolism and has an anti-inflammatory effect.9 A soluble form of αKl, which is produced by shedding the transmembrane form, is detected in serum.10 Serum levels of soluble αKl (sαKl) show several protective effects and is reported to relate with inflammatory cytokines.11,12
Focusing on the protective aspect of sαKl, we previously reported the unique association of serum levels of sαKl with smoking and psychological stress.13,14 We demonstrated that smoking and stressed condition increased serum levels of sαKl in men. Moreover, sαKl levels associated with interleukin (IL)-6, suggesting that sαKl regulates IL-6. As sαKl has protective effects, we concluded that increased serum levels of sαKl might be a compensatory response to protect against the harmful effects of smoking and psychological stress.
sαKl levels are reduced in some diseases, including type 2 diabetes mellitus and coronary artery disease.6,15 However, the association of sαKl with pre-clinical disorders has not been well studied. Assessing the development of disorders is important to prevent diseases and disease progression. Thus, in the present study, we evaluated the role of sαKl in preventing disease progression by assessing the association of serum levels of sαKl with several pre-clinical disorders.
Methods
Study Subjects
This study was designed as a prospective cohort study of employees at the Osaka University. The subjects were individuals who underwent an annual health checkup in the Osaka University Health and Counseling Center during the entry period between April 2005 and March 2008. The inclusion criteria for this study were as follows: (1) male subjects, (2) ages of 40 and 60 years, (3) with no underlying disease. Information on their medical history, current treatments, and smoking status was obtained via questionnaires. From 874 individuals who met the inclusion criteria, 88 subjects were randomly selected for the study. Among the 88 subjects, eight were excluded because of no follow-up between their baseline visit and March 2018. The final study cohort consisted of 80 men with at least one follow-up visit (Figure 1). This study was carried out in accordance with the Declaration of Helsinki and the ethics guidelines for clinical research from the Ministry of Health, Labour and Welfare and the Ministry of Education, Culture, Sports, Science and Technology. All experimental protocols in this study were approved by the Ethics Committee of Health and Counseling Center, Osaka University and written informed consent was obtained from all subjects prior to participation in the study.
 |
Figure 1 Flow diagram of inclusion and exclusion of the study subjects. |
Physical and Biochemical Parameters
As the baseline data, physical and biochemical parameters were obtained from all subjects at their first visit during the entry period. Body mass index (BMI: body weight (kg) divided by squared height (m2)), waist circumference (WC) at the umbilical level, systolic blood pressure (SBP), and diastolic blood pressure (DBP) were measured as physical parameters.
Serum was collected from subjects between 9 and 11 AM after an overnight fast and kept at ≤ –20°C until assayed. Serum concentrations of creatinine (Cr), uric acid (UA), triglycerides (TG), low-density lipoprotein-cholesterol (LDL-C), fasting plasma glucose (FPG), and sαKl were measured as biochemical parameters. Serum levels of sαKl were measured with a sandwich enzyme-linked immunoassay system according to the manufacturer’s instructions (Immuno-Biological Laboratories, Takasaki, Japan).
Pre-Clinical Disorder Assessment
Pre-clinical disorders were categorized as follows: overweight was defined as BMI ≥ 25 kg/m2 according to the recommendations of the World Health Organization; abdominal obesity was defined as WC ≥ 85 cm according to the guideline for abdominal obesity in Japanese;16 high blood pressure was defined as SBP ≥ 135 mmHg and/or DBP ≥ 85 mmHg using the guideline for hypertension in Japanese.17 In the final stages of the study, we used SBP ≥ 140 mmHg and/or DBP ≥ 90 mmHg to define hypertension; a high Cr level was defined as Cr ≥ 1.05 mg/dL using the upper normal value limit; and a high UA level was defined as UA ≥ 8 mg/dL by Japanese guideline for the management of hyperuricemia.18 According to the criteria of the Japan Atherosclerosis Society, hypertriglyceridemia and hyper-LDL cholesterolemia were defined as TG ≥ 150 mg/dL and LDL-C ≥ 140 mg/dL, respectively.19 A high FPG level was defined as FPG ≥ 100 mg/dL according to the criteria of the Japan Diabetes Society.20
Each pre-clinical disorder category was assessed to all study subjects. When assessing each disorder, we excluded subjects whose baseline data exceeded the values defined above. Therefore, among the 80 subjects, overweight: 20, abdominal obesity: 32, high blood pressure: 29, high Cr level: 7, high UA level: 7, hypertriglyceridemia: 17, hyper-LDL cholesterolemia: 24, and high FPG level: 10 subjects were excluded regarding each pre-clinical disorder assessment. The onset of each disorder was defined as the time when values measured at the follow-up visit exceeded the defined values or those at the initiation of treatment for the disorder. Hypertension, hypertriglyceridemia, and high FPG were used as factors for evaluating multiple pre-clinical disorders.
The term “progression of pre-clinical disorders” or “pre-clinical order progression” was used when the measurement values at the follow-up visit were progressing from normal range to the defined pre-clinical disorder range.
Statistical Analyses
All statistical analyses were performed using STATA 14 (STATA Corp LLC, College Station, TX, USA). The distribution of continuous variables was tested by the Shapiro–Wilk test. Normally distributed variables are presented as means ± standard deviation; non-normally distributed variables are reported as medians with the interquartile range. Student’s t-test, Mann–Whitney U-test, or chi-squared test were used to compare the difference between the two groups. Multivariable logistic regression analysis was performed to evaluate the predictive risk factors for pre-clinical disorders progression. Variables were entered into the multivariable model using forward selection (p < 0.2 included). To compare the predictive risk factors, we constructed the Receiver operator characteristic (ROC) curves and determined their area under the curve (AUC).21 For multi-group comparisons, Dunnett’s test was used to compare groups and the nptrend command of STATA software was used for the trend test. Statistical significance was set at P < 0.05.
Results
Serum Levels of sαKl Were Low in Subjects Developing Pre-Clinical Disorders
As shown in Table 1, we confirmed that there were no significant differences in baseline characteristics between the study subjects (n = 80) and individuals who met the inclusion criteria (n = 874). The mean duration of follow-up of the study subjects was 6.3 ± 3.1 years. The median age at the time of enrollment was 48 (42−52) years. The mean serum level of sαKl was 500 ± 172 pg/mL.
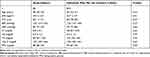 |
Table 1 Baseline Characteristics |
Table 2 shows the associations of serum levels of sαKl with the progression of each pre-clinical disorder. Baseline serum levels of sαKl were significantly lower in subjects developing a high FPG level than in subjects not developing a high FPG level (p = 0.004). In subjects with the progression of abdominal obesity, high blood pressure, high Cr level, high UA level, hypertriglyceridemia, and hyper-LDL cholesterolemia, baseline sαKl levels tended to be lower than in subjects without progression of those disorders, however significant differences were not shown. It is suggested that low serum levels of sαKl might promote the progression of pre-clinical disorders, especially hyperglycemia.
 |
Table 2 Baseline Serum Levels of Soluble Alpha-Klotho (sαKl) in the Subjects with or without Pre-Clinical Disorder Progression |
During their follow-up period, 20 subjects were diagnosed with some diseases; including hypertension, dyslipidemia, type 2 diabetes mellitus, hyperuricemia, and cardiac disease. We evaluate the difference of sαKl levels in subjects with diseases (539 ± 262 pg/mL) between the subjects without diseases (509 ± 166 pg/mL) and confirmed that there was no significant difference (p = 0.64).
Low sαKl Levels is Suggested to Predict Hyperglycemia
We further evaluate the relationship between serum levels of sαKl and the progression of a high FPG level. Table 3 shows the baseline characteristics of subjects with or without progression of a high FPG level. Multivariable logistic regression analysis showed that low level of sαKl and high baseline FPG level were independent predictive factors for the progression of a high FPG level (Table 4). Evaluating each AUC, both FPG and sαKl levels showed moderate accuracy with the progression of a high FPG level (Figure 2). Adding both factors slightly increased the AUC to 0.84, however a significant difference was not shown.
 |
Table 3 Baseline Characteristics in Subjects with or without Progression of High Fasting Plasma Glucose Level |
 |
Table 4 Multivariable Logistic Analysis: Risk Factors for High Fasting Plasma Glucose Level |
 |
Table 5 Baseline Serum Levels of sαKl Subject with or without Hypertension Among Never-Smokers and Smokers |
Moreover, we divided the study subjects into two groups according to the sαKl levels and compared the frequency of developing a high FPG level in the low (310 ± 76 pg/mL) and high (609 ± 112 pg/mL) sαKl groups. Individuals in the low sαKl group were significantly more likely to develop a high FPG level than those in the high sαKl group (χ2 (1) = 8.34, p = 0.0004).
These results suggest that decreased serum levels of sαKl would be a predictive factor for the progression of hyperglycemia.
Association of sαKl Levels with Disorders in Never-Smokers
We previously reported that a smoking habit upregulated the serum levels of sαKl.13 Therefore, we assessed the smoking population in the study subjects and the association of serum levels of sαKl with hypertension using the hypertension definition: SBP ≥ 140 mmHg and/or DBP ≥ 90 mmHg (Table 5). Among the subjects studied, there were 44 smokers and 36 never-smokers. The baseline serum level of sαKl in the smoker group was 541 ± 158 pg/mL. This was significantly higher than that in the never-smoker group (455 ± 175 pg/mL, p = 0.029). Serum levels of sαKl in subjects developing hypertension were significantly higher in smokers than in never-smokers (p = 0.041). Among never-smoker group, serum levels of sαKl tended to be lower in subjects with the progression of hypertension than in subjects without progression of hypertension. As smoking status might affect sαKl levels, we confirmed the association between sαKl levels and each pre-clinical disorder in the never-smoker group. We found that serum levels of sαKl in the never-smoker group were significantly lower for subjects developing a high FPG level (322 ± 94 pg/mL) than in subjects without this change (509 ± 174 pg/mL, p = 0.0005).
Serum Levels of sαKl in Subjects with Multiple Disorders
As the serum levels of sαKl were low in subjects with the progression of pre-clinical disorders, we evaluated the sαKl levels in subjects with multiple disorders: hypertension, hypertriglyceridemia, and high FPG. To exclude the influence of smoking, the never-smoker group was used for comparison. As shown in Figure 3, subjects with multiple disorders progression tended to have lower serum levels of sαKl. We also found that sαKl levels in subjects with more than two disorders progression tended to be lower than those in subjects with no disorder.
Discussion
The number of people developing pre-clinical states of disorders, such as hypertension, dyslipidemia, and hyperglycemia, is increasing in modern society. Since the morbidity and mortality of diseases caused by these disorders are still high,22 assessing their progression is important. The present study identified a relationship between sαKl levels and the progression of pre-clinical disorders.
αKl functions as a co-receptor for fibroblast growth factor 23 and has a central role in maintaining phosphate homeostasis.23 As αKl influences adipose cell maturation and glucose metabolism, αKl is regarded to have an essential role in whole-body energy metabolism.24 Furthermore, serum levels of sαKl decrease in obese, chronic kidney disease, and diabetic patients.6,25,26
In the present study, we analyzed the association of sαKl levels with the progression of several pre-clinical disorders. We found that baseline serum levels of sαKl were low in subjects progressing to a high FPG level. Serum levels of sαKl also exhibited a similar relationship with other pre-clinical disorders. We further identified that a low level of sαKl was independent predictive factor for the progression of a high FPG level. Moreover, in subjects with low baseline, sαKl were more likely to progress to a high FPG level. These results suggest that subjects with low serum levels of sαKl are likely to develop the pre-clinical state of disorders.
We used BMI and WC as pre-clinical disorder assessment for overweight and abdominal obesity, respectively. The relationship between sαKl levels and WC showed the same tendency as other disorders; however, BMI showed a different tendency. Since WC relates strongly to visceral fat accumulation, WC might associate with other disorders more strongly than BMI.27,28
We previously reported that serum levels of sαKl were upregulated by smoking and psychological stress in healthy subjects.13,14 As αKl levels are reported to be reduced in patients with chronic obstructive pulmonary disease and depressive symptoms; diseases which are caused by smoking and psychological stress, respectively,29,30 increased serum levels of sαKl may be a protective response to prevent disease progression. sαKl levels in never-smoker subjects progressing to hypertension tended to be lower than in subjects without such progression. Moreover, in the never-smoker group, there was a larger difference of sαKl levels in those progressing to a high FPG level than in the entire subjects. Excluding the influence of smoking, the difference of sαKl levels between the subjects with and without progression of pre-clinical disorders was more obvious.
αKl has several protective effects. For example, αKl regulates vascular function by improving endothelial dysfunction. It is reported that delivery of the Klotho gene decreased blood pressure and reduced medial hypertrophy of the aorta, and perivascular fibrosis of the coronary artery, in an animal model that displayed multiple atherogenic risk factors.31 Moreover, αKl exerts an anti-inflammatory effect by suppressing the TNF-α-induced expression of adhesion molecules, NF-kB activation, and RIG-I-mediated activation of IL-6 and IL-8.9,32 In addition, serum levels of sαKl revealed to have protective effects by suppressing cellular apoptosis, oxidative damage, and associating with pro-inflammatory status.11,12 We previously demonstrated that serum levels of sαKl were related with IL-6 levels, and that increased sαKl levels could be a compensatory response to inflammatory stress.13 Considering these protective aspects of αKl and sαKl levels, it is quite possible that increased sαKl levels would prevent the progression of various disorders and disease.
As with metabolic syndrome, people tend to suffer multiple disorders simultaneously. Therefore, we assessed differences in serum levels of sαKl in subjects according to the number of disorders they displayed. Although we could not find a significant difference, subjects with multiple disorders progression tended to show lower serum levels of sαKl, suggesting that extremely low levels of sαKl might enable or cause the progression of multiple disorders. This finding also supports our hypothesis that high serum levels of sαKl prevent the progression of several pre-clinical disorders.
This study has some limitations. Since the study was exploratory research to find a new predictive marker for the pre-clinical disorder, the sample size was set in small size. Even though the sample size was small, we could detect a statistically significant effect of sαKl. We are willing to confirm the result in a lager sample size in our further study. Another limitation is that all subjects were recruited from a single center. To confirm the effect of sαKl, a multicenter study is preferable in the future study.
Conclusion
We evaluated the association between serum levels of sαKl and the progression of several pre-clinical disorders. Subjects with low baseline serum levels of sαKl developed a high FPG level. Moreover, sαKl levels in subjects with multiple disorders progression tended to present lower than those in subjects with no disorder progression.
Acknowledgments
The authors would like to thank all the nurses and technicians who helped collect the data. The authors would also like to thank all the study participants.
Funding
This work was supported by JSPS KAKENHI (Grant Number JP18K17923).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wilson PW, D’Agostino RB, Parise H, Sullivan L, Meigs JB. Metabolic syndrome as a precursor of cardiovascular disease and type 2 diabetes mellitus. Circulation. 2005;112(20):3066–3072. doi:10.1161/CIRCULATIONAHA.105.539528
2. Chei CL, Yamagishi K, Tanigawa T, et al. Metabolic syndrome and the risk of ischemic heart disease and stroke among middle-aged Japanese. Hypertens Res. 2008;31(10):1887–1894. doi:10.1291/hypres.31.1887
3. Wu J, Qiu L, Cheng XQ, et al. Hyperuricemia and clustering of cardiovascular risk factors in the Chinese adult population. Sci Rep. 2017;7(1):5456. doi:10.1038/s41598-017-05751-w
4. Martin-Nunez E, Donate-Correa J, Muros-de-Fuentes M, Mora-Fernandez C, Navarro-Gonzalez JF. Implications of klotho in vascular health and disease. World J Cardiol. 2014;6(12):1262–1269. doi:10.4330/wjc.v6.i12.1262
5. Zhou HJ, Li H, Shi MQ, et al. Protective effect of klotho against ischemic brain injury is associated with inhibition of RIG-I/NF-κB signaling. Front Pharmacol. 2017;8:8. doi:10.3389/fphar.2017.00950
6. Nie F, Wu D, Du H, et al. Serum klotho protein levels and their correlations with the progression of type 2 diabetes mellitus. J Diabetes Complications. 2017;31(3):594–598. doi:10.1016/j.jdiacomp.2016.11.008
7. Kuro-o M, Matsumura Y, Aizawa H, et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997;390(6655):45–51. doi:10.1038/36285
8. Suga T, Kurabayashi M, Sando Y, et al. Disruption of the klotho gene causes pulmonary emphysema in mice. Defect in maintenance of pulmonary integrity during postnatal life. Am J Respir Cell Mol Biol. 2000;22(1):26–33. doi:10.1165/ajrcmb.22.1.3554
9. Maekawa Y, Ishikawa K, Yasuda O, et al. Klotho suppresses TNF-alpha-induced expression of adhesion molecules in the endothelium and attenuates NF-kappaB activation. Endocrine. 2009;35(3):341–346. doi:10.1007/s12020-009-9181-3
10. Yamazaki Y, Imura A, Urakawa I, et al. Establishment of sandwich ELISA for soluble alpha-Klotho measurement: age-dependent change of soluble alpha-Klotho levels in healthy subjects. Biochem Biophys Res Commun. 2010;398(3):513–518. doi:10.1016/j.bbrc.2010.06.110
11. Ravikumar P, Ye J, Zhang J, et al. Alpha-klotho protects against oxidative damage in pulmonary epithelia. Am J Physiol Lung Cell Mol Physiol. 2014;307(7):L566–575. doi:10.1152/ajplung.00306.2013
12. Martin-Nunez E, Donate-Correa J, Ferri C, et al. Association between serum levels of klotho and inflammatory cytokines in cardiovascular disease: a case-control study. Aging. 2020;12(2):1952–1964. doi:10.18632/aging.102734
13. Nakanishi K, Nishida M, Harada M, et al. Klotho-related molecules upregulated by smoking habit in apparently healthy men: a cross-sectional study. Sci Rep. 2015;5(1):14230. doi:10.1038/srep14230
14. Nakanishi K, Nishida M, Taneike M, et al. Implication of alpha-klotho as the predictive factor of stress. J Investig Med. 2019;67(7):1082–1086. doi:10.1136/jim-2018-000977
15. Navarro-Gonzalez JF, Donate-Correa J, Muros de Fuentes M, Pérez-Hernández H, Martínez-Sanz R, Mora-Fernández C. Reduced klotho is associated with the presence and severity of coronary artery disease. Heart. 2014;100(1):34–40. doi:10.1136/heartjnl-2013-304746
16. Examination Committee of Criter T, Examination Committee of Criteria for ‘Obesity Disease’ in J, Japan Society for the Study of O. New criteria for ‘obesity disease’ in Japan. Circ J. 2002;66(11):987–992. doi:10.1253/circj.66.987
17. Shimamoto K, Ando K, Fujita T, et al. The Japanese society of hypertension guidelines for the management of hypertension (JSH 2014). Hypertens Res. 2014;37(4):253–390.
18. Yamanaka H. Japanese guideline for the management of hyperuricemia and gout: second edition. Nucleosides Nucleotides Nucleic Acids. 2011;30(12):1018–1029. doi:10.1080/15257770.2011.596496
19. Kinoshita M, Yokote K, Arai H, et al. Japan atherosclerosis society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2017. J Atheroscler Thromb. 2018;25(9):846–984. doi:10.5551/jat.GL2017
20. Haneda M, Noda M, Origasa H, et al. Japanese clinical practice guideline for diabetes 2016. J Diabetes Investig. 2018.
21. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–845. doi:10.2307/2531595
22. Mendis S, Davis S, Norrving B. Organizational update: the world health organization global status report on noncommunicable diseases 2014; one more landmark step in the combat against stroke and vascular disease. Stroke. 2015;46(5):e121–122. doi:10.1161/STROKEAHA.115.008097
23. Kuro OM. The klotho proteins in health and disease. Nat Rev Nephrol. 2019;15(1):27–44. doi:10.1038/s41581-018-0078-3
24. Razzaque MS. The role of klotho in energy metabolism. Nat Rev Endocrinol. 2012;8(10):579–587. doi:10.1038/nrendo.2012.75
25. Amitani M, Asakawa A, Amitani H, et al. Plasma klotho levels decrease in both anorexia nervosa and obesity. Nutrition. 2013;29(9):1106–1109. doi:10.1016/j.nut.2013.02.005
26. Shimamura Y, Hamada K, Inoue K, et al. Serum levels of soluble secreted alpha-klotho are decreased in the early stages of chronic kidney disease, making it a probable novel biomarker for early diagnosis. Clin Exp Nephrol. 2012;16(5):722–729. doi:10.1007/s10157-012-0621-7
27. Neeland IJ, Ross R, Despres JP, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. 2019;7(9):715–725. doi:10.1016/S2213-8587(19)30084-1
28. Despres JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881–887. doi:10.1038/nature05488
29. Gao W, Yuan C, Zhang J, et al. Klotho expression is reduced in COPD airway epithelial cells: effects on inflammation and oxidant injury. Clin Sci (Lond). 2015;129(12):1011–1023. doi:10.1042/CS20150273
30. Prather AA, Epel ES, Arenander J, et al. Longevity factor klotho and chronic psychological stress. Transl Psychiatry. 2015;5(6):e585. doi:10.1038/tp.2015.81
31. Saito Y, Nakamura T, Ohyama Y, et al. In vivo klotho gene delivery protects against endothelial dysfunction in multiple risk factor syndrome. Biochem Biophys Res Commun. 2000;276(2):767–772. doi:10.1006/bbrc.2000.3470
32. Liu F, Wu S, Ren H, Gu J. Klotho suppresses RIG-I-mediated senescence-associated inflammation. Nat Cell Biol. 2011;13(3):254–262. doi:10.1038/ncb2167
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


