Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 13
Serum CA125 Level Is Associated with Diabetic Retinopathy in Chinese Patients with Type 2 Diabetes
Authors Yao L, Zhong Y, He L, Wang Y , Wu J, Geng J, Zhou Y, Zhang J, Chen J, Shan Z, Teng W, Xu Y , Chen L, Liu L
Received 23 February 2020
Accepted for publication 7 May 2020
Published 22 May 2020 Volume 2020:13 Pages 1803—1812
DOI https://doi.org/10.2147/DMSO.S250928
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Konstantinos Tziomalos
Litong Yao,1,2,* Yifan Zhong,3,* Lingzi He,1,* Yan Wang,4,* Jingyang Wu,3 Jin Geng,3 Yun Zhou,3 Jiahua Zhang,3 Jun Chen,3 Zhongyan Shan,5 Weiping Teng,5 Yingying Xu,2 Lei Chen,3 Lei Liu3
1China Medical University, Shenyang 110001, People’s Republic of China; 2Department of Breast Surgery, The First Hospital of China Medical University, Shenyang 110001, People’s Republic of China; 3Department of Ophthalmology, The First Hospital of China Medical University, Shenyang 110001, People’s Republic of China; 4Department of Radiology, General Hospital of Liaohe Oil Field, Panjin 124010, People’s Republic of China; 5Department of Endocrinology and Metabolism, Institute of Endocrinology, Liaoning Provincial Key Laboratory of Endocrine Diseases, The First Affiliated Hospital of China Medical University, Shenyang 110001, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lei Chen; Lei Liu Email [email protected]; [email protected]
Background: To investigate the association between serum carbohydrate antigen 125 (CA125) and the presence as well as severity of diabetes retinopathy (DR) in Chinese adult patients with type 2 diabetes.
Methods: A hospital-based cross-sectional study was conducted from February 2012 to November 2018. DR was assessed using Early Treatment Diabetic Retinopathy Study criteria. Vision-threatening DR (VTDR) was diagnosed if subjects had severe non-proliferative DR (NPDR), proliferative DR (PDR), or clinically significant macular edema (CSME). Multivariate logistic regression models were applied to explore the associations.
Results: Among the 2696 participants, the overall prevalence of DR was 25.1%, of which the prevalence of mild NPDR, moderate NPDR, and VTDR was 10.8%, 4.5%, and 9.9%, respectively. Serum CA125 level was significantly higher in participants with DR and increased with the severity of DR (P = 0.013). After accounting for age, gender, smoking, drinking, duration of diabetes, anti-diabetic agents use, systolic blood pressure, pulse pressure, weight, hemoglobin A1c and fasting plasma glucose levels, CA125 level was significantly associated with subjects in any-severity DR (odds ratio [OR] 1.006 [95% confidence interval CI: 1.002– 1.010], P = 0.006) and VTDR (1.008 [1.003– 1.013], P = 0.001). When CA125 was treated as categorized variables, the prevalence of VTDR might increase as improving CA125 quartiles (P value for trend = 0.017).
Conclusion: In this study, serum CA125 level was associated with the presence and severity of DR in Chinese patients with type 2 diabetes. Further prospective studies should be warranted to validate the feasible role of CA125 as well as other biomarkers.
Keywords: type 2 diabetes, diabetes retinopathy, tumor biomarker, CA125, association
Introduction
Diabetes retinopathy (DR) is one of the prevailing chronic complications of diabetes caused by accumulated impairments of retinal microvasculature, and leads to severe or even perpetual visual loss.1,2 Statistically, nearly 80% diabetic patients tracing for over ten years were expected to suffer from this complication, and DR has been proven to be the dominant cause of blindness worldwide in adults of working age.3,4 Accompanying with the prevalence of type 2 diabetes has reached global epidemic proportions, the morbid risk of DR will continue to rise. Based on current epidemiologic evidences and clinical investigations, factors such as the duration of diabetes, the control of glycemia and blood pressure significantly influenced the presence of DR, but the mechanisms responsible for DR remained unclear.5,6 In addition, researchers also indicated that maintenance of those factors could not decelerate the progression of retinopathy.7 The benefit of timely screening and treatment in the early stage of DR is observable for patient’s outcome. Currently, the diagnosis and evaluation of DR depends on fundus examination by retina photographs, and no exact parameter for predicting presence of DR has been identified.8,9
Several investigations have provided valuable evidence independent of well-documented related risk factors of diabetes, that cancer was also linked to the prevalence of diabetes.10,11 Previous studies illustrated that some status correlated to diabetes and retinopathy might be postulated as a key role in cancer’s deterioration or mortality, such as dysglycemia, insulin resistance and inflammatory cytokines.12 Of note, Azuma et al recently identified DR as a risk factor associated with the development of hepatocellular carcinoma, further supporting the possible associations between DR and cancer.13 Tumor biomarkers are a series of substances which reflect the occurrence and proliferation of cancers, and widely applied in clinic for tumor early detection, diagnosis, monitoring and prognostic evaluation.14 Limited researches have evaluated tumor biomarkers in connection to diabetes and corresponding complications, and an ascending or descending trend of markers were observed in patients with diabetes.
A retrospective analysis based on Chinese population suggested that the elevation of tumor biomarkers existed obviously in patients with type 2 diabetes compared to normal controls, especially carbohydrate antigen 125 (CA125) levels which increased in type 2 diabetes patients with multiple complications.15 CA125, one of the most common tumor biomarkers, was found to be produced mainly by coelomic epithelium-derived tissues, including thoracic, pericardial and peritoneal mesothelium, and Mullerian duct epithelial structures. Esteghamati et al identified the CA125 level with regard to metabolic syndrome and diabetes, and investigated the relationship between CA125 level and various associated anthropometric and biochemical parameters.16,17 Researchers demonstrated that CA125 was inversely related to diabetes status and associated variables, while the result was inconsistent with the finding in a study based on Chinese population. Serum CA125 level was higher in patients with diabetes and related complications, especially diabetic foot and compound conditions.18 To date, no data has been reported that there is any correlation between the level of serum CA125, and the presence as well as severity of DR. Thus, we conducted a hospital-based cross-sectional study to investigate the association between CA125 and DR in Chinese adult patients with type 2 diabetes.
Materials and Methods
Study Population
Between February 2012 to November 2018, 2880 patients aged 18 years and over were recruited as having confirmed diabetes at the First Affiliated Hospital of China Medical University (CMU1h). The CMU1h is located in Shenyang City, situated in the northern-east part of China, and functions as a National key discipline for both Endocrine and Metabolic disease. All patients were registered in the hospital-based diabetes registry and followed-up database system. Each participant was requested to complete a comprehensive questionnaire at the time of his/her initial admission. This diabetes registry records clinical findings and information for all patients admitted to the CMU1h. Type 2 diabetes was defined as fasting plasma glucose (FPG) ≥ 7.0 mmol/l, hemoglobin A1c (HbA1c) level ≥ 6.5% (48 mmol/mol), self-reported previous clinician-diagnosed diabetes or use of anti-diabetic agents, according to the criteria of the American Diabetes Association Standards of diabetes care.9 Exclusion criteria removed participants with type 1 diabetes, pregnancy, diabetic ketoacidosis, malignancy tumors such as cervical, endometrial, ovarian, fallopian tube and breast cancer; benign diseases including ovarian chocolate cysts, endometriosis, pelvic inflammatory disease, appendicitis, peritonitis and so on; cardiovascular diseases such as chronic heart failure, atrial fibrillation, aortic stenosis and coronary heart disease; and a history of mental disorders, autoimmune disease such as systemic lupus erythematosus or other connective tissue disease, and severe liver or kidney insufficiency. Participants with unrecorded retinal fundus photographs in one or both eyes were also excluded.
Measurements of Anthropometric and Biochemical Parameters
At baseline, the following anthropometric and clinical parameters of participants were achieved: age, gender, height, weight, body mass index (BMI), status of smoking and alcohol-drinking, heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), pulse pressure (PP), duration of diabetes and use of anti-diabetic agents. BMI was calculated as body weight (kg) divided by square height (m). Smokers were defined as patients with self-reported previous or current history of smoking, regardless of their smoking frequency, and the definition of drinkers was similar to smokers. BP was measured three times by trained nurses using standard manual mercury sphygmomanometers and the average was recorded. PP was calculated as SBP minus DBP.
Blood biochemical parameters including HbA1c, FPG, serum total cholesterol (TC), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C) levels were measured at the Endocrinology Laboratory of China Medical University, using blood samples collected in the morning from individuals after an overnight fasting (at least 8 hours without calorie intaking). HbA1c was evaluated by using high-performance liquid chromatography (HLC-723 G7; TOSHO, Tokyo, Japan). FPG was determined by applying the glucose oxidase methods. Lipid parameters were measured by using standard enzymatic methods through biochemical analyzers (7600-120; Hitachi, Tokyo, Japan). We use Roche Cobas E601 or E602 electrochemiluminescence immunoassay analyzer to determine tumor biomarkers including carcinoembryonic antigen (CEA), alpha-fetoprotein (AFP), CA125, CA153 and CA199 at the Department of Clinical Laboratory of the First Affiliated Hospital of China Medical University. This assay had a wide working range from 0.5 mmol/L to 1000 mmol/L. It was sensitive (limit of detection: 0.5 mmol/L), precise and reproducible with low inter- and intra-coefficient of variances (< 20%).
Assessment of DR
Two-field fundus photographs which centered on the optic disk and fovea were assigned to each eye by trained photographers, using nonmydriatic fundus camera (CR6-45NM; Canon, Inc., Tokyo, Japan). The assessment of DR was performed by ophthalmologists in blinded manners at the Department of Ophthalmology, the First Affiliated Hospital of China Medical University, according to the Early Treatment for Diabetic Retinopathy Study (ETDRS) standards.19,20 DR was considered to be present if one or more retinal microaneurysms or retinal blot hemorrhages existed with or without more severe lesions including cotton wool spots, intraretinal microvascular abnormalities, hard or soft exudates, venous bleeding and retinal new vessels. And, if the binocular DR was diagnosed unequally, the result of the more advanced eye should be analyzed. The severity of DR was graded following the modified Airlie House classification system, and categorized as non-DR, mild non-proliferative DR (NPDR), moderate NPDR, severe NPDR and proliferative DR (PDR).19 Vision-threatening DR (VTDR) was defined as the presence of severe NPDR, PDR, or clinically significant macular edema.
Statistical Analysis
Statistical analyses were performed using SPSS (IBM, version 23). Data are described as mean ± SD (continuous variables) or percentage (categorical variables) unless otherwise indicated. Normal distribution test was performed by Kolmogorov–Smirnov test. For the continuous variables, if the variable was normal distribution, one-way ANOVA or Jonckheere-Terpstra test for continuous variables with homogeneous or heterogeneous variance, while Kruskal–Wallis H-test was performed for abnormal distribution. For categorical variables, the comparisons were used Chi-square test and Fisher exact test. Quartiles were defined as the 25%, 50% and 75% percentiles of CA125 level, then, its level was treated as categorized variables according to four quartiles (quartile 1 or Q1: < 6.97 U/mL; quartile 2 or Q2: 6.97–9.72 U/mL; quartile 3 or Q3: 9.73–13.48 U/mL; quartile 4 or Q4: ≥ 13.49 U/mL), with the highest tertile being the reference tertile. Further, multivariate logistic regression models were conducted to estimate associations between DR severity strata (dependent variable) and CA125 levels (independent variable) individually in each stratified method. Model 1 adjusted with age and gender. In Model 2, confounder variables identified as significant in the univariate analyses including duration of diabetes, anti-diabetic agents use, SBP, PP, weight, HbA1c and FPG levels, were entered into logistic regression analyses. Model 3 included all of the confounders mentioned above as well as status of smoking and alcohol-drinking, which had shown potential association with disease severity strata during the literature search. In all 3 models, each of four DR subgroups was assigned with a number according to DR severity strata: 0 for non-DR, 1 for mild NPDR, 2 for moderate NPDR and 3 for VTDR. A P-value less than 0.05 (two-tailed) was considered statistically significant.
Results
Complete information was available for 2696 subjects (93.6% of the total sample). Table 1 listed basic characteristics of involved participants with different DR severities. Among all enrolled subjects with type 2 diabetes, the prevalence of DR was 25.1% (n = 677), in which mild NPDR, moderate NPDR and VTDR to be 10.8% (n = 290), 4.5% (n = 121), and 9.9% (n = 266), respectively. The results illustrated that DR severity strata was related to longer duration of diabetes, anti-diabetic agent use, SBP, PP, weight, BMI, HbA1c and FBG levels significantly (all P < 0.05). Among five observed tumor biomarkers, only CA125 levels were higher in participants with DR compared to non-DR subjects and increased significantly with severity of DR (P = 0.013). CEA, AFP, CA153 and CA199 did not vary significantly across different groups of DR severity strata. Table 2 categorized patients into four groups according to quartiles of CA125 levels. Clinical characteristics were compared among quartiles as shown in Table 2. The prevalence of DR severity increased with ascending CA125 quartiles (P = 0.016), especially in VTDR group. Thus, further analysis was carried out principally around CA125. Figure 1 illustrated the serum levels of CA125 in various stages of DR as box-and-whisker diagram.
 |
Table 1 Characteristics of Study Participants by the Presence and Severity of DR |
 |
Table 2 Characteristics of Study Participants by Quartiles of CA125 |
Table 3 showed the associations between CA125 and various stages of DR. In a binary logistic regression model, non-DR group was set as a control group, and a significant (odds ratio [OR] 1.007 [95% CI: 1.003–1.012], P = 0.001) association between DR and CA125 level was revealed after adjusting age and gender. Then, using a multivariate logistic regression model, the relation of CA125 level to the prevalence of DR severity strata was explored, and the results showed that the relationships were significant for moderate NPDR group (1.008 [1.002–1.014], P = 0.014) and VTDR group (1.009 [1.005–1.014], P < 0.001), but not significant for mild NPDR group (1.002 [0.995–1.010], P = 0.534). Further adjusting confounding factors in Model 3, VTDR group was still correlated with CA125 level significantly (1.008 [1.003–1.013], P = 0.001), as well as any-severity DR (1.006 [1.002–1.010], P = 0.006).
 |
Table 3 Associations Between CA125 and Various Stages of DR After Controlling for Confounding Factors |
When CA125 was regarded as a categorized variable in logistic regression model, Table 4 studied the association between CA125 quartiles and DR severity, setting Q4 as reference. Increases of CA125 quartiles correlated significantly with VTDR (Q1: 0.647 [0.457–0.917], P = 0.014; Q2: 0.595 [0.419–0.846], P = 0.004; Q3: 0.554 [0.387–0.794], P = 0.001) after adjusting age and gender in Model 1, and trend test was then carried out with P value equal to 0.003. The statistical significance did not remain in association between Q1 with VTDR in Model 2 or Model 3, but still existed in Q2 (Model 2: 0.630 [0.439–0.905], P = 0.012; Model 3: 0.629 [0.438–0.904], P = 0.012) and Q3 (Model 2: 0.630 [0.436–0.911], P = 0.014; Model 3: 0.639 [0.442–0.924], P = 0.017). Both P values for trend were significant (Model 2: P value for trend = 0.017; Model 3: P value for trend = 0.018), from which we can infer that prevalence of VTDR might increase as ascending CA125 quartiles.
 |
Table 4 Associations Between Quartiles of CA125 and Various Stages of DR After Controlling for Confounding Factors |
Discussion
Known as retinal microcirculation disorders, DR would attack more than half of diabetic patients with 10 years or longer duration, disregarding its varying severity, and the global prevalence of DR rises continuously.21 A meta-analysis based on the data from 35 studies including 22,896 participants with diabetes, revealed that the overall prevalence of any DR was 34.6%, and 28.4% in patients with type 2 diabetes.5 Among the Chinese population, a recent systemic review integrating available epidemiological studies since 1990 estimated that the pooled prevalence of any DR, NPDR and PDR in diabetic patients were 18.5%, 15.1% and 0.99%, respectively.22 In this large sample, cross-sectional, hospital-based study, similar to the published findings, 25.1% of the participants with type 2 diabetes were expected to have DR, 15.2% and 9.9% of whom suffered from NPDR and VTDR, supporting the reliability of methodology and effectiveness of population samples. Our findings underscored the importance of optimal glycemia management, monitoring of blood pressure and early detection of DR in preventing the progression of microvascular complications and reducing the risk of visual impairment.
Considering the observed associations of cancer and diabetes, both cause-and-effect relationship or confounding from common risk factors, we suggested that there may be a connection between cancer and the progression of diabetes complications.23,24 Elevated levels of tumor biomarkers were found in patients with certain types which reflected the carcinogenic status.25 On this basis, we collected the serum level of CA125 in patients with type 2 diabetes and examined possible associations between this tumor biomarker and the presence as well as severity of DR. Serum CA125 level was significantly related to the presence of DR in Chinese adult patients with type 2 diabetes. In addition, when DR was analyzed categorically, a statistically significant association between CA125 level and different DR severity strata was also observed. CA125 level was exceedingly higher in participants with more advanced DR such as VTDR, and there was no difference in mild or moderate NPDR participants inversely. After using multiple adjusted models and accounting for clinical confounders, there was still an increasing trend of VTDR risk, accompanying with the improvement of CA125 quartiles. Therefore, it was speculated that elevated CA125 level was consider as an independent predictor for the presence of DR and VTDR. Serum CA125 is an easily assessed laboratory parameter in clinical practice and could be expected to become a potential indicator to early detection of DR, enabling the clinicians to make timely treatment, and prevent the development of vision impairment.
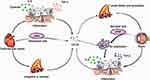
Previous study reported that CA125 was inversely associated with diabetes status and related variables.17 However, a research based on Chinese population illustrated that patients with diabetic foot and compound conditions expressed a higher serum CA125 level.18 While the relationship between DR and CA125 has not been explored till now. Pathological angiogenesis is the common characteristic of cancer and retinopathy.12 Our study illustrated that CA125 level was significantly higher in patients with DR, especially VTDR and the independent association was further confirmed. Consequently, the actual relationship between CA125 and the diabetic microvascular complication DR has not been explained fundamentally. Pathological angiogenesis is the common characteristic of cancer and retinopathy.26 Previous studies have discussed the potential mechanisms of serum CA125 elevation in cardiovascular diseases including chronic heart failure, atrial fibrillation, aortic stenosis, coronary heart disease, etc,27 and CA125 were produced from mesothelial cells mainly in response to inflammatory stimuli and fluid overload.25 Considering DR is a kind of hyperplastic lesion of micro-vessels, we put forward the theory that proliferative vessels might undergo similar process with above-mentioned cardiovascular diseases, such as vascular wall expansion and congestion, leading to overload of blood vessels. As for inflammation, a study on patients without ovarian tumor analyzed chronic clinical conditions, and CA125 was significantly higher in coronary heart disease and inflammatory bowel disease.28 Researchers discovered that those diseases would result in cytokine overexpression and activation, which may raise CA125.29 In a research on DR, obvious phosphorylation of c-Jun N-terminal kinase (JNK) signaling pathway was observed in microglial cells of retina, leading to polarization to M1 cell and triggering excessive inflammation Coincidently, CA125 synthesis is achieved through JNK signaling pathway of mesothelial cells.30 However, whether inflammation in retina associates with CA125 expression through the JNK pathway still needs further exploration. Potential signal pathway for the association between CA125 and DR was shown in Figure 2.
The major strengths of this hospital-based, cross-sectional study included large sample sizes with comprehensive variables and objective categorizations for DR severity strata, which could improve the authenticity and reliability of our findings. Nevertheless, several potential limitations should be taken into account. First, the cross-sectional design of our study limited the causal interferon between tumor biomarkers levels and the presence of DR. Whether elevated CA125 levels lead to the development of DR or act as the consequence of DR were still unknown. Further prospective researches with a similar conception were required to validate the associations and elucidate the mechanisms. Second, differences in study methodologies, ascertainment and classification of DR and population characteristics, could lead to comparisons between studies. The assessment and monitoring of DR in this study was based on two-field fundus photographs and it may contribute to underestimation of the presence of low-grade DR, especially mild and moderate NPDR, if the pathological lesions were in the peripheral regions of retina. The highly sensitive seven-field fundus photographs, which have been used in several investigations including the Early Treatment Diabetic Retinopathy Study (ETDRS), can play a critical role in detecting a more accurate perception of development of DR. And it should be noted that serum CA125 was a non-specific marker, benign conditions would influence the expression level of serum CA125, we have included related condition in exclusion criteria, however other uncommon conditions might lead to limited deviation. Additionally, the individuals participating in our study were hospital-based Chinese patients with type 2 diabetes. According to characteristic features, our findings might not be applicable to other ethnic and non-hospitalized patients.
Conclusion
In conclusion, we provided evidences that serum CA125 level was associated with the prevalence and severity of DR in patients with type 2 diabetes, especially VTDR. As a risk factor, CA125 level increased with the advanced DR severity in co-direction. The findings suggested that CA125 might be a reliable marker that represents metabolic states in the pathophysiology of retinopathy, as well as a possible predictor for monitoring DR or VTDR. Further prospective studies should be warranted to validate the feasible role of CA125 as well as other biomarkers and then elucidate related mechanisms of disease onset and development in DR.
Data Sharing Statement
L.L is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Ethics Approval and Informed Consent
This study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional review board (IRB) of the First Affiliated Hospital of China Medical University (AF-SOP-07-1.1-01/2019-13). Written informed consents were achieved for the entire study population included in this study.
Acknowledgments
Thanks to other investigators in the Department of Endocrinology and Metabolism, First Hospital of China Medical University. Thanks to Yuedong Hu, Kaibo Yang and Lele Huang for data collection who are working in Department of Ophthalmology, First Hospital of China Medical University.
Author Contributions
L.Y. and Yifan Zhong contributed to the study design, data collection and analysis, and manuscript writing. L.H., Y.W. and J.W. researched data interpretation and contributed to paper revision and editing. J.G., Yun Zhou, J.Z., J.C. contributed to data collection and management, and discussions. Z.S. and W.T. critically reviewed and edited the manuscript, and contributed to study design. Y.X., L.C. and L.L. were responsible for the conception, funding and final version. All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet. 2010;376(9735):124–136. doi:10.1016/S0140-6736(09)62124-3
2. Stitt AW, Curtis TM, Chen M, et al. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res. 2016;51:156–186. doi:10.1016/j.preteyeres.2015.08.001
3. Frank RN. Diabetic retinopathy. N Engl J Med. 2004;350(1):48–58. doi:10.1056/NEJMra021678
4. Kempen JH, O’Colmain BJ, Leske MC, et al. The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol. 2004;122(4):552–563.
5. Yau JW, Rogers SL, Kawasaki R, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556–564. doi:10.2337/dc11-1909
6. Nathan DM, Genuth S, Lachin J, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977–986.
7. Wong TY, Cheung CMG, Larsen M, Sharma S, Simó R. Diabetic retinopathy. Nat Rev Dis Primers. 2016;2:16012. doi:10.1038/nrdp.2016.12
8. Ferris FL. How effective are treatments for diabetic retinopathy? JAMA. 1993;269(10):1290–1291. doi:10.1001/jama.1993.03500100088034
9. American Diabetes Association. Standards of medical care in diabetes–2014. Diabetes Care. 2014;37(Suppl 1):S14–S80. doi:10.2337/dc14-S014
10. Shlomai G, Neel B, LeRoith D, Gallagher EJ. Type 2 diabetes mellitus and cancer: the role of pharmacotherapy. J Clin Oncol. 2016;34(35):4261–4269. doi:10.1200/JCO.2016.67.4044
11. Badrick E, Renehan AG. Diabetes and cancer: 5 years into the recent controversy. Eur J Cancer. 2014;50(12):2119–2125.
12. Giovannucci E, Harlan DM, Archer MC, et al. Diabetes and cancer: a consensus report. Diabetes Care. 2010;33(7):1674–1685. doi:10.2337/dc10-0666
13. Azuma S, Asahina Y, Kakinuma S, et al. Diabetic retinopathy as a risk factor associated with the development of hepatocellular carcinoma in nonalcoholic fatty liver disease. Dig Dis. 2019;37(3):247–254. doi:10.1159/000493580
14. Sturgeon C. Practice guidelines for tumor marker use in the clinic. Clin Chem. 2002;48(8):1151–1159. doi:10.1093/clinchem/48.8.1151
15. Yu C, Lei Q, Wang J, et al. Serum carbohydrate antigen 125 levels and incident risk of type 2 diabetes mellitus in middle-aged and elderly Chinese population: the Dongfeng-Tongji cohort study. Diab Vasc Dis Res. 2019;16(5):424–430. doi:10.1177/1479164119843095
16. Diamandis EP, Bast RC, Gold P, Chu TM, Magnani JL. Reflection on the discovery of carcinoembryonic antigen, prostate-specific antigen, and cancer antigens CA125 and CA19-9. Clin Chem. 2013;59(1):22–31. doi:10.1373/clinchem.2012.187047
17. Esteghamati A, Seyedahmadinejad S, Zandieh A, et al. The inverse relation of CA-125 to diabetes, metabolic syndrome, and associated clinical variables. Metab Syndr Relat Disord. 2013;11(4):256–261. doi:10.1089/met.2012.0058
18. Xie XY, Zhong RH. The relationship between serum tumor biomarker levels and complication of diabetes patients. Chin J Health Lab Technol. 2015;25:1005–1007.
19. Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs–an extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991;98(5 Suppl):786–806. doi:10.1016/S0161-6420(13)38012-9
20. Sasongko MB, Widyaputri F, Agni AN, et al. Prevalence of diabetic retinopathy and blindness in Indonesian adults with type 2 diabetes. Am J Ophthalmol. 2017;181:79–87. doi:10.1016/j.ajo.2017.06.019
21. Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. III. Prevalence and risk of diabetic retinopathy when age at diagnosis is 30 or more years. Arch Ophthalmol. 1984;102(4):527–532. doi:10.1001/archopht.1984.01040030405011
22. Song P, Yu J, Chan KY, Theodoratou E, Rudan I. Prevalence, risk factors and burden of diabetic retinopathy in China: a systematic review and meta-analysis. J Glob Health. 2018;8(1):010803. doi:10.7189/jogh.08.010803
23. Silidis KK, Kasimis JC, Lopez DS, Ntzani EE, Ioannidis JP. Type 2 diabetes and cancer: umbrella review of meta-analyses of observational studies. BMJ (Clinical Research Ed). 2015;350:g7607.
24. Hwangbo Y, Kang D, Kang M, et al. Incidence of diabetes after cancer development: a Korean National Cohort Study. JAMA Oncol. 2018;4(8):1099–1105. doi:10.1001/jamaoncol.2018.1684
25. Oubaha M, Miloudi K, Dejda A, et al. Senescence-associated secretory phenotype contributes to pathological angiogenesis in retinopathy. Sci Transl Med. 2016;8(362):362ra144. doi:10.1126/scitranslmed.aaf9440
26. Falcao F, de Oliveira FRA, da Silva M, Sobral Filho DC. Carbohydrate antigen 125: a promising tool for risk stratification in heart diseases. Biomark Med. 2018;12(4):367–381. doi:10.2217/bmm-2017-0452
27. Huang F, Chen J, Liu Y, Zhang K, Wang J, Huang H. New mechanism of elevated CA125 in heart failure: the mechanical stress and inflammatory stimuli initiate CA125 synthesis. Med Hypotheses. 2012;79(3):381–383. doi:10.1016/j.mehy.2012.05.042
28. Akinwunmi BO, Babic A, Vitonis AF, Cramer DW, Titus L, Tworoger SS. Chronic medical conditions and CA125 levels among women without ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2018;27(12):1483–1490.
29. De Gennaro L, Brunetti ND, Montrone D, De Rosa F, Cuculo A, Di Biase M. Inflammatory activation and carbohydrate antigen-125 levels in subjects with atrial fibrillation. Eur J Clin Invest. 2012;42(4):371–375. doi:10.1111/j.1365-2362.2011.02592.x
30. Li J, Song SJ, Liu FL, et al. Carbohydrate antigen 125 levels and clinical outcomes after off-pump coronary artery bypass grafting. Coron Artery Dis. 2015;26(5):432–436. doi:10.1097/MCA.0000000000000262
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.