Back to Journals » Veterinary Medicine: Research and Reports » Volume 11
Seroprevalence of Rift Valley Fever and West Nile Fever in Cattle in Gambella Region, South West Ethiopia
Authors Asebe G , Mamo G , Michlmayr D, Abegaz WE, Endale A , Medhin G, Larrick JW, Legesse M
Received 26 August 2020
Accepted for publication 27 October 2020
Published 19 November 2020 Volume 2020:11 Pages 119—130
DOI https://doi.org/10.2147/VMRR.S278867
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Young Lyoo
Getahun Asebe,1,2 Gezahegne Mamo,1 Daniela Michlmayr,3 Woldaregay Erku Abegaz,4 Adugna Endale,5,6 Girmay Medhin,5 James W Larrick,7 Mengistu Legesse5
1Department of Veterinary Microbiology, Immunology and Public Health, College of Veterinary Medicine, Addis Ababa University, Bishoftu, Ethiopia; 2College of Agriculture and Natural Resources, Gambella University, Gambella, Ethiopia; 3Division of Infectious Diseases and Vaccinology, School of Public Health, University of California Berkeley, Berkeley, CA, USA; 4Department of Microbiology, Immunology & Parasitology, School of Medicine, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 5Aklilu Lemma Institute of Pathobiology, Addis Ababa University, Addis Ababa, Ethiopia; 6School of Medicine, College of Medicine and Health Sciences, Dire Dawa University, Dire Dawa, Ethiopia; 7Panorama Research Institute, Sunnyvale, CA, USA
Correspondence: Getahun Asebe Email [email protected]
Introduction: Rift Valley fever (RVF) and West Nile fever (WNF) are re-emerging mosquito-borne zoonotic diseases that cause public health and economic crises. Ethiopia shares borders with South Sudan and Kenya, where these diseases are often documented. The free movement of animals and humans across these borders expects to increase the spread of these diseases. The current study was conducted to assess the occurrence of these diseases in the Gambella region of Ethiopia.
Methodology: We collected a total of 368 cattle serum samples from the Lare district on the border of South Sudan and measured the presence of IgG antibody against RVF and WNF virus infections using enzyme-linked immunosorbent assays (ELISA).
Results: The prevalence of anti-RVF virus IgG antibody was 7.6% (95% CI: 5.3– 10.82%), while that of anti-WNF virus IgG antibody was 5.4% (95% CI: 3.52– 8.29%). In this study higher seroprevalence of IgG antibodies to RVF virus infection was observed comparing to the WNF virus in cattle. There was no significant association between the prevalence and the cattle age, sex or sampled locations.
Conclusion: The detection of IgG antibody to RVF and WNF virus infections in the Gambella region warrants further study of active case findings and the dynamics of transmission.
Keywords: serology RVF; WNF; cattle; Gambella, Ethiopia
Background
Arboviruses are among the 72% of emerging infectious viruses that originate from wildlife.1 They transmit to different vertebrate hosts by arthropod species such as mosquitoes, ticks, sand flies and midges.2 Climatic changes influence the transmission of the viruses associated with the life cycle of the transmitting vectors.3
Rift Valley fever virus (RVFV) and West Nile virus (WNV) are among the re-emerging viruses transmitted through the bite of infected Aedes and Culex mosquitoes respectively.4,5 RVFV belongs to the family Bunyavirida, genus Phlebovirus.6 RVFV was first identified in 1931 near the Great Rift Valley of Kenya.6 Since then, this virus has caused many documented outbreaks in humans and livestock in many sub-Saharan African countries, in the Arabian Peninsula, and some Indian Ocean Islands.7,8 RVFV infects a range of vertebrate hosts including humans, livestock, and wildlife, with ruminants being among the most affected hosts.9 Infection with RVFV can result in severe disease that can lead to high morbidity and mortality in animals including abortions and stillbirths.10,11
In humans, the virus causes neonatal mortalities and moderate influenza-like illness. Similarly, severe complications and small proportion of deaths may occur among some groups of infected people.11–14
Unusually, heavy and sustained rainfall leads to outbreaks of the disease by favoring breeding of the mosquito vectors.15 Study reports documented presence of RVFV in some East African countries: South Sudan,16 Kenya10,17 Somalia, Tanzania and Sudan.18 where some of them are neighbors of Ethiopia.
WNV is a single-stranded RNA virus that belongs to the family of Flaviviridae, genus Flavivirus,19 and was first isolated from a febrile patient in the West Nile district of Northern Uganda in 1937. The virus causes disease mainly in birds, equines and humans.20,21 Birds are important vertebrate amplifying hosts and their migratory pattern have greatly influenced the re-emergence and global spread of WNV. The virus infects the mosquitoes when they suck blood-meal from infected birds. Thereafter, the mosquitoes spread the virus on the next susceptible host they bite.22 Humans and other mammals, especially horses, may develop clinical illness but usually are incidental or dead-end hosts because they do not produce significant viremia, and thus do not contribute to the transmission cycle.23
WNV also infects domestic animals such as cows.24 WNV infections typically cause a mild febrile illness in humans. The main risk factors for WNV transmission includes environmental and anthropological factors that increase mosquitoes’ population during heavy rain and flooding, irrigation, global warming and formation of ecologic niches that favor mass breeding of mosquitoes.25
WNV has been documented in 83 countries around the world, including several countries in Africa (e.g. Uganda, Kenya, Democratic Republic of Congo and Sudan26,27), the Middle East, Asia, and Australia.28–30 In Ethiopia very few studies have been conducted regarding arboviruses infections in humans,31–34 in spite of ample evidence regarding the importance of animals and human movement between neighboring countries in the spread of arboviruses.
The transmission dynamics between the hosts and reservoir, behavior of intermediate insect vectors population, animals and humans’ movement and urbanizations are among the key factors that alter the epidemiology of many emerging and re-merging viral diseases. Similarly, the challenge of many arboviruses in the absence of adequate diagnostic facility and capacity in livestock is supposed to be significantly increased, especially in remote areas. Hence, due to many known and unknown factors, the arboviruses remain under-recognized and underreported in many developing worlds such as Africa including Ethiopia. Some of the factors so far mentioned as low awareness by health-care providers, the presence of other prevalent febrile illnesses, lack of diagnostic testing and regular systematic surveillance, and many other factors. The main hypothesis of selecting the present study area were: a) because of its proximity to South Sudan in which these viruses are already confirmed; b) the hot and humid climate favoring arboviral transmission, and conducive for vector breeding and c) the lack of RVF vaccine administration: Importantly, to date, the RVFV vaccine has not been previously administered to cattle in Ethiopia. Henceforth, the aim of this study was to determine the seoprevalence of RVF and WNF to assess possible RVFV and WNV circulation in Gambella Region, Southwest Ethiopia.
Materials and Methods
Study Area and Study Design
We conducted a cross-sectional study in the Gambella Region of Ethiopia between December 2018 and January 2019. Gambella is one of the nine regions of Ethiopia. The geographical location lies between latitudes 6° 22ʹ and 8° 30ʹ N, and longitudes 33° 10ʹ and 35° 50ʹ E. The elevation of Gambella region ranges from ~1000 to ~2000 m above sea level (masl) in the east, from ~500–900 masl in the center, and ~300–500 masl in the west.35 Recent human population of the region is estimated to be 478,000.36 The livestock census conducted in the region in the year 2018 indicated 285,102 cattle, 35,285 sheep, 107,083 goats, and 301,531 poultry.37
In the present study area, the Abigar cattle breed are the most dominant both in the Nuer and Anywaa zones of the region.38 Among the indigenous genotypes known in the country, these cattle are known as “Sanga” located normally in the border area between Ethiopia and Sudan with larger extension in Ethiopia covering the Akobo area of Gambella.39 Geographically this breed is located around the White Nile of Sudan and Ethiopia. The breed is mainly distributed in the neighboring lowlands of Southwest of Ethiopia. This breed is mainly reared and kept by the Nuer ethnic groups.40,41 This breed of cattle is characterized by the unique features that are used in identifying the breed from other indigenous breeds. The breed has large body size, long-curved horns, gray and white dominant coat colors as well as ease of management by all gender groups due to its recognized docile character. In addition, the breed has better production and reproduction capabilities despite the high heat load, recurrent drought and repeated disease prevalence in the region.38 The African cattle breeds in general are known as more resistant to diseases including RVFV compare to European breeds.42
Lare is one of the 13 districts of the Gambella Region belonging to the Nuer Zone that borders South Sudan (Figure 1). The district was selected based on (a) its high cattle population, and (b) its adjacent to South Sudan where different arboviruses cases are previously reported including RVFV.43 The district hosts pastoralists migrating from South and North Sudan (commonly called “Fallata” or “Fulani”) in search of pasture and water for their livestock.44 The indigenous pastoralists also travel far from their villages to the adjacent territories of South Sudan in search of pasture. Pastoralists have a tradition of presenting and accepting cattle to or from the South Sudan community during marriage ceremonies which have the potential to introduce diseases in the study area.
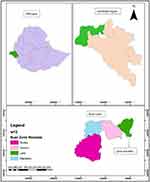 |
Figure 1 Map of study area (top left Ethiopia; Top right Gambella Region; bottom right Nuer zone and, Lare woreda with green color) “Woreda” is the local name representing the district. |
Sample Size Estimation and Data Collection
The sample size was determined using the following formula: 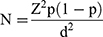 , where Z = 1.96 corresponding value to the desired 95% confidence level, p is an estimate of the expected prevalence of infection, and d is the margin of error in the estimate.45 In designing this study we took the estimate of p from Southwestern Uganda with a RVFV prevalence of 27%.13 We added 20% to account for non-response of cattle owners about cattle during sampling, questioning and dropping of aggressive animals. These assumptions resulted in a minimum sample size of 368 cattle. Four pastoral sub districts or “kebeles” (the lowest government administrative structure in Ethiopia) were selected for the study. These kebeles were Pal Bol, Bilimkun, Kechi and Tandor. Each selection was based on the cattle population, proximity to South Sudan and their frequent contact with the pastoralists from North and South Sudan. Inclusion criteria for recruitment of study were cattle of both sexes and a minimum age of one year.
, where Z = 1.96 corresponding value to the desired 95% confidence level, p is an estimate of the expected prevalence of infection, and d is the margin of error in the estimate.45 In designing this study we took the estimate of p from Southwestern Uganda with a RVFV prevalence of 27%.13 We added 20% to account for non-response of cattle owners about cattle during sampling, questioning and dropping of aggressive animals. These assumptions resulted in a minimum sample size of 368 cattle. Four pastoral sub districts or “kebeles” (the lowest government administrative structure in Ethiopia) were selected for the study. These kebeles were Pal Bol, Bilimkun, Kechi and Tandor. Each selection was based on the cattle population, proximity to South Sudan and their frequent contact with the pastoralists from North and South Sudan. Inclusion criteria for recruitment of study were cattle of both sexes and a minimum age of one year.
Based upon the age of puberty in local cattle breeds in the area, the study animals were classified into two groups: 1–3.5 years of age and 3.5+ years.38 After obtaining the owners’ consent, 5 mL blood sample was collected from selected cattle in the herd via the jugular venipuncture using a sterile vacutainer tube. Information such as age, sex, parity, herd size, history of mass abortion, history of mass death of young animals and some selected clinical signs regarding each animal (identified by a temporary paper-based identification code) was recorded separately using a checklist prepared as a mini questionnaire at the time of blood collection. Serum was separated and stored at −20°C until screening for IgG antibody to RVFV and WNV infections by ELISA.
Laboratory Analysis
The indirect ELISA-based type detection of IgG antibody against RVFV and WNV infections was conducted as per the manufacturer’s protocol ready for each individual (Abbexa Ltd, Cambridge UK).46 The diagnostic sensitivity and specificity have not been determined for these specific kits where they are only ready for research purposes. All the tests were conducted in duplicates. The results were read at an optical density (OD) of 450 nm using a 96-well ELISA plate reader (Multiskan™ FC Microplate Photometer), and interpreted as positive or negative based on the manufacturer’s recommended cut-off values. Test validity was verified according to the manufacturer’s manual when the mean OD of the positive control was ≥1 and when the mean OD of the negative control was ≤0.15. The cut-off value for each plate was calculated independently calculated where the cut-off value = negative control +0.15. Hence, any sample value greater than or equal to this value labelled as positive and any value less than the cut-off value scored as negative.
Statistical Analysis
All collected data cleared and entered into EpiData Software v.3.1 and analyzed using STATA Version 13.0. In the study herds, age, sex, and parity were used as explanatory parameters, while the history of mass abortion and the history of mass death of young animals together with other signs such as excess salivation, and loss of appetite and diarrhea were used to investigate clinical signs mainly about the RVF. In this study, the outcome variables were ELISA IgG antibody positive and negative.
The seroprevalence/apparent prevalence of IgG antibody elicited towards both RVFV and WNV were estimated by dividing the number of cattle with positive test results by the total number of tested cattle. Here the cattle included in the study were apparently healthy and sample was not taken from diseased animals where true prevalence could be determined. Univariable logistic regression was used to assess the crude association between the seropositivity of IgG antibody and the hypothesized individual potential risk factors such as age, sex, parity, and site, calculated with descriptive and analytical analysis using chi-square (χ2) test. Multivariable logistic regression analysis was used to assess the effect of each of the independent variables on the outcome variable (seropositivity) after adjusting each independent variable for all other variables. A p-value below 0.05 was considered indicative of a statistically significant association.
Ethical Approval and Consent to Participate
The livestock owners were informed and aware about the purpose of the study, and that verbal informed consent was approved by the Institutional Review Board of the College of Veterinary Medicine and Agriculture, Addis Ababa University with the certificate reference no: VM/ERC/07/05/10/2018. Besides, permission to visit the study sites and to collect the blood sample from cattle was obtained from the Gambella Regional Livestock and Fishery Office, district administration Office and community leaders of each study site. Blood sample collection was carried out under aseptic conditions by experienced veterinary laboratory technicians.
Results
Characteristics of the Study Cattle and Owners’ Responses on Selected Clinical Signs
A total of 275 (74.7%) female and 93 (25.3%) male cattle were included in the current study. The majority were in the age groups of 1–3.5 years, the minimum and maximum age of the sampled cattle population was 1 and 20 years, respectively, and the average age was 3.9 years with a standard deviation of 3.23. Among 121 cows the overall mean of the birth rate was 3.0 calves per cow (standard deviation = 2.2). The number of births per cow ranged from 1 to 14 calves.
The animals came from 53 herds with a minimum and a maximum herd size of 7 and 67 animals, respectively, and a mean herd size of 23.2. Just over half (50.9%) of the herd owners described mass abortion in the area and 43% mentioned the deaths of young animals within their herd within the past three to five years. Among the herd owners, 43.4% mentioned the historical presence of mass death in their animals (Table 1).
 |
Table 1 Characteristics of the Study Cattle and Owners’ Responses About Selected Clinical Signs |
Seroprevalence of RVFV Infection
Out of 368 analyzed serum samples, 28 (7.6%) (95% CI: 5.29–10.81%) were positive for IgG antibody to RVFV infection, which was significantly associated with a history of abortion (14.8%; p=0.041) compared to those cows had no history of abortion. However, there was no significant association between IgG positivity to the virus and the study kebeles (villages) (Table 2).
 |
Table 2 Seropositivity for RVFV Infection in Cattle |
Seroprevalence of WNV Infection
Table 3 presents the seroprevalence of WNV infection stratified by different background characteristics. The seroprevalence of WNV-specific IgG antibody was detected in serum samples collected from 20 cattle (5.4%) (95% CI: 3.52–8.28%). No significant associations were observed between the findings of WNV antibody IgG and past recorded clinical signs (Table 3). However, significantly higher proportions of anti-WNV IgG antibody were observed in serum samples collected from cattle with a history of abortion as compared to those without a history of abortion (11.1% Vs 3.8%, p= 0.04)
 |
Table 3 Seropositivity for WNV Infection in Cattle |
Independent Predictors of Seropositivity for RVFV Infection
In a multivariate logistic regression analysis where sex, age, parity, sub districts, and selected clinical signs considered were taken as independent predictors, no significant association between the RVFV specific IgG antibody and these predictor variables were observed (Table 4).
 |
Table 4 Univariate and Multivariate Analysis of Risk Factors for RVFV Seropositivity |
Independent Predictors of Seropositivity for WNV Infection
Similar to the finding observed in modeling the odds of getting RVFV positive IgG, all the variables included in the multivariate logistic regression that investigated factors associated with the odds of getting WNV positive IgG did not result in statistically significant association with any one of the investigated risk factors (Table 5).
 |
Table 5 Univariate and Multivariate Analyses of Risk Factors for WNV Seropositivity |
Discussion
A limited number of studies on arboviruses in human31–34 and livestock47 have been carried out in Ethiopia. This seroprevalence study focused on RVFV and WNV infections in cattle in the Gambella region of southwestern Ethiopia. This region and the study districts were selected due to their proximity to South Sudan, which serves as the entry point for many refugees to Ethiopia as well as the main route for livestock movement between the adjacent territories.
The present study revealed 28 (7.6%) (95% CI: 5.29–10.81%) animals were seropositive for RVFV infection, while 20 (5.4%) (95% CI: 3.52–8.28%) were seropositive for WNV infection. Antibody to both viruses was found in cattle from all the four sub-district “kebeles”. Comparing the anti-IgG of WNV and RVFV in this study, it has been found high prevalence of RVFV than WNV.
The seroprevalence of RVFV infection in cattle previously reported in another African country is 6.2% in the eastern region of the Democratic Republic of the Congo48 which is similar to the result of current study. On the other hand, the seroprevalence observed in our study is lower than a report from the eastern parts of Ethiopia, Somalia region which is 17.9%47 and from other African countries like Cameroon (9.4%),49 Rwanda (16.8%);10 Madagascar (25.8%)50 and Mozambique (36.6%).51
There are no reports on the clinical importance of WNV infection in cattle. Cases of WNF in cattle are rarely documented and serologic findings of WNV-specific IgG antibodies have been reported in other countries like Nigeria. In the Nigeria study, WNV IgG-antibody was documented in different animal sera including camels, goats, cattle and sheep.52 WNV IgG-antibody was also reported in cattle in Senegal,53 in Trinidad,54 in Turkey,55 and in Palestine.56 A number of other studies in African countries have also reported anti- RVFV IgG and anti-WNV among apparently healthy animals,57 including Kenya,58,59 Tanzania,58,60 Mozambique and Senegal.49,51,59,61
In Ethiopia in general and in Gambella in particular, there have been no reports of outbreaks or specific studies on RVFV and WNV in animals. Likely, this is due to the lack of virus screening measurements for the surveillance. Asymptomatic or unnoticed or mild form of the diseases may contribute to under-reporting of cases. However, few reports have documented RVFV and WNV infections in apparently healthy cattle.48,62
Many East African countries depend on pastoral agriculture for milk and meat production. Therefore, livestock diseases, including RVF may impact international livestock marketing and may harm the economics of a country like Ethiopia which depends on mainly on the livestock besides the direct impact on the human health.63
The current study showed the presence of RVFV and WNV-specific IgG antibodies and did not measure the level of anti-IgM antibodies, which would be an indicator of acute infection. Thus, this study cannot confirm recent infection, and the results may show only the exposure rate in the area or antibody cross-reactivity due to other arbovirus infections. The high IgG seropositivity to RVFV and WNV infections among the age group of 3.5+ years may show that these viruses have been occurred in the study area. However, WNV seroprevalence seems to have increased with time compared to RVFV because seropositivity in the age groups of 1–3.5 years was higher in the former than in the same age groups in the latter, where relatively higher seropositivity was observed among the age groups of 3.5+ years.
The seropositivity and sex of the cattle showed no significant association indicating that both males and females are affected by RVFV and WNV. However, male cattle were more likely affected by WNV infection than females, while the opposite was true for RVFV infection. Further studies are necessary to look at the reason for this result.
Other factors such as age and sampling sites did not significantly affect the occurrences of both viruses. In contrast to the present study, other studies have found a significant association between seropositivity of these viruses and two factors (age and sex).13,60,64,65 For example, a study conducted in Madagascar reported higher prevalence of RVFV in male cattle compared to female cattle.50 In regards to the association of age-seropositivity of IgG antibodies in an endemic country, the occurrence of IgG antibodies in older animals generally supports the contention that most animals are exposed to the virus during their lifetimes. Infection of younger animals permits to pinpoint the time window of the infection and how recent the infection occurred.50,60 Based on the multivariate logistic regression analysis, our study revealed 1.5 times higher seroprevalence of RVFV infection in the age group of 3.5+ years and a high seroprevalence of WNV infection in the age group between 1 and 3.5 years, which is in line with earlier studies showing a high proportion of IgG in similar age groups.48,50,60 Enrolling a larger sample size might reveal a different association of the IgG result with age and sex.
Upon blood sample taking, livestock owners were also interviewed on some selected clinical signs related to RVFV infection since the WNF clinical signs in cattle have not been described. Almost half (50.9%) of the owners reported the occurrence of mass abortion in their herds, a finding significantly associated with RVFV seropositivity. Many (56.0%) of the livestock owners did not recall the history of the mass death of young animals. No-specific symptoms such as excess salivation were reported by 14.1% of respondents and loss of appetite, weakness, and diarrhea by 26.1% of participants together with signs of mass death of young animals and abortion. However, there was no significant difference between these variables and the serological documentation of WNV or RVFV infection. The statistical insignificance association of these signs and symptoms with outcomes of anti-RVFV and anti-WNV IgG, the signs and symptoms might arise due to other diseases.
In the present study, anti-WNV IgG-antibodies presence using ELISA was not confirmed by confirmatory to detect actual viral infection. Direct detection of the WNV infection from samples from various animal species would give an improved understanding of the epidemiological situation in east Africa. Serologically measured anti-WNV IgG antibodies in cattle in other African countries also support the possibility that infection causes clinical disease in these hosts like in birds, horses, crocodiles, and humans where many clinical signs shown. WNV occurrences in other species besides cattle elsewhere indicate the virus to be prevalent in birds, equine as well as in human populations.66 The result of this study suggests the possible circulation of WNV and warrants performing further studies in Ethiopia with other species.
Limitations of the Study
In this study, the screening of the viruses with ELISA test was not supported with more sensitive and specific confirmatory tests like reverse transcription-polymerase chain reaction (RT-PCR). Another setback of this study is that it was conducted only on cattle, excluding other species of livestock in the study that would enable the comparison of the viruses’ distributions in the available potential hosts. Finally, this study could not present antibody titers of the positive samples that might explain the results in advance. Cattle are the main means of economic importance in the area among the livestock population where pastoralists mainly depend on. In line with their importance, this is the first serologic study in cattle in the area which gives evidence of WNF and RVF presence, and encourages further studies. Besides, we believe that our study would provide baseline data on the epidemiology of WNF and RVF in the area.
Conclusions
The present antibody-based ELISA study revealed the possible occurrence of RVF and WNF among the cattle population in the study area. Significantly high IgG antibodies to both viruses was observed among those with a history of abortion. An increased trend of IgG-antibody positivity was also observed among animals in the age 3.5 years and above. Henceforth, the results warrant further studies toward active case detection of antigens of these viruses in susceptible hosts, including other livestock animals and humans, their transmission dynamics and their public health significance to predict and reduce possible future outbreaks in the area.
Abbreviations
RVFV, Rift Valley fever virus; RVF, Rift Valley fever; WNF, West Nile fever; WNV, West Nile virus; RT-PCR, reverse transcriptase-polymerase chain reaction; ELISA, enzyme-linked immunosorbent assay; PRNT, plaque reduction neutralization test; IgG, immunoglobulin G; IgM, immunoglobulin M; OD, optical density; RNA, ribonucleic acid.
Data Sharing Statement
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We would like to thank Gambella Livestock and Fishery Bureau and Lare district livestock and fishery Office, livestock owners, data collectors and community leaders for their kind cooperation during this study.
Author Contributions
All authors made a significant contribution to this work, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was financially supported by Addis Ababa University, Vice President for Research and Technology Transfer. The ELISA kits were donated by LaRuke Development and the Panorama Research Institute, California USA. The research funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Disclosure
The authors declare that they have no competing interests.
References
1. Jones KE, Patel NG, Levy MA, et al. Global trends in emerging infectious diseases. Nature. 2008;451(7181):990–993. doi:10.1038/nature06536
2. Pfeffer M, Dobler G. Emergence of zoonotic arboviruses by animal trade and migration. Parasit Vectors. 2010;3(1):1–15. doi:10.1186/1756-3305-3-35
3. world health organization. Arthropod-borne and rodent-borne viral diseases. Report of a WHO scientific group. World Health Tech Rep Ser. 1985:1–114.
4. Turell MJ. Members of the culex pipiens complex as vectors of viruses1. J Am Mosq Control Assoc. 2012;28(suppl 4):123–126. doi:10.2987/8756-971X-28.4.123
5. Harbach RE. Culex pipiens: species versus species complex – taxonomic history and perspective. J Am Mosq Control Assoc. 2012;28(Suppl 4s):10–23. doi:10.2987/8756-971X-28.4.10
6. Daubney R, Hudson J, Garnham P. Enzootic hepatitis or rift valley fever. An undescribed virus disease of sheep, cattle and man from East Africa. J Pathol Bacteriol. 1931;34:545–579. doi:10.1002/path.1700340418
7. Bird BH, Nichol ST. Breaking the chain: rift valley fever virus control via livestock vaccination. Curr Opin Virol. 2012;2(3):315–323. doi:10.1016/j.coviro.2012.02.017
8. Chevalier V, De La Rocque S, Baldet T, Vial L, Roger F. Epidemiological processes involved in the emergence of vector-borne diseases: West Nile fever, Rift Valley fever, Japanese encephalitis and crimean-congo haemorrhagic fever. Rev Sci Tech. 2004;23(2):535–555. doi:10.20506/rst.23.2.1505
9. Clark MHA, Warimwe GM, Di Nardo A, Lyons NA, Gubbins S, Barker CM. Systematic literature review of rift valley fever virus seroprevalence in livestock, wildlife and humans in Africa from 1968 to 2016. PLoS Negl Trop Dis. 2018;12(7):e0006627. doi:10.1371/journal.pntd.0006627
10. Umuhoza T, Berkvens D, Gafarasi I, Rukelibuga J, Mushonga B, Biryomumaisho S. Seroprevalence of rift valley fever in cattle along the Akagera Nyabarongo rivers, Rwanda. J S Afr Vet Assoc. 2017;88:a1379. doi:10.4102/jsava.v88.1379
11. Wilson M. Rift valley fever virus ecology and the epidemiology of disease emergence. Ann NY Acad Sci. 1994;740:169–180. doi:10.1111/j.1749-6632.1994.tb19867.x
12. Baudin M, Jumaa A, Jomma H, et al. Association of rift valley fever virus infection with miscarriage in Sudanese women: a cross-sectional study. Lancet Glob Health. 2016;4(1):
13. Nyakarahuka L, Maurice AD, Purpura L, et al. Prevalence and risk factors of rift valley fever in humans and animals from Kabale district in Southwestern Uganda, 2016. PLoS Negl Trop Dis. 2018;12(5):e0006412.
14. Javelle E, Lesueur A, Pommier De Santi V, et al. The challenging management of rift valley fever in humans: literature review of the clinical disease and algorithm proposal. Ann Clin Microbiol Antimicrob. 2020;19(1):1–18. doi:10.1186/s12941-020-0346-5
15. Bosworth A, Ghabbari T, Dowall S, et al. Serologic evidence of exposure to rift valley fever virus detected in Tunisia. New Microbes New Infect. 2016;9:1–7. doi:10.1016/j.nmni.2015.10.010.
16. Hassan OA, Ahlm C, Sang R, Evander M, Brooker S. The 2007 rift valley fever outbreak in Sudan. PLoS Negl Trop Dis. 2011;5(9):e1229. doi:10.1371/journal.pntd.0001229
17. Sang R, Kioko E, Lutomiah J, et al. Rift valley fever virus epidemic in Kenya, 2006/2007: the entomologic investigations. Am J Trop Med Hyg. 2010;83(Suppl 2):28–37. doi:10.4269/ajtmh.2010.09-0319
18. Himeidan YE, Kweka EJ, Mahgoub MM, El Rayah EA, Ouma JO. Recent outbreaks of rift valley fever in East Africa and the Middle East. Front Public Health. 2014;2. doi:10.3389/fpubh.2014.00169
19. Daubney R, Hudson JR. Rift valley fever. Lancet. 1932;219(5664):611–612. doi:10.1016/S0140-6736(01)24634-0
20. Nyamwaya D, Wang’ondu V, Amimo J, et al. Detection of West Nile virus in wild birds in Tana River and Garissa Counties, Kenya. BMC Infect Dis. 2016;16(1):1–10. doi:10.1186/s12879-016-2019-8
21. CDC. West Nile virus | West Nile Virus | CDC. 2019. Available from: https://www.cdc.gov/westnile/index.html.
22. Sang RC, Dunster LM. The growing threat of arbovirus transmission and outbreaks in Kenya: a review. East Afr Med J. 2001;74(12):655–661.
23. CDC. West Nile Virus. 2020. Available from: https://www.cdc.gov/westnile/resources/pdfs/13_240124_west_nile_lifecycle_birds_plainlanguage_508.pdf.
24. Philip CB, Smadel JE. Transmission of West Nile Virus by infected aedes albopictus. Exp Biol Med. 1943;53(1):49–50. doi:10.3181/00379727-53-14180
25. Hubálek Z, Halouzka J. West Nile fever a reemerging mosquito-borne viral disease in Europe. Emerg Infect Dis. 1999;5(5):643–650. doi:10.3201/eid0505.990505
26. Atoni E, Waruhiu C, Nganga S, Xia H, Yuan Z. Arboviruses of human health significance in Kenya Atoni. Afr J Health Sci. 2018;31(1):122–141.
27. Smithburn K, Jacobs H. Neutralization-tests against neurotropic viruses with sera collected in central Africa. J Immunol. 1942;44:9–23.
28. Eybpoosh S, Fazlalipour M, Baniasadi V, et al. Epidemiology of West Nile virus in the Eastern Mediterranean region: a systematic review. PLoS Negl Trop Dis. 2019;13:1. doi:10.1371/journal.pntd.0007081
29. Hayes EB, Komar N, Nasci RS, Montgomery SP, Leary DRO, Campbell GL. Epidemiology and transmission dynamic of West Nile Virus disease. Emerg Infect Dis. 2005;11(8):1167.
30. Gideononline. West Nile Fever. Sign in West Nile Fever. Available from: https://www.gideononline.com/cases/westnilefever/. 2020.
31. Ardoin P, Rodhain F, Hannoun C. Epidemiologic study of arboviruses in the Arba-Minch district of Ethiopia. Trop Geogr Med. 1976;28(4):309–315.
32. Woyessa A, Mengesha M, Kassa W, Kifle E, Wondabeku M, Girmay A. The first acute febrile illness investigation associated with dengue fever in Ethiopia, 2013: a descriptive analysis. Ethiop J Health Dev. 2013;28(3):155–161.
33. Mengesha Tsegaye M, Beyene B, Ayele W, et al. Sero-prevalence of yellow fever and related flavi viruses in Ethiopia: a public health perspective. BMC Public Health. 2018;18(1):1–10. doi:10.1186/s12889-018-5726-9
34. Lilay A, Asamene N, Bekele A, et al. Reemergence of yellow fever in Ethiopia after 50 years, 2013: epidemiological and entomological investigations. BMC Infect Dis. 2017;17(1):343.
35. Woube M. Flooding and sustainable land–water management in the lower Baro–Akobo river basin, Ethiopia. App Geogr. 1999;19:235–251. doi:10.1016/S0143-6228(99)00004-1
36. Central Statistical Agency. Population Projections for Ethiopia 2007–2037; 2013.
37. CSA. Federal Democratic Republic of Ethiopia Central Statistical Agency. Report on Livestock and Livestock Characteristics (Private Peasant Holdings); 2018.
38. Nakachew M, Girma A, Tadelle D. On-farm description and status of Nuer (Abigar) cattle breed in Gambella Regional State, Ethiopia. Int J Biodivers Conserv. 2018;10(6):292–302. doi:10.5897/ijbc2017.1168
39. Alberro M, Haile-Mariam S. The indigenous cattle of Ethiopia. Part I. World Anim Rev. 1982;41:2–10.
40. Gambella Regional State. Gambella Regional Land-Use and Land Allotment Study. Amended Draft Final Report, Vol. II. Yeshi-Ber Con; 2003.
41. Information Systems Domestic Animal Genetic Resources. Domestic Animal Genetic Resources Information Systems (DAGRIS). (Rege; J. E. O.; O. Hanotte; Biruk Asrat and Tadelle Dessie (Eds)); 2007.
42. Fischer EA, Boender GJ, Nodelijk G, De Koeijer AA, Van Roermund HJ. The transmission potential of rift valley fever virus among livestock in the Netherlands: a modelling study. Vet Res. 2013;44:1. doi:10.1186/1297-9716-44-58
43. Lewis M. Yellow fever outbreak in Sudan. N Engl J Med. 2013;368(8):689–691. doi:10.1056/NEJMp1300772
44. Miller C, Manga A, Amin A. The West African communities in Gedaref state: processes of settlement and local integration. Africa (Lond). 2005;2005:375–424.
45. Thrustfield M. Veterinary Epidemiology.
46. Abbexa Ltd. Available from: www.abbexa.com.
47. Ibrahim M, Schelling E, Zinsstag J, et al. Sero-prevalence of brucellosis, Q-fever and rift valley fever in humans and livestock in Somali region, Ethiopia. bioRxiv. 2020.
48. Georges TM, Justin M, Victor M, Marie KJ, Mark R, Léopold MMK. Seroprevalence and virus activity of rift valley fever in Cattle in Eastern region of democratic Republic of the Congo. J Vet Med. 2018;2018.
49. Zeller HG, Bessin R, Thiongane Y, et al. Rift valley fever antibody prevalence in domestic ungulates in Cameroon and several West African countries (1989–1992) following the 1987 mauritanian outbreak. Res Virol. 1995;146(1):81–85. doi:10.1016/0923-2516(96)80593-0
50. Jeanmaire EM, Rabenarivahiny R, Biarmann M, et al. Prevalence of rift valley fever infection in ruminants in Madagascar after the 2008 outbreak. Vector Borne Zoonotic Dis. 2011;11:395–402. doi:10.1089/vbz.2009.0249
51. Lagerqvist N, Moiane B, Mapaco L, Fafetine J, Vene S, Falk KI. Antibodies against rift valley fever virus in cattle, mozambique. Emerg Infect Dis. 2013;19(7):1177–1179. doi:10.3201/eid1907.130332
52. Olaleye O, Omilabu S, Ilomechina E, Fagbami A. A survey for haemagglutination-inhibiting antibody to West Nile virus in human and animal sera in Nigeria. Comp Immunol Microbiol Infect Dis. 1990;13:35–39. doi:10.1016/0147-9571(90)90006-F
53. Davoust B, Maquart M, Roqueplo C, et al. Serological survey of west nile virus in domestic animals from Northwest Senegal. Vector Borne Zoonotic Dis. 2016;16:359–361. doi:10.1089/vbz.2015.1881
54. Thompson NN, Auguste AJ, Coombs D, et al. Serological evidence of flaviviruses and alphaviruses in livestock and wildlife in Trinidad. Vector Borne Zoonotic Dis. 2012;12(11):969–978. doi:10.1089/vbz.2012.0959
55. Ozkul A, Yildirim Y, Pinar D, Akcali A, Yilmaz V, Colak D. Serological evidence of West Nile Virus (WNV) in mammalian species in Turkey. Epidemiol Infect. 2006;134(4):826–829. doi:10.1017/S0950268805005492
56. Darwish MA, Hoogstraal H, Roberts TJ, Ahmed IP, Omar F. A sero-epidemiological survey for certain arboviruses (Togaviridae) in Pakistan. Trans R Soc Trop Med Hyg. 1983;77(4):442–445. doi:10.1016/0035-9203(83)9010
57. Fafetine J, Neves L, Thompson PN, Paweska JT, Rutten VPMG, Coetzer JAW. Rift valley fever in Uganda: seroprevalence and risk factor surveillance vis-`a-vismosquito vectors, anti-RVF virus IgG and RVF virus neutralizing antibodies in goats. PLoS Negl Trop Dis. 2013;7.
58. Labeaud AD, Muchiri M, Ndzovuz M, et al. Interepidemic rift valley fever virus seropositivity, northeastern Kenya. Emerg Infect Dis. 2008;14(8):1240–1246. doi:10.3201/eid1408.080082
59. Munyua P, Murithi RM, Wainwright S, et al. Rift valley fever outbreak in livestock in Kenya, 2006–2007. Am J Trop Med Hyg. 2010;83(2):58–64. doi:10.4269/ajtmh.2010.09-0292
60. Sumaye RD, Geubbels E, Mbeyela E, Berkvens D, Bird B. Inter-epidemic transmission of rift valley fever in livestock in the Kilombero river valley, Tanzania: a cross-sectional survey. PLoS Negl Trop Dis. 2013;7(8):e2356. doi:10.1371/journal.pntd.0002356
61. Lef`evre PC. Impact des arboviroses d’int´erˆet v´et´erinaire. Le cas de la fi`evre de la vall´ee du Rift. M´edecine Trop. 2000;60(2):27–30.
62. Soumare B, Tempia S, Cagnolati V, Mohamoud A, Huylenbroeck G, Berkvens D. Screening for rift valley fever infection in northern Somalia: a GIS based survey method to overcome the lack of sampling frame. Vet Microbiol. 2007;121(3–4):249–256. doi:10.1016/j.vetmic.2006.12.017
63. Pratt N, Bonnet P, Jabbar M, Ehui S, DeHaan C Benefits and costs of compliance in sanitary regulations in livestock markets: the case of rift valley fever in Ethiopia. In:
64. Ould El Mamy AB, Baba MO, Barry Y, et al. Unexpected rift valley fever outbreak, Northern Mauritania. Emerg Infect Dis. 2011;17:1984.
65. Halawi AA, Saasa N, Pongombo BL, et al. Seroprevalence of rift valley fever in cattle of smallholder farmers in Kwilu Province in the Democratic Republic of Congo. Trop Anim Health Prod. 2019;51(8):2619–2627.
66. Glaser A. West Nile virus and North America: an unfolding story. Rev Sci Tech. 2004;23(2):557–568. doi:10.20506/rst.23.2.1504
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
