Back to Journals » The Application of Clinical Genetics » Volume 16
Screening Y Chromosome Microdeletion in 1121 Men with Low Sperm Concentration and the Outcomes of Microdissection Testicular Sperm Extraction (mTESE) for Sperm Retrieval from Azoospermic Patients
Authors The Trinh S , Nguyen NN , Thi Thu Le H, Thi My Pham H, Tien Trieu S , Tran NTM , Sy Ho H , Van Tran D , Van Trinh T, Trong Hoang Nguyen H, Pham Minh N, Duc Dang T, Huu Dinh V, Thi Doan H
Received 26 May 2023
Accepted for publication 4 August 2023
Published 28 August 2023 Volume 2023:16 Pages 155—164
DOI https://doi.org/10.2147/TACG.S420030
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Martin Maurer
Son The Trinh,1 Nhat Ngoc Nguyen,1 Hien Thi Thu Le,2 Hanh Thi My Pham,2 Sang Tien Trieu,3 Ngoc Thao My Tran,4 Hung Sy Ho,5 Danh Van Tran,6 Tam Van Trinh,2 Hiep Trong Hoang Nguyen,2 Ngoc Pham Minh,2 Trinh Duc Dang,7 Viet Huu Dinh,2 Hang Thi Doan1
1Military Institute of Clinical Embryology and Histology, Vietnam Military Medical University, Hanoi, Vietnam; 2Department of Andrology, Andrology and Fertility Hospital of Hanoi, Hanoi, Vietnam; 3Department of Biology and Genetics, Vietnam Military Medical University, Hanoi, Vietnam; 4Department of Life Sciences, Biosciences Division, College of Health, Medicine and Life Sciences, Brunel University London, Uxbridge, UK; 5Department of Obstetrics and Gynecology, Hanoi Medical University, Hanoi, Vietnam; 6Respiratory Center, Military Hospital 103, Vietnam Military Medical University, Hanoi, Vietnam; 7Faculty of Mathematics and Computer Science, Vietnam Military Medical University, Hanoi, Vietnam
Correspondence: Hang Thi Doan, Military Institute of Clinical Embryology and Histology, Vietnam Military Medical University, Hanoi, 12108, Vietnam, Email [email protected] Viet Huu Dinh, Andrology and Fertility Hospital of Hanoi, Hanoi, 100000, Vietnam, Email [email protected]
Background: The Y chromosome has a specific region, namely the Azoospermia Factor (AZF) because azoospermia is typically reported in the microdeletion of the AZF region. This study aims to assess the characteristics of AZF microdeletion after screening a massive number of low sperm concentration men; and the Microdissection testicular sperm extraction (mTESE) outcomes for retrieving sperm from azoospermic patients.
Materials and Methods: This retrospective multiple-center study enrolled a total of 1121 men with azoospermia, cryptozoospermia, and severe oligozoospermia from December 2016 to June 2022. An extension analysis used a total of 17 STSs to detect the position-occurring microdeletion in the AZF region (AZFa, b, c, and/or d loci). Microdissection testicular sperm extraction (mTESE) was performed to retrieve sperm in azoospermic men diagnosed AZFc microdeletion.
Results: One hundred and fifty-three men carried AZF microdeletion were detected in the 1121 participants (13.64%). The incidences of AZF microdeletion were confined to AZF a, c, and d regions, both individual and concurrence, with the most common in the AZFc region accounting for 49.67%; There was no significant difference in clinical and paraclinical characteristics between the deleted regions, except FSH level (highest in AZFa microdeletion, p = 0.043). The AZFc region was the most common type of AZF microdeletion (49.67%), including complete microdeletion (4 patients) and gr/gr partial microdeletion (39 patients) with 50.00% and 63.63% in the success rate of mTESE, separately.
Conclusion: The absence of AZFa and/or AZFb regions often express the most severe phenotype – azoospermia and the increasing FSH level. The AZFc region played the most common microdeletion. Microdissection testicular sperm extraction (mTESE) was the possible therapy for sperm retrieval from the testis of azoospermia men having AZFc microdeletion.
Keywords: Y chromosome microdeletion, AZF microdeletion, azoospermia, mTESE
Introduction
Male infertility is defined as the inability to make their female partner get pregnant.1 A previous study reported that 90% of infertility problems in males were related to abnormal sperm count. Low sperm concentration was categorized into azoospermia, cryptozoospermia, and oligozoospermia.2,3 Azoospermia is the situation with no sperm in the semen, which was diagnosed by two consecutive confirmations of lacking sperm in centrifuged ejaculated semen samples. Meanwhile, cryptozoospermia or virtual azoospermia seems to have no sperm in the semen ejaculated, but a few sperm can be found in the pellet after centrifugation.4 Oligozoospermia occurs in men having less than 15 million sperm/mL in the ejaculate, and patients having sperm count less than 5 million sperms/mL were defined as severe oligozoospermia.3,5–7
Among multifactorial causes of male infertility, genetic disorders have been associated with severe oligozoospermia and azoospermia. Severe oligozoospermia or azoospermia are at increased risk for getting a genetic abnormality compared to others.8,9 Y chromosome microdeletions (YCMD) play the second most common genetic factor of male infertility after Klinefelter’s syndrome.10 Generally, Y chromosome microdeletion is present in 1:4000 men, and the disorder significantly rises in the infertile male group accounting for 2–5% in severe oligospermia and 5–10% in azoospermic males.11 The Y chromosome is a dominant sexual factor owing to the presence of SRY (sex-determining region on Y) and also harbors essential genes related to spermatogenesis located in the distal end of the long arm (Yq11).11,12 The long arm of the Y chromosome is characterized by many ampliconic and palindromic sequences making self-recombination. As a result, intra-chromosomal deletions often occur during spermatogenesis.5,13,14 When Y chromosome microdeletion occurs in Yq11, it is referred to as AZF microdeletion.15 After the first time introduced in 1976, the Yq11 microdeletion was further divided into the proximal (AZFa), middle (AZFb), and distal deletion (AZFc) separately depending on the association with specific spermatogenic disruption phenotype manifestations.16 The AZF microdeletions may occur individually (AZFa, b, c loci) or concurrently (AZFbc, AZFabc), which can lead to severe oligozoospermia or even azoospermia.16,17 AZFc (∼80%) was the most popular Y chromosome microdeletion, followed by AZFa (0.5–4%), AZFb (1–5%), and AZFbc (1–3%) deletion. Furthermore, AZFabc was often reported as related to abnormal karyotypes such as iso (Y) or 46,XX male.18 Generally, the AZFa deletion could lead to Sertoli cell‐only syndrome (SCOS), characterized by the only presence of Sertoli cells in seminiferous tubules, while in the AZFb deletion, germ cells often get maturation arrest at the spermatocyte stage. In contrast, the AZFc deletion was associated with hypospermatogenesis, which tends to have more potential for presenting sperm in the testes.19 Analyzing the AZF regions and their association with genotype and phenotype, a fourth AZF region between AZFb and AZFc was identified, which was termed AZFd. Men affected by AZFd microdeletion may get mild oligozoospermia or even normozoospermia.20
As the most prevalent, the complete absence of AZFc region leads to disappearance of three coding protein multicopy gene families, namely DAZ (deleted in azoospermia), CDY1 (CDY1a and CDY1b; chromodomain protein, Y chromosome 1), BPY2 (a basic protein on Y chromosome 2).21 DAZ was reported that have an essential role in the pathogenesis of nonobstructive azoospermia; hence, primer sets of two sequence-tagged sites (STSs) were used for basic analysis of the presence or absence of DAZ gene at sY254, sY255 loci.10,22–24 With the development of molecular techniques, extension analysis of AZFc microdeletions was performed by using more STSs consisting of sY1191, sY1291, BPY2, and sY160 (heterochromatic region).21,25 Six STS loci analyzed AZFc microdeletions in complete AZFc microdeletions (sY254 + sY255), gr/gr partial deletion type (sY1191 is positive; sY1291 is negative), and b2/b3 partial deletion type (sY1291 is positive; sY1191 is negative). Testing for these AZFc microdeletions (partial, complete) could have diagnostic and prognostic value for infertility treatment by Microdissection testicular sperm extraction (mTESE), which is the most powerful technique enabling the exploration of the testicular parenchyma and is therefore considered the gold-standard surgical technique to retrieve sperm in azoospermic carriers. According to the European Academy of Andrology (EAA) and the European Molecular Genetics Quality Network (EMQN) guideline, mTESE should not be proposed in AZFa deletions and maybe eventually attempted in AZFb or AZFbc deletions, while it is considered as a possible method for retrieving sperm in AZFc azoospermic men.10 Much effort has been made in this multicenter study to assess the distribution of AZF microdeletion after screening a large number of azoospermic, cryptozoospermic, and oligozoospermic Vietnamese men, in detail in analyzing AZFc region prevalences and mTESE outcome.
Materials and Methods
Subjects
One thousand one hundred and twenty-one Vietnamese patients with severe abnormal sperm concentration, including azoospermia, cryptozoospermia, and severe oligozoospermia, were recruited from the Military Institute of Clinical Embryology and Histology and the Andrology and Fertility Hospital of Hanoi. One hundred and fifteen patients carried AZF microdeletion detected by sequence-tagged sites (STSs) analysis from December 2016 to June 2022. Ten STSs were first chosen for founding AZF microdeletion (AZFa, b, c, and/or d loci) in 38 patients. Then, extension analysis using a total of 17 STSs detected 115 men having AZF microdeletion. The semen analysis was performed according to the guidance of the World Health Organization (WHO) 2010.3 Physical examination, karyotyping, and Y chromosome microdeletion reports were adequate while having a few deficiencies in the reproductive hormone results.
DNA Extraction
From the collected samples, genomic DNA was extracted using the G-spin™ Total DNA Extraction Mini Kit (iNtRON Bio, Korea), following the manufacturer’s instructions. After that, DNA’s quantity and purity were analyzed with a SpectraMax QuickDrop.
Screening AZF Microdeletions by Multiplex PCR
Based on the published guidelines for diagnosing microdeletion on the Y chromosome by the European Academy of Andrology (EAA) and the European Molecular Genetics Quality Network (EMQN), basic primer sets of sequence tagged sites (STSs) were suggested to ensure the test’s accuracy.10 It was advised that each AZF region include at least two STS loci for diagnostic reliability. For the detection of AZFa deletions, the STSs were chosen (with some modifications) as sY84, sY86, USP9Y, and DBY. In AZFb and AZFc, these sites were sY127, sY134, and sY254, sY255, respectively. In addition to the classical suggestion, the AZFd region was also used to detect microdeletion consisting of two markers, sY152, and sY153.
The diagnosis utilized the sY14 (SRY) marker as an internal control to prevent any technical failures. Both positive and negative controls were used during the test. Based on the lengths of possible PCR products, the primer sets were divided into different tubes to carry out the Multiplex PCR. Then, the PCR reaction mixture of 12.5 ul was prepared as follows: 6.25 ul GoTaq Green Master Mix (Promega), 0.25ul forward primer, 0.25ul reverse primer (IDT), 2–3 ul genomic DNA, nuclease-free water (Promega). After that, the amplification process was carried out on a thermal cycler with the condition as follows: 94°C in 11 minutes, 35 cycles of denaturing at 94°C in 45 seconds, annealing at 57°C in 45 seconds, then extending at 72°C in 1 minute, with a final extension at 72°C in 6 minutes. The final PCR products would then undergo electrophoresis on 2% agarose gel (Cleaver Scientific) at 100V and 100mA in one hour. The gel would then be visualized on the omniDOC Gel Documentation System (Cleaver Scientific) to analyze the results. The tests were repeated (at least once) until the results were clear.
Performing Extension Diagnosis on AZFb and AZFc Regions
For extension analysis, our genetic laboratory has chosen more STSs for extension analysis. In terms of AZFb, sY105, sY121, and sY1192 would be chosen for the extension analysis. Regarding AZFc, sY160, sY1191, sY1291 and BPY2 were the markers identifying the extension deletion. The PCR ingredients and conditions of the extension diagnosis were carried out as described above and repeated at least once.
Microdissection Testicular Sperm Extraction (mTESE) Procedure
General anesthesia was performed before the surgery to avoid painful experiences for our patients. A scrotal incision of 3–4 cm in length was done to ensure that in parallel to the subcutaneous vessels.26,27 Following the albugineal incision, the testicular parenchyma is surveyed at high magnification (×40) under the operating microscope to search carefully dilated seminiferous tubules that probably contain spermatozoa in 90% of cases.28 The average duration for our surgery operation is about 1–2 hours.
Statistical Analysis
Patient characteristics were presented as mean-standard deviation and proportion. Differences in variables were statistically analyzed with the Fisher’ exact test, Chi square’ test, and Kruskal Wallis test, when appropriate. The STATA software (Version 16.0; Stata Corporation) was used to conduct all data. The difference was considered statistically significant when p-value <0.05.
Results
Table 1 shows that AZFa microdeletion may be the most severe type, as azoospermia is always expressed in all 3 AZFa patients. We did not find any subjects with AZFb microdeletion. The absence of the AZFc region was the most popular microdeletion in the Y chromosome, with 49.67%. The percentages of severe oligozoospermia and azoospermia men in AZFc microdeletion were 28.95% and 31.58%, respectively; Meanwhile, the rate of cryptozoospermia was highest at 39.47%. The difference was statistically significant, with p < 0.05. These three phenotypes could be seen in 6 men deleted in the AZFd region.
 |
Table 1 The AZF Microdeletions: Distribution, Karyotype, and Sperm Count Manifestations |
The second most popular microdeletion occurred in the AZFbcd region, with 28.10%, and there was no statistically significant difference between the three phenotypes of AZFbcd microdeletion. While the AZFbc occupied 7.84% of all AZF microdeletions and was a more common azoospermia expression (accounted for 83.33%) than cryptozoospermia or severe oligozoospermia phenotypes (p < 0.05). Interestingly, 2 cases found with AZFabcd deletion were 46, XX male, and both were azoospermic. We also found 35 subjects having other chromosomal disorders, in which 22 cases carried disorder in the sex chromosome. Five patients of AZF microdeletions also got Klinefelter syndrome (47, XXY), and they all suffered from azoospermia.
Table 2 shows three groups depending on the deleted region in AZFa, AZFc, or AZFd loci. The AZFb microdeletion was not found in this study. The levels of FSH hormonal serum between the three groups showed a significant difference, with p < 0.05. The subjects with AZFa deletion had the highest level of FSH, with 36.67 ± 12.70 mIU/mL, followed by AZFc (14.63 ± 11.44) and AZFd (8.25 ± 3.20). Meanwhile, we did not find significant differences in other paraclinical characteristics and testis volume measurements.
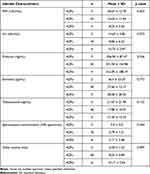 |
Table 2 Hormonal Profile and Other Specific Symptoms of Men with AZF Individual Microdeletions |
Table 3 describes the complete and partial AZFc microdeletion detail in the prevalence and sperm concentration. The effectiveness of Microdissection testicular sperm extraction (mTESE) was also evaluated for retrieving sperm from azoospermic men. Four cases with complete AZFc microdeletion were detected (sY254+sY255) at 9.30%, 2 subjects of them consented to go under mTESE, and one successfully accounted for 50%.
 |
Table 3 AZFc Microdeletion and mTESE Outcome for Azoospermia |
Thirty-nine patients had gr/gr partial AZFc microdeletion (90.70%). Among them, 13 men had severe oligozoospermia, 8 had cryptozoospermia, and 18 had azoospermia. Notably, over half of the patients with gr/gr microdeletion had sperm through self-ejaculation. Of the 18 others with azoospermia, 11 were treated by Microdissection testicular sperm retrieval, and sperm retrieval was successful in 7 of those patients (63.63%).
Discussion
The Y chromosome classically includes the pseudoautosomal regions (PAR1 and PAR2) and the male-specific region.25 Pseudoautosomal areas are located at the distal ends of the chromosome. The other, namely the male-specific region of the Y Chromosome (MSY) containing essential genes responsible for sex determination and spermatogenesis. The MSY region consists of the short-arm para-centromeric region, the centromere, and the long-arm para-centromeric component. Three ingredient sequences were found in this region consisting of X-transposed (99% similar to the X chromosome), X-degenerate (single-copy genes or pseudogene X-linked genes), and amplicons. Ampliconic sequences are characterized by sequence pairs organized in eight large palindromes.25 Y chromosomal genetic variations involving point mutations, microdeletions, or alterations of genetic copy number, may affect decreased or arrested spermatogenesis.5,29 When Y chromosome microdeletion occurs in Yq11, it is referred to as AZF microdeletion.15 Indeed, most of the MSY amplicons euchromatin located in the Yq11 region recombine through a non-reciprocal transfer of sequences occurring between duplicated sequences within the chromosome, the biological process maintaining the gene content between repeated sequences organized in pairs in inverted orientation within palindromes.14,21 However, the same orientation through Non-Allelic Homologous Recombination (NAHR) is widely supposed that can lead to AZF microdeletions. This abnormal recombination process with the same orientation can occur between rich homologous repeated sequences leading to genetic material loss.10,30,31
Y-Chromosome Microdeletion (YCM) appended within the proximal part of Y long-arm (Yq11) is highly suggestive of spermatogenic disorder and low sperm concentration. The prevalence of YCM was suggested as high as 2% in the general population by the Practice Committee of the American Society for Reproductive Medicine (ASRM), while this prevalence was far lower than that in the European Academy of Andrology (EAA) report.6,10 In contrast, it is widely accepted that the disorders are far more prevalent among men affected by abnormal semen parameters.11 The frequency of AZF microdeletion ranges from 2% in Germany, 10.02% in India, 12% in America, and 24.2% in Iran.17,32 In a retrospective study performed in 1274 azoospermic or oligozoospermic Chinese men, 164 patients having Y chromosome microdeletion was shown at 12.87%.33 AZF microdeletion has been studied globally; their frequency within infertile men populations presents vast heterogeneity depending on geography and ethnicity, so more studies on this failure need to be done. All Vietnamese patients with azoospermia, cryptozoospermia, and severe oligozoospermia were screened for finding AZF microdeletion in this retrospective multicenter study. One hundred and fifty-three men carried AZF microdeletion detected in 1121 screened patients by sequence-tagged sites (STSs) analysis. Consequently, the proportion of men with AZF microdeletion in severe abnormal sperm count was 13.64%. This proportion was slightly higher than that in most recommended countries because stringent inclusion criteria recruited severe oligozoospermia with less than a 5 million sperm/mL threshold.
The AZF microdeletions were first classified into three distinct regions, namely the proximal (AZFa), middle (AZFb), and distal deletion (AZFc), depending on the reflexing spermatogenic failure phenotype manifestations.16 Then, a putative fourth region (AZFd) between AZFb and AZFc was hypothesized.20 For two decades, reliable duplex and/or multiplex PCR diagnostic testing was performed to identify the presence or absence of AZF regions. Multiplex PCR permits the concomitant amplification of some targets in the same reaction by using more than 1 pair of primers. It was approved that at least two non-polymorphic STS loci should be amplified for diagnostic reliability in each AZF region. The EAA/EMQN guidelines recommend a list of primers for AZFa (sY84, sY86), AZFb (sY127, sY134), AZFc (sY254, sY255). This primer set will enable the detection of over 95% of the microdeletions reported, so extension determination of the microdeletions should be performed.10 Both sY84 and sY86 makers are appended upstream of the DDX3Y and USPY9 genes which are essential genes of the AZFa region. The deletion of both sY84 and sY86 is often considered a complete deletion. However, partial AZFa deletions have been reported despite both sY84 and sY86 loci absence, and their effect is milder than the complete situation. Consequently, the AZFa region should be analyzed in detail by 4 specific STS (sY84, sY86, USP9Y, DBY).34 In the first period of this study, 38 patients were first diagnosed by checking the 10 AZF-specific STSs makers, including AZFa (sY84, sY86, USP9Y, DBY), AZFb (sY127, sY134), AZFc (sY254, sY255) and AZFd (sY152, sY153). Then, according to updated guidelines, second-choice markers have been performed for extension analyses of AZFb and AZFc microdeletions.10,35,36 Our laboratory has chosen further novel markers: sY105, sY121, and sY1192 (for the AZFb region); and sY160, sY1191, sY1291, and BPY2 (for the AZFc region), which have an important function to candidate AZF-specific genes.14,37
In the present study, we identified microdeletions of the AZFa, AZFb, AZFc, and AZFd plus several concurrences, which were AZFabc, AZFabcd, AZFbc, AZFbcd, AZFbd, and AZFcd. AZFa microdeletion was detected in 3 out of 153 patients (1.96%), and all of them were azoospermic. Because of losing 2 genes in the AZFa region, which were USP9Y, DBY resulted in Sertoli cell only syndrome (SCOS), having no matured sperm formed in seminiferous tubes.38–41 The diagnosis of the AZFa microdeletion mostly implies spermatozoa retrieval failure from the testis.42 Nobody with AZFb microdeletion was seen in this study, which was difficult in the prevalence of others.33,43 The AZFb region was supposed to be necessary for spermatozoa maturation; thus, a testicular phenotype of maturation arrest can be found in patients with complete AZFb microdeletion.44 The highest rate of microdeletion was found at the AZFc region (49.67%), followed by 3.92% at the AZFd locus. The AZFd microdeletion often be supposed to get mild oligozoospermia or even normozoospermia.20 However, more information about the AZFd region has been added, including the prevalence and the manifestation in both azoospermia (1 case), cyptozoospermia (2 cases), and severe oligozoospermia phenotypes (3 cases). The AZFbc, AZFbcd, AZFbd, and AZFcd combination phenotypes had a variety of manifestations and did not correlate with semen quality (except AZFbc, expressed in mostly azoospermia). In contrast, all males with AZFabc and AZFabcd genotypes were in the azoospermia phenotype. Consequently, the different type of AZF microdeletions in males reflects different clinical manifestations. About 25–55% of men carrying AZF microdeletions had pathological testicular such as hypospermatogenesis, sperm maturation arrest, or SCOS.45,46
Our data also indicated 35 men presented chromosomal abnormalities (22.88%) in 153 patients. Interestingly, 5 patients of AZF microdeletions also had Klinefelter syndrome (47, XXY), which was reported in some previous studies.47,48 Although the variety of degrees of AZF microdeletions can be seen, the combination of the microdeletions with Klinefelter syndrome clearly made semen abnormalities severe, mostly in azoospermia.49,50 There were 2 patients affected by AZFabcd microdeletion. In this case (all the Yq markers are absent), the interpretation of the control marker (SRY) is of outstanding importance. After karyotype analysis, they all were diagnosed in 46, XX male syndrome because of Sex-determining region Y (SRY) translocating from the Y to X chromosome during paternal meiosis. The 46 XX male, known as de la Chapelle syndrome, presents primary infertility and azoospermia.51,52 Generally, the infertile severity was most remarkable in AZFa deletions and decreased with subsequently deleted intervals AZFb, AZFc, and AZFd, perspectively. In addition, the severity of infertility phenotypes may correlate to the size of AZF microdeletion (partial, complete, or concurrence) and the abnormal karyotype situation. Besides spermatogenesis failure, patients with AZF microdeletions typically present with higher FSH serum levels and inconsistent effects of LH, prolactin, estradiol, and testis volume, which were compared between AZFa, AZFc, and AZFd microdeletion groups (AZFb microdeletion was not seen in this study). Physiological serum FSH level is necessary for the appropriate induction and maintenance of spermatogenesis. In this study, the average FSH serum levels in AZFa and AZFc microdeletion groups were higher than the normal range.53 Men affected by AZFa deletion had the highest level of FSH, with 36.67 ± 12.70 mIU/mL, followed by AZFc (14.63 ± 11.44) and AZFd (8.25 ± 3.20). The difference had clinical significance. It has been reported that azoospermic patients with high FSH levels ≥20 IU/L have lower chances of having live-born children with assisted reproductive technologies,54 so FSH levels might be considered as a predictor of mTESE outcome in azoospermic men.
Azoospermic men carrying AZF microdeletions are commonly received specific therapy by microdissection testicular sperm extraction (mTESE), which plays the gold-standard surgical technique to retrieve sperm for intracytoplasmic sperm injection (ICSI). According to former studies, mTESE is not recommended for AZFa, AZFb, AZFbc, and AZFabc patients, because of seldom retrieve spermatozoa.33 Another reason for spending much effort in the AZFc region was the highest rate of AZF microdeletions, accounting for 49.67%, which was still less than previous reports, such as 62.20% and 77.78%.33,55 When both sY254 and sY255 loci are deleted, a diagnosis of complete AZFc microdeletion can be made. Four cases with complete AZFc microdeletion were detected (sY254+sY255), leading to azoospermic/cryptozoospermic male infertility. The chance of sperm retrieval during surgical or microsurgical procedures may increase to more than 50%.56,57 There was only one type of partial AZFc microdeletion in this study: gr/gr deletion (sY1191 is positive; sY1291 is negative), accounting for 39 patients. The partial deletions reduce the copy number of gene families, resulting in the loss of one of the two CDY1, two of the four DAZ gene copies, and one of the three BPY2 gene copies. The b2/b3 microdeletion removes almost the same gene content as gr/gr.5,14 The partial deletions were common in African and Asian men from 10% to 15%, but less than 5% in other populations.5 AZFc is the most common region of the AZF locus associated with impaired spermatogenesis, but the partial AZFc microdeletion roles in spermatogenesis are controversial. Notably, over half of the patients with gr/gr microdeletion had sperm through self-ejaculation. Of the 18 others with azoospermia, 11 were treated by microdissection testicular sperm retrieval, and sperm retrieval was successful in 7 of those patients (63.63%). These results indicated that partial AZFc microdeletion was the lightest AZF microdeletion type, with variant manifestations in the semen ejaculated and a high possibility of sperm retrieval from semen and testis. Although several previous studies reported that the gr/gr deletions also occurred in fertile males with normozoospermia, the association of gr/gr microdeletion and male infertility depends on ethnicity, such as Asian, Australian, and African men.5,58
Conclusion
The prevalence of AZF microdeletions was 13.64% in Vietnamese men with azoospermia, cryptozoospermia, and severe oligozoospermia. The absence of AZFa and/or AZFb regions often expresses azoospermia and the increasing FSH serum. The AZFc region played the most widespread type, including complete microdeletion and gr/gr partial microdeletion (but no deletions were found in the b2/b3 region) with various changes in semen analysis. Microdissection testicular sperm extraction (mTESE) was an effective method for sperm retrieval from the testis of azoospermia men in the AZFc group.
Data Sharing Statement
The data that support our findings are available from the corresponding author upon reasonable request.
Ethical Statements
All patients provided informed written consent to be participants in this study. The study’s protocol was approved by the Ethical Review Committee of the Andrology and Fertility Hospital of Hanoi (No.779/2021/IRB-AFHANOI) and appropriate to the Helsinki Declaration.
Acknowledgment
We gave sincere thanks to the Military Institute of Clinical Embryology and Histology and the Andrology and Fertility Hospital of Hanoi for supporting this study.
Author Contributions
All authors contributed significantly to this work, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declared no conflicts of interest for this work.
References
1. Cooper TG, Noonan E, von Eckardstein S, et al. World Health Organization reference values for human semen characteristics. Hum Reprod Update. 2010;16(3):231–245. doi:10.1093/humupd/dmp048
2. Kumar N, Singh AK. Trends of male factor infertility, an important cause of infertility: a review of literature. J Hum Reprod Sci. 2015;8(4):191–196. doi:10.4103/0974-1208.170370
3. World Health Organization. World Health Organization laboratory manual. Google Scholar. Available from: https://scholar.google.com/scholar_lookup?title=WHO+Laboratory+Manual+for+the+Examination+and+Processing+of+Human+Semen&publication_year=2010&.
4. Alrabeeah K, Wachter A, Phillips S, Cohen B, Al-Hathal N, Zini A. Sperm retrieval outcomes with microdissection testicular sperm extraction (micro-TESE) in men with cryptozoospermia. Andrology. 2015;3(3):462–466. doi:10.1111/andr.12000
5. Colaco S, Modi D. Genetics of the human Y chromosome and its association with male infertility. Reprod Biol Endocrinol. 2018;16:14. doi:10.1186/s12958-018-0330-5
6. Practice Committee of the American Society for Reproductive Medicine. Diagnostic evaluation of the infertile male: a committee opinion. Fertil Steril. 2015;103(3):e18–25. doi:10.1016/j.fertnstert.2014.12.103
7. Rabinowitz MJ, Huffman PJ, Haney NM, Kohn TP. Y-chromosome microdeletions: a review of prevalence, screening, and clinical considerations. Appl Clin Genet. 2021;14:51–59. doi:10.2147/TACG.S267421
8. Chandley AC. Chromosome anomalies and Y chromosome microdeletions as causal factors in male infertility. Hum Reprod. 1998;13:45–50. doi:10.1093/humrep/13.suppl_1.45
9. Foresta C, Garolla A, Bartoloni L, Bettella A, Ferlin A. Genetic abnormalities among severely oligospermic men who are candidates for intracytoplasmic sperm injection. J Clin Endocrinol Metab. 2005;90(1):152–156. doi:10.1210/jc.2004-1469
10. Krausz C, Hoefsloot L, Simoni M, Tüttelmann F. European Academy of Andrology, European Molecular Genetics Quality Network. EAA/EMQN best practice guidelines for molecular diagnosis of Y-chromosomal microdeletions: state-of-the-art 2013. Andrology. 2014;2(1):5–19. doi:10.1111/j.2047-2927.2013.00173.x
11. Krausz C, Casamonti E. Spermatogenic failure and the Y chromosome. Hum Genet. 2017;136(5):637–655. doi:10.1007/s00439-017-1793-8
12. Sadeghi-Nejad H, Farrokhi F. Genetics of azoospermia: current knowledge, clinical implications, and future directions. Part II: y chromosome microdeletions. Urol J. 2007;4(4):192–206.
13. Yen P. The fragility of fertility. Nat Genet. 2001;29(3):243–244. doi:10.1038/ng1101-243
14. Repping S, Skaletsky H, Brown L, et al. Polymorphism for a 1.6-Mb deletion of the human Y chromosome persists through balance between recurrent mutation and haploid selection. Nat Genet. 2003;35(3):247–251. doi:10.1038/ng1250
15. Tiepolo L, Zuffardi O. Localization of factors controlling spermatogenesis in the nonfluorescent portion of the human Y chromosome long arm. Hum Genet. 1976;34(2):119–124. doi:10.1007/BF00278879
16. Vogt PH, Edelmann A, Kirsch S, et al. Human Y chromosome azoospermia factors (AZF) mapped to different subregions in Yq11. Hum Mol Genet. 1996;5(7):933–943. doi:10.1093/hmg/5.7.933
17. Simoni M, Tüttelmann F, Gromoll J, Nieschlag E. Clinical consequences of microdeletions of the Y chromosome: the extended Münster experience. Reprod Biomed Online. 2008;16(2):289–303. doi:10.1016/s1472-6483(10)60588-3
18. Lange J, Skaletsky H, van Daalen SKM, et al. Isodicentric Y chromosomes and sex disorders as byproducts of homologous recombination that maintains palindromes. Cell. 2009;138(5):855–869. doi:10.1016/j.cell.2009.07.042
19. Navarro-Costa P, Plancha CE, Gonçalves J. Genetic dissection of the AZF regions of the human Y chromosome: thriller or filler for male (in)fertility? J Biomed Biotechnol. 2010;2010:936569. doi:10.1155/2010/936569
20. Kent-First M, Muallem A, Shultz J, et al. Defining regions of the Y-chromosome responsible for male infertility and identification of a fourth AZF region (AZFd) by Y-chromosome microdeletion detection. Mol Reprod Dev. 1999;53(1):27–41. doi:10.1002/(SICI)1098-2795(199905)53:1<27::AID-MRD4>3.0.CO;2-W
21. Kuroda-Kawaguchi T, Skaletsky H, Brown LG, et al. The AZFc region of the Y chromosome features massive palindromes and uniform recurrent deletions in infertile men. Nat Genet. 2001;29(3):279–286. doi:10.1038/ng757
22. Dutta S, Paladhi P, Pal S, et al. Prevalence of Y chromosome microdeletion in azoospermia factor subregions among infertile men from West Bengal, India. Mol Genet Genomic Med. 2021;9(10):e1769. doi:10.1002/mgg3.1769
23. Rastgar Rezaei Y, Zarezadeh R, Nikanfar S, et al. microRNAs in the pathogenesis of non-obstructive azoospermia: the underlying mechanisms and therapeutic potentials. Syst Biol Reprod Med. 2021;67(5):337–353. doi:10.1080/19396368.2021.1951890
24. Fu XF, Cheng SF, Wang LQ, Yin S, De Felici M, Shen W. DAZ family proteins, key players for germ cell development. Int J Biol Sci. 2015;11(10):1226–1235. doi:10.7150/ijbs.11536
25. Skaletsky H, Kuroda-Kawaguchi T, Minx PJ, et al. The male-specific region of the human Y chromosome is a mosaic of discrete sequence classes. Nature. 2003;423(6942):825–837. doi:10.1038/nature01722
26. Colpi GM, Caroppo E. Performing microdissection testicular sperm extraction: surgical pearls from a high-volume infertility center. J Clin Med. 2021;10(19):4296. doi:10.3390/jcm10194296
27. Flannigan R, Bach PV, Schlegel PN. Microdissection testicular sperm extraction. Transl Androl Urol. 2017;6(4):745–752. doi:10.21037/tau.2017.07.07
28. Caroppo E, Colpi EM, Gazzano G, et al. The seminiferous tubule caliber pattern as evaluated at high magnification during microdissection testicular sperm extraction predicts sperm retrieval in patients with non-obstructive azoospermia. Andrology. 2019;7(1):8–14. doi:10.1111/andr.12548
29. Walsh TJ, Pera RR, Turek PJ. The genetics of male infertility. Semin Reprod Med. 2009;27(2):124–136. doi:10.1055/s-0029-1202301
30. Vergnaud G, Page DC, Simmler MC, et al. A deletion map of the human Y chromosome based on DNA hybridization. Am J Hum Genet. 1986;38(2):109–124.
31. Li G, Davis BW, Raudsepp T, et al. Comparative analysis of mammalian Y chromosomes illuminates ancestral structure and lineage-specific evolution. Genome Res. 2013;23(9):1486–1495. doi:10.1101/gr.154286.112
32. Waseem AS, Singh V, Makker GC, et al. AZF deletions in Indian populations: original study and meta-analyses. J Assist Reprod Genet. 2020;37(2):459–469. doi:10.1007/s10815-019-01661-0
33. Liu T, Song YX, Jiang YM. Early detection of Y chromosome microdeletions in infertile men is helpful to guide clinical reproductive treatments in southwest of China. Medicine. 2019;98(5):e14350. doi:10.1097/MD.0000000000014350
34. Kamp C, Huellen K, Fernandes S, et al. High deletion frequency of the complete AZFa sequence in men with Sertoli-cell-only syndrome. Mol Hum Reprod. 2001;7(10):987–994. doi:10.1093/molehr/7.10.987
35. Vogt PH, Bender U. Human Y chromosome microdeletion analysis by PCR multiplex protocols identifying only clinically relevant AZF microdeletions. Methods Mol Biol. 2013;927:187–204. doi:10.1007/978-1-62703-038-0_17
36. Khabour OF, Fararjeh AS, Alfaouri AA. Genetic screening for AZF Y chromosome microdeletions in Jordanian azoospermic infertile men. Int J Mol Epidemiol Genet. 2014;5(1):47–50.
37. Repping S, van Daalen SKM, Korver CM, et al. A family of human Y chromosomes has dispersed throughout northern Eurasia despite a 1.8-Mb deletion in the azoospermia factor c region. Genomics. 2004;83(6):1046–1052. doi:10.1016/j.ygeno.2003.12.018
38. Foresta C, Ferlin A, Moro E. Deletion and expression analysis of AZFa genes on the human Y chromosome revealed a major role for DBY in male infertility. Hum Mol Genet. 2000;9(8):1161–1169. doi:10.1093/hmg/9.8.1161
39. Ditton HJ, Zimmer J, Kamp C, Rajpert-De Meyts E, Vogt PH. The AZFa gene DBY (DDX3Y) is widely transcribed but the protein is limited to the male germ cells by translation control. Hum Mol Genet. 2004;13(19):2333–2341. doi:10.1093/hmg/ddh240
40. Kishi K, Uchida A, Takase HM, et al. Spermatogonial deubiquitinase USP9X is essential for proper spermatogenesis in mice. Reproduction. 2017;154(2):135–143. doi:10.1530/REP-17-0184
41. Sun C, Skaletsky H, Birren B, et al. An azoospermic man with a de novo point mutation in the Y-chromosomal gene USP9Y. Nat Genet. 1999;23(4):429–432. doi:10.1038/70539
42. Liu XY, Wang RX, Fu Y, Luo LL, Guo W, Liu RZ. Outcomes of intracytoplasmic sperm injection in oligozoospermic men with Y chromosome AZFb or AZFc microdeletions. Andrologia. 2017;49(1). doi:10.1111/and.12602
43. Simoni M, Bakker E, Krausz C. EAA/EMQN best practice guidelines for molecular diagnosis of y-chromosomal microdeletions. State of the art 2004. Int J Androl. 2004;27(4):240–249. doi:10.1111/j.1365-2605.2004.00495.x
44. Kleiman SE, Yogev L, Lehavi O, et al. The likelihood of finding mature sperm cells in men with AZFb or AZFb-c deletions: six new cases and a review of the literature (1994–2010). Fertil Steril. 2011;95(6):2005–2012. doi:10.1016/j.fertnstert.2011.01.162
45. Bansal SK, Jaiswal D, Gupta N, et al. Gr/gr deletions on Y-chromosome correlate with male infertility: an original study, meta-analyses, and trial sequential analyses. Sci Rep. 2016;6:19798. doi:10.1038/srep19798
46. Foresta C, Moro E, Ferlin A. Y chromosome microdeletions and alterations of spermatogenesis. Endocr Rev. 2001;22(2):226–239. doi:10.1210/edrv.22.2.0425
47. Li LX, Dai HY, Ding XP, et al. Investigation of AZF microdeletions in patients with Klinefelter syndrome. Genet Mol Res. 2015;14(4):15140–15147. doi:10.4238/2015.November.25.2
48. Mitra A, Dada R, Kumar R, Gupta NP, Kucheria K, Gupta SK. Y chromosome microdeletions in azoospermic patients with Klinefelter’s syndrome. Asian J Androl. 2006;8(1):81–88. doi:10.1111/j.1745-7262.2006.00083.x
49. Hadjkacem-Loukil L, Ghorbel M, Bahloul A, Ayadi H, Ammar-Keskes L. Genetic association between AZF region polymorphism and Klinefelter syndrome. Reprod Biomed Online. 2009;19(4):547–551. doi:10.1016/j.rbmo.2009.05.006
50. Golin AP, Neil S, Flannigan R. Case - Severely oligozoospermic patient with both mosaic Klinefelter syndrome and a complete azoospermia factor c (AZFc) Y chromosome microdeletion. Can Urol Assoc J. 2020;14(5):E224–E226. doi:10.5489/cuaj.6255
51. Majzoub A, Arafa M, Starks C, Elbardisi H, Al Said S, Sabanegh E. 46 XX karyotype during male fertility evaluation; case series and literature review. Asian J Androl. 2017;19(2):168–172. doi:10.4103/1008-682X.181224
52. Terribile M, Stizzo M, Manfredi C, et al. 46,XX testicular Disorder of Sex Development (DSD): a case report and systematic review. Medicina. 2019;55(7):371. doi:10.3390/medicina55070371
53. Bahmanimehr A, Zeighami S, Namavar Jahromi B, et al. Detection of Y chromosome microdeletions and hormonal profile analysis of infertile men undergoing assisted reproductive technologies. Int J Fertil Steril. 2018;12(2):173–177. doi:10.22074/ijfs.2018.5244
54. Zitzmann M, Nordhoff V, von Schönfeld V, et al. Elevated follicle-stimulating hormone levels and the chances for azoospermic men to become fathers after retrieval of elongated spermatids from cryopreserved testicular tissue. Fertil Steril. 2006;86(2):339–347. doi:10.1016/j.fertnstert.2005.12.058
55. Sun K, Chen XF, Zhu XB, et al. A new molecular diagnostic approach to assess Y chromosome microdeletions in infertile men. J Int Med Res. 2012;40(1):237–248. doi:10.1177/147323001204000124
56. Park SH, Lee HS, Choe JH, Lee JS, Seo JT. Success rate of microsurgical multiple testicular sperm extraction and sperm presence in the ejaculate in Korean men with y chromosome microdeletions. Korean J Urol. 2013;54(8):536–540. doi:10.4111/kju.2013.54.8.536
57. Huang IS, Chen WJ, Li LH, Brannigan RE, Huang WJ. The predictive factors of successful sperm retrieval for men with Y chromosome AZFc microdeletion. J Assist Reprod Genet. 2022;39(10):2395–2401. doi:10.1007/s10815-022-02601-1
58. Stouffs K, Lissens W, Tournaye H, Haentjens P. What about gr/gr deletions and male infertility? Systematic review and meta-analysis. Hum Reprod Update. 2011;17(2):197–209. doi:10.1093/humupd/dmq046
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
“Dancing Coins?” Unexpected Finding During microsurgery and Potential Risk of Sperm Damage: Intrascrotal Calculi: A Retrospective Analysis
Liu H, Zhang Z, Lv W, Li Q, Qing Z, Zhong X, Peng S, Lin H, Zheng H
International Journal of General Medicine 2022, 15:8755-8766
Published Date: 29 December 2022
