Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
Schizophrenia Treatment with Second-Generation Antipsychotics: A Multi-Country Comparison of the Costs of Cardiovascular and Metabolic Adverse Events and Weight Gain
Authors Kearns B , Cooper K , Cantrell A, Thomas C
Received 19 September 2020
Accepted for publication 18 November 2020
Published 20 January 2021 Volume 2021:17 Pages 125—137
DOI https://doi.org/10.2147/NDT.S282856
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Benjamin Kearns, Katy Cooper, Anna Cantrell, Chloe Thomas
School of Health and Related Research, The University of Sheffield, Sheffield, UK
Correspondence: Benjamin Kearns Email [email protected]
Purpose: Cardiovascular and metabolic adverse events are costly to treat, and their incidence is increased amongst people with schizophrenia, with different rates observed for second-generation antipsychotics. To inform treatment choice, this study sought to estimate the lifetime costs associated with antipsychotic choice, and how these costs varied across European countries.
Methods: Systematic searches were conducted to identify evidence on effectiveness and costs. A Markov model was developed to assess the costs of ten antipsychotics: aripiprazole, brexpiprazole, cariprazine, lumateperone, lurasidone, olanzapine, paliperidone, quetiapine, risperidone and ziprasidone. Costs were obtained for seven countries: Italy, Hungary, France, Slovenia, Spain, Sweden and the UK. The costs considered were adverse events (including diabetes, myocardial infarction, stroke and weight gain), drug costs, relapse, treatment discontinuation and schizophrenia management. Two adult populations were modelled; initiating either acute or maintenance treatment, with a lifetime horizon for both.
Results: Lurasidone was associated with the lowest lifetime costs amongst patients initiating acute treatment compared to all other atypical antipsychotics considered. The second lowest costs were for ziprasidone. These results were observed for all seven countries. The main drivers of cost differences were rates of diabetes and cardiovascular diseases, which were lowest for lurasidone, followed by ziprasidone then lumateperone. Costs for managing weight gain were lowest for lurasidone and ziprasidone. Similar results were observed for patients initiating maintenance treatment.
Conclusion: Diabetes and cardiovascular events are large drivers of lifetime costs for people with schizophrenia. Lurasidone is predicted to have the lowest rates of these adverse events, and so the lowest costs amongst patients initiating acute treatment in seven European countries compared to nine other antipsychotics. Future research should investigate the individual costs of relapse management, including differences in the costs and proportions of hospitalizations.
Keywords: cost analysis, side-effects, Europe, acute treatment, maintenance treatment
Plain Language Summary
People with schizophrenia can be treated with antipsychotics. These are often effective at controlling the symptoms of schizophrenia but can also result in a number of side effects. These include diabetes, weight gain, and conditions that affect the heart or blood vessels (cardiovascular diseases). The rates of these side effects vary with choice of antipsychotic, and the costs of treating these side effects is also different in different countries. The aim of our research was to identify the full costs of using antipsychotics to treat people with schizophrenia. The results of this research will help to guide the choice of antipsychotic for schizophrenia treatment.
We considered ten antipsychotics and looked at the costs in seven different European countries: Italy, Hungary, France, Slovenia, Spain, Sweden and the UK. We included the costs of the antipsychotic treatment, the costs of treating side effects, and the costs of managing schizophrenia (such as when people relapse). We searched for published evidence for both antipsychotics (how well they work and how many side effects people have) and country-specific costs. We used a mathematical model to bring all this evidence together and estimate likely lifetime costs.
We found that, of the ten antipsychotics, Lurasidone and Ziprasidone had the lowest lifetime costs in all seven countries. This was mainly because their use led to lower rates of diabetes, cardiovascular diseases, and weight gain.
Introduction
Treating cardiovascular and metabolic (cardiometabolic) diseases can result in large costs to healthcare systems. In Europe, the annual cost of cardiovascular diseases in the general population was estimated to be €111 billion in 2015, whilst the global cost of diabetes is projected to reach $490 billion by 2030.1,2 There is an elevated risk of cardiometabolic disease amongst patients with schizophrenia; compared with the general population they are twice as likely to have diabetes,3 and twice as likely to be diagnosed and die with CVD,4 contributing to a reduction in life expectancy of 15 years.5 The risk factors for cardiometabolic disease are affected by choice of antipsychotic treatment.6
As schizophrenia is a life-long disorder it is important to consider the lifetime costs and adverse health effects associated with the choice of antipsychotic. This is a particular concern for second-generation antipsychotics which have similar efficacy for improving the acute symptoms of schizophrenia but differ in their side effect profiles. Clinical guidelines recommend that the choice of antipsychotic is driven primarily by rates of side effect, of which weight gain and associated cardiometabolic diseases play a prominent role.7 The treatment of side effects is not the only driver of lifetime costs. Relapses and treatment discontinuation will also vary by antipsychotic and contribute to overall costs.8,9 Overall costs will vary by country due to differences in healthcare systems, treatment pathways, and the distributions of cardiometabolic risk factors. Existing studies have sought to quantify the lifetime costs for people with schizophrenia.10–16 However, these studies only consider selected side effects for a small number of antipsychotics and are restricted to a single country.
The aim of this study was to estimate, for several European countries and several second-generation antipsychotics, the total lifetime costs associated with the management of schizophrenia and the treatment of side effects including cardiometabolic diseases. To support this research, systematic searches of the literature were conducted to identify both country-specific costs and antipsychotic-specific effectiveness and side effect data. To predict the incidence of long-term cardiometabolic diseases, risk models were used that are adjusted to be specific to a population with schizophrenia.
Methods
An economic model was developed to synthesise evidence on the effectiveness of specific second-generation antipsychotics, country-specific costs (including costs of antipsychotics and treating cardiometabolic diseases), country-specific risk factors for cardiometabolic diseases, and risk models to estimate long-term outcomes. Ten antipsychotics were included: aripiprazole, brexpiprazole, cariprazine, lumateperone, lurasidone, olanzapine, paliperidone, quetiapine, risperidone, and ziprasidone. To limit the number of antipsychotics to a feasible amount, only second- and third-generation atypical antipsychotics that are available and have the highest market shares on average in Europe were considered. Seven European countries were included: France, Hungary, Italy, Slovenia, Spain, Sweden, and the UK. The choice of country was based on the availability of country-specific cost data. Two adult populations; initiating either acute or maintenance antipsychotic treatment were modelled and followed-up for 50 years (assumed to represent a lifetime horizon) to capture all relevant differences in outcomes and costs.
Review of Clinical Evidence
A literature review was undertaken to identify existing systematic reviews, meta-analyses, network meta-analyses (NMAs) and indirect treatment comparisons of randomised controlled trials (RCTs). MEDLINE, Embase and the Cochrane Database of Systematic Reviews were searched in February 2020 based on the following combinations of search concepts: schizophrenia AND (antipsychotics OR 10 individual drugs) AND relevant outcome terms AND systematic review or meta-analysis or NMAs or indirect comparison. A full set of synonyms was used for each concept; the full MEDLINE search terms are provided in Appendix 1, with further details, including the inclusion criteria, provided in Appendix 2. The search covered publications from the year 2000 onwards. The most up-to-date analyses covering relevant interventions and reporting relevant outcomes were selected for full data extraction.
The evidence review considered a long list of 33 pre-defined outcomes including cardiometabolic risk factors and outcomes as well as effectiveness outcomes; the full list is replicated in Appendix 2. Consistent with existing economic evaluations, it was assumed that antipsychotics had similar efficacy for change in symptoms, so evidence on these was not collected.17–21 Both short-term outcomes (up to 13 weeks, typically six weeks) and longer-term outcomes (available up to three years) were considered. The majority of the data was obtained from NMAs of RCTs of multiple antipsychotics (Table 1). There remained some evidence gaps after conducting the review of NMAs and meta-analyses. Hence, additional searches in PubMed and ClinicalTrials.gov were undertaken to identify individual RCTs. Systematic reviews were also checked for relevant trials. Individual trials were sought and identified for lumateperone (short-term outcomes) and cariprazine and brexpiprazole (long-term outcomes).22–25 No long-term outcomes were identified for lumateperone, hence for the model these outcomes were assumed to be the average of the other antipsychotics. A total of 12 outcomes were used from the review of clinical evidence including change in lipids and fasting plasma glucose, weight gain, treatment discontinuation and relapse rates; the values and data sources for each are provided in Table 1. An overview of the key studies identified for the relevant outcomes is provided in Appendix 2. Blood pressure was identified as being a key outcome for which there was a lack of evidence. Targeted literature reviews, and contacting authors of key NMAs, did not identify any evidence that blood pressure outcomes differed by antipsychotic, hence it was assumed that there was no treatment effect. No evidence for specific antipsychotics was identified for rates of type 2 diabetes, stroke, myocardial infarction and metabolic syndrome. Risk models were used to estimate the first three outcomes as described subsequently (and in more detail in Appendix 3), whilst Appendix 4 provides details on how rates of metabolic syndrome were estimated.
 |
Table 1 Clinical Effectiveness Evidence: Short-Term and Long-Term Outcomes |
Risk Models
Risk models were used to link short-term changes in weight, cholesterol (total and high-density lipoprotein) and fasting plasma glucose to the probability of developing type 2 diabetes, stroke and myocardial infarction. For this analysis, QDiabetes was used to estimate the risk of developing type 2 diabetes, whilst QRisk was used to estimate the risk of developing a cardiovascular event (stroke or myocardial infarction).26,27 In contrast to other risk models,28,29 both of these risk models have been independently validated, and were derived on a European (UK) cohort whilst also including adjustment for both antipsychotic use and having schizophrenia/severe mental illness.
Where possible the risk models were populated using mean country-specific values as inputs. Details on all the inputs used, and their sources, are provided in Appendix 3.
Evidence on Costs
A systematic search was performed to identify country-specific costs relating to the management of schizophrenia and the treatment of side effects. MEDLINE was searched in February 2020, full details of the search terms used are provided in Appendix 1. As with the searches for clinical effectiveness, the cost search originally considered the long list of 33 pre-defined outcomes. Country-specific costs used in the analysis are provided in Table 2. All costs were inflated to 2018 costs. For comparability across countries, Table 2 reports costs in a common currency (US $), whereas for the analysis country-specific currencies are used; when country-specific costs were not available purchasing power parities were used (see Appendix 4 for more details). The cost perspective was direct medical costs. An overview is provided here; further details (including full references) are provided in Appendix 4, which also provides details on the search process.
 |
Table 2 Country-Specific Annual per Patient Costs (US $) |
In general, preference was given to the most recent studies. Clinically relevant weight gain was defined as an increase of at least 7% from baseline weight. The majority of studies costed cardiometabolic outcomes based on costs from the general population, this approach was used here. Most studies provided an overall cost of treating diabetes and its complications. This will lead to double-counting as some of the complication costs will be for cardiovascular diseases, hence these costs were multiplied by 0.78.30 The costs of cardiovascular disease included country-specific costs of stroke and myocardial infarction. For both outcomes separate costs were obtained for fatal events as well as for the first and subsequent years for non-fatal events. The overall cardiovascular costs were a weighted average of stroke and myocardial infarction costs, assuming that myocardial infarction events contributed to 61% of fatal and 47% of non-fatal cardiovascular events. These proportions were based on those observed in a large cohort of individuals with serious mental illness (of those with a recorded diagnosis, 39% had schizophrenia and 30% had bipolar disorder).28 Due to a lack of country-specific costs for treating metabolic syndrome, costs from a Swedish study were used for all countries.21 The development of hyperprolactinaemia was costed as requiring a GP visit every three months and one endocrinologist visit a year. Management of QT prolongation was assumed to require one additional visit each to a psychiatrist and a cardiologist every six months.
For the costs of disease management (relapse and stable disease), the costs of antipsychotic treatment were excluded to avoid double-counting. The costs of relapse were a weighted average of the costs of relapses requiring and not requiring hospitalisations, using country-specific weights where available. Treatment switching was assumed to require three psychiatrist visits.18
Country-specific costs of the public prices paid for antipsychotics were provided by Angelini (an Italian group present in Pharmaceuticals and Mass-Market internationally, with core work in rare diseases, mental health, inflammation and consumer health) and are confidential. This evidence did not include the cost of lumateperone, so a monthly cost of $1,320 was assumed based on a press release.31
Model Structure and Methods
A cohort Markov model was developed in Microsoft Excel to synthesise data on clinical effectiveness, costs, and risk models.32 Costs were discounted at 3.5% per year. As both risk models provided separate estimates for males and females, all analyses were performed separately by sex. Overall results were obtained as a country-specific weighted average of the sex-specific results. Two populations were considered: patients initiating acute treatment for schizophrenia, and patients initiating maintenance treatment. For consistency with the main evidence sources, the patient population was defined as adults with schizophrenia or a related disorder (such as schizoaffective or schizophreniform).6,9
For schizophrenia both acute and longer-term outcomes are important. To align with the clinical evidence, the model included an initial six-week acute treatment period, followed by a longer-term period of maintenance treatment and remission, with a cycle length of one year. Patients initiating acute treatment began in the acute phase, and moved into the longer-term phase following successful treatment. Treatment effects for those initiating maintenance treatment began on entering the long-term phase. The model had nine health states, as shown in Figure 1. In addition, the treatment outcomes of relapse, discontinuation and remission were also modelled, as were changes in risk factors over time. Patients who discontinued treatment or experienced a relapse received subsequent treatment with a different antipsychotic. It was assumed that subsequent treatment represented a blended average of the available antipsychotics, and so the average effectiveness (and costs) for these was used. Due to its high cost, lumateperone was not included when calculating the blended cost. Patients were assumed to be in remission, and so not require treatment with antipsychotics, after experiencing two years of maintenance treatment without either a relapse or treatment discontinuation.7 Hence changes in long-term changes in weight gain and fasting plasma glucose levels could only occur for up to two years. For the cardiometabolic health states in Figure 1, costs were based on the proportion of the cohort in each health state at each time cycle. The remaining costs (for weight gain, hyperprolactinaemia, QT prolongation, treatment discontinuation and relapses) were based on the proportion of the cohort experiencing these events per time cycle (for more details, see Appendix 4).
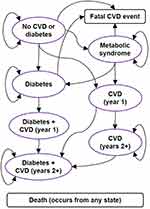 |
Figure 1 Model schematic (patient health states). Abbreviation: CVD, cardiovascular disease. |
Country-specific age and sex life tables were used to model the probability of non-cardiovascular mortality. To account for the increased probability of death amongst people with schizophrenia, standardised mortality ratios were applied. Country-specific values were identified for Hungary (2.4 for both sexes),33 Sweden (2.8 men, 2.4 women)10 and the UK (2.9 men, 2.8 women).34 For the remaining countries values from a European meta-analysis were used: 3.0 for men and 2.4 for women.35
To assess uncertainty, both probabilistic and deterministic sensitivity analyses were undertaken. For the probabilistic sensitivity analysis 1,000 iterations were undertaken; proportions were modelled with a beta distribution, a Dirichlet distribution was used for multinomial data (ethnicity and smoking status), costs were modelled with a gamma distribution (drug costs were assumed known and so fixed), with the remaining parameters modelled via a Normal distribution. The following deterministic sensitivity analyses were conducted:
- For the treatment effect on weight gain during the acute period use results from the network meta-analysis of Pillinger and colleagues.6
- For the treatment effect on relapses during the maintenance period use results from the network meta-analysis of Millier and colleagues where available (as no data were available for paliperidone it was assumed to have the same effectiveness as olanzapine).8
- Alternative estimates for the cost of relapse and stable disease from the European SOHO study.36
- Alternative weight gain costs for France,37 Spain,20 Sweden,10 and the UK18 (in the absence of evidence, costs for Hungary and Slovenia were both derived from the UK costs, whilst costs for Italy were derived from the Spanish costs).
- Use the same starting age for all countries, based on the meta-analysis of Huhn and colleagues.9
- Use the same percent of females for all countries, based on the meta-analysis of Huhn and colleagues.9
- Use starting values for metabolic parameters from an alternative study.38
- Reducing all-cause mortality rates by 37% to account for having cardiovascular deaths as a separate heath state.39
Results
An overview of the lifetime costs for the cohort starting with acute treatment is provided in Table 3, whilst results for the cohort starting with maintenance treatment are provided in Table 4. For both tables results are ordered by total costs for the UK and are reported using country-specific currencies. A full breakdown of costs is provided in Appendix 5 Tables 6 and 7.
 |
Table 3 Lifetime Costs, Patients Initiating Acute Treatment (Country-Specific Currencies) |
 |
Table 4 Lifetime Costs, Patients Initiating Maintenance Treatment (Country-Specific Currencies) |
Lurasidone was associated with the lowest lifetime costs amongst patients initiating acute treatment compared to all other atypical antipsychotics considered. This result was similarly observed in all 7 countries considered. The second lowest costs were for ziprasidone. The main drivers of cost differences were rates of diabetes and cardiovascular diseases, which were lowest for lurasidone, followed by ziprasidone then lumateperone. On average, cardiometabolic outcomes accounted for approximately two thirds of the total lifetime costs, although it varied by country being the highest in Italy and lowest in Slovenia. Costs for managing weight gain were lowest for ziprasidone and lurasidone. Total costs were very similar for aripiprazole, brexpiprazole and risperidone and these drugs always have the lowest overall costs after lurasidone and ziprasidone. The drugs resulting in the highest overall cost were olanzapine and lumateperone for all of the countries considered. For lumateperone, the main cost driver was the cost of the drug which contributed to between 21% and 46% of total costs. For the other antipsychotics, drug costs varied from 1% of the total cost (olanzapine in France and Spain) to 10% (lurasidone in Hungary). If lumateperone cost the same as lurasidone, it would have the third lowest overall cost for all countries (behind lurasidone and ziprasidone).
For patients initiating maintenance treatment lurasidone and ziprasidone were associated with the lowest lifetime costs, in all countries considered. The highest costs were again generally observed for olanzapine and lumateperone. Reducing the cost for lumateperone to be the same as for lurasidone led to its total cost being between the sixth lowest (Sweden) and third lowest (Spain). More variation in the ranking of the remaining antipsychotics was observed for the maintenance population compared with the acute population. For example, aripiprazole had the third lowest costs for four countries (France, Italy, Hungary, UK), but the second highest costs for Slovenia, and the fourth and fifth highest costs for Spain and Slovenia, respectively. The relative contribution of cardiometabolic outcomes to overall costs was almost identical amongst those initiating maintenance and acute treatments.
Results from probabilistic sensitivity analyses are demonstrated in Figure 2, which demonstrates the distribution of lifetime costs, and Appendix 5 Table 7 which provides the probability that each treatment has the lowest overall costs, per country. Figure 2 demonstrates substantial variability across iterations. For each country lurasidone always had the highest probability of having the lowest overall costs. For Slovenia and Sweden, the probability was 35.4% and 38.4%, respectively. For the remaining five countries lurasidone had the lowest overall costs in the majority of the iterations (range: 54.8% in Spain to 77.8% in Italy). Ziprasidone always had the second highest probability (range: 19.2% in Italy to 77.8% in Sweden); no other treatment had a probability above 8.6% (risperidone in Slovenia).
 |
Figure 2 Distribution of lifetime costs (acute population). |
Results for the seven deterministic sensitivity analyses are provided in Appendix 5 Table 8. In general results for these were very similar to the main analysis, with lurasidone and ziprasidone being the two drugs with the lowest lifetime costs in the majority of analysis, whilst olanzapine and lumateperone generally had the two highest costs. The main exception was for the second sensitivity analysis (alternative values for long-term relapse), for which brexpiprazole had the lowest lifetime costs for Slovenia (lurasidone had the lowest costs for all other countries), and the second lowest costs for Spain and Sweden.
Discussion
Lurasidone and ziprasidone were associated with the lowest total lifetime costs when initiated as acute or maintenance treatments for all seven countries considered. This was primarily due to the avoidance of diabetes and cardiovascular events, although costs of weight gain were also lowest for these two drugs. The rankings of the remaining antipsychotics varied by country, emphasising the importance of considering country-specific costs.
Existing evaluations of lifetime costs amongst people with schizophrenia only considered a small number of antipsychotics or a limited range of cardiometabolic outcomes. This limits the comparability of their results to this study. The most comprehensive previous evaluation was that conducted for the UK NICE clinical guidelines on the treatment and management of schizophrenia in adults.18 This evaluation included four of the ten antipsychotics considered here (aripiprazole, olanzapine, paliperidone, risperidone), as well as the main cardiometabolic outcomes (diabetes, stroke and myocardial infarction). It found that olanzapine and paliperidone (which had the lowest relapse rates) had the lowest lifetime costs but did not provide a cost breakdown. The results are in contrast with this analysis, for which olanzapine and paliperidone still had the lowest relapse rates (0.24 and 0.26 respectively, compared to 0.30 for lurasidone) but also had some of the highest costs in the UK. These differences may be due to the estimated cost of relapse, which were about four times greater in the NICE evaluation. As discussed in Appendix 4, the relapse costs used by NICE are much larger than any of the other relapse costs identified, and so may lack face validity. Two other studies were identified which evaluated the long-term impact of cardiometabolic outcomes, both of which included aripiprazole.21,29 In a Swedish study the comparator was olanzapine, in a UK study the comparator was standard of care. Both studies found that the lower rates of cardiometabolic outcomes with aripiprazole led to lower lifetime costs, consistent with the results of this analysis.
To the authors’ knowledge, this is the first study to comprehensively consider multi-country costs for a large number of second-generation antipsychotics. The large number of treatments and outcomes considered is a particular strength of this work. The identification of effectiveness data and costs were also based on systematic searches of the literature, supplemented by targeted literature searches, to ensure that the most relevant evidence was identified. The use of an economic model with validated risk equations allowed for the synthesis of evidence, with extrapolation of longer-term outcomes. Another strength of this study is that, where possible, effectiveness data were obtained from NMAs of multiple antipsychotics, representing high level evidence. Of the identified studies that estimated the costs of any cardiometabolic outcomes, only two included an NMA.18,40
There are limitations with this project. High quality evidence about the short-term impact on triglycerides was available,6 but it was not possible to link this to costs or to differences in long-term outcomes. Of the drugs considered, lurasidone and brexpiprazole were particularly effective at controlling triglyceride levels, so the lifetime costs for these treatments may be under-estimated. There were also some key evidence gaps; including evidence on the long-term effectiveness of lumateperone. Further, as the NMAs did not always include all ten antipsychotics, they were supplemented by trial evidence. This may lead to inconsistent estimates, so future research should seek to incorporate this evidence within NMAs. Whilst treatment-specific differences in the rates of relapse were modelled, treatment-specific differences in the proportions of relapses requiring hospitalisation were not modelled due to an absence of evidence. Future research should investigate the proportions of relapses that lead to hospitalisations, by antipsychotic. When possible, inputs for risk models were for a schizophrenia population, but where this data was lacking it was obtained from the general population. This will affect the absolute accuracy of country-specific estimates, but not the relative outcomes by antipsychotic. In addition, the costs of treating neurological side effects, such as extrapyramidal symptoms and tardive dyskinesia, were not included. Relative to the costs of chronic cardiometabolic disease, including the costs of treating acute neurological side effects is not anticipated to greatly influence the findings of this study. The use of standardised mortality ratios applied to life-table data in addition to the explicit modelling of CVD deaths may lead to some double-counting of deaths. However, this was explored in a sensitivity analyses, which suggests that results are robust to any double-counting. As with any analysis which uses evidence from the published literature, there is the possibility of publication bias.
Clinical guidelines for antipsychotic treatment in schizophrenia emphasise the importance of considering the adverse event profile of drugs.7 This study has estimated the rates of major cardiometabolic outcomes for ten antipsychotics, as well as quantifying the contribution of these adverse events to lifetime treatment costs for seven European countries. The results indicate that the development of diabetes and cardiovascular events are the primary cost drivers, and so the risk of developing these should be a key consideration in the choice of antipsychotic.
To conclude, when making decisions about the choice of antipsychotic for treating schizophrenia, it is important to consider lifetime outcomes. Cardiometabolic outcomes are a key long-term outcome which typically have a bigger impact on total costs than other schizophrenia treatment costs, such as managing relapses. Amongst the ten antipsychotics considered, lurasidone and ziprasidone had the lowest rates of cardiometabolic outcomes and lowest lifetime costs in seven European countries.
Data Sharing Statement
The data used in this manuscript are provided in Appendices 1–5.
Ethics Approval and Informed Consent
Neither ethical approval nor informed consent were relevant to this research.
Acknowledgments
The authors would like to acknowledge the input of Dr Penny Breeze.
Funding
Funding for this work was provided by Angelini.
Disclosure
Benjamin Kearns, Katy Cooper, Anna Cantrell, and Chloe Thomas report grants from Angelini, during the conduct of the study and outside the submitted work. The authors report no other potential conflicts of interest in this work.
References
1. Ryder S, Fox K, Rane P, et al. A systematic review of direct cardiovascular event costs: an international perspective. Pharmacoeconomics. 2019;37(7):895–919. doi:10.1007/s40273-019-00795-4
2. Zhang P, Zhang X, Brown J, et al. Global healthcare expenditure on diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87(3):293–301. doi:10.1016/j.diabres.2010.01.026
3. Bresee LC, Majumdar SR, Patten SB, Johnson JA. Prevalence of cardiovascular risk factors and disease in people with schizophrenia: a population-based study. Schizophr Res. 2010;117(1):75–82. doi:10.1016/j.schres.2009.12.016
4. Correll CU, Solmi M, Veronese N, et al. Prevalence, incidence and mortality from cardiovascular disease in patients with pooled and specific severe mental illness: a large-scale meta-analysis of 3,211,768 patients and 113,383,368 controls. World Psychiatry. 2017;16(2):163–180. doi:10.1002/wps.20420
5. Kelly DL, McMahon RP, Liu F, et al. Cardiovascular disease mortality in chronic schizophrenia patients treated with clozapine. J Clin Psychiatry. 2010;71(3):304. doi:10.4088/JCP.08m04718yel
6. Pillinger T, McCutcheon RA, Vano L, et al. Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: a systematic review and network meta-analysis. Lancet Psychiatry. 2020;7(1):64–77. doi:10.1016/S2215-0366(19)30416-X
7. Keating D, McWilliams S, Schneider I, et al. Pharmacological guidelines for schizophrenia: a systematic review and comparison of recommendations for the first episode. BMJ Open. 2017;7(1):e013881. doi:10.1136/bmjopen-2016-013881
8. Millier A, Zineddine O, Zhou J, Aballea S, Taieb V, Toumi M. A network meta-analysis to compare the efficacy and safety of antipsychotics as maintenance treatment for patients with schizophrenia. Value Health. 2017;20(9):A709. doi:10.1016/j.jval.2017.08.1866
9. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939–951. doi:10.1016/S0140-6736(19)31135-3
10. Mehnert A, Nicholl D, Pudas H, Martin M, McGuire A. Cost effectiveness of paliperidone palmitate versus risperidone long-acting injectable and olanzapine pamoate for the treatment of patients with schizophrenia in Sweden. J Med Econ. 2012;15(5):844–861. doi:10.3111/13696998.2012.681531
11. Geitona M, Kousoulakou H, Ollandezos M, Athanasakis K, Papanicolaou S, Kyriopoulos I. Costs and effects of paliperidone extended release compared with alternative oral antipsychotic agents in patients with schizophrenia in Greece: a cost effectiveness study. Ann Gen Psychiatry. 2008;7:16. doi:10.1186/1744-859X-7-16
12. Davies A, Vardeva K, Loze JY, G J L, Sennfalt K, Baardewijk M. Cost-effectiveness of atypical antipsychotics for the management of schizophrenia in the UK. Curr Med Res Opin. 2008;24(11):3275–3285. doi:10.1185/03007990802507547
13. Treur M, Baca E, Bobes J, et al. The cost-effectiveness of paliperidone extended release in Spain. J Med Econ. 2012;15(Suppl 1):26–34. doi:10.3111/13696998.2012.734884
14. O’Day K, Rajagopalan K, Meyer K, Pikalov A, Loebel A. Long-term cost-effectiveness of atypical antipsychotics in the treatment of adults with schizophrenia in the US. Clinicoecon Outcomes Res. 2013;5:459–470. doi:10.2147/CEOR.S47990
15. Garcia-Ruiz AJ, Perez-Costillas L, Montesinos AC, Alcalde J, Oyaguez I, Casado MA. Cost-effectiveness analysis of antipsychotics in reducing schizophrenia relapses. Health Econ Rev. 2012;2(1):8. doi:10.1186/2191-1991-2-8
16. Citrome L, Kamat SA, Sapin C, et al. Cost-effectiveness of aripiprazole once-monthly compared with paliperidone palmitate once-monthly injectable for the treatment of schizophrenia in the United States. J Med Econ. 2014;17(8):567–576. doi:10.3111/13696998.2014.917089
17. Obradovic M, Mrhar A, Kos M. Cost-effectiveness of antipsychotics for outpatients with chronic schizophrenia. Int J Clin Pract. 2007;61(12):1979–1988. doi:10.1111/j.1742-1241.2007.01431.x
18. Excellence N. Psychosis and schizophrenia in adults: prevention and management. NICE Guidelines. 2014.
19. Lindstrom E, Eberhard J, Fors BM, Hansen K, Sapin C. A pharmacoeconomic analysis of sertindole in the treatment of schizophrenia in Sweden. Nord J Psychiatry. 2011;65(6):403–413. doi:10.3109/08039488.2011.590603
20. Marazuela R, Rubio-Terrés C, Giner J, Jiménez FJ. Pharmacoeconomic analysis of the treatment of schizophrenia with quetiapine, olanzapine, risperidone or haloperidol in Spain. J Med Econ. 2008;9(1–4):101–119. doi:10.3111/200609101119
21. Kasteng F, Eriksson J, Sennfalt K, Lindgren P. Metabolic effects and cost-effectiveness of aripiprazole versus olanzapine in schizophrenia and bipolar disorder. Acta Psychiatr Scand. 2011;124(3):214–225. doi:10.1111/j.1600-0447.2011.01716.x
22. Correll CU, Davis RE, Weingart M, et al. Efficacy and safety of lumateperone for treatment of schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2020;77(4):349. doi:10.1001/jamapsychiatry.2019.4379
23. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952–961. doi:10.1016/j.biopsych.2015.08.026
24. Durgam S, Earley W, Li R, et al. Long-term cariprazine treatment for the prevention of relapse in patients with schizophrenia: a randomized, double-blind, placebo-controlled trial. Schizophr Res. 2016;176(2–3):264–271. doi:10.1016/j.schres.2016.06.030
25. Fleischhacker WW, Hobart M, Ouyang J, et al. Efficacy and safety of brexpiprazole (OPC-34712) as maintenance treatment in adults with schizophrenia: a randomized, double-blind, placebo-controlled study. Int J Neuropsychopharmacol. 2017;20(1):11–21. doi:10.1093/ijnp/pyw076
26. Hippisley-Cox J, Coupland C. Development and validation of QDiabetes-2018 risk prediction algorithm to estimate future risk of type 2 diabetes: cohort study. BMJ. 2017;359:j5019. doi:10.1136/bmj.j5019
27. Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. BMJ. 2017;357:j2099. doi:10.1136/bmj.j2099
28. Zomer E, Osborn D, Nazareth I, et al. Effectiveness and cost-effectiveness of a cardiovascular risk prediction algorithm for people with severe mental illness (PRIMROSE). BMJ Open. 2017;7(9):e018181. doi:10.1136/bmjopen-2017-018181
29. Barnett AH, Millar HL, Loze JY, L’Italien GJ, van Baardewijk M, Knapp M. UK cost-consequence analysis of aripiprazole in schizophrenia: diabetes and coronary heart disease risk projections (STAR study). Eur Arch Psychiatry Clin Neurosci. 2009;259(4):239–247. doi:10.1007/s00406-008-0863-2
30. Nerat T, Kos M. Burden of type 2 diabetes from the healthcare payer perspective in Slovenia/Breme Sladkorne Bolezni Tipa 2 S Stališča Plačnika Zdravstvenega Varstva V Sloveniji. SJPH. 2013;52(3):162–180. doi:10.2478/sjph-2013-0018
31. Reuters. BRIEF-intra-cellular says wholesale acquisition price for caplyta is $1,320/month. 2020. Available from: https://www.reuters.com/article/brief-intra-cellular-says-wholesale-acqu/brief-intra-cellular-says-wholesale-acquisition-price-for-caplyta-is-1320-month-idUSFWN2BG0N6.
32. Siebert U, Alagoz O, Bayoumi AM, et al. State-transition modeling: a report of the ISPOR-SMDM modeling good research practices task force–3. Med Decis Making. 2012;32(5):690–700. doi:10.1177/0272989X12455463
33. Németh B, Bendes R, Nagy B, et al. Cost-utility analysis of cariprazine compared to risperidone among patients with negative symptoms of schizophrenia. Health Policy Technol. 2019;8(1):84–91. doi:10.1016/j.hlpt.2019.01.004
34. Rajagopalan K, Trueman D, Crowe L, Squirrell D, Loebel A. Cost-utility analysis of lurasidone versus aripiprazole in adults with schizophrenia. Pharmacoeconomics. 2016;34(7):709–721. doi:10.1007/s40273-016-0405-0
35. Saha S, Chant D, McGrath J. A systematic review of mortality in schizophrenia: is the differential mortality gap worsening over time? Arch Gen Psychiatry. 2007;64(10):1123–1131. doi:10.1001/archpsyc.64.10.1123
36. Hong J, Windmeijer F, Novick D, Haro JM, Brown J. The cost of relapse in patients with schizophrenia in the European SOHO (Schizophrenia Outpatient Health Outcomes) study. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(5):835–841. doi:10.1016/j.pnpbp.2009.03.034
37. Arteaga Duarte C, Colin X, De Moor R, Guillon P, van Gils CW. Three-monthly long-acting formulation of paliperidone palmitate is a dominant treatment option, cost saving while adding qalys, compared to the one-monthly formulation in the treatment of schizophrenia in France. Value Health. 2016;19(7):A378. doi:10.1016/j.jval.2016.09.181
38. Colombo GL, Caruggi M, Di Matteo S, Rossi A. An economic evaluation of aripiprazole vs olanzapine adapted to the Italian setting using outcomes of metabolic syndrome and risk for diabetes in patients with schizophrenia. Neuropsychiatr Dis Treat. 2008;4(5):967. doi:10.2147/NDT.S3701
39. Eurostat. Cardiovascular diseases statistics. Published 2020. Available from: https://ec.europa.eu/eurostat/statistics-explained/index.php/Cardiovascular_diseases_statistics.
40. Druais S, Doutriaux A, Cognet M, et al. Cost effectiveness of paliperidone long-acting injectable versus other antipsychotics for the maintenance treatment of schizophrenia in France. Pharmacoeconomics. 2016;34(4):363–391. doi:10.1007/s40273-015-0348-x
41. Zhao YJ, Lin L, Teng M, et al. Long-term antipsychotic treatment in schizophrenia: systematic review and network meta-analysis of randomised controlled trials. BJPsych Open. 2016;2(1):59–66. doi:10.1192/bjpo.bp.115.002576
42. Zhang Y, Liu Y, Su Y, et al. The metabolic side effects of 12 antipsychotic drugs used for the treatment of schizophrenia on glucose: a network meta-analysis. BMC Psychiatry. 2017;17(1):373. doi:10.1186/s12888-017-1539-0
43. Kishimoto T, Hagi K, Nitta M, Kane JM, Correll CU. Long-term effectiveness of oral second-generation antipsychotics in patients with schizophrenia and related disorders: a systematic review and meta-analysis of direct head-to-head comparisons. World Psychiatry. 2019;18(2):208–224. doi:10.1002/wps.20632
44. Kishi T, Ikuta T, Matsuda Y, Sakuma K, Iwata N. Aripiprazole vs. brexpiprazole for acute schizophrenia: a systematic review and network meta-analysis. Psychopharmacology. 2020;30:30.
45. Kishi T, Ikuta T, Matsunaga S, Matsuda Y, Oya K, Iwata N. Comparative efficacy and safety of antipsychotics in the treatment of schizophrenia: a network meta-analysis in a Japanese population. Neuropsychiatr Dis Treat. 2017;13:1281–1302. doi:10.2147/NDT.S134340
46. Maman K, Krause T, Pedersen AM, et al. Pmh4 an indirect treatment comparison of brexpiprazole versus lurasidone in patients undergoing acute and maintenance phase treatment for schizophrenia. Value Health. 2019;22(Supplement 2):S226. doi:10.1016/j.jval.2019.04.1050
47. Zhu Y, Krause M, Huhn M, et al. Antipsychotic drugs for the acute treatment of patients with a first episode of schizophrenia: a systematic review with pairwise and network meta-analyses. Lancet Psychiatry. 2017;4(9):694–705. doi:10.1016/S2215-0366(17)30270-5
48. Ng-Mak D, Tongbram V, Ndirangu K, Rajagopalan K, Loebel A. Efficacy and metabolic effects of lurasidone versus brexpiprazole in schizophrenia: a network meta-analysis. J Comp Eff Res. 2018;7(8):737–748. doi:10.2217/cer-2018-0016
49. Kishi T, Oya K, Matsui Y, et al. Comparison of the efficacy and safety of 4 and 2 mg/day brexpiprazole for acute schizophrenia: a meta-analysis of double-blind, randomized placebo-controlled trials. Neuropsychiatr Dis Treat. 2018;14:2519–2530. doi:10.2147/NDT.S176676
50. Leucht S, Leucht C, Huhn M, et al. Sixty years of placebo-controlled antipsychotic drug trials in acute schizophrenia: systematic review, bayesian meta-analysis, and meta-regression of efficacy predictors. Am J Psychiatry. 2017;174(10):927–942. doi:10.1176/appi.ajp.2017.16121358
51. Zhao MJ, Qin B, Wang JB, et al. Efficacy and acceptability of cariprazine in acute exacerbation of schizophrenia: meta-analysis of randomized placebo-controlled trials. J Clin Psychopharmacol. 2018;38(1):55–59. doi:10.1097/JCP.0000000000000834
52. Zheng W, Cai DB, Yang XH, et al. Short-term efficacy and tolerability of lurasidone in the treatment of acute schizophrenia: a meta-analysis of randomized controlled trials. J Psychiatr Res. 2018;103:244–251. doi:10.1016/j.jpsychires.2018.06.005
53. Asmal L, Flegar SJ, Wang J, Rummel-Kluge C, Komossa K, Leucht S. Quetiapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2013;11:CD006625.
54. Hutton P, Taylor PJ, Mulligan L, Tully S, Moncrieff J. Quetiapine immediate release v. placebo for schizophrenia: systematic review, meta-analysis and reappraisal. Br J Psychiatry. 2015;206(5):360–370. doi:10.1192/bjp.bp.114.154377
55. Marder SR, Hakala MJ, Josiassen MK, et al. Brexpiprazole in patients with schizophrenia: overview of short-and long-Term Phase 3 controlled studies. Acta Neuropsychiatr. 2017;29(5):278–290. doi:10.1017/neu.2016.57
56. Rattehalli RD, Zhao S, Li BG, Jayaram MB, Xia J, Sampson S. Risperidone versus placebo for schizophrenia. Cochrane Database Syst Rev. 2016;12:CD006918.
57. Nussbaum A, Stroup TS. Paliperidone for schizophrenia. Cochrane Database Syst Rev. 2008;(2):CD006369.
58. Samara MT, Dold M, Gianatsi M, et al. Efficacy, acceptability, and tolerability of antipsychotics in treatment-resistant schizophrenia: a network meta-analysis. JAMA Psychiatry. 2016;73(3):199–210. doi:10.1001/jamapsychiatry.2015.2955
59. Cortesi PA, Mencacci C, Luigi F, et al. Compliance, persistence, costs and quality of life in young patients treated with antipsychotic drugs: results from the COMETA study. BMC Psychiatry. 2013;13(1):98. doi:10.1186/1471-244X-13-98
60. Fefeu M, De Maricourt P, Cachia A, et al. One-year mirror-image study of the impact of olanzapine long-acting injection on healthcare resource utilization and costs in severe schizophrenia. Psychiatry Res. 2018;270:205–210. doi:10.1016/j.psychres.2018.09.041
61. Hodgson RE. Evaluating the cost and clinical effectiveness of long-acting, injectable aripiprazole and paliperidone palmitate once a month in a real-world setting. Clinicoecon Outcomes Res. 2019;11:517–524. doi:10.2147/CEOR.S191198
62. Dernovsek M, Rupel VP, Rebolj M, Tavcar R. Quality of life and treatment costs in schizophrenic outpatients, treated with depot neuroleptics. Eur Psychiatry. 2001;16(8):474–482. doi:10.1016/S0924-9338(01)00609-5
63. Sicras-Mainar A, Maurino J, Ruiz-Beato E, Navarro-Artieda R. Impact of negative symptoms on healthcare resource utilization and associated costs in adult outpatients with schizophrenia: a population-based study. BMC Psychiatry. 2014;14(1):225. doi:10.1186/s12888-014-0225-8
64. Exchange GHD. GBD results tool. Institute for Health Metrics and Evaluation Seattle; 2019. Available from: http://ghdx.healthdata.org/gbd-results-tool.
65. OECD. Health risks - Daily smokers - OECD data. 2020. Available from: https://data.oecd.org/healthrisk/daily-smokers.htm.
66. Bank W. GDP deflator. 2018. Available from: https://data.worldbank.org/indicator/NY.GDP.DEFL.ZS.
67. OECD. Purchasing power parities. 2020. Available from: https://data.oecd.org/conversion/purchasing-power-parities-ppp.htm.
68. de Arce Cordón R, Eding E, Marques-Teixeira J, Milanova V, Rancans E, Schreiner A. Descriptive analyses of the aripiprazole arm in the risperidone long-acting injectable versus quetiapine relapse prevention trial (ConstaTRE). Eur Arch Psychiatry Clin Neurosci. 2012;262(2):139–149. doi:10.1007/s00406-011-0220-8
69. Gaebel W, Schreiner A, Bergmans P, et al. Relapse prevention in schizophrenia and schizoaffective disorder with risperidone long-acting injectable vs quetiapine: results of a long-term, open-label, randomized clinical trial. Neuropsychopharmacology. 2010;35(12):2367–2377. doi:10.1038/npp.2010.111
70. Addington DE, Labelle A, Kulkarni J, Johnson G, Loebel A, Mandel FS. A comparison of ziprasidone and risperidone in the long-term treatment of schizophrenia: a 44-week, double-blind, continuation study. Can J Psychiatry. 2009;54(1):46–54. doi:10.1177/070674370905400108
71. Meyer JM, Mao Y, Pikalov A, Cucchiaro J, Loebel A. Weight change during long-term treatment with lurasidone: pooled analysis of studies in patients with schizophrenia. Int Clin Psychopharmacol. 2015;30(6):342. doi:10.1097/YIC.0000000000000091
72. Leal J, Luengo-Fernandez R, Gray A, Petersen S, Rayner M. Economic burden of cardiovascular diseases in the enlarged European Union. Eur Heart J. 2006;27(13):1610–1619. doi:10.1093/eurheartj/ehi733
73. Ray JA, Valentine WJ, Secnik K, et al. Review of the cost of diabetes complications in Australia, Canada, France, Germany, Italy and Spain. Curr Med Res Opin. 2005;21(10):1617–1629. doi:10.1185/030079905X65349
74. Ekman M. Economic evidence in stroke: a review. Eur J Health Econ. 2004;5:S74. doi:10.1007/s10198-005-0292-3
75. Truelsen T, Ekman M, Boysen G. Cost of stroke in Europe. Eur J Neurol. 2005;12:78–84. doi:10.1111/j.1468-1331.2005.01199.x
76. Lamotte M, Annemans L, Evers T, Kubin M. A multi-country economic evaluation of low-dose aspirin in the primary prevention of cardiovascular disease. Pharmacoeconomics. 2006;24(2):155–169. doi:10.2165/00019053-200624020-00005
77. Brodszky V, Beretzky Z, Baji P, et al. Cost-of-illness studies in nine Central and Eastern European countries. Eur J Health Econ. 2019;20(Suppl 1):155–172. doi:10.1007/s10198-019-01066-x
78. Terent A, Marke L-A, Asplund K, Norrving B, Jonsson E, Wester P-O. Costs of stroke in Sweden. A national perspective. Stroke. 1994;25(12):2363–2369. doi:10.1161/01.STR.25.12.2363
79. Kanavos P, van den Aardweg S, Schurer W. Diabetes expenditure, burden of disease and management in 5 EU countries. LSE Health Social Care. 2012.
80. Jönsson B. Revealing the cost of type II diabetes in Europe. Diabetologia. 2002;45(1):S5–S12. doi:10.1007/s00125-002-0858-x
81. Vera-Llonch M, Delea TE, Richardson E, Rupnow M, Grogg A, Oster G. Outcomes and costs of risperidone versus olanzapine in patients with chronic schizophrenia or schizoaffective disorders: a Markov model. Value Health. 2004;7(5):569–584. doi:10.1111/j.1524-4733.2004.75008.x
82. Furiak NM, Ascher-Svanum H, Klein RW, et al. Cost-effectiveness model comparing olanzapine and other oral atypical antipsychotics in the treatment of schizophrenia in the United States. Cost Eff Resour Alloc. 2009;7:4. doi:10.1186/1478-7547-7-4
83. Oxford Health NHS Foundation Trust. Antipsychotic-induced hyperprolactinaemia - Trust guideline for identification, monitoring and management. 2015. Available from: http://www.oxfordhealthformulary.nhs.uk/docs/AntipsychoticinducedhyperprolactinaemiaguidelineJuly2015.pdf.
84. Grigg J, Worsley R, Thew C, Gurvich C, Thomas N, Kulkarni J. Antipsychotic-induced hyperprolactinemia: synthesis of world-wide guidelines and integrated recommendations for assessment, management and future research. Psychopharmacology. 2017;234(22):3279–3297. doi:10.1007/s00213-017-4730-6
85. Dilla T, O’Donohoe P, Moller J, et al. Cost-effectiveness of long-acting olanzapine versus long-acting risperidone in patients with schizophrenia in Spain. Value Health. 2011;14(7):A292. doi:10.1016/j.jval.2011.08.331
86. Almond S, Knapp M, Francois C, Toumi M, Brugha T. Relapse in schizophrenia: costs, clinical outcomes and quality of life. Br J Psychiatry. 2004;184(4):346–351. doi:10.1192/bjp.184.4.346
87. Bagnall AM, Jones L, Ginnelly L, et al. A systematic review of atypical antipsychotic drugs in schizophrenia. Health Technol Assess. 2003;7(13). doi:10.3310/hta7130.
88. Tilden D, Aristides M, Meddis D, Burns T. An economic assessment of quetiapine and haloperidol in patients with schizophrenia only partially responsive to conventional antipsychotics. Clin Ther. 2002;24(10):1648–1667. doi:10.1016/S0149-2918(02)80069-8
89. Leucht S, Tardy M, Komossa K, et al. Antipsychotic drugs versus placebo for relapse prevention in schizophrenia: a systematic review and meta-analysis. Lancet. 2012;379(9831):2063–2071. doi:10.1016/S0140-6736(12)60239-6
90. World Health Organisation. ATC/DDD Index 2020. 2020. Available from: https://www.whocc.no/atc_ddd_index/.
91. Committee JF. British national formulary (BNF) 66: aripiprazole. Vol 66. Pharmaceutical Press; 2013. Available from: https://bnf.nice.org.uk/drug/aripiprazole.html.
92. Moher D, Liberati A, Tetzlaff J, Altman DG, Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS med. 2009. Jul 21;6(7):e1000097.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
