Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Respiratory Mechanics and Diaphragmatic Dysfunction in COPD Patients Who Failed Non-Invasive Mechanical Ventilation
Authors Marchioni A, Tonelli R , Fantini R, Tabbì L, Castaniere I, Livrieri F, Bedogni S , Ruggieri V, Pisani L, Nava S, Clini E
Received 11 June 2019
Accepted for publication 23 September 2019
Published 22 November 2019 Volume 2019:14 Pages 2575—2585
DOI https://doi.org/10.2147/COPD.S219125
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Alessandro Marchioni,1 Roberto Tonelli,1,2 Riccardo Fantini,1 Luca Tabbì,1 Ivana Castaniere,1,2 Francesco Livrieri,1,3 Sabrina Bedogni,4 Valentina Ruggieri,1 Lara Pisani,5 Stefano Nava,5 Enrico Clini1
1University Hospital of Modena, Pneumology Unit and Center for Rare Lung Diseases, Department of Medical and Surgical Sciences, University of Modena and Reggio Emilia, Modena, Italy; 2PhD Course in Clinical and Experimental Medicine, University of Modena and Reggio Emilia, Modena, Italy; 3Respiratory Disease Unit, Hospital Carlo Poma, Mantova, Italy; 4School of Medicine, University of Modena and Reggio Emilia, Modena, Italy; 5Department of Specialistic, Diagnostic and Experimental Medicine (DIMES), University of Bologna, Bologna, Italy
Correspondence: Roberto Tonelli
University Hospital of Modena, Pneumology Unit and Center for Rare Lung Diseases, Department of Medical and Surgical Sciences, University of Modena and Reggio Emilia, Via del Pozzo, 71 – 41125, Modena, Italy
Tel +39-342-7241672
Email [email protected] Skype roberto.tonelli150288
Background: Although non-invasive mechanical ventilation (NIV) is the gold standard treatment for patients with acute exacerbation of COPD (AECOPD) developing respiratory acidosis, failure rates still range from 5% to 40%. Recent studies have shown that the onset of severe diaphragmatic dysfunction (DD) during AECOPD increases risk of NIV failure and mortality in this subset of patients. Although the imbalance between the load and the contractile capacity of inspiratory muscles seems the main cause of AECOPD-induced hypercapnic respiratory failure, data regarding the influence of mechanical derangement on DD in this acute phase are lacking. With this study, we investigate the impact of respiratory mechanics on diaphragm function in AECOPD patients experiencing NIV failure.
Methods: Twelve AECOPD patients with respiratory acidosis admitted to the Respiratory ICU of the University Hospital of Modena from 2017 to 2018 undergoing mechanical ventilation (MV) due to NIV failure were enrolled. Static respiratory mechanics and end-expiratory lung volume (EELV) were measured after 30 mins of volume control mode MV. Subsequently, transdiaphragmatic pressure (Pdi) was calculated by means of a sniff maneuver (Pdisniff) after 30 mins of spontaneous breathing trial. Linear regression analysis and Pearson’s correlation coefficient served to assess associations.
Results: Average Pdisniff was 23.3 cmH2O (standard deviation 29 cmH2O) with 3 patients presenting bilateral diaphragm palsy. Pdisniff was directly correlated with static lung elastance (r=0.69, p=0.001) while inverse correlation was found with dynamic intrinsic PEEP (r=−0.73, p=0.007). No significant correlation was found with static intrinsic PEEP (r=−0.55, p=0.06), EELV (r=−0.4, p=0.3), airway resistance (r=−0.2, p=0.54), chest wall, and total elastance (r=−0-01, p=0.96 and r=0.3, p=0.36, respectively). Significant linear inverse correlation was found between Pdisniff and the ratio between Pdi assessed at tidal volume and Pdi sniff (r=−0.82, p=0.02).
Conclusion: The causes of extreme DD in AECOPD patients who experienced NIV failure might be predominantly mechanical, driven by a severe dynamic hyperinflation that overlaps on an elastic lung substrate favoring volume overload.
Keywords: acute exacerbation of COPD, non-invasive mechanical ventilation, respiratory failure, transdiaphragmatic pressure
Introduction
Non-invasive mechanical ventilation (NIV) is the gold standard for the treatment of patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD) with respiratory acidosis.1 In this population, a considerable amount of data show that NIV is able to reduce mortality and the need for tracheal intubation compared to standard medical therapy, although the failure rate ranges from 5% to 40% depending on the severity of respiratory failure.2 Recent studies have shown that, among patients with severe AECOPD admitted to an intensive care unit (ICU), 32% have severe bilateral diaphragmatic dysfunction, which exposes them to an increased risk of NIV failure and an almost fivefold higher risk of dying.3,4 Although, during AECOPD, the imbalance between the load and the contractile capacity of inspiratory muscles is the main cause of hypercapnic respiratory failure, data related to the maximal diaphragmatic force developed in the acute phase are sparse.5–7 Some studies have investigated the pressure-generating capacity of respiratory muscles during AECOPD requiring NIV, demonstrating a reduction in the force generated by the diaphragm with respect to the maximal contractile capacity developed under conditions of clinical stability.8,9 The reasons underlying the reduction in diaphragm strength during exacerbation are not fully understood, although several factors such as hyperinflation, systemic inflammation, septic shock and sepsis, alterations in the biochemical environment (hypercapnia, acute respiratory acidosis, hypoxia), and pharmacological factors (use of steroids) can contribute to this result in various ways.10,11 Although it is known that lung volume influences the contractile force of the diaphragm, studies comparing the maximal force generated by the diaphragm with lung volume in the acute phase of AECOPD are lacking.
Using a twitch tracheal pressure in response to magnetic phrenic nerve stimulation (Ptrstim), Demoule et al have shown that 64% of the patients admitted to ICU already have diaphragmatic dysfunction at the onset of the critical illness, with sepsis as a major independent risk factor.12 Therefore, since the pathophysiology underlying AECOPD is extremely complex, the degree of diaphragm dysfunction and the mechanisms involved may be different from the “general” ICU population. The main physiological factor related to a reduction in diaphragm muscle strength during AECOPD is still thought to be an altered force–volume relationship, due to the phenomenon of hyperinflation, despite the fact that, to our knowledge, no patients have so far demonstrated this in vivo.
In the present study, we wanted to investigate this issue in chronic obstructive pulmonary disease (COPD) patients who had failed an NIV trial and therefore were intubated, monitoring both transdiaphragmatic pressure during a sniff maneuver and static and dynamic respiratory mechanics, in order to verify a possible correlation.
Materials and Methods
This prospective physiological study was carried out in a single respiratory intensive care unit (RICU) at the University Hospital of Modena (Italy) over a 18-month period (from January 2017 to June 2018). The study was conducted according to the guidelines of the Declaration of Helsinki. Approval from the local ethics committee of Modena was obtained (registered protocol number 839/C.E.). Written informed consent was obtained from each patient before enrollment in the study. (NCT clinicaltrials.gov NCT03852394).
Patients
The eligibility criteria for the study were as follows: age over 18 years, AECOPD with respiratory acidosis admitted to the RICU undergoing endotracheal intubation due to NIV failure. Exclusion criteria were any of the following: history of neuromuscular disease, presence of chest wall deformities, coexistence of interstitial lung disease, presence of pulmonary edema, severe hemodynamic instability, septic shock, evidence of lobar pneumonia or bilateral parenchymal consolidation at chest X-ray on admission, contraindication to NIV, previously assessed diaphragmatic palsy, intracranial hypertension, known pregnancy, need for immediate endotracheal intubation, neurologic impairment, lack of collaboration, unreliable Pdisniff maneuver to calculate maximal transdiaphragmatic pressure. Patients with sublobar consolidation and signs of infection were classified as septic according to the most recent consensus document.13
From January 2017 to June 2018, 130 patients with AECOPD were admitted to the RICU for respiratory acidosis requiring NIV. Forty-eight patients were considered non-eligible due to the presence of exclusion criteria (Figure 1). Out of 82 patients with AECOPD, 30 were eligible as they experienced NIV failure. They were intubated and subjected to invasive ventilation. Propofol (2–3 mg/kg/hr) was the only medication allowed for sedation of eligible patients. After 24 hrs of invasive mechanical ventilation, sedation was suspended once measurements of respiratory mechanical proprieties had been performed. Enrollment criteria for patients after suspension of sedation were established as follows: presence of stable clinical conditions (no need for amine infusion, absence of respiratory acidosis or severe hypercapnia [pCO2 < 60 mmHg], absence of major arrhythmias), absence of neurological and cognitive deficits, Glasgow Coma Scale of 15, ability to tolerate a spontaneous breathing trial with T-tube for 30 mins, valid collaboration, Pdisniff deemed reliable after the execution of three maximal maneuvers. Eighteen patients were excluded from the study due to the impossibility of performing a reliable Pdisniff maneuver (Figure 1), while 12 patients were included in the study and underwent the physiological analyses required by the protocol.
 |
Figure 1 Flow chart for study population. |
Protocol
On admission, clinical severity was assessed with the Glasgow Coma Scale (GCS), Kelly score, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, the Simplified Acute Physiology Score (SAPS II) score, and the Subsequent Organ Failure Assessment (SOFA) score. Arterial blood gases (PaO2–PaCO2), pH, PaO2/FiO2 ratio, and blood lactate levels were recorded before NIV and within 2 hrs after NIV initiation. The presence of pneumonia,13 previous treatment with systemic steroids, and relevant comorbidities were also recorded. The diagnosis of COPD was confirmed by clinical history and previous pulmonary function tests.
The baseline ventilatory settings were established by the attending physician according to standard criteria: tidal volume 6–8 mg/kg of ideal body weight, respiratory rate 12–15 breaths/min, positive end-expiratory pressure (PEEP) 6–8 cmH2O. In all patients, a multifunctional nasogastric tube equipped with an esophageal and gastric balloon was placed (NutriVent nasogastric polyfunctional catheter; SIDAM, Mirandola, Italy), which was subsequently connected to a pressure transducer (OptiVent monitor; SIDAM) to allow the monitoring of esophageal (Pes) and gastric pressure (Pga). An occlusion test was performed to assess the validity of Pes measurements.14 The study protocol consisted of two consecutive phases. Phase I was performed within 24 hrs of mechanical ventilation in volume control mode with constant inspiratory flow. In this first phase, measurements of static respiratory mechanics were performed after 30 mins of zero end-expiratory pressure (ZEEP). Phase II was performed after stopping sedation during a spontaneous breathing trial with T-tube, once the neurological, cognitive, and clinical conditions of the patient had been judged compatible with the inclusion criteria (see above). In this phase, the physiological measurements were performed 30 mins after initiation of the spontaneous breathing trial.15
Physiologic Measurements
Flow (V’) was measured with a heated pneumotachograph (Fleish2, Fleisch, Lausanne, Switzerland) placed between the Y-piece of the ventilator and the artificial airway. Volume was measured by integration of the flow. Airway pressure (Paw) was measured proximally to the endotracheal tube. The validity of the Pes and Pga measurements was assessed with the methods described earlier. All traces were sampled at 100 Hz and processed on a dedicated data acquisition system (Colligo; Elekton, Milan, Italy). End-expiratory lung volume (EELV) measurement was based on the nitrogen washout/washin technique through dedicated software (FRC Inview, GE Healthcare). The principle of this technique is as follows: The volume of lung gas includes a volume of nitrogen (V(1)N2) that is determined by the alveolar fraction of nitrogen (FAN2(1)) and by the EELV:
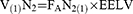
The alveolar fraction of nitrogen can change by changing the administered FiO2 (FAN2(2)), resulting in a new volume of nitrogen (V(2)N2) in the lung after a period of balance.
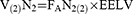
Assuming that, after the change of FiO2, the EELV does not change until a new balance of the alveolar gas is reached, the following equation can be written:
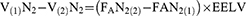
As the changes in FAN2 mirror changes in FiO2, the EELV can be calculated as:
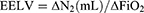
where ΔN2 is equal to the exhaled nitrogen after the change in FiO2 once equilibrium has been reached (20 breaths). The algorithm used by the Engstrom Carestation (FRC Inview, GE Healthcare) employs tidal concentration of oxygen and carbon dioxide to obtain an estimate of nitrogen concentration in expired and inspired air. For more details, see Ref. 16. Static respiratory mechanics and airway resistances were calculated according to standard formulae.17,18 In phase I of the study protocol, measurements of static respiratory mechanics were performed using the end-inspiratory occlusion (EIO) technique during constant flow inflation17 and the end-expiratory occlusion (EEO) method.18 All patients were studied in the semi-recumbent position and measurements were performed after 30 mins of mechanical ventilation with 0 cmH2O of end-expiratory pressure (ZEEP).
In phase II of the study, transdiaphragmatic pressure (Pdi) was calculated after a tidal inspiration. The maximal transdiaphragmatic pressure was calculated by a sniff maneuver (Pdisniff), and the best of three efforts was considered for data analysis.19 Dynamic intrinsic positive end-expiratory pressure (PEEPidyn) was estimated from the decrease in pleural pressure (Ppl) preceding the start of the inspiratory flow and was corrected for the activity of the expiratory muscles by subtracting the negative deflection of the abdominal pressure (Pab).6 For more details about physiological measurements, see Supplemental Materials.
Statistical Analysis
The statistical package GraphPad Prism 7.0 (GraphPad Software, Inc., La Jolla, CA, USA) was used for analysis. The study population has been characterized through descriptive statistics and data are presented as mean and interquartile ranges for continuous variables, and as numbers and percentages for dichotomous variables. The association between Pdisniff values and static and dynamic mechanical features was assessed through linear regression analysis and Pearson’s correlation coefficient. The association between Pdisniff and Pdi/Pdisniff was assessed through linear regression analysis and Pearson’s correlation coefficient. A p value lower than 0.05 was considered significant.
Results
Demographics and relevant clinical features of the patients on admission are shown in Table 1. All patients were classified as COPD GOLD D20 according to clinical and functional data.
 |
Table 1 Demographics and Relevant Clinical Features of the Patients On Admission |
Table 2 shows the physiological characteristics of each of the enrolled patients. Of note, the average Pdisniff value was 2.3 (standard deviation [SD]=29) cmH2O and three patients presented bilateral diaphragm palsy. The average EELV measured during controlled mechanical ventilation and static intrinsic PEEP was 2900 (SD=975) mL and 8.8 (SD=5.7) cmH2O, respectively.
 |
Table 2 Physiological Characteristics of Enrolled Patients |
Dynamic intrinsic PEEP was considerably higher with values ranging from 1 to 22 cmH2O. Analysis of the mechanical properties of the respiratory system showed low levels of lung elastance (average value 6.7 [SD=2.9] cmH2O/L) and high values of airway resistance (average value 33 [SD=10.2] cmH2O/L*sec).
Linear regression analysis showed a high inverse correlation between maximal diaphragmatic strength and dynamic intrinsic PEEP (r=–0.73, p=0.007) (Figure 2, panel A) while no significant correlation was found with static indices of hyperinflation (static intrinsic PEEP r=–0.55, p=0.06, EELV r=–0.4, p=0.3, respectively) and airway resistance (r=–0.2, p=0.54) (Figure 3, panel A and D). A high direct correlation was found between Pdisniff values and static lung elastance (r=0.69, p=0.01) (Figure 2, panel B) while no correlation was demonstrated between Pdisniff values and either total elastance or chest wall elastance (r=0.3, p=0.36 and r=−0.01, p=0.96, respectively) (Figure 3, panel B and C). A significant linear inverse correlation was found between Pdi/Pdisniff and Pdisniff (r=–0.88, p=0.001, Figure 2, panel C).
Discussion
With this prospective physiological study, we have shown that COPD patients who fail NIV have a marked diaphragmatic dysfunction that can even lead to muscle paralysis. The maximal contraction force generated by the diaphragm is mainly influenced by mechanical factors, while metabolic- and infection-related alterations are probably less relevant in this subset of patients. In particular, the linear correlation between static elastance and maximum contraction force of the diaphragm suggests that high elasticity of the lung at baseline constitutes a susceptibility factor to the development of diaphragmatic dysfunction during AECOPD. Moreover, the inverse correlation between PEEPidyn and Pdisniff (Figure 2A) suggests that dynamic hyperinflation is the main physiological mechanism which, acting on a predisposing mechanical substrate, determines diaphragmatic dysfunction during spontaneous breathing in severe AECOPD requiring NIV. Therefore, the results of this study allow us to speculate the existence of a physiological phenotype of COPD prone to NIV failure, which is characterized by high elastic properties of the lung at baseline that make it susceptible to a higher volume overload during dynamic hyperinflation.
Maximal Transdiaphragmatic Pressure in AECOPD Patients Who Experience NIV Failure
There are only a few studies evaluating the maximum contraction force generated by the diaphragm during AECOPD. In a recent study, Ceriana et al evaluated changes in respiratory mechanics and diaphragmatic contractile force in a cohort of COPD patients during severe exacerbation requiring NIV and during the “recovery” period.8 The maximal contractile capacity of the diaphragm, measured with the Pdisniff technique, was significantly reduced in the AECOPD period compared to the measurements performed during clinical stability. In this study, the mean Pdisniff during AECOPD treated with NIV was 43 cmH2O, a value that is about twice that which we recorded in our series. In another study, Purro et al showed an increase in load associated with respiratory muscle weakness during the early phase of AECOPD resulting in a load/capacity imbalance.9 Nine patients required ventilator support because of acute respiratory acidosis due to AECOPD (NPPV group), while seven patients were successfully managed with medical therapy alone (SB group). In that study, the data collected showed that the maximal transdiaphragmatic pressure (Pdimax) generated was significantly lower in patients with more severe AECOPD requiring ventilatory support (NPPV group, Pdimax: 40.1 cmH2O) than in patients treated with medical therapy alone (SB group, Pdimax: 68.9 cmH2O). In our series, Pdisniff was significantly lower than in patients enrolled in other studies evaluating the AECOPD phase. These data indicate that AECOPD patients who experience NIV failure may have more severe diaphragmatic impairment, which might differentiate them from patients with AECOPD successfully managed with NIV. In support of this hypothesis is the finding of three patients with bilateral diaphragmatic paralysis in our series.
Finally, the linear correlation between Pdisniff and Pdi/Pdisniff suggests that muscle dysfunction might be considered to be a critical factor affecting the load/capacity imbalance.
EELV and Maximal Transdiaphragmatic Pressure
As already known, the contractile capacity of the diaphragm is closely related to the length–tension relationship of the muscle. Some experimental studies have shown that the active tension developed by the muscle during contraction is a function of the rest length of the muscle before stimulation.21–23 Being the length of the diaphragm influenced by lung volume, lung hyperinflation is one of the main determinants of the ability of the muscle to generate pressure.24–26 Moreover, several studies have shown how the flattening and shortening of the diaphragm can result in a structural modification of muscle fibers after a variable time. Indeed, the mechanical disadvantage of the diaphragm produced by hyperinflation determines the implementation of adaptation mechanisms designed to restore the optimal muscle length.27 This structural alteration is an adaptive mechanism that partly allows conservation of the diaphragm’s ability to generate force.28 More recently, Lindqvist et al have shown that mechanical ventilation with PEEP determines similar adaptive mechanisms by reducing the number of sarcomeres in series in whole-length diaphragm fibers, suggesting that this mechanism could contribute to the rapid development of diaphragm weakness in critically ill patients.29,30
Since lung volume is the “critical factor” for diaphragm contractile capacity, in our series, we have analyzed EELV and correlated it with patient-generated Pdisniff. The mean measured EELV was 2900 mL (Table 2), which is quite similar to previous assessments that were performed in heterogeneous populations undergoing invasive mechanical ventilation.31,32 The EELV measured in our COPD population might seem to be not as high as one would expect, but we must consider that most of the studies that analyzed the EELV used different methodologies and the measurements were performed after PEEP application.16,33 In our protocol, the measurement of EELV was performed at ZEEP during controlled mechanical ventilation, so the lung volume obtained may not be comparable with that measured in other studies. Furthermore, the results of our study do not show a significant correlation between baseline EELV and maximal contractile capacity of the diaphragm measured by the sniff maneuver. This result can be explained by various considerations. First, the measured lung volume may be underestimated and may not accurately represent static hyperinflation, due to various factors: (1) the presence of derecruitment phenomena of the lung-dependent areas during controlled ventilation and ZEEP, (2) reduction of about 25% of the EELV in the semi-supine position with respect to the volume that can be measured in orthostatism.34 Second, the ventilatory strategy and the pharmacological approach might be able to empty the lung. For this reason, the lung volume measured during controlled ventilation might represent static hyperinflation, but it does not correspond to the lung volume achieved during the spontaneous breathing trial, which is subject to dynamic hyperinflation. The difficulty in obtaining reliable values of EELV during the spontaneous breathing trial limits the conclusions of our study, but it is likely that the volume achieved during dynamic hyperinflation is the key factor influencing diaphragmatic contractility for at least two reasons: (1) volume overload during dynamic hyperinflation could reach an area very close to total lung capacity (TLC) with severe shortening of diaphragmatic fibers; (2) acute volume overload does not allow the establishment of the compensating molecular phenomena that serve to maintain the contractile capacity of the muscle.
Respiratory Mechanics and Contractile Capacity of the Diaphragm
One of the study’s most important findings is the significant linear correlation between lung static elastance and Pdisniff. These data may not seem easy to interpret, and this therefore requires some consideration. During AECOPD, dynamic hyperinflation shifts the patient’s tidal volume to an area of the pressure–volume curve where the lung is less distensible, resulting in an increase in dynamic lung elastance and elastic load.35 Along with the increased resistive load, this can contribute to determine the imbalance between respiratory muscle effort and load that occurs when hypercapnic respiratory failure develops. Measurement of static lung elastance during controlled mechanical ventilation does not correspond to dynamic lung elastance that the patient develops in spontaneous breathing during AECOPD, but it describes the lung elastic condition at baseline. This consideration has some relevance; indeed, it can be deduced that the lung static elastance can be considered to be the measure of how much the lung is susceptible to volume overload during dynamic hyperinflation. Patients with very low lung elastance at baseline will develop a noticeable increase in end-expiratory volume once PEEPdyn is applied. Considerable volume overload will result in a rapid significant shortening of diaphragmatic fibers with the consequences already discussed on muscle strength. The static lung elastance measured in our series is lower than that measured in other studies that evaluated small COPD populations. Ranieri et al performed measurements of static respiratory mechanics at various PEEP levels (from ZEEP to 15 cmH2O) in 7 COPD patients undergoing mechanical ventilation.36 The mean lung elastance at ZEEP was 9.8 cmH2O/L, significantly higher than that detected in our patients (6.7 cmH2O/L). A subsequent study compared the static respiratory mechanics of 8 mechanically ventilated AECOPD patients with 9 patients without pulmonary disease who underwent mechanical ventilation for surgical reasons.37 The mean lung static elastance in COPD patients was 9.2 cmH2O/L and did not differ significantly from that measured in surgical patients. These data indicate that patients enrolled in our study have a severe alteration of lung elasticity, as shown by the values of static lung elastance definitely lower than those measured in previous studies. Another result of our study is the significant inverse correlation between PEEPdyn and Pdisniff during the spontaneous breathing trial. This correlation is easily understandable and confirms that dynamic hyperinflation is one of the most important physiological phenomena that limits the contractile capacity of the diaphragm during AECOPD. Therefore, by evaluating these physiological data as a whole, we can hypothesize that the portion of AECOPD patients who fail NIV constitute a “physiological phenotype” characterized by low lung elastance at baseline over which dynamic hyperinflation is superimposed. The result is a significant volume overload with shortening of the diaphragm and consequent muscle dysfunction.
Limitations of the Study
This prospective physiological study presents some methodological limitations that must be highlighted. First, the limited number of patients enrolled raises concerns about the statistical appropriateness of linear regression on outcomes assessment. Given the physiological nature of the study, the regression model can reasonably be used to produce estimates that may be considered quite stable despite outliers.38 These estimates need to be confirmed by larger clinical trials. Second, diaphragm maximal force was measured by a volitional test (Pdisniff) in intubated patients at the end of the spontaneous breathing trial. Although we performed a rigorous selection of patients and verified the reliability of the results of the Pdisniff maneuvers performed, a non-volitional test carried out with magnetic or electrical stimulation of the phrenic nerve could prove to be more accurate. Third, due to technical difficulties, it was not possible to measure the EELV reached at the end of the spontaneous breathing trial; therefore, it is not possible to know the EELV reached at the time of Pdisniff measurement. Moreover, in a population of patients with severe COPD, the EELV as assessed by nitrogen washout/washin technique might result quite inaccurate due to close volume. Considering this limitation, we do not know the volume overload due to dynamic hyperinflation; therefore, the effect of PEEPdyn and lung static elastance on lung volume at the end of the spontaneous breathing trial remains speculative.
Conclusions
With this prospective physiological study, we have shown that AECOPD patients who have experienced NIV failure present with severe diaphragmatic dysfunction that can lead to muscle paralysis. The causes of extreme diaphragmatic weakness might be probably predominantly mechanical, determined by a severe dynamic hyperinflation that overlaps on an elastic substrate of the lung favoring volume overload. The results of the study might suggest the existence of a physiological phenotype of AECOPD prone to NIV failure, characterized by low lung elastance and development of high values of PEEPdyn during acute exacerbation. Despite its obvious limitations, we believe that this study can increase our understanding of the complex physiological mechanisms underlying NIV failure in AECOPD.
Abbreviations
AECOPD, acute exacerbation of chronic obstructive pulmonary disease; COPD, chronic obstructive pulmonary disease; MV, mechanical ventilation; Pab, abdominal pressure; Paw, airway pressure; Pdi, transdiaphragmatic pressure; Pes, esophageal pressure; Pga, gastric pressure; Ppl, pleural pressure; Pdisniff, maximal transdiaphragmatic pressure calculated by sniff maneuver; NIV, non-invasive mechanical ventilation; TLC, total lung capacity; ZEEP, zero end-expiratory pressure; SD, standard deviation.
Ethics Approval and Consent to Participate
Approval from the local ethics committee of Modena was obtained (registered protocol number 839/C.E.). Written informed consent to participate was obtained from all patients enrolled, or their relatives, when appropriate.
Data Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
Consent for publication was obtained from all patients enrolled.
Acknowledgment
We thank Professional Editor, Colin Woodham, for language editing.
Author Contributions
AM and RT designed the study, analyzed data, and wrote the manuscript. RF, LT, FL and SB made substantial contributions to literature review, data collection and interpretation and paper writing. IC and LP reviewed the literature, made substantial contribution in data acquisition, wrote the manuscript and produced the figures. SN and EC designed the study and critically reviewed and edited the manuscript. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors have no competing interests with any organization or entity with a financial interest in competition with the subject, matter or materials discussed in the manuscript, and they report no conflicts of interest in this work.
References
1. Rochwerg B, Brochard L, Elliott MW, et al. Official ERS/ATS clinical practice guidelines: noninvasive ventilation for acute respiratory failure. Eur Respir J. 2017;50(2):1602426. doi:10.1183/13993003.02426-2016
2. Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med. 1995;333(13):817–822. doi:10.1056/NEJM199509283331301
3. Marchioni A, Castaniere I, Tonelli R, et al. Ultrasound-assessed diaphragmatic impairment is a predictor of outcomes in patients with acute exacerbation of chronic obstructive pulmonary disease undergoing noninvasive ventilation. Crit Care. 2018;22(1):109. doi:10.1186/s13054-018-2033-x
4. Antenora F, Fantini R, Iattoni A, et al. Prevalence and outcomes of diaphragmatic dysfunction assessed by ultrasound technology during acute exacerbation of COPD: a pilot study. Respirology. 2017;22(2):338–344. Epub 2016 Oct 14. doi:10.1111/resp.12916
5. Bellemare F, Grassino A. Force reserve of the diaphragm in patients with chronic obstructive pulmonary disease. J Appl Physiol Respir Environ Exerc Physiol. 1983;55(1 Pt 1):8–15. doi:10.1152/jappl.1983.55.1.8
6. Appendini L, Purro A, Patessio A, et al. Partitioning of inspiratory muscle workload and pressure assistance in ventilator-dependent COPD patients. Am J Respir Crit Care Med. 1996;154(5):1301–1309. doi:10.1164/ajrccm.154.5.8912740
7. Vassilakopoulos T, Zakynthinos S, Roussos C. The tension-time index and the frequency/tidal volume ratio are the major pathophysiologic determinants of weaning failure and success. Am J Respir Crit Care Med. 1998;158(2):378–385. doi:10.1164/ajrccm.158.2.9710084
8. Ceriana P, Vitacca M, Carlucci A, Paneroni M, Pisani L, Nava S. Changes of respiratory mechanics in COPD patients from stable state to acute exacerbations with respiratory failure. COPD. 2017;14(2):150–155. Epub 2016 Dec 20. doi:10.1080/15412555.2016.1254173
9. Purro A, Appendini L, Polillo C, et al. Mechanical determinants of early acute ventilatory failure in COPD patients: a physiologic study. Intensive Care Med. 2009;35(4):639–647. Epub 2008 Nov 26. doi:10.1007/s00134-008-1353-8
10. Gayan-Ramirez G, Decramer M. Mechanisms of striated muscle dysfunction during acute exacerbations of COPD. J Appl Physiol. 2013;114(9):1291–1299. Epub 2013 Jan 31. doi:10.1152/japplphysiol.00847.2012
11. Michelet P, Carreira S, Demoule A, et al. Effects of acute respiratory and metabolic acidosis on diaphragm muscle obtained from rats. Anesthesiology. 2015;122(4):876–883. doi:10.1097/ALN.0000000000000574
12. Demoule A, Jung B, Prodanovic H, et al. Diaphragm dysfunction on admission to the intensive care unit. Prevalence, risk factors, and prognostic impact - a prospective study. Am J Respir Crit Care Med. 2013;188(2):213–219. doi:10.1164/rccm.201209-1668OC
13. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):801–810. doi:10.1001/jama.2016.0287
14. Baydur A, Behrakis PK, Zin WA, Jaeger M, Milic-Emili J. A simple method for assessing the validity of the esophageal balloon technique. Am Rev Respir Dis. 1982;126(5):788–791. doi:10.1164/arrd.1982.126.5.788
15. Sklar MC, Burns K, Rittayamai N, et al. Effort to breathe with various spontaneous breathing trial techniques. A physiologic meta-analysis. Am J Respir Crit Care Med. 2017;195(11):1477–1485. doi:10.1164/rccm.201607-1338OC
16. Olegård C, Söndergaard S, Houltz E, Lundin S, Stenqvist O. Estimation of functional residual capacity at the bedside using standard monitoring equipment: a modified nitrogen washout/washin technique requiring a small change of the inspired oxygen fraction. Anesth Analg. 2005;101(1):206–212. doi:10.1213/01.ANE.0000165823.90368.55
17. Bates JHT, Baconnier P, Milic-Emili J. A theoretical analysis of interrupter technique for measuring respiratory mechanics. J Appl Physiol. 1988;64:2204–2214. doi:10.1152/jappl.1988.64.5.2204
18. Pepe PE, Marini JJ. Occult positive end-expiratory pressure in mechanically ventilated patients with airflow obstruction. Am Rev Respir Dis. 1982;126:166–170. doi:10.1164/arrd.1982.126.1.166
19. Steier J, Kaul S, Seymour J, et al. The value of multiple tests of respiratory muscle strength. Thorax. 2007;62(11):975–980. Epub 2007 Jun 8. doi:10.1136/thx.2006.072884
20. Global Initiative for Chronic Obstructive Lung Disease. 2019.
21. Gordon AM, Huxley AF, Julian FJ. The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J Physiol. 1966;184(1):170–192. doi:10.1113/jphysiol.1966.sp007909
22. ter Keurs HE, Iwazumi T, Pollack GH. The sarcomere length-tension relation in skeletal muscle. J Gen Physiol. 1978;72(4):565–592. doi:10.1085/jgp.72.4.565
23. McCully KK, Faulkner JA. Length-tension relationship of mammalian diaphragm muscles. J Appl Physiol Respir Environ Exerc Physiol. 1983;54(6):1681–1686. doi:10.1152/jappl.1983.54.6.1681
24. Farkas GA, Rochester DF. Functional characteristics of canine costal and crural diaphragm. J Appl Physiol. 1988;65(5):2253–2260. doi:10.1152/jappl.1988.65.5.2253
25. Braun NM, Arora NS, Rochester DF. Force-length relationship of the normal human diaphragm. J Appl Physiol Respir Environ Exerc Physiol. 1982;53(2):405–412. doi:10.1152/jappl.1982.53.2.405
26. Smith J, Bellemare F. Effect of lung volume on in vivo contraction characteristics of human diaphragm. J Appl Physiol. 1987;62(5):1893–1900. doi:10.1152/jappl.1987.62.5.1893
27. Orozco-Levi M, Gea J, Lloreta JL, et al. Subcellular adaptation of the human diaphragm in chronic obstructive pulmonary disease. Eur Respir J. 1999;13(2):371–378. doi:10.1183/09031936.99.13237199
28. Similowski T, Yan S, Gauthier AP, Macklem PT, Bellemare F. Contractile properties of the human diaphragm during chronic hyperinflation. N Engl J Med. 1991;325(13):917–923. doi:10.1056/NEJM199109263251304
29. Lindqvist J, van den Berg M, van der Pijl R, et al. Positive end-expiratory pressure ventilation induces longitudinal atrophy in diaphragm fibers. Am J Respir Crit Care Med. 2018;198(4):472–485. doi:10.1164/rccm.201709-1917OC
30. Ottenheijm CA, van Hees HW, Heunks LM, Granzier H. Titin-based mechanosensing and signaling: role in diaphragm atrophy during unloading? Am J Physiol Lung Cell Mol Physiol. 2011;300(2):L161–6. Epub 2010 Nov 12. doi:10.1152/ajplung.00288.2010
31. Neumann P, Zinserling J, Haase C, Sydow M, Burchardi H. Evaluation of respiratory inductive plethysmography in controlled ventilation: measurement of tidal volume and PEEP-induced changes of end-expiratory lung volume. Chest. 1998;113(2):443–451. doi:10.1378/chest.113.2.443
32. Patroniti N, Saini M, Zanella A, et al. Measurement of end-expiratory lung volume by oxygen washin-washout in controlled and assisted mechanically ventilated patients. Intensive Care Med. 2008;34(12):2235–2240. Epub 2008 Jul 23. doi:10.1007/s00134-008-1218-1
33. Bikker IG, van Bommel J, Reis Miranda D, Bakker J, Gommers D. End-expiratory lung volume during mechanical ventilation: a comparison with reference values and the effect of positive end-expiratory pressure in intensive care unit patients with different lung conditions. Crit Care. 2008;12(6):R145. Epub 2008 Nov 20. doi:10.1186/cc7125
34. Ibanez J, Raurich JM. Normal values of functional residual capacity in the sitting and supine positions. Intensive Care Med. 1982;8:173–177. doi:10.1007/BF01725734
35. Ferguson GT. Why does the lung hyperinflate? Proc Am Thorac Soc. 2006;3(2):176–179. doi:10.1513/pats.200508-094DO
36. Ranieri VM, Giuliani R, Mascia L, et al. Chest wall and lung contribution to the elastic properties of the respiratory system in patients with chronic obstructive pulmonary disease. Eur Respir J. 1996;9(6):1232–1239. doi:10.1183/09031936.96.09061232
37. Musch G, Foti G, Cereda M, Pelosi P, Poppi D, Pesenti A. Lung and chest wall mechanics in normal anaesthetized subjects and in patients with COPD at different PEEP levels. Eur Respir J. 1997;10(11):2545–2552. doi:10.1183/09031936.97.10112545
38. National Academies of Sciences, Engineering, and Medicine. Improving Health Research on Small Populations: Proceedings of a Workshop. Washington, DC: The National Academies Press; 2018. doi:10.17226/25112
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


