Back to Journals » OncoTargets and Therapy » Volume 11
Primary clear cell adenocarcinoma of the pancreas: a case report and literature update
Received 8 August 2018
Accepted for publication 31 October 2018
Published 16 November 2018 Volume 2018:11 Pages 8197—8200
DOI https://doi.org/10.2147/OTT.S183054
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tohru Yamada
Pi-Jiang Sun,1 Yan-Hua Yu,2 Xi-Jun Cui1
1Department of Hepatobiliary Surgery, Weihai Central Hospital, Weihai 264400, Shandong Province, People’s Republic of China; 2Department of Dermatology, Weihai Central Hospital, Weihai 264400, Shandong Province, People’s Republic of China
Abstract: Primary clear cell carcinoma of pancreas is extremely rare. We present a case of a 64-year-old male with a mass in the distal body and tail of the pancreas. He underwent a distal pancreatectomy. The histopathology of tumor cells showed features with abundant clear cytoplasm and prominent cell boundaries. Immunohistochemical analysis of neoplastic cells showed reactions to antibodies against cytokeratin-7 and showed no reactions to antibodies against hepatocyte nuclear factor-1β, carbonic anhydrase 9, synaptophysin, and chromogranin A. The patient was subsequently diagnosed with a primary clear cell carcinoma of the pancreas. This is the first time we have encountered it. We report this rare case and update the current literature of this tumor.
Keywords: clear cell carcinoma, pancreas, rare tumor
Introduction
According to the WHO classification, primary clear cell carcinoma of the pancreas is classified as a rare “miscellaneous” carcinoma;1 morphologically it is similar to the clear cell carcinoma of renal. It was first described by Cubilla in 1980.2 Because only few cases have been reported in the literature, the systematic overview of this tumor entity is insufficient. We report a rare case of primary clear cell carcinoma of the pancreas, review, and update the medical literature reported of this neoplasm.
Case report
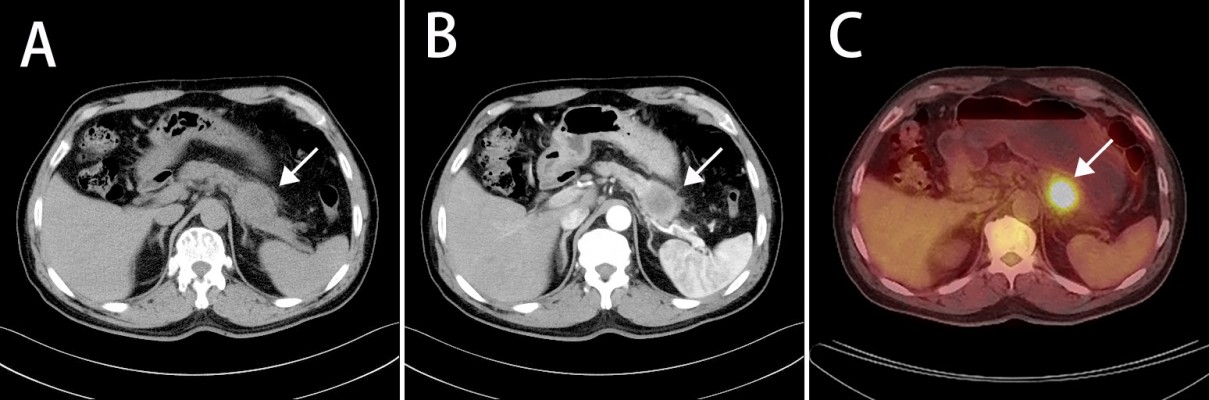
A 64-year-old male presented to our hospital with a year history of epigastric pain, radiating to the back, and 6 kg weight loss. He had no history of carcinoma and had a history of laparoscopic cholecystectomy 10 years ago. He had smoked 20 cigarettes per day for 30 years and drank alcohol on occasion. The family history was negative for malignant diseases. Physical examination revealed tenderness over the epigastric area. There were no enlarged superficial lymph nodes nor any palpable mass in the abdomen; the liver and spleen were not enlarged. The laboratory findings were all within the normal ranges except tumor markers. Tumor marker assay showed that serum carcinoembryonic antigen (CEA) was 18.56 ng/mL (normal: 0–5 ng/mL), the serum cancer antigen (CA) 19–9 was 649.15 U/mL (normal: 0–37 U/mL), CA125 was 132.97 U/L (normal: 0–35 kU/L), CA242 was 393.93 U/L (normal: 0–20 kU/L), neuron-specific enolase was 11 ng/mL (normal: 0–25 ng/mL), CYFRA21–1 was 1.7 ng/L (normal: 0–5 ng/L). Abdominal computed tomography (CT) showed a 40 × 45 mm heterogeneous irregular hypodense mass in the distal body and tail of the pancreas (Figure 1A), and it was enhanced after infusion of contrast material (Figure 1B). Positron emission tomography-CT showed diffuse abnormal uptake only in the pancreatic mass (Figure 1C).
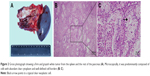
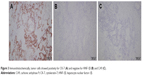
Preoperatively, it was diagnosed as an adenocarcinoma of the pancreas. Distal pancreatectomy and splenectomy were performed, and the specimen was sent for histopathological examination (Figure 2A). Microscopically, highly atypical glands were composed of round or oval bulky cells with abundant clear cytoplasm and well-defined cell borders (Figure 2B and C). These tumor cells were arranged as solid trabeculae, cords, and tubules. The hobnail appearance of cell arrangement was seen in some tumor cells. Nuclei were small, moderately pleomorphic with irregular borders, and often were eccentrically placed. These features were present in more than 90% of the tumor cells. Immunohistochemistry showed strong and diffuse staining for cytokeratin-7 (CK-7) (Figure 3A), and the immunohistochemistry staining was negative for chromogranin A (CGA), synaptophysin (SYN), hepatocyte nuclear factor-1β (HNF-1β) (Figure 3B), and carbonic anhydrase 9 (CA9) (Figure 3C). The patient recovered well and was discharged 10 days after surgery. CT scan revealed multiple hepatic metastasis 3 months after surgery, then he received gemcitabine chemotherapy and died 2 months later.
Discussion
Clear cell carcinoma occurs mostly in the kidney, lung, ovary, and thyroid, but this tumor extremely rarely originates from the pancreas.3 This is also the first time we have encountered primary pancreatic clear cell carcinoma, diagnosed by pathology and immunohistochemistry. We summarized our case and the available 12 cases of pancreatic clear cell carcinoma reported previously (Table 1). The age of the patients (nine males and three females) was 46–75 years, an average age of 64.4 years. The clinical symptoms are not specific, mainly manifested as abdominal pain, radiating to the back, jaundice, anorexia weight loss, and so on. Tumor biomarkers in these patients, such as serum CA19–9, CA125, CA242, CEA, can be elevated or normal. There are no specific diagnostic criteria for it. The diagnosis relies mainly on typical pathological features with abundant clear cytoplasm and prominent cell boundaries. Immunohistochemistry is a complementary method to distinguish from other tumors. Immunohistochemistry almost showed reactions to antibodies against CK-7 in all cases, while showed no reactions to neuroendocrine markers such as CGA, SYN in all cases. Sequencing of the K-ras oncogene was analyzed in three cases,4–6 and a mutation at codon 12 in K-ras oncogene was detected in two cases,5,6 providing molecular evidence of ductal origin. Based on the above characteristics, it was believed that pancreatic clear cell carcinoma is a variation of pancreatic ductal adenocarcinoma.5 HNF-1β may be a useful marker to identify these clear cell carcinomas, and its overexpression may aid in stratifying survival rate,7 but in our case, HNF-1β was negative. Primary pancreatic clear cell carcinoma must be distinguished from the pancreatic neuroendocrine tumor, solid pseudopapillary tumor, mixed ductal-endocrine carcinoma, and pancreatic metastasis from clear cell renal cell carcinoma.8 Of these, the most confusing tumor seems to be metastatic renal cell carcinoma.9,10 However, it is not very difficult to distinguish this tumor from it. History of renal tumors, preoperative CT, and MRI findings contribute to differential diagnosis. Postoperative immunohistochemistry of renal clear cell carcinoma showed positive reactions to antibodies against CD10, CA9, and Pax2, while immunonegative to CK-7.11–13 Because it is a rare tumor with very few cases reported previously, the incidence and prognosis are not well known for this neoplasm. However, we found that most of the reported cases showed aggressive behavior portending poor outcome, especially when the tumor was located in the tail of the pancreas.
Conclusion
We presented an extremely rare case of pancreatic clear cell ductal adenocarcinoma, which can be regarded as a rare variant of ductal adenocarcinoma in the proper morphology context supported by immunohistochemistry. Genetic analysis for detecting K-ras mutations and biomarker studies, such as HNF-1β, would aid in the identification of this rare neoplasm, but it is not absolute.
Acknowledgments
Written informed consent was acquired from the patient with approval of case detail and related patient information for publication use. Institutional approval was not required to publish this case report.
Disclosure
The authors report no conflicts of interest in this work.
References
Hamilton SR, Aaltonen LA. World Health Organization classification of tumours. Pathology and Genetics of Tumours of the Digestive System. Lyon, France: IARC Press; 2000:221–250. | ||
Cubilla AL, Fitzgerald PJ. Cancer (non-endocrine) of the pancreas. A suggested classification. Monogr Pathol. 1980;21:82–110. | ||
Modi Y, Shaaban H, Gauchan D, Maroules M, Parikh N, Guron G. Primary clear cell ductal adenocarcinoma of the pancreas: a case report and clinicopathologic literature review. J Cancer Res Ther. 2014;10(3):773–776. | ||
Sasaki A, Ishio T, Bandoh T, et al. Clear cell carcinoma of the pancreas: an adenocarcinoma with unusual phenotype of duct cell origin. J Hepatobiliary Pancreat Surg. 2004;11(2):140–144. | ||
Lüttges J, Vogel I, Menke M, Henne-Bruns D, Kremer B, Klöppel G. Clear cell carcinoma of the pancreas: an adenocarcinoma with ductal phenotype. Histopathology. 1998;32(5):444–448. | ||
Ray S, Lu Z, Rajendiran S. Clear cell ductal adenocarcinoma of pancreas: a case report and review of the literature. Arch Pathol Lab Med. 2004;128(6):693–696. | ||
Kim L, Liao J, Zhang M, et al. Clear cell carcinoma of the pancreas: histopathologic features and a unique biomarker: hepatocyte nuclear factor-1beta. Mod Pathol. 2008;21(9):1075–1083. | ||
Lee HY, Lee DG, Chun K, Lee S, Song SY. Clear cell carcinoma of the pancreas – a case report and review of the literature. Cancer Res Treat. 2009;41(3):175–181. | ||
Benhaim R, Oussoultzoglou E, Saeedi Y, Mouracade P, Bachellier P, Lang H. Pancreatic metastasis from clear cell renal cell carcinoma: outcome of an aggressive approach. Urology. 2015;85(1):135–140. | ||
Hoshino Y, Shinozaki H, Kimura Y, et al. Pancreatic metastases from renal cell carcinoma: a case report and literature review of the clinical and radiological characteristics. World J Surg Oncol. 2013;11:289. | ||
Tan PH, Cheng L, Rioux-Leclercq N, et al. Renal tumors: diagnostic and prognostic biomarkers. Am J Surg Pathol. 2013;37(10):1518–1531. | ||
Zianne M, Takahashi N, Tsujibata A, Miwa K, Goto Y, Matano Y. Asymptomatic pancreatic metastasis from renal cell carcinoma diagnosed 21 years after nephrectomy. Case Rep Gastrointest Med. 2017;2017:1–6. | ||
Pickering LM, Larkin J. Kidney cancer: carbonic anhydrase IX in resected clear cell RCC. Nat Rev Urol. 2015;12(6):309–310. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.