Back to Journals » Clinical Epidemiology » Volume 6
Potential of prescription registries to capture individual-level use of aspirin and other nonsteroidal anti-inflammatory drugs in Denmark: trends in utilization 1999–2012
Authors Schmidt M , Hallas J , Friis S
Received 13 December 2013
Accepted for publication 14 January 2014
Published 12 May 2014 Volume 2014:6 Pages 155—168
DOI https://doi.org/10.2147/CLEP.S59156
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Morten Schmidt,1 Jesper Hallas,2 Søren Friis1,3
1Department of Clinical Epidemiology, Aarhus University Hospital, Aarhus, Denmark; 2Department of Clinical Pharmacology, University of Southern Denmark, Odense, Denmark; 3Danish Cancer Society Research Center, Danish Cancer Society, Copenhagen, Denmark
Background: Due to over-the-counter availability, no consensus exists on whether adequate information on nonsteroidal anti-inflammatory drug (NSAID) use can be obtained from prescription registries.
Objectives: To examine utilization of aspirin and nonaspirin NSAIDs in Denmark between 1999 and 2012 and to quantify the proportion of total sales that was sold on prescription.
Method: Based on nationwide data from the Danish Serum Institute and the Danish National Prescription Registry, we retrieved sales statistics for the Danish primary health care sector to calculate 1-year prevalences of prescription users of aspirin or nonaspirin NSAIDs, and to estimate the corresponding proportions of total sales dispensed on prescription.
Results: Both low-dose aspirin and nonaspirin NSAIDs were commonly used in the Danish population between 1999 and 2012, particularly among elderly individuals. The 1-year prevalence of prescribed low-dose aspirin increased throughout the study period, notably among men. Nonaspirin NSAID use was frequent in all age groups above 15 years and showed a female preponderance. Overall, the prevalence of prescribed nonaspirin NSAIDs decreased moderately after 2004, but substantial variation according to NSAID subtype was observed; ibuprofen use increased, use of all newer selective cyclooxygenase-2 inhibitors nearly ceased after 2004, diclofenac use decreased by nearly 50% after 2008, and naproxen use remained stable. As of 2012, the prescribed proportion of individual-level NSAID sales was 92% for low-dose aspirin, 66% for ibuprofen, and 100% for all other NSAIDs.
Conclusion: The potential for identifying NSAID use from prescription registries in Denmark is high. Low-dose aspirin and nonaspirin NSAID use varied substantially between 1999 and 2012. Notably, use of cyclooxygenase-2 inhibitors nearly ceased, use of diclofenac decreased markedly, and naproxen use remained unaltered.
Keywords: drug utilization, NSAID, registries, over-the-counter
Introduction
In Danish pharmacoepidemiological studies, use of aspirin and nonaspirin nonsteroidal anti-inflammatory drugs (NSAIDs) are typically identified from prescription databases.1–3 However, there is no consensus on whether adequate information on NSAID use can be obtained from such databases.4 Stratification by4 or adjustment for5,6 aspirin use has been abandoned or dismissed by some authors, who argue that since aspirin is available over the counter (OTC), use of this drug or other NSAIDs cannot be captured reliably in the Danish prescription registries.4
The potential of prescription registries to capture individual-level use of aspirin and other NSAIDs is of interest in several contexts. One is whether these registries can be used to survey utilization of nonaspirin NSAIDs,7 which are associated with an increased risk of cardiovascular disease.8,9 The current evidence indicates that all nonaspirin NSAIDs increase the risk of heart failure, whereas the risk of thrombotic events varies according to type of drug.9 Use of selective cyclooxygenase(COX)-2 inhibitors (coxibs) is associated with the highest vascular risk, whereas naproxen appears to have the least harmful cardiovascular risk profile.9,10 Moreover, increasing evidence supports that traditional nonaspirin NSAIDs with high COX-2 selectivity, in particular diclofenac, have thrombogenic properties similar to coxibs.9 Despite these important differences in cardiovascular toxicity, no study has evaluated whether use of naproxen or diclofenac has changed since the concern about cardiovascular toxicity associated with COX-2 inhibiting agents was first raised in 2004.8
We examined the utilization of NSAIDs in Denmark from 1999 to 2012, with specific focus on trends in nonaspirin NSAID use and the potential of prescription registries to capture individual-level use of aspirin and nonaspirin NSAIDs.
Methods
We ascertained individual use of low-dose aspirin and nonaspirin NSAIDs using data from the Danish National Prescription Registry, with focus on trends in overall utilization, age and sex distribution, volume, and the proportion of total sales that was sold on prescription.
Setting
The Danish National Health Service provides universal tax-supported health care, guaranteeing free and equal access to general practitioners and hospitals and partial reimbursement for prescribed medications, including NSAIDs.11 Individual-level linkage of all Danish databases is possible using the unique Danish personal identification number, which is assigned to each Danish citizen at birth and to residents upon immigration.12
Pharmacies in Denmark are equipped with electronic accounting systems, which are primarily used to secure reimbursement from the National Health Service.13,14
A detailed account of variables registered in the prescription registries has previously been described.13 Briefly, for each redeemed prescription, the patient’s personal identification number, the type of drug prescribed according to the Anatomical Therapeutic Chemical (ATC) classification system,15 pack size (numbers of pills and daily defined doses), and the date of drug dispensing are transferred electronically from the pharmacies to prescription registries.13,14 Different dose units for the same pharmaceutical entity can also be identified separately in the prescription registries by use of product codes.13,14
We used the web facility Medstat (http://www.medstat.dk) to retrieve data on NSAID sales in Denmark.16 This publicly available webpage from the Danish Serum Institute provides aggregate statistics on the sale of pharmaceutical preparations in Denmark since 1995 based on the data reported to the Danish National Prescription Registry.14,16 This reporting is mandatory, and Medstat statistics are complete from 1999 onwards. The registration of total drug sales (including OTC sales) facilitates computations of descriptive statistics, including for example the proportion of total sales sold on prescription, and allows for stratification by age, sex, region, and health care sector (primary or secondary).16
OTC use in Denmark
OTC NSAIDs include aspirin in all preparations, diclofenac (during the period July 16, 2007 to December 14, 2008), and low-dose ibuprofen (200 mg tablets) since March 27, 1989.17,18 Regular users of aspirin or nonaspirin NSAIDs have an economic incentive to obtain the drugs by prescription due to the reimbursement through the Danish National Health Service’s insurance program.
In an effort to reduce suicide attempts by overdoses of analgesics,19 the Danish Health Authorities have implemented several restrictions in the dispensing of OTC drugs since 2001.20–22 First, packages of aspirin and paracetamol containing ≥30 tablets were labeled with red box warnings alerting parents to read the warnings in the package leaflet and to store the drugs in a safe place (October 1, 2004).20 More recently (March 7, 2011),21 OTC sales of aspirin, paracetamol, and ibuprofen were restricted to persons aged ≥18 years and at maximum one package per person per day. Just recently (September 20, 2013), each dispensing of OTC analgesics has been restricted to pack sizes containing a maximum of 10 g of aspirin (ie, 20 high-dose tablets), 10 g of paracetamol (ie, 20 tablets), or 4 g of ibuprofen (ie, 20 tablets).22
Aspirin
Aspirin (acetylsalicylic acid) has the characteristic analgesic, antipyretic, and anti-inflammatory properties of nonselective NSAIDs.23,24 In high doses (500 mg), the main indication for aspirin is pain relief (ATC group: N02BA01, N02BA51). At low doses (75–150 mg), aspirin is not an effective analgesic, but the drug has an antithrombotic effect conferred by inhibition of platelet aggregation by irreversible blockage of the COX-1 enzyme.23,24 Accordingly, the main indication for low-dose aspirin (ATC group: B01AC06) is prevention and treatment of occlusive vascular events in patients with coronary artery disease or ischemic stroke.25
Nonaspirin NSAIDs
The main indications for nonaspirin NSAIDs are inflammatory conditions and pain (ATC group: M01A).23 We excluded glucosamine (ATC: M01AX05) from the main group (M01A), as this agent does not possess the pharmacodynamic properties of nonaspirin NSAIDs.26
We identified all individual drugs from each NSAID class on the Danish market; ie, butylpyrazolidines, acetic acids, enolic acids, proprionic acids, fenamic acids, nonacidics, and coxibs.17 We furthermore identified the six most frequently used nonaspirin NSAIDs, which, according to their COX-selectivity, could be classified as nonselective NSAIDs (ibuprofen and naproxen), older COX-2 inhibitors (diclofenac and etodolac), and coxibs (celecoxib and rofecoxib). The newer and older COX-2 inhibitors are almost similar in COX-2 selectivity when comparing the concentration of the drugs (IC50) required to inhibit COX-1 and COX-2 activity by 50%.27 For instance, the COX-1/COX-2 IC50 is 29 for diclofenac and 30 for celecoxib.27
Statistical analyses
We obtained sales statistics for the entire Danish population (5.6 million inhabitants as of 2012) from January 1, 1999 to December 31, 2012. The retrieval of sales statistics was restricted to the primary health care sector; ie, sales outside the hospital setting. In addition to pharmacies and nonpharmacy outlets, drug sales in the primary health care sector comprised sales from the Danish Serum Institute and in general practices.16
First, we calculated and illustrated graphically the 1-year prevalence of low-dose aspirin users and nonaspirin NSAID users, overall and by sex and age groups (15–19 years, 20–39 years, 40–64 years, 65–79 years, and ≥80 years). Age was defined as age at first redeemed prescription each year. Results were calculated for nonaspirin NSAIDs overall as well as separately for each of the six most frequently used types. Secondly, we identified the proportion of all aspirin and nonaspirin NSAID sales that was dispensed on prescription each year in the study period.
Results
Aspirin
Aspirin was prescribed almost exclusively in low doses for cardiovascular prevention. The annual number of low-dose aspirin prescription users increased steadily from 232,213 (4.4%) in 1999 to 408,555 (7.3%) in 2012 (Table 1 and Figure 1). Age- and sex-stratified analyses for prescribed low-dose aspirin showed an equal sex distribution in 1999, but over time slightly more men than women redeemed prescriptions for aspirin (8.0% vs 6.7% in 2012) (Figure 1). Practically no use of low-dose aspirin occurred in individuals younger than 40 years, but the prevalence of use increased to around 5% in those between the ages of 40–64 years, 25% in those aged 65–79 years, and 40% in those aged 80 years or more in 2012 (Figure 2A). The proportion of total low-dose aspirin sales that was prescribed on an individual level increased steadily from 62% in 1999 to 92% in 2012 (Table 2 and Figure 3).
  | Figure 1 The 1-year prevalence of the Danish population redeeming a prescription for low-dose aspirin or nonaspirin nonsteroidal anti-inflammatory drugs (NSAIDs) during 1999–2012, overall and by sex. |
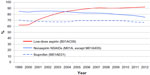  | Figure 3 The percentage of total nonsteroidal anti-inflammatory drug (NSAID) sales sold on prescription in Denmark between 1999 and 2012. |
High-dose aspirin was sold alone or in combination with codeine (9.6 mg) or caffeine (50 mg). As a single-compound product, high-dose aspirin was prescribed to 3,233 persons (0.06%) in 1999 and 521 (0.009%) in 2012 (Table 1). As a combination product, high-dose aspirin was prescribed to 6,340 (0.1%) in 1999 and 8,398 (0.2%) in 2012. The vast majority of total sales of high-dose aspirin both alone (90%–93%) or in combination tablets (97%–99%) were sold OTC (Table 2).
Nonaspirin NSAIDs
Each year, around 13%–15% of the total Danish population redeemed at least one prescription of nonaspirin NSAID between 1999 and 2012 (Table 1). From age 10–15 years, the prevalence of use increased markedly with age (Figure 2B). The overall prescription pattern of nonaspirin NSAIDs varied during the study period. Thus, the 1-year prevalence of individuals redeeming at least one prescription increased from 13.6% (n=723,325) in 1999 to 15.5% (n=836,072) in 2004, before declining steadily to 13.1% (n=731,667) in 2012 (Table 1 and Figure 1). A similar pattern was observed among men and women (Figure 1). Individuals aged 80 years or above constituted the most frequent users of prescription nonaspirin NSAIDs until 2003, after which their use decreased to below that of individuals aged 40–79 years (Figure 2B). All nonaspirin NSAIDs were used more frequently among women than among men.
The decrease in nonaspirin NSAID use from 2004 was seen for all three types of nonaspirin NSAIDs; ie, nonselective NSAIDs, older COX-2 inhibitors, and coxibs (Table 1 and Figure 4). An exception was ibuprofen, for which the 1-year prevalence increased steadily from 6.5% in 1999 to 9.9% in 2012. Among other commonly used nonaspirin agents, use of naproxen decreased slightly from 1999 to 2004 (1.1% in 1999 and 0.7% in 2004) and remained stable thereafter. Diclofenac was consistently prescribed to around 4% of the Danish population until 2008, after which the prevalence decreased and reached 2% in 2012. Etodolac was prescribed to approximately 0.5% throughout the period (0.7% in 1999 and 0.5% in 2012). Celecoxib and rofecoxib comprised almost the entire sale of coxibs. These agents displayed a fairly similar pattern of use, increasing steeply after their introduction (on November 15, 1999 for rofecoxib and on May 15, 2000 for celecoxib) to surpass both naproxen and etodolac (Figure 4). After 2002, the use of coxibs began to decrease; after September 30, 2004, when rofecoxib was withdrawn from the market, celecoxib use decreased sharply, and it was only used by 0.04% of the population in 2012 (Table 1 and Figure 4). Stratified analyses according to age and sex (Table S1) revealed that coxibs and etodolac were used almost entirely among individuals above 40 years, whereas ibuprofen, naproxen, and diclofenac constituted the most frequently used nonaspirin NSAIDs among younger individuals.
The proportion of nonaspirin NSAIDs dispensed on prescription in Denmark decreased from 85% in 1999 to 75% in 2012 (Table 2 and Figure 3). The OTC availability of diclofenac in part of 2007–2008 did not influence the overall prescription/OTC relation materially (Table 2). Thus, low-dose (200 mg) ibuprofen accounted for practically all OTC use of nonaspirin NSAIDs between 1999 and 2012. Specifically, OTC use of low-dose ibuprofen accounted for 30%–35% of total ibuprofen sales and 15%–25% of total nonaspirin NSAID sales between 1999 and 2012 (Figure 3). The overall decrease in the proportion of nonaspirin NSAIDs dispensed by prescription reflected that OTC ibuprofen use increased more than the prescribed use of ibuprofen.
Discussion
Both low-dose aspirin and nonaspirin NSAIDs were commonly used in the Danish population between 1999 and 2012. The proportions of total sales of low-dose aspirin or nonaspirin NSAIDs dispensed by prescription and thus captured in prescription registries were high: as of 2012, 92% for low-dose aspirin, 66% for ibuprofen and 100% for all other nonaspirin NSAIDs. The 1-year prevalence of prescribed low-dose aspirin increased throughout the period, particularly among men. Except for ibuprofen, the 1-year prevalence of nonaspirin NSAID use decreased after 2004. The decline was independent of age or sex, but there was a consistently higher prevalence of use in older age groups and among women. Use of coxibs decreased to near null after 2004. Interestingly, the use of diclofenac was reduced by half between 2008 and 2012, but no substantial change occurred in use of naproxen and etodolac.
OTC use of low-dose aspirin and nonaspirin NSAIDs is far less common in Denmark than in many other countries.28 Therefore, the potential for identification of NSAID use from prescription registries is high. Indeed, with the most recent restriction on pack sizes of OTC analgesics in Denmark,22 OTC use of NSAIDs is likely to decrease even further. In this context, it should be noted that while low-dose aspirin is used continuously for prevention of cardiovascular disease and a substantial proportion of high-dose nonaspirin NSAID therapy is also used on a chronic basis, high-dose aspirin and low-dose ibuprofen are mainly used as short-term treatment of transient pain conditions such as headaches, sports injuries, or backaches. Danish prescription registry data are thus reliable data sources for research on the effects of aspirin and nonaspirin NSAID exposure, especially when indicated for chronic use.
Assessment of low-dose aspirin use has the advantage, compared with many other medications, that the daily defined dose equals one pill per day.17 Thus, the expected number of exposure days from a prescription refill can be modeled from the number of pills per package.1 Using a more accurate exposure window in this way (rather than a fixed exposure-window) may help to reduce misclassification of aspirin use.
Because the prescription data are prospectively recorded, any misclassification of NSAID use due to nonadherence or OTC use of aspirin or ibuprofen would generally bias measures of associations towards the null. The magnitude of misclassification bias due to OTC use can be illustrated from a hypothetical cohort study examining the effect of drug exposure on a given outcome. Assuming that 15% of the population uses the drug every day, only two-thirds obtain the drug on prescription (as for ibuprofen), and an equal age distribution among new and long-term users, there will be no misclassification of the apparently exposed and only 5% (nondifferential) misclassification of the apparently nonexposed (as one-third of 15% will be OTC users who are not captured by the prescription registry). Considering another scenario with an exposure prevalence of 10% and prescription coverage of 50%, the misclassification will still be only 5%. Unless the relative risk measure is very high, misclassification of this magnitude has no practical impact on the relative risk estimate, rate difference, or etiologic fraction among the exposed. Only the etiologic fraction in the background population will be substantially underestimated from such misclassification. Moreover, the misclassification would be even less for drugs with a prevalence of use below 10%–15% (which is often the case). Finally, the bias generated by such misclassification would be even smaller if the drug with OTC availability is a confounder drug and not the primary exposure.
The withdrawal of rofecoxib contributed to the subsequent reduction in use of all coxibs following 2004, including celecoxib.8 The increased focus on the cardiovascular toxicity associated with nonaspirin NSAID use in general, and COX-2 selective NSAIDs in particular, may also have contributed to the overall decrease in prescribed nonaspirin NSAID use from 2004 onwards. The decrease in diclofenac use after 2008 may be a direct consequence of recommendations from the Danish Medicines Agency in 200829 and Danish Society for Cardiology in 200930 to use diclofenac with caution due to an increased risk of cardiovascular disease. Surprisingly, use of naproxen did not increase during the study period despite several studies pointing to a markedly lower cardiovascular risk profile of naproxen than of other nonaspirin NSAID agents.9,10 In contrast, both prescribed and OTC use of ibuprofen increased substantially from 1999. These patterns are difficult to explain as a rational response to concerns about cardiovascular toxicity of nonaspirin NSAIDs. Until 2004, however, the dominant discourse on NSAID toxicity concerned gastrointestinal bleeding. Among the traditional NSAIDs, ibuprofen has a well-established low gastrointestinal risk, whereas the gastrointestinal safety is lower for naproxen.31,32 Although the magnitude of cardiovascular versus gastrointestinal risks for individual NSAIDs is controversial,33 it is possible that preferences to a large extent still are driven mainly by the perceived gastrointestinal risks.
Whereas low-dose ibuprofen therapy seems safe,34,35 high-dose ibuprofen has also been associated with adverse cardiovascular events.9 Even though OTC ibuprofen is only available in 200 mg tablets, it is not possible to monitor the consumed number of pills and hence the daily dose. This is a concern, especially before September 20, 2013,22 because OTC drugs are often used in higher doses than recommended and with little attention to potential side effects.28
Strengths and limitations
Using Medstat, we were able to obtain complete data on prescribed and total sales of all marketed NSAIDs for the entire Danish population during a 14-year period.14,16 We did not have information on the exact number of OTC users, but we had information on the proportions of total sales of aspirin or nonaspirin NSAID that were dispensed by prescription. The proportion of prescription use of all nonaspirin NSAIDs, except low-dose ibuprofen, equals almost all use in Denmark.
Conclusion
The potential for identification of individual-level use of low-dose aspirin and nonaspirin NSAIDs from prescription registries in Denmark is high. This is of vast importance for analytical studies addressing NSAIDs as either primary or secondary exposures. The pattern of NSAID use in Denmark varied substantially between 1999 and 2012. Use of coxibs nearly ceased and diclofenac use decreased by half since 2008, whereas naproxen use remained stable and did not increase despite its less harmful cardiovascular risk profile.
Research Ethics and Informed Consent
As this study was based solely on register data and did not involve any contact with patients, no approval was required from the Danish Scientific Ethical Committee.
Disclosure
MS has no disclosures. JH has participated in research projects funded by Novartis, Pfizer, Menarini, MSD, Nycomed, and Astellas, with grants paid to the institution where he was employed. JH has personally received fees for teaching or consulting from the Danish Association of Pharmaceutical Manufacturers, Nycomed, Pfizer, Novartis, AstraZeneca, Lundbeck, Menarini, Leo Pharmaceuticals, and Ferring. SF has personally received fees for teaching or consulting from the Danish Association of Pharmaceutical Manufacturers. The authors declare no other conflicts of interest.
References
Schmidt M, Johansen MB, Lash TL, Christiansen CF, Christensen S, Sørensen HT. Antiplatelet drugs and risk of subarachnoid hemorrhage: a population-based case-control study. J Thromb Haemost. 2010;8(7):1468–1474. | |
Schmidt M, Christiansen CF, Mehnert F, Rothman KJ, Sørensen HT. Non-steroidal anti-inflammatory drug use and risk of atrial fibrillation or flutter: population based case-control study. BMJ. 2011;343:d3450. | |
Gaist D, García-Rodríguez LA, Sørensen HT, Hallas J, Friis S. Use of low-dose aspirin and non-aspirin nonsteroidal anti-inflammatory drugs and risk of glioma: a case-control study. Br J Cancer. 2013;108(5):1189–1194. | |
Lidegaard ø. Reply to: Stroke and myocardial infarction with hormonal contraception. N Engl J Med. 2012;367:1264. | |
Caporaso NE. Statins and cancer-related mortality – let’s work together. N Engl J Med. 2012;367(19):1848–1850. | |
Dawwas MF. Statin use and reduced cancer-related mortality. N Engl J Med. 2013;368(6):574–575. | |
Fosbøl EL, Gislason GH, Jacobsen S, et al. The pattern of use of non-steroidal anti-inflammatory drugs (NSAIDs) from 1997 to 2005: a nationwide study on 4.6 million people. Pharmacoepidemiol Drug Saf. 2008;17(8):822–833. | |
Bresalier RS, Sandler RS, Quan H, et al; Adenomatous Polyp Prevention on Vioxx (APPROVe) Trial Investigators. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med. 2005;352(11):1092–1102. | |
Bhala N, Emberson J, Merhi A, et al; Coxib and traditional NSAID Trialists’ (CNT) Collaboration. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013;382(9894):769–779. | |
Trelle S, Reichenbach S, Wandel S, et al. Cardiovascular safety of non-steroidal anti-inflammatory drugs: network meta-analysis. BMJ. 2011;342:c7086. | |
Ministry of Health and Prevention. Health Care in Denmark. 2008. Available from: http://www.sum.dk/Aktuelt/Publikationer/~/media/Filer%20-%20Publikationer_i_pdf/2008/UK_Healthcare_in_dk/pdf.ashx. Accessed December 1, 2013. | |
Pedersen CB. The Danish Civil Registration System. Scand J Public Health. 2011;39(Suppl 7):22–25. | |
Johannesdottir SA, Horváth-Puhó E, Ehrenstein V, Schmidt M, Pedersen L, Sørensen HT. Existing data sources for clinical epidemiology: The Danish National Database of Reimbursed Prescriptions. Clin Epidemiol. 2012;4:303–313. | |
Kildemoes HW, Sørensen H, Hallas J. The Danish National Prescription Registry. Scand J Public Health. 2011;39(Suppl 7):38–41. | |
World Health Organization Collaborating Centre for Drug Statistics Methodology. ATC Classification Index with DDDs. World Health Organization Collaborating Centre for Drug Statistics Methodology, Oslo, 2013. Available from: http://www.whocc.no/atc_ddd_index/. Accessed December 1, 2013. | |
Danish Serum Institute. Statistics on the annual sale of medicines in Denmark based on the data from the Register of Medicinal Product Statistics. Available from: http://www.medstat.dk/en. Accessed December 1, 2013. | |
Danish Pharmaceutical Information. Drugs and package sizes on the Danish drug market. Available for search from http://pro.medicin.dk. Accessed December 1, 2013. | |
Danish Health and Medicines Authority. Over-the-counter medicines in Denmark. Available from: http://sundhedsstyrelsen.dk/en/medicines/sale/over-the-counter-medicines. Accessed December 1, 2013. | |
Hawton K, Bergen H, Simkin S, et al. Long term effect of reduced pack sizes of paracetamol on poisoning deaths and liver transplant activity in England and Wales: interrupted time series analyses. BMJ. 2013;346:f403. | |
Danish Health and Medicines Authority. Special warning on the labelling of pain relievers. October 1, 2004. Available from: http://laegemiddelstyrelsen.dk/en/topics/authorisation-and-supervision/licensing-of-medicines/summaries-of-product-characteristics,-pa---labelling/special-warning-on-the-labelling-of-pain-relievers. Accessed December 1, 2013. | |
Danish Health and Medicines Authority. Age limit of 18 years introduced for buying painkillers. March 7, 2011. Available from: http://laegemiddelstyrelsen.dk/en/service-menu/news/age-limit-of-18-years-introduced-for-buy--ainkillers. Accessed December 1, 2013. | |
Danish Health and Medicines Authority. Painkillers to be prescription only in Denmark from September 30, 2013. Available from: http://sundhedsstyrelsen.dk/en/news/2013/painkillers-to-be-prescription-only-in-denmark-from-30-september-2013. Accessed December 1, 2013. | |
Day RO, Graham GG. Non-steroidal anti-inflammatory drugs (NSAIDs). BMJ. 2013;346:f3195. | |
Fuster V, Sweeny JM. Aspirin: a historical and contemporary therapeutic overview. Circulation. 2011;123(7):768–778. | |
Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002;324(7329):71–86. | |
Anderson JW, Nicolosi RJ, Borzelleca JF. Glucosamine effects in humans: a review of effects on glucose metabolism, side effects, safety considerations and efficacy. Food Chem Toxicol. 2005;43(2):187–201. | |
Capone ML, Tacconelli S, Di Francesco L, Sacchetti A, Sciulli MG, Patrignani P. Pharmacodynamic of cyclooxygenase inhibitors in humans. Prostaglandins Other Lipid Mediat. 2007;82(1–4):85–94. | |
Wilcox CM, Cryer B, Triadafilopoulos G. Patterns of use and public perception of over-the-counter pain relievers: focus on nonsteroidal antiinflammatory drugs. J Rheumatol. 2005;32(11):2218–2224. | |
Danish Health and Medicines Authority. New information about cardiovascular adverse reactions from the use of NSAID. Danish Medicines Agency. November 20, 2008. Available from: http://sundhedsstyrelsen.dk/en/news/2008/new-information-about-cardiovascular-adverse-reactions-from-the-use-of-nsaid. Accessed December 1, 2013. | |
Gislason GH, Fosbøl EL, Grande P, et al. Danish Society of Cardiology. Recommendations on NSAID use in patients with cardiovascular disease. 2009. Available from: http://www.cardio.dk/rapporter/holdningspapir-menu. Accessed December 1, 2013. | |
Henry D, Lim LL, Garcia Rodriguez LA, et al. Variability in risk of gastrointestinal complications with individual non-steroidal anti-inflammatory drugs: results of a collaborative meta-analysis. BMJ. 1996;312(7046):1563–1566. | |
Castellsague J, Riera-Guardia N, Calingaert B, et al; Safety of Non-Steroidal Anti-Inflammatory Drugs (SOS) Project. Individual NSAIDs and upper gastrointestinal complications: a systematic review and meta-analysis of observational studies (the SOS project). Drug Saf. 2012;35(12):1127–1146. | |
van Staa TP, Smeeth L, Persson I, Parkinson J, Leufkens HG. What is the harm-benefit ratio of Cox-2 inhibitors? Int J Epidemiol. 2008;37(2):405–413. | |
Fosbøl EL, Gislason GH, Jacobsen S, et al. Risk of myocardial infarction and death associated with the use of nonsteroidal anti-inflammatory drugs (NSAIDs) among healthy individuals: a nationwide cohort study. Clin Pharmacol Ther. 2009;85(2):190–197. | |
European Medicines Agency. Assessment report for non-steroidal anti-inflammatory drugs (NSAIDs) and cardiovascular risk. 2012. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Report/2012/11/WC500134717.pdf. Accessed December 1, 2013. |
Supplementary materials
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





