Back to Journals » OncoTargets and Therapy » Volume 13
Overexpression of a Long Non-Coding RNA BC037916 is Associated with Pancreatic Tumorigenesis and Poor Prognosis
Authors Chen G, Xu L, Ye G, Lin J, Meng Z, Shen Y
Received 21 September 2020
Accepted for publication 8 December 2020
Published 5 January 2021 Volume 2020:13 Pages 13451—13463
DOI https://doi.org/10.2147/OTT.S282350
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Leo Jen-Liang Su
Gang Chen,1 Litao Xu,2,3 Guanxiong Ye,4 Junhua Lin,2,3 Zhiqiang Meng,2,3 Yehua Shen2,3
1Department of Pediatric Cardiothoracic Surgery, Children’s Hospital of Fudan University, Shanghai, People’s Republic of China; 2Department of Integrative Oncology, Fudan University Shanghai Cancer Center, Shanghai, China; 3Department of Oncology, Shanghai Medical College, Fudan University, Shanghai, People’s Republic of China; 4Department of Hepatobiliary Surgery, People’s Hospital of Lishui, The Sixth Affiliated Hospital of Wenzhou Medical University, Lishui, People’s Republic of China
Correspondence: Yehua Shen 270 Dong’An Road, Shanghai 200032, People’s Republic of China
Tel + (86)21-64175590-83625
Email [email protected]
Background: Pancreatic cancer is one of the most lethal malignancies. Accumulating evidence supports for the critical contribution of long noncoding RNAs (lncRNAs) to the cancer development and progression. We tried to identify novel lncRNAs involved in the pancreatic carcinogenesis.
Materials and Methods: Two independent datasets (Gene Expression Omnibus datasets: GSE16515 and GSE32688) were obtained from the Gene Expression Omnibus (GEO). The level of BC037916 was detected in pancreatic cancer tissues and adjacent no-tumorous tissues (n=86) by qRT-PCR. Effects of BC037916 on proliferation, apoptosis, and invasion of pancreatic cancer cells were examined.
Results: We identified a novel lncRNA BC037916 involved in the pancreatic carcinogenesis by analyzing GEO datasets. Quantitative RT-PCR analysis showed that 86.0% (74/86) pancreatic cancer tissues had increased BC037916 expression as compared with normal counterparts. Further, positive correlation was observed between BC037916 expression and clinical stage, primary tumor, and regional lymph node invasion. Importantly, BC037916 was an independent prognostic factor of pancreatic cancer. Functionally, knockdown of BC037916 repressed cell proliferation, inhibited cell invasion, halted cell cycle progression, and promoted apoptosis in both PANC-1 and SW1990 cells. In contrast, overexpression of BC037916 in CAPAN-1 had opposite effects. Moreover, silencing of BC037916 significantly inhibited the tumor growth of xenografted SW1990 cells in vivo. Results of Western blot assays suggested that BC037916 knockdown also suppressed the activation of JAK2/STAT3 and TGF-β signaling. Further experiments suggested that BC037916 positively regulated the expression of Twist through miR-3145-3p.
Conclusion: BC037916 exhibited oncogenic potential in pancreatic cancer development.
Keywords: lncRNA, pancreatic cancer, JAK2/STAT3, TGF-β
Background
Pancreatic carcinoma is the fourth leading disease-related cause of death globally.1 Although the underlying causes of pancreatic cancer have not been fully understood, certain risk factors, including smoking, obesity, diabetes, age, and genetic conditions, have been identified.2 Due to the lack of useful biomarkers and effective early detection methods, most cases of pancreatic cancer are diagnosed in advanced stages, which lead to the devastatingly poor prognosis of pancreatic cancer patients.1,3,4 Hence, there is an urgency for identification of novel prognostic biomarkers and targets and development of better therapeutic options to accelerate the diagnosis and improve the treatment of pancreatic carcinoma.
Recent next-generation sequencing studies have identified that the majority of human transcriptome is recognized as non-coding RNAs (ncRNAs), which are further categorized into small (<200 nt in length) and long non-coding RNAs (lncRNAs, >200 nt in length).5 LncRNAs have attracted increasing scientific interest and increasing data have shown the important role of lncRNAs in a spectrum of cellular processes such as cell survival,6 cell cycle,7,8 migration,9,10 and metabolism.11 Dysregulated lncRNAs have been detected in many complicated human diseases including human cancers, indicating that lncRNAs may be critical players in the development and progression of cancer.12–20
In pancreatic cancer, aberrant expression of lncRNAs, such as HOTAIR,19 MALAT1,20 and CRNDE21 have been shown. LncRNA MALAT1 was up-regulated and overexpression of MALAT1 serves a marker for an unfavorable prognosis.20 Knockdown of lncRNA HOTAIR19 or HOTTIP12 inhibited proliferation and inhibited invasion of pancreatic cancer cells. However, it is estimated that there are approximately 15,000 lncRNAs in humans22 and lncRNA profiles in pancreatic cancer remain largely unknown. In the current study, two independent datasets (GSE1651523 and GSE32688) were obtained from the Gene Expression Omnibus (GEO), and lncRNAs profiling was accomplished by Genomic and Transcriptomic Explorer (GATExplorer).24 lncRNA BC037916 was identified as a candidate pro-oncogene in pancreatic cancer. We then explored its expression profiles and clinical implication in pancreatic cancer tissues and investigated its biological function during tumor progression.
Materials and Methods
Microarray and lncRNA Profile Mining
Microarray datasets from GSE1651523 and GSE32688 were acquired from GEO (http://www.ncbi.nlm.nih.gov/geo/). GSE16515 consisted of 36 pancreatic tumor samples and 16 normal samples, while GSE32688 consisted of 25 pancreatic tumor samples and 7 normal samples. GATExplorer24 was used to process microarrays for gene expressions of lncRNAs as previously described.25
Tissue Collection and RNA Extraction
This study was in accordance with the local Research Ethics Committee approval. A total of 86 patients admitted at People Hospital of Lishui, the Sixth Affiliated Hospital of Wenzhou Medical University (Zhejiang, China) from January 2007 to June 2010 were enrolled in this study after written informed consent was acquired. Patients had never undergone chemotherapy, radiation therapy, or any other types of anti-cancer therapies prior to surgery. The clinical stages were determined in compliance with the American Joint Committee on Cancer (AJCC) staging system. Pairs of on-site tumor tissues and adjacent non-tumor tissues were preserved and stored at −80°C. Overall survival (OS) was calculated from the dates of surgery to decrease (in month). RNA was isolated from the tissue samples using Trizol (Invitrogen, Carlsbad, CA USA) as per manufacturer’s guidance.
Quantitative Real-Time PCR (qRT-PCR)
qRT-PCR was conducted to determine expression levels of BC037916, miRNAs, and Twist using the SYBR Green Kit (Thermo Fisher Scientific, Waltham, MA, USA) and ABI 7300 System (Applied Biosystems, Foster City, CA, USA). The primers were listed in Supplemental Table S1. LncRNA and Twist expression was normalized to GAPDH, while miRNA expression was normalized to U6 small nuclear 1 (RNU6-1) using the formula 2−ΔΔCT.
Cell Lines
A normal pancreatic duct epithelial cell line, HPDE6-C7, and five pancreatic cancer cell lines (BXPC3, SW1990, PANC-1, CAPAN-1, and ASPC-1) were acquired from the Shanghai Cell Bank (Shanghai, China). Cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM, Invitrogen) containing 10% fetal bovine serum (FBS, Hyclone, Logan, UT, USA) and antibiotics in tissue culture incubators.
RNA Interference and Plasmid Transfection
Four siRNAs targeting human BC037916 (siRNA1, GAC GCT ATT CTA AGG ACC ATA; siRNA2, GTA GAA GAG CTA AGT GAA ATG; siRNA3, GCA GTC TTA GAG GGT TTG AGG; siRNA4, GCA GAG GGT AAT ACC AAT ACT) and one negative control siRNA (siNC) were obtained from Genepharma (Shanghai, China). The BC037916 full sequence was cloned into the pcDNA3.1 vector (Invitrogen). Knockdown and overexpression of BC037916 in pancreatic cancer cells was performed by transfection using Lipofectamine 2000 (Invitrogen) as per the manufacturer’s instruction.
Western Blotting
Protein lysates were prepared in RIPA buffer supplemented with protease inhibitor cocktails (Sigma, St. Louis, MO, USA) by centrifugation. A total of 30–40 μg of total protein resolved by 10% or 15% SDS-PAGE gels was blotted onto nitrocellulose membranes. After 30 min of blocking with 5% nonfat milk at room temperature, membranes were incubated with primary antibodies at 4°Covernight. Following incubation with horseradish peroxidase (HRP)-conjugated secondary antibody (Beyotime, Shanghai, China), enhanced chemiluminescence (ECL) (Bio-Rad, Hercules, CA, USA) was added to membrane for developing. Primary antibodies against PCNA, Cyclin D, Twist, MMP-2, MMP-9, pSTAT3, STAT3, and psmad2/3 were purchased from Abcam (Cambridge, United Kingdom). Antibodies for E-cadherin, b-catenin, pJAK2, JAK2 and smad2/3 and GAPDH were obtained from Cell Signaling Technology (Danvers, MA, USA). Anti-Bcl-2 and anti-Bax were from Santa Cruz Biotechnology (Dallas, TX, USA).
Cell Proliferation Assay
Cells were plated in 96-well plates (3×103/well), and either siRNA or lncRNA expression plasmid was transfected to cells. 0 h, 24 h, 48 h, and 72 h after transfection, Cell Count Kit-8 (CCK-8, Dojindo Laboratories, Tokyo, Japan) reagent was added and incubated at 37°C for 1 h. Absorbance at 450 nm was recorded using a plate reader (BioRad, Hercules, CA, USA).
Flow Cytometry Analyses of Cell Cycle and Apoptosis
At 48 h after transfection, cells were collected and washed once with ice-cold PBS. To assess cell cycle distribution, the cells were fixed in cold 70% ethanol at −20°C overnight, and washed twice with ice-cold PBS, then stained with 0.1 mg/mL propidium iodide (PI, Sigma) and 1 mg/mL RNase A at 37°C for 30 min. For cell apoptosis assay, the collected cells were stained with PI and annexin V-fluorescein isothiocyanate (FITC) (KeyGEN Biotech, Nanjing, China) at room temperature for 20 min. Cell cycle distribution and cell apoptosis were evaluated by the FACScan flow cytometer (BD Biosciences, San Jose, CA, USA).
Cell Invasion Assay
Cells were harvested 24 hours post-transfection, resuspended with serum-free DMEM medium, and plated in 24-well Matrigel invasion chambers (8 μm; BD Biosciences) at a density of 5×104/well. DMEM supplemented with 10% FBS as a chemoattractant was added to the bottom chamber. The noninvasive cells were carefully removed using a cotton swab at 48 h post incubation. After fixing with 4% paraformaldehyde, invasive cells were enumerated by staining with 0.2% crystal violet and microscopy.
miRNA Transfection
miR3145-3p mimic (5ʹ-AGAUAUUUUGAGUGUUUGGAAUUG-3ʹ), inhibitor (5ʹ-CAAUUCCAAACACUCAAAAUAUCU-3ʹ) and control (5ʹ-CAGUACUUUUGUGUAGUACAA-3ʹ) were synthesized by Genepharm Technologies (Shanghai, China). Transfection was carried out using the Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer’s instructions.
Luciferase Reporter Assay
The full-length human Twist wild-type (WT) 3ʹ-untranslated region (3ʹ-UTR) fragment and mutant 3ʹ UTR fragment with mutation in the putative miR-3145-3p binding sites were inserted into pGL3 Vector (Promega, Madison, WI). Luciferase activity was detected using the Dual-Luciferase Reporter Assay System (Promega).
Subcutaneous Xenografts in Nude Mice
Animal studies were conducted in compliance with the guidelines of the Animal Care and Use Committee of Fudan University Shanghai Cancer Center. Eight weeks old male athymic BALB/c nude mice were acquired from Slac Laboratory Animal Co. Ltd. (Shanghai, China). SW1990 cells transfected with siNC or siRNA4 were collected and subcutaneously (s.c.) injected into flanks of the mice (5×106/0.1 mL PBS). The mice were administered with siNC or siRNA4 containing formulations, respectively, via tail vein twice a week. Subcutaneous tumor volumes (V) were measured every three days with digital calipers and determined from the equation: V= (length × width2)/2. After 46 days, all mice were euthanized and tumors were harvested.
Statistical Analysis
Data were analyzed by SPSS software (v16.0, SPSS, Inc., Chicago, IL, USA) and shown as mean ± standard deviation (SD). Student’s t-test was carried out to compare the differences between the two groups. The clinical characteristics of tumors with differential BC037916 expression were determined by the Chi-square tests. Cox regression model was employed to determine univariate and multivariate hazard ratios. Patients’ survival analyses were carried out using Kaplan-Meier analysis and Log-rank test. P<0.05 represents statistical significance, with significance annotated as follows: *P<0.05, **P<0.01, and **P<0.001.
Results
Screen of Pancreatic Cancer-Specific LncRNA
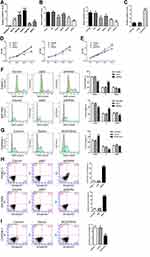
To characterize differentially expressed lncRNAs between pancreatic cancer tissues and normal tissues, GSE16515 (Figure 1A) and GSE32688 datasets (Figure 1B) were analyzed by Arraytools. The expression of lncRNA BC037916 is extremely higher in pancreatic cancer tissues than in normal tissues in both datasets (P<0.001).
BC037916 Was Overexpressed in Pancreatic Cancer
We then performed qRT-PCR to assess the expression of lncRNA BC037916 in 86 pairs of pancreatic tumor tissues and adjacent normal tissues collected from our own hospital and found that BC037916 expression was dramatically elevated in 86.0% (74/86) cancerous tissues in comparison with corresponding normal tissues (P<0.0001) (Figure 1C).
Correlation Analysis Between BC037916 Expression and the Clinical Parameters of Pancreatic Cancer
The 86 pancreatic cancer tissues were divided into BC037916 high expression group and BC037916 low expression group as values above and below the median, respectively (Figure 1D). We then explored the clinical relevance of BC037916 expression in pancreatic cancer by examining the association between its expression and patients’ clinical characteristics. As shown in Table 1, increased BC037916 expression was strongly related to clinical stage (P = 0.0202), primary tumor (P = 0.0164) and regional lymph node invasion (P = 0.0225), but not with other clinical parameters.
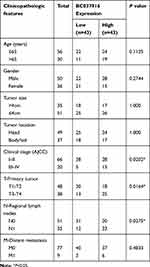 |
Table 1 Correlations Between BC037916 Expression and Clinicopathologic Features in Patients with Pancreatic Cancer |
BC037916 Serves as an Independent Prognostic Factor for Pancreatic Cancer
Univariate analysis was conducted and four prognostic factors, BC037916 expression (high vs low), clinical stage (I–II vs III–IV), primary tumor (T1-2 vs T3-4) and regional lymph node invasion (N0 vs N1) were identified as statistically indicative prognosis factors (Table 2). Further multivariate analysis revealed that these four factors may serve as prognostic factors for poor OS independently. Kaplan-Meier survival curves in Figure 1E showed that patients with higher BC037916 expression had inferior OS compared with those with lower expression (P<0.0001).
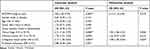 |
Table 2 Cox Regression Analysis of BC037916 Expression with Cancer-Specific Mortality in Pancreatic Cancer |
BC037916 Expression in Pancreatic Cancer Cells
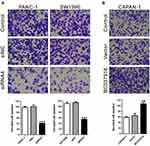
BC037916 expression in pancreatic cancer cell lines (BXPC3, SW1990, PANC-1, CAPAN-1, and ASPC-1) and a normal pancreatic duct epithelial cell line (HPDE6-C7) was assessed by qRT-PCR. As illustrated in Figure 2A, BC037916 expression was remarkably elevated in pancreatic cancer cells than in HPDE6-C7. Two cell lines, SW1990 and PANC-1, showing higher BC037916 expression were chosen for RNAi experiment. On the contrary, CAPAN-1 that has lower BC037916 expression was chosen for overexpression experiment.
Effect of BC037916 Expression Level on Cell Proliferation
Four siRNAs specific to BC037916 (siRNA1-4) and a control siRNA (siNC) were transfected into SW1990 and PANC-1cells. qRT-PCR assay revealed that siRNAs transfection significantly reduced the expression of BC037916 in both cell lines (Figure 2B). siRNA4 was the most effective one and chosen for the subsequent experiments. BC037916 was forced overexpressed in CAPAN-1 cells by transfection with plasmid expressing BC037916 (Figure 2C).
To investigate the potential function of BC037916 in pancreatic carcinogenesis, CCK-8 assay was conducted to assess the cell proliferation based on BC037916 expression levels. The results demonstrated that silencing of BC037916 by siRNA4 (Figure 2D) or siRNA3 (Supplemental Figure S1A) significantly repressed cell proliferation in both SW1990 and PANC-1 cells when compared with the wild-type (WT) cells and siNC transfected cells (Figure 2D). On the contrary, cell proliferation of CAPAN-1 cells was increased by BC037916 overexpression (Figure 2E).
Effects of BC037916 Expression Level on Cell Cycle Distribution and Cell Apoptosis
Cell cycle status was then evaluated in BC037916 knocked down and BC037916 overexpressed cells. Flow cytometry (FACS) analyses demonstrated that the percentage of G0/G1 phase was significantly reduced by 31.9% (P<0.01), while the percentage of S phase was notably increased by 85.5% in PANC-1 cells transfected with BC037916 siRNA4, compared with siNC transfected cells (Figure 2F). Similar results were obtained in SW1990 cells, while complementary results were observed in CAPAN-1 cells overexpressing BC037916 (Figure 2G).
Annexin V-FITC/PI staining and FACS analysis demonstrated that apoptotic ratio was significantly increased in BC037916 knocked down PANC-1 and SW1990 cells (Figure 2H), while BC037916 overexpressed CAPAN-1 cells showed reduced apoptotic ratio (Figure 2I).
Effect of BC037916 Expression Level on Cell Invasion
Next, we examined the invasive potential of PANC-1 and SW1990 cells transfected with siRNA4 or siRNA3 (Figure 3A and Supplemental Figure S1B), and CAPAN-1 cells overexpressing BC037916 (Figure 3B) by Matrigel invasion assays. The number of invasive cells was remarkably lower in siRNA4 or siRNA3 transfected cells, but notably higher in BC037916 overexpressed CAPAN-1 cells compared with control cells. These data suggested that BC037916 potentially contributed to the invasion of pancreatic cancer cells.
Downstream Affected by lncRNA BC037916
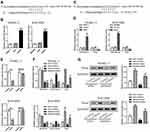
To analyze the expression of genes downstream of lncRNA BC037916, Western blotting was performed (Figure 4). In line with in vitro functional characterization, silencing of BC037916 led to notable decreases in the expression of genes related to cell growth (PCNA and Cyclin D), anti-apoptosis (Bcl-2), metastasis (MMP-2 and 9), epithelial–mesenchymal transition (EMT) related factors (Twist and β-catenin), and a remarkable up-regulation in the expression of apoptotic protein Bax and the main factor of EMT (E-cadherin) in PANC-1 and SW1990 cells compared to the control cells.
Moreover, TGF-β expression, as well as phosphorylation of JAK2, STAT3, and smad2/3 were significantly decreased in the BC037916 knocked down cells compared with the controls, while JAK2, STAT3, and smad2/3 protein expression were not affected, which indicated that lncRNA BC037916 may exert its function by inhibition JAK2/STAT3 and TGF-β/smad2/3-mediated signaling pathway.
BC037916 Positively Regulated the Expression of Twist Through miR-3145-3p
Accumulated evidence indicated that lncRNAs may sequester miRNAs, thereby reducing the binding of endogenous miRNAs to target genes.26 By using FINDTAR3 (http://bio.sz.tsinghua.edu.cn/), we found that miR-3145-3p, miR-4482-3p, and miR-12,136 have putative binding sites with BC037916 (Figure 5A and Supplemental Figure S2A). qRT-PCR analysis showed that BC037916 knockdown increased the expression of miR-3145-3p (Figure 5B), but had little effect on miR-4482-3p and miR-12,136 expression (Supplemental Figure S2B and 2C).
We then explored whether miR-3145-3p targeted the above downstream genes affected by lncRNA BC037916. By using TargetScan (http://www.targetscan.org/), Twist, the EMT related marker was found as the only potential target of miR-3145-3p (Figure 5C). In qRT-PCR analysis, miR-3145-3p mimic significantly suppressed Twist expression in PANC-1 and SW1990 cells (Figure 5D). Further luciferase reporter assay showed that miR-3145-3p mimic inhibited luciferase activity of Twist 3ʹUTR (WT), but had no effects on that of mutant 3ʹUTR (Figure 5E). These data suggested that miR-3145-3p directly bound to Twist 3ʹUTR, which resulted in decreased Twist expression.
We then explored whether inhibition of miR-3145-3p could reverse BC037916 knockdown-decreased Twist expression. As expected, downregulation of miR-3145-3p with miR-3145-3p inhibitor efficiently reversed the inhibition of Twist mRNA (Figure 5F) and protein levels (Figure 5G) by BC037916 knockdown. These analyses show that BC037916 promoted Twist expression through downregulating miR-3145-3p.
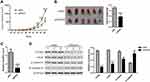
BC037916 Knockdown Suppressed Tumor Growth in Nude Mice
Xenograft experiments were conducted to verify the effect of lncRNA BC037916 in vivo. As shown in Figure 6A, BC037916 knockdown in SW1990 cells significantly reduced tumor burden. At 46 days after cell injection, the average weight of tumors established from BC037916 knockdown cells was 45.5% lower as compared with that from control cells (Figure 6B). BC037916 expression decreased 77.2% in tumors derived from BC037916 knockdown cells (Figure 6C). These results indicated that downregulation of BC037916 hampered tumor growth in vivo. Western blotting confirmed that PCNA, Twist, and β-catenin expressions were also significantly decreased, and E-cadherin was remarkably increased in tumors derived from BC037916 knocked down cells (Figure 6D).
Discussion
Gene expression profiles using microarray analyses have provided genome-wide biological information to help distinguish the pancreatic cancer subtypes.27–30 LncRNAs, as a novel class of functional molecules involved in the development and progression of cancer, may also supply valuable information to reveal the mechanisms of the progression of pancreatic cancer.12,19–21 In this study, we identified BC037916 as the most significantly up-regulated lncRNA in pancreatic cancer tissues by GATExplorer on publicly available datasets. BC037916, a novel lncRNA transcript, was previously recognized as a Homo sapiens cDNA clone.31 The biological functions of lncRNA BC037916 remain unknown. In the present study, we confirmed that lncRNA BC037916 is extremely elevated in 86 pancreatic cancer tissues, compared with normal tissues (Figure 1). The relationship between BC037916 expression levels and certain clinicopathologic parameters was then evaluated (Table 1). Our study revealed that BC037916 expression in pancreatic cancer was related to the clinical stage, primary tumor, and regional lymph node invasion. Importantly, Kaplan-Meier survival curves (Figure 1E) and univariate analysis (Table 2) showed increased BC037916 expression was remarkably correlated with poor OS rate of pancreatic cancer patients. Multivariate analysis implied BC037916 as an independent risk indicator for poor prognosis of pancreatic cancer (Table 2). Collectively, our study suggests that BC037916 serves as a novel prognostic marker for pancreatic cancer patients.
Next, we characterized the biological functions of BC037916 by gain-of-function and loss-of-function experiments. We demonstrated that BC037916 facilitated cell proliferation, disrupted cell cycle progression, and facilitated cell invasion, but inhibited cell apoptosis in pancreatic cancer. BC037916 overexpression in CAPAN-1 cells induced G0/G1 arrest, suppressed cell apoptosis, and promoted cell proliferation, which suggested that increased cell growth may be ascribed to the suppressed cell apoptosis. This is the first study on the functional significance of BC037916 in pancreatic cancer, showing that BC037916 has oncogenic potential in the pancreatic cancer development.
lncRNAs have been suggested to promote cancer progression and metastasis by regulating the expression of protein-coding genes.6,8,12 We then determine whether lncRNA BC037916 regulates the protein expression of cancer-related molecules. As expected, our data suggested that knockdown BC037916 can modulate cell growth, apoptosis, metastasis, and EMT related factors, including down-regulating PCNA, Bcl-2, MMP-2, MMP-9, Twist, and β-catenin, as well as up-regulating Bax and E-cadherin (Figure 4). Currently, JAK/STAT signaling is responsible for regulating multiple cellular activities in tumorigenesis, such as cell cycle progression, apoptosis, angiogenesis, and tumor cell invasion.32 As a member of JAK/STAT signaling pathway, STAT3 is phosphorylated in response to extracellular signals. Once phosphorylated, STAT3 dimers translocate to the nucleus and activates the transcription of target genes.33 Constitutive activation of STAT3 is frequently found in various human tumors including pancreatic cancer.34–37 Moreover, TGF-β signaling has been demonstrated to trigger EMT and promote tumor invasion.38 Here, we observed lower levels of TGF-β, psmad2/3, pJAK2, and pSTAT3 in BC037916 knockdown cells than in control cells. Our data suggested that activity of JAK2/STAT3 and TGF-β/smad2/3 signaling pathways can be suppressed by BC037916 knockdown, which partially triggers cell proliferation, EMT, and invasion. Moreover, it has been recently shown that lncRNAs regulate miRNA targets by interfering with miRNA pathways.26 Our study has shown that BC037916 knockdown increased the expression of miR-3145-3p, which may target Twist. Downregulation of miR-3145-3p efficiently reversed the suppression of Twist induced by BC037916 knockdown (Figure 5). Although our data suggested lncRNA BC037916 being an oncogene and targeting miR-3145-3p/Twist in pancreatic cancer, other direct downstream targets of BC037916 could not be ruled out. Therefore, further studies such as immunoprecipitation are needed to identify the proteins or microRNAs that directly bind to BC037916.
Conclusion
In summary, our findings suggested that lncRNA BC037916 could be a useful prognostic marker in pancreatic cancer patients. LncRNA BC037916 could promote cell proliferation and invasion via JAK2/STAT3 and TGF-β/smad2/3 signaling in pancreatic cancer. Our findings demonstrate that BC037916 is a potential oncogene participating in pancreatic cancer pathogenesis.
Abbreviations
GEO, Gene Expression Omnibus; lncRNAs, long noncoding RNAs; AJCC, American Joint Committee on Cancer; OS, overall survival; qRT-PCR, quantitative real-time PCR; RNU6-1, U6 small nuclear 1; DMEM, Dulbecco’s modified Eagle’s medium; FBS, fetal bovine serum; HRP, horseradish peroxidase; ECL, enhanced chemiluminescence; FITC, fluorescein isothiocyanate; PI, propidium iodide; WT, wild-type; UTR, untranslated region fragment; SD, standard deviation; EMT, epithelial–mesenchymal transition.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
This study was approved by the Ethics Review Committee of People Hospital of Lishui, the Sixth Affiliated Hospital of Wenzhou Medical University (Zhejiang, China). Animal studies were conducted in compliance with the guidelines of the Animal Care and Use Committee of Fudan University Shanghai Cancer Center.
Consent for Publication
Not applicable.
Acknowledgments
This study was supported by grant from the National Natural Science Foundation of China (81573757).
Disclosure
The authors declare that they have no competing interests.
References
1. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. doi:10.3322/caac.21208
2. Ryan DP, Hong TS, Bardeesy N. Pancreatic adenocarcinoma. N Engl J Med. 2014;371(11):1039–1049. doi:10.1056/NEJMra1404198
3. The Lancet O. Pancreatic cancer in the spotlight. Lancet Oncol. 2014;15(3):241. doi:10.1016/S1470-2045(14)70097-X
4. Stathis A, Moore MJ. Advanced pancreatic carcinoma: current treatment and future challenges. Nat Rev Clin Oncol. 2010;7(3):163–172. doi:10.1038/nrclinonc.2009.236
5. Consortium EP. The ENCODE (ENCyclopedia Of DNA Elements) project. Science. 2004;306(5696):636–640.
6. Huang JL, Ren TY, Cao SW, et al. HBx-related long non-coding RNA DBH-AS1 promotes cell proliferation and survival by activating MAPK signaling in hepatocellular carcinoma. Oncotarget. 2015;6(32):33791–33804. doi:10.18632/oncotarget.5667
7. Bida O, Gidoni M, Ideses D, Efroni S, Ginsberg D. A novel mitosis-associated lncRNA, MA-linc1, is required for cell cycle progression and sensitizes cancer cells to Paclitaxel. Oncotarget. 2015;6(29):27880–27890. doi:10.18632/oncotarget.4944
8. Liu Y, Zhao J, Zhang W, et al. lncRNA GAS5 enhances G1 cell cycle arrest via binding to YBX1 to regulate p21 expression in stomach cancer. Sci Rep. 2015;5:10159. doi:10.1038/srep10159
9. Yao J, Zhou B, Zhang J, et al. A new tumor suppressor LncRNA ADAMTS9-AS2 is regulated by DNMT1 and inhibits migration of glioma cells. Tumour Biol. 2014;35(8):7935–7944. doi:10.1007/s13277-014-1949-2
10. Gupta RA, Shah N, Wang KC, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature. 2010;464(7291):1071–1076. doi:10.1038/nature08975
11. Kino T, Hurt DE, Ichijo T, Nader N, Chrousos GP. Noncoding RNA gas5 is a growth arrest- and starvation-associated repressor of the glucocorticoid receptor. Sci Signal. 2010;3(107):ra8. doi:10.1126/scisignal.2000568
12. Cheng Y, Jutooru I, Chadalapaka G, Corton JC, Safe S. The long non-coding RNA HOTTIP enhances pancreatic cancer cell proliferation, survival and migration. Oncotarget. 2015;6(13):10840–10852. doi:10.18632/oncotarget.3450
13. Nie Y, Liu X, Qu S, Song E, Zou H, Gong C. Long non-coding RNA HOTAIR is an independent prognostic marker for nasopharyngeal carcinoma progression and survival. Cancer Sci. 2013;104(4):458–464. doi:10.1111/cas.12092
14. Sun J, Chen X, Wang Z, et al. A potential prognostic long non-coding RNA signature to predict metastasis-free survival of breast cancer patients. Sci Rep. 2015;5:16553. doi:10.1038/srep16553
15. Tsai MC, Spitale RC, Chang HY. Long intergenic noncoding RNAs: new links in cancer progression. Cancer Res. 2011;71(1):3–7. doi:10.1158/0008-5472.CAN-10-2483
16. Matouk IJ, DeGroot N, Mezan S, et al. The H19 non-coding RNA is essential for human tumor growth. PLoS One. 2007;2(9):e845. doi:10.1371/journal.pone.0000845
17. Nagano T, Fraser P. No-nonsense functions for long noncoding RNAs. Cell. 2011;145(2):178–181. doi:10.1016/j.cell.2011.03.014
18. Wilusz JE, Sunwoo H, Spector DL. Long noncoding RNAs: functional surprises from the RNA world. Genes Dev. 2009;23(13):1494–1504. doi:10.1101/gad.1800909
19. Kim K, Jutooru I, Chadalapaka G, et al. HOTAIR is a negative prognostic factor and exhibits pro-oncogenic activity in pancreatic cancer. Oncogene. 2013;32(13):1616–1625. doi:10.1038/onc.2012.193
20. Pang EJ, Yang R, Fu XB, Liu YF. Overexpression of long non-coding RNA MALAT1 is correlated with clinical progression and unfavorable prognosis in pancreatic cancer. Tumour Biol. 2015;36(4):2403–2407. doi:10.1007/s13277-014-2850-8
21. Ellis BC, Molloy PL, Graham LD. CRNDE: A long non-coding RNA involved in cancer, neurobiology, and development. Front Genet. 2012;3:270. doi:10.3389/fgene.2012.00270
22. Derrien T, Johnson R, Bussotti G, et al. The GENCODE v7 catalog of human long noncoding RNAs: analysis of their gene structure, evolution, and expression. Genome Res. 2012;22(9):1775–1789. doi:10.1101/gr.132159.111
23. Pei H, Li L, Fridley BL, et al. FKBP51 affects cancer cell response to chemotherapy by negatively regulating Akt. Cancer Cell. 2009;16(3):259–266. doi:10.1016/j.ccr.2009.07.016
24. Risueno A, Fontanillo C, Dinger ME. GATExplorer: genomic and transcriptomic explorer; mapping expression probes to gene loci, transcripts, exons and ncRNAs. BMC Bioinform. 2010;11:221. doi:10.1186/1471-2105-11-221
25. Pang KC, Stephen S, Engstrom PG, et al. RNAdb–a comprehensive mammalian noncoding RNA database. Nucleic Acids Res. 2005;33(Databaseissue):D125–130.
26. Paraskevopoulou MD, Hatzigeorgiou AG. Analyzing miRNA–lncRNA interactions. In: Long Non-Coding RNAs. Methods Mol Biol. 2016;1042:271–286.
27. Nakamura T, Furukawa Y, Nakagawa H, et al. Genome-wide cDNA microarray analysis of gene expression profiles in pancreatic cancers using populations of tumor cells and normal ductal epithelial cells selected for purity by laser microdissection. Oncogene. 2004;23(13):2385–2400. doi:10.1038/sj.onc.1207392
28. Logsdon CD, Simeone DM, Binkley C, et al. Molecular profiling of pancreatic adenocarcinoma and chronic pancreatitis identifies multiple genes differentially regulated in pancreatic cancer. Cancer Res. 2003;63(10):2649–2657.
29. Harada T, Chelala C, Crnogorac-Jurcevic T, Lemoine NR. Genome-wide analysis of pancreatic cancer using microarray-based techniques. Pancreatology. 2009;9(1–2):13–24. doi:10.1159/000178871
30. Grutzmann R, Saeger HD, Luttges J, et al. Microarray-based gene expression profiling in pancreatic ductal carcinoma: status quo and perspectives. Int J Colorectal Dis. 2004;19(5):401–413. doi:10.1007/s00384-003-0563-3
31. Strausberg RL, Feingold EA, Grouse LH, et al. Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences. Proc Natl Acad Sci U S A. 2002;99(26):16899–16903.
32. Greten FR, Karin M. Peering into the aftermath: jAKi rips STAT3 in cancer. Nat Med. 2010;16(10):1085–1087. doi:10.1038/nm1010-1085
33. Bowman T, Garcia R, Turkson J, Jove R. STATs in oncogenesis. Oncogene. 2000;19(21):2474–2488. doi:10.1038/sj.onc.1203527
34. Dhir R, Ni Z, Lou W, DeMiguel F, Grandis JR, Gao AC. Stat3 activation in prostatic carcinomas. Prostate. 2002;51(4):241–246. doi:10.1002/pros.10079
35. Grandis JR, Drenning SD, Zeng Q, et al. Constitutive activation of Stat3 signaling abrogates apoptosis in squamous cell carcinogenesis in vivo. Proc Natl Acad Sci U S A. 2000;97(8):4227–4232. doi:10.1073/pnas.97.8.4227
36. Niu G, Bowman T, Huang M, et al. Roles of activated Src and Stat3 signaling in melanoma tumor cell growth. Oncogene. 2002;21(46):7001–7010. doi:10.1038/sj.onc.1205859
37. Scholz A, Heinze S, Detjen KM, et al. Activated signal transducer and activator of transcription 3 (STAT3) supports the malignant phenotype of human pancreatic cancer. Gastroenterology. 2003;125(3):891–905. doi:10.1016/S0016-5085(03)01064-3
38. Heldin CH, Landstrom M, Moustakas A. Mechanism of TGF-beta signaling to growth arrest, apoptosis, and epithelial-mesenchymal transition. Curr Opin Cell Biol. 2009;21(2):166–176. doi:10.1016/j.ceb.2009.01.021
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.