Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Optimizing stem cell functions and antibacterial properties of TiO2 nanotubes incorporated with ZnO nanoparticles: experiments and modeling
Authors Liu W, Su P, Gonzales III A, Chen S, Wang N, Wang J, Li H , Zhang Z, Webster T
Received 16 September 2014
Accepted for publication 11 November 2014
Published 12 March 2015 Volume 2015:10(1) Pages 1997—2019
DOI https://doi.org/10.2147/IJN.S74418
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lei Yang
This paper has been retracted.
Wenwen Liu,1–3 Penglei S Su,2 Arthur Gonzales III,3 Su C Chen,1 Na Wang,1 Jinshu Wang,2 Hongyi Li,2,4 Zhenting Zhang,1
Thomas J Webster3,5
1Laboratory of Biomaterials and Biomechanics, Beijing Key Laboratory of Tooth Regeneration and Function Reconstruction, School of Stomatology, Capital Medical University, 2Photoelectrochemical Research Group, Key Laboratory of Advanced Functional Materials, School of Materials Science and Engineering, Beijing University of Technology, Beijing, People’s Republic of China; 3Chemical Engineering Department, Northeastern University, Boston, MA, USA; 4Guangxi Research Institute of Chemical Industry, Nanning, People’s Republic of China; 5Center of Excellence for Advanced Materials Research, King Abdulaziz University, Jeddah, Saudi Arabia
Abstract: To optimize mesenchymal stem cell differentiation and antibacterial properties of titanium (Ti), nano-sized zinc oxide (ZnO) particles with tunable concentrations were incorporated into TiO2 nanotubes (TNTs) using a facile hydrothermal strategy. It is revealed here for the first time that the TNTs incorporated with ZnO nanoparticles exhibited better biocompatibility compared with pure Ti samples (controls) and that the amount of ZnO (tailored by the concentration of Zn(NO3)2 in the precursor) introduced into TNTs played a crucial role on their osteogenic properties. Not only was the alkaline phosphatase activity improved to about 13.8 U/g protein, but the osterix, collagen-I, and osteocalcin gene expressions was improved from mesenchymal stem cells compared to controls. To further explore the mechanism of TNTs decorated with ZnO on cell functions, a response surface mathematical model was used to optimize the concentration of ZnO incorporation into the Ti nanotubes for stem cell differentiation and antibacterial properties for the first time. Both experimental and modeling results confirmed (R2 values of 0.8873–0.9138 and 0.9596–0.9941, respectively) that Ti incorporated with appropriate concentrations (with an initial concentration of Zn(NO3)2 at 0.015 M) of ZnO can provide exceptional osteogenic properties for stem cell differentiation in bone cells with strong antibacterial effects, properties important for improving dental and orthopedic implant efficacy.
Keywords: titanium nanotubes, ZnO nanoparticles, mesenchymal stem cells, antibacterial effect, modeling
Introduction
Titanium (Ti) and its oxide equivalent, titania, have been widely used for numerous dental and orthopedic applications.1 For dental and orthopedic implantation, early bone formation and osseointegration are essential for the success of the implant. However, insufficient cytocompatibility properties leading to initial and prolonged bone growth have been frequently observed when applying Ti for dental/orthopedic applications.2 It is for these reasons that numerous researchers have been trying to improve properties of Ti for orthopedic applications, such as altering its chemistry, crystallinity, or even roughness. For example, titania (TiO2) forms on the surface of Ti in oxygen environments, and the anatase phase of TiO2 has been shown to possess better cytocompatibility properties than the rutile phase of TiO2.3
On the other hand, roughening of the Ti surface can be another strategy to promote bone cell functions. Specifically, nanotopographical features on Ti can provide a similar structure with bone’s natural hierarchical nanostructure.4 In fact, our previous studies suggest that TiO2 nanotubes (TNTs) with a diameter of 70 nm (fabricated by electrochemical anodization) can promote bone regeneration in vivo.5 To further improve the osteogenic properties of TiO2, bioactive molecules (such as bone growth factors and bone morphogenetic proteins) can be embedded into TNTs.6–8 However, growth factors and proteins are easily degraded at high temperatures used during the sterilization processes.9 Similarly, the release of bioactive proteins from TNTs may not be stable enough in vivo and may result in large initial release profiles unhealthy for new bone formation.
To obtain a more stable release of bone growing agents, metals in nanoparticle form can be added into TNTs. For example, strontium (Sr) was reported10 to promote osseoinduction with anabolic activity. However, Sr is not a common metallic element in the body, and doses of Sr should be low (20 μg/mL) to avoid toxicity or any deleterious effect on bone mineralization.11
In contrast, Zn is an important trace element in the human body and can improve DNA synthesis, enzyme activity, and nucleic acid metabolic activity.12 Zn ions have the ability to promote osteoblast proliferation and enhance biomineralization.13,14 Nano-sized zinc oxide (ZnO) particles exhibit unique biological properties different from their bulk micron counterparts, due to their increased specific surface area.15 Moreover, the nanosize of ZnO increases the possibility of cellular uptake interactions between ZnO and biological molecules or tissues.16,17 Nano ZnO-decorated TNTs also have antibacterial effects, which is important for various implant applications.18
However, a high number of Zn ions will lead to cytotoxicity.18 To maintain cell functions and balance an antibacterial effect, it is becoming important to identify an optimal amount of ZnO. To the best of our knowledge, there are no reported literature studies focusing on modeling and optimization of the amount of ZnO used in Ti-based implants. In this study, quadratic and cubic response surface models, according to response surface methodology (Supplementary materials), were identified and fitted to the experimental data. The optimum conditions were also evaluated using these models to estimate the best combination of several parameters. The goals were combined into an overall desirability function. The program then maximized this function. The goal begins at a random starting point and proceeds up the steepest slope to a maximum. There may be two or more maximums because of curvature in the response surfaces and their combination into the desirability function.
Different concentrations of ZnO nanoparticles in TNTs were used here to investigate osteogenic properties. Osteogenic-related gene expression and related osteogenic proteins were measured to elucidate the osteogenic properties of the ZnO-incorporated samples; the results provided much promise for the continued investigation of ZnO-incorporated TNTs. Both the experimental and modeling results confirmed that TNTs incorporated with nano-sized ZnO can improve cell functions and antibacterial properties.
Experimental section
Sample preparation and characterization
Preparation of TNTs
Pure Ti sheets (Aldrich, 10×10×0.3 mm3) were used as substrates. After ultrasonically cleaning the samples with acetone, ethyl alcohol, and deionized water, anodization was carried out at 30 V for 2 hours to create TNTs on the surface of Ti sheets. The electrolyte was ethylene glycol containing 0.3 wt% ammonium fluoride (NH4F) and 2 vol% distilled water. Pure Ti was used as a control sample. The anatase crystalline phase of TiO2 has a higher corrosion resistance and better biocompatibility than amorphous TNTs. Thus, all amorphous TNTs phases were transformed into an anatase crystalline structure by annealing the samples at 450°C for 3 hours.
Incorporation of ZnO nanoparticles into TNTs
Samples were hydrothermally reacted at 70°C for 2 hours in a solution that contained 2 mg of citric acid, and zinc nitrate (Zn(NO3)2) and hexamethylenetetramine ((CH2)6N4) at a 1:2 molar ratio. Four different concentrations of Zn(NO3)2 (0.005 M, 0.015 M, 0.03 M, and 0.075 M) were used to introduce nano-sized ZnO into TNTs (TNT-Zn). All samples were annealed at 300°C for 100 minutes and then sterilized in an autoclave at 120°C for 40 minutes before in vitro experiments.
Characterization of samples
Field-emission scanning electron microscopy (SU8000 Series UHR Cold-Emission FE-SEM; Hitachi Ltd., Tokyo, Japan), atomic force microscopy (AutoProbe CP; Park Scientific Instruments, Sunnyvale, CA, USA), and transmission electron microscopy (JEM-2100F, JEOL, Japan) were used to observe the surface morphologies of the samples. X-ray diffraction (XRD; Philips X’Pert PRO, MA, USA) and energy-dispersive X-ray spectrometry (EDS; Hitachi) were used to analyze the sample phase compositions and distribution of Zn. X-ray photoelectron spectroscopy (XPS; ESCALAB MK-II; VG Scientific, UK) was used to identify the bonding states of the surface constituents.
Zn ion release and TNT incorporating capacity
The TNT-Zn samples were immersed in 4 mL of phosphate-buffered saline (PBS) for 9 days to monitor the amount of Zn2+ released from the samples. Different time points (1 day, 3 days, 5 days, 7 days, and 9 days) were used, and the amount of Zn2+ released was determined by inductively coupled plasma atomic emission spectrometry (ICP-AES; Varian Vista AX, MA, USA). The total amount of Zn ions in the TNTs was determined by an X-ray fluorescence spectrometer (X-Supreme8000; Oxford instruments, Austin, USA) and ICP-AES. After being measured by an X-ray fluorescence spectrometer, samples were dissolved in a solution of 1% HF and 1.5% HNO3 for ICP-AES experiments.
Polarization curve measurements
Polarization curves were determined in a three-electrode system to test the anticorrosion ability of the materials. For this, a 500-mL test solution was constructed from Essential Medium Alpha (MEM α; product line of Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (FBS, product line of Thermo Fisher Scientific). A saturated calomel electrode served as the reference electrode, and a platinum electrode served as the counter electrode. Samples were immersed into the solution vertically. The polarization curves were then recorded.
Contact angle and surface roughness analysis
A contact angle analysis system (Model OCA20; Data Physics Co., Ltd.) was used to determine surface contact angles for the samples. The contact angles at three different locations on each sample were calculated to obtain the mean value. Distilled water, diiodomethane, and ethylene glycol were used as the test fluids whose surface energy was known. Based on the surface energy parameters of these different liquids, surface energy was calculated according to previously described methods.5,19
The surface roughness of the samples was measured by a surface profiler (DektakXT profiler; Bruker) at a 2 mm scan distance and a scan rate of 0.2 mm/s. The Ra values were reported as the mean ± standard deviation (n=3).
Protein adsorption assay
Bovine serum albumin (BSA; Sigma-Aldrich Co., St Louis, MO, USA) and fibronectin (FN; Sigma-Aldrich Co.) were used as model proteins. All samples were soaked in a 200 μL protein solution (1 mg/mL BSA or FN). After 2 hours and 24 hours at 37°C, the samples were transferred to a new 24-well plate (one sample per well) and were washed three times with PBS. In all, 200 μL of a 2% sodium dodecyl sulfate (SDS; Sigma-Aldrich Co.) solution was added to the wells and was shaken for 2 hours to detach proteins from the sample surfaces. The SDS solution with the collected proteins was assessed for protein content by a Micro BCA Protein Assay Kit (Pierce, Rockford, USA), and the protein concentrations were quantified using a microplate spectrophotometer (SpectraMax Plus 384) at 562 nm.
Mesenchymal stem cell assays
Mesenchymal stem cell culture: Six-week-old male Sprague Dawley rats were used to extract bone marrow for bone mesenchymal stem cells (BMSCs). BMSCs were cultured using Minimum Essential Medium α (MEM α, product line of Thermo Fisher Scientific, Waltham, MA, USA) with 10% FBS (Gibco BRL) and 1% penicillin/streptomycin in a 5% CO2 incubator at 37°C. Cell culture media were changed every 2 or 3 days. BMSCs at passage numbers 2–3 were used in the experiments. The experimental protocol in this study was reviewed and approved by the Animal Care and Use Committee of the Capital Medical University, People’s Republic of China.
Stem cell morphology
BMSCs were seeded at a density of 1×104 cells/well. After 24 hours of incubation, the cells on samples were washed with PBS and fixed with 4% formaldehyde. Then, the cells were treated with 1% Triton X-100 and blocked with 2% BSA. The samples were incubated with anti-vinculin mAb (V9264; Sigma-Aldrich Co.) as the primary antibody for 1 hour. After washing in PBS, fluorescein isothiocyanate (F2562, Sigma-Aldrich Co.) was added to the surfaces and incubated for 2 hours. The actin cytoskeleton was labeled by incubating the cells with Alexa Fluor 594 Phalloidin (Invitrogen). At last, the cell nuclei were contrast-labeled by DAPI (4’,6-diamidino-2-phenylindole) (Thermo Fisher Scientific). Cells were then observed using confocal laser scanning microscopy.
The morphology of the cells on the samples was also observed using SEM. Cells were seeded at a density of 1×104 cells/well in the same manner as described above. After culturing for 24 hours, the cells on the samples were fixed, dehydrated, coated with gold, and observed using FE-SEM.
Stem cell lactate dehydrogenase activity assay
To determine the cytotoxicity of the samples toward BMSCs, the activity of lactate dehydrogenase (LDH) in the culture media was used as an index. For this, 1×104 BMSCs/well were cultured on the surface of the samples for 1 day and 4 days. The culture media was collected and centrifuged. The supernatant was then tested using an LDH kit following manufacturer’s instructions (Sigma-Aldrich Co.). LDH activity was calculated by the relative optical density (OD) values:
| (1) |
Stem cell proliferation assays
The proliferation of BMSCs on the samples was investigated with the Cell Counting Kit-8 (CCK-8; Dongjing Molecular Technologies, Tabaru, Japan) after 1 day, 4 days, and 7 days of culturing. For this, the cells were inoculated on the samples in 24-well plates at a density of 2×104 cells/well. After each time point, the cells were rinsed with PBS and transferred to a new 24-well plate (each sample per well). In all, 300 μL of media (MEM α with 10% FBS and 1% penicillin/streptomycin) with 30 μL of a CCK-8 solution were added to each well. The samples were cultured for another 2 hours at 37°C. At last, 150 μL of the solution from each well was transferred to a 96-well plate. The absorbance from each solution was measured at a wavelength of 450 nm with a microplate spectrophotometer (Model SpectraMax Plus 384).
Stem cell alkaline phosphatase activity and intracellular total protein synthesis
Cells were seeded on samples at a density of 2×104/well and were incubated in osteogenic differentiation medium (MEM, 10% FBS, 5 μg/mL gentamicin, and 2 mM GlutaMAX™; product line of Thermo Fisher Scientific). The osteogenic medium was replaced every other day. After culturing for 3 days and 7 days, the cells were lysed in a 2% SDS solution using ultrasonication in ice water. Then, alkaline phosphatase (ALP) activity was determined by the ALP reagent, which contained p-nitrophenyl phosphate (Sigma-Aldrich Co.). The ALP activity was assayed by measuring OD values at 405 nm. The intracellular total protein content was determined by a Micro BCA Protein Assay Kit, and the ALP activity was normalized to it.
Stem cell osteogenic-related gene expression
In a further attempt to determine the mechanism by which stem cell functions were influenced by the substrates of interest of the present study, stem cell osteogenic gene expression was determined. For this, BMSCs were seeded on the samples at a density of 2×104/well and were cultured in osteogenic differentiation medium (MEM, 10% FBS, gentamicin, and 2 mM GlutaMAX™). The samples with adherent cells were collected after 3 days, 7 days, and 14 days of culture for real-time polymerase chain reaction (RT-PCR) analysis.
The total RNA was isolated and collected by TRIzol (Thermo Fisher Scientific). Rat mRNA encoding genes for ALP, collagen-I (col-I), osteocalcin (OC), and osterix (Osx) were determined, and the housekeeping gene GAPDH was used as the internal control gene to normalize the quantities of the target genes. Reverse transcription and RT-PCR were used as previously reported.5 Primers used for the target and housekeeping genes are shown in Table 1. Lastly, the results of the RT-PCR were determined by the 2−ΔΔCt method.
Antibacterial assay
In bacterial cultures, the ability of the proposed materials to reduce bacterial growth was evaluated using Streptococcus mutans (S. mutans, UA159) and Porphyromonas gingivalis (P. gingivalis, ATCC33277). The culture and analysis method was followed according to a previous study.18
Modeling
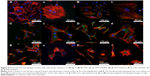
Each response (Tables S1–S8) was correlated to the experimental parameters and fitted to the surface defined by
| (2) |
where Y is the predicted response; xi, xj, and xk are the coded experimental parameters; and a0, bi, cij, and dijk are the constant, linear, quadratic, and cubic coefficients, respectively, where dijk =0 when the model is quadratic. When i≠j or i≠k, they are called the interaction coefficients.
The experimental data were fitted to the model using Design-Expert 7.0 software (Stat-Ease Inc., Minneapolis, MN, USA). It was used for regression analysis, evaluation of the statistical significance of the equation, and searching for the optimal combination of parameters for a best local maximum. By starting from several points in the design space, chances improved for finding the best local maximum. The default was 30 starting points. The cell proliferation (OD) value data with respect to time (days), the initial Zn(NO3)2 concentration (M), the total Zn2+ concentration (ppm), the reduction rates of S. mutans and P. gingivalis bacteria with respect to time (days), the initial Zn(NO3)2 concentration (M), and the total Zn2+ concentration (ppm) were analyzed. The experimental design matrix is given in the supplementary material.
Statistical analysis
All experiments were conducted in triplicate. Statistically significant differences (P<0.05) were measured using one-way ANOVA combined with the Student–Newman–Keuls post hoc test. Data were expressed as the mean ± standard deviation.
Results and discussions
Substrate characterization
As SEM and transmission electron microscopy analysis showed, the diameter of the TNTs was about 70 nm and the length was about 2 μm (Figure 1). Surface features from the Ti, TNTs, and TNT-Zn0.015 samples are shown in the atomic force microscopy images (Figure S1), which confirmed the expected nanotubular dimensions.
The ratio of Ti to O for all the TNTs showed that the TNTs mainly consisted of TiO2, as expected (Table 2). For the ZnO-incorporated samples, EDS results showed that the ZnO content from the samples increased along with increasing concentrations of Zn(NO3)2.
The XRD spectra (Figure 2A) of the samples displayed Ti, anatase TiO2 peaks, and some small peaks of rutile phase for the TNTs and TNT-Zn0.015. There were no feature peaks of Zn2+ compounds in the TNT-Zn0.015; most likely this is because the content of ZnO was too low to be detected by XRD.
Further XPS studies revealed that Ti, O, and Zn peaks were found for all the ZnO-incorporated samples (Figure 2B). The C signals are most likely present due to contamination. The XPS depth profile showed the distribution of ZnO in the TNTs (Figure 2C).
The results of contact angle and surface energy measurements showed significant differences for the samples of interest to this study (Figure S2). The samples decorated with ZnO had lower roughness values than that of plain Ti and TNT. The surface energies of the plain Ti and ZnO-decorated samples were lower than that of TNT.
The corrosion potential and corrosion current density of pure Ti was −0.35 V (vs saturated calomel electrode) and 10−6 A/cm2, respectively (Figure S3A). Meanwhile, for the anodized TiO2 samples, the corrosion potential was almost equal to that of pure Ti; however, the corrosion current density was about 10−8 A/cm2.
The Zn2+ release rate followed the order TNT-Zn0.075>TNT-Zn0.03>TNT-Zn0.015>TNT-Zn0.005 (Figure S3B). As expected, the amount of released Zn2+ diminished with time. The accumulated amount increased steadily during the first 9 days.
ZnO nanoparticles were distributed homogeneously along the walls of TNT. EDS results showed that the amount of ZnO content in samples TNT-Zn0.03 and TNT-Zn0.075 was greater than that of the other two. The results indicated that ZnO was embedded into the TNTs. In the XPS spectra of ZnO from TNT-Zn0.015, energies of the Zn 2p3 peak position located at 1,021.7 eV and 1,044.8 eV were assigned to the Zn 2p1 peak position, and this position did not shift with the depth of investigation. The binding energy of the Zn 2p3/2 peak was located at 1,021.7 eV, a perfect fit with Zn2+ in ZnO.
EDS and XPS spectrum further verified that nano-sized ZnO was successfully incorporated into TNTs. The ZnO embedded dose in TNTs can be tailored according to the concentrations of Zn(NO3)2 in the precursor. As expected, the surface modification of TiO2 with nanotubes improved hydrophilicity. Further, it was found that the surface energy decreased with the addition of ZnO nanoparticles in TNTs; however, this decrease was not obvious when concentrations of the Zn(NO3)2 in the precursor were below 0.015 M. The surface energy of TiO2 increased after anodization, which is important for attracting initial protein adsorption for promoting stem cell adhesion. Although the surface energy of TNTs decorated with ZnO nanoparticles decreased, TNT-Zn0.015 and TNT-Zn0.030 were still higher than that of pure Ti. The anticorrosion properties of the anodized TiO2 samples improved, which can be attributed to the TNT films formed on the surface of the Ti substrate. The TiO2 formed on the Ti surface provided a barrier, which prevented metal ion release from the metal matrix into the electrolyte. In our study, it was found that the TNT surface had significantly higher anticorrosion properties in α-MEM containing 10% FBS compared to pure Ti. It was further found that the anticorrosion properties of the anodized samples improved when ZnO was inserted into the nanotubes. When the concentration of Zn(NO3)2 in the precursor was 0.015 M and 0.03 M, the best anticorrosion properties were achieved. These results suggested that the ZnO incorporated into the TNTs can improve its anticorrosion properties, indicating a better biosafety and long-term performance.
Protein adsorption
Results from the present study showed that more BSA than FN adsorbed onto the TNT samples (Figure S4A and B). More importantly, all the experimental samples had greater BSA adsorption than control samples. The adsorption of BSA decreased with more ZnO incorporated into the TNT samples. In contrast, there was no significant difference in the adsorption of FN after 24 hours between all samples. However, after 2 hours of adsorption, TNTs and TNT-Zn0.015 had slightly more FN adsorption compared to the other samples, which may positively influence stem cell functions, especially adhesion due to the presence of the cell-adhesive arginine-glycine-aspartic acid amino acid sequence in FN.
LDH activity
It was found that (Figure 3A) after culturing for 1 day and 4 days, both TNT-Zn0.03 and TNT-Zn0.075 had significantly different LDH activities compared to all other samples. The other samples presented no trend of cytotoxicity. The same samples on different days had no significant difference. Clearly, a high enough concentration of ZnO will lead to cytotoxicity.
The cytotoxicity tests showed that TNT-Zn0.03 and TNT-Zn0.075 had significant cytotoxicity as LDH was used as a marker released from cells. The other groups had similar LDH values, and the results are consistent in terms of Zn2+ release. It can be concluded from the above statement that the Zn2+ concentration in TNT-Zn0.015 is the upper limit for biosafety among these samples.
Cell proliferation
Stem cell proliferation was significantly lower for the TNT-Zn0.030 and TNT-Zn0.075 samples than for any other groups after 1 day of culture (Figure 3B). In fact, after 4 days of culture, all cells died on the TNT-Zn0.075 sample. Keeping with this trend, the number of cells on TNT-Zn0.03 was significantly lower than controls after 4 days of culture. After 7 days of culture, all the cells on TNT-Zn0.03 died. After 7 days of culture, cell proliferation was significantly higher on control Ti and TNT-Zn0.015 than on TNTs and TNT-Zn0.005. Cell proliferation on pure Ti, TNT, TNT-Zn0.005, and TNT-Zn0.015 after 1 day, 4 days, and 7 days was significantly different.
The cells on pure Ti had higher proliferation rates than others, in which it has been reported that a higher cell density improves proliferation while a lower cell density (but confluent) promotes osteoblastic differentiation.5 The relatively lower cell densities on TNT-Zn0.005 and 0.015 samples may be good for cell differentiation.
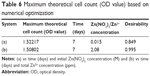
Cell morphology
On pure Ti, the stem cells spread relatively poorly with round shapes (Figure 4) after 1 day of culture. In contrast, the stem cells cultured on the surface of TNT-Zn0.005 and TNT-Zn0.015 had polygonal morphologies and more spread filopodia than all the other samples. The vinculin cell membrane protein was expressed more clearly on TNT-Zn0.005 and TNT-Zn0.015 than all the other samples. Vinculin formed dot-shaped structures on TNT-Zn samples, which indicated the formation of focal contacts between the cells and sample surfaces. On TNT-Zn0.030 and TNT-Zn0.075, some of the cells formed extensive filopodia; however, some other cells spread poorly and vinculin was not expressed that clearly.
Similarly, and in contrast to controls, according to SEM (Figure S5), after 1 day of culture, stem cells were polygonal and had an elongated shape on ZnO-incorporated samples. Stem cell filopodia were longer on TNT-Zn0.015 than all the other samples.
The SEM images showed that cells on the TNT-Zn0.005 and TNT-Zn0.015 samples had more connections and cellular extensions (such as filopodia and lamellipodia) than on the others. However, on the TNT-Zn0.03 and TNT-Zn0.075 samples, cells were smaller and shrunk (even appearing apoptotic) because of the cytotoxicity of ZnO. Thus, this study confirmed that ZnO is regarded to be cytocompatible at relatively low concentrations, whereas high doses of ZnO can induce cytotoxicity.
The morphology of the mesenchymal stem cells (MSCs) on the samples demonstrated that TNT-Zn0.005 and TNT-Zn0.015 had a greater ability to induce MSCs differentiation. From the fluorescence images, the red actin cytoskeleton images showed that TNT-Zn0.005 and TNT-Zn0.015 elicited stem cell elongation, which induced cytoskeletal stress and selective differentiation into an osteoblast-like cell.20 The vinculin protein was marked with green fluorescence in this study. Vinculin is an intracellular protein involved in the linkage among cell adhesion membranous molecules, integrins, and the actin skeleton.21 Some vinculin was distributed along the edge of the cell and filopodia, which may have played a key role in the cytoskeletal development and focal adhesion of the cell.22 The formation of focal adhesion contacts and cell adhesion is an important starting point for cell proliferation, differentiation, and some cellular behaviors, which is activated by the formation of vinculin adhesion spots.23,24 The higher expression exhibited better adhesion. It has been reported that various kinds of physical stresses can accelerate stem cell differentiation into specific cells.24–28
ALP activity
The results (Figure 5) also demonstrated that stem cell ALP activity increased for TNT-Zn0.005 and TNT-Zn0.015 compared to all other samples. Compared to TNT-Zn0.005 and TNT-Zn0.015, the addition of ZnO to TNT-Zn0.030 and TNT-Zn0.075 decreased ALP activity from the stem cells.
ALP is a marker for early osteogenic differentiation, which participates in the process of pre-mineralization and promotes the formation of mineral nodules.29 The BMSCs cultured on TNT-Zn0.015 had the highest ALP values after both days 3 and 7, which demonstrated that BMSCs on these samples had a greater early osteogenic tendency, which is in good agreement with the confocal laser scanning microscopy image in Figure 5.
Osteogenic-related gene expression
Compared with pure Ti and TNTs, the expression of Osx, ALP, col-I, and OC was the highest on TNT-ZnO0.015 after 3 days, 7 days, and 14 days of culture (Figure 6). The expression of ALP, OC, and col-I from stem cells on TNTs was higher than pure Ti. The Osx genetic expression from stem cells on the surfaces of Ti and TNTs had a different tendency: on the TNTs, Osx was higher after 3 days and 14 days than on Ti.
Osteogenesis is the result of a complex sequence of events that involve the differentiation of MSCs into osteoblasts.30 Osteogenic-related genes all have a unique expression profile and are regulated strictly in a specific chronological order.31–33 The ALP gene guides protein synthesis, which implies that the ZnO in the TNTs can improve ALP activity and early cell differentiation. In this study, the TNTs also had a slightly higher ALP expression compared with the control sample. Osx is a zinc finger transcription factor expressed by osteoblasts, which plays a key role in osteogenic differentiation and has been shown to be fundamental for bone homeostasis and osteoblast differentiation by preosteoblasts to immature osteoblasts.30,34 In this study, TNT-Zn0.015 improved the expression of Osx at all time periods. The cells on TNTs had higher Osx expression after 3 days and 14 days than control samples, which suggests that the surface of TNTs had a greater potential to induce BMSCs to preosteoblasts than that of pure Ti. Col-I is an early osteogenitor marker, which is the most abundant collagen of the human body, heavily distributed in a mineralized extracellular matrix.34,35 Col-I gene expression was upregulated at all intervals on the TNT-Zn0.015 samples, suggesting that TNT-Zn0.015 can promote matrix formation. The TNTs had higher expression levels of col-I at early time periods (days 3 and 7). The nanotubes may have the ability to improve matrix formation earlier than pure Ti.
OC is secreted by osteoblasts and plays a role in pre-osteoblastic and bone-building processes.33–36 As a bone extracellular matrix vesicle, OC is mainly related to the formation of mineralized tissue and can protect the environment for mineralization when connected to calcium ions and hydroxyapatite. OC is used as a marker of osteogenic tendency, which correlates with the maturation of the osteoblast population and subsequent spontaneous mineralization.22 The surface of the TNT-Zn0.015 can stimulate high expression levels of OC in this study, which suggests that ZnO can improve MSCs matrix protein production and osteogenesis. All the gene expression results demonstrated that TNT-Zn0.015 can improve the differentiation of the MSCs into osteoblasts.
Zn ion concentration is relatively high in bone, cartilage, and teeth. Accordingly, Zn ions are believed to be critical to control bone homeostasis in the human body.37,38 It was revealed from recent studies that Zn ions can be mediated by Zn transporters that exert multiple functions in cellular events.39 In this study, the TNT-Zn0.015 sample had significant osteogenic properties that are dependent on proper Zn ion release. Zn ion uptake into cells and subsequent intracellular Zn2+ are essential for bone growth in mammals. MacDonald40 reported that Zn2+ accumulation affects hormone or growth factor signaling cascades important for growth hormone production and chondrocyte differentiation. In genetic expression studies, transcription factors containing zinc fingers as DNA-binding motifs are the largest family of transcriptional regulators. The zinc proteins require zinc to maintain their structural integrity and function.41 Runx2 and RANK expression can be reduced by Zn ion deficiency through a decrease in MITF and SP-1 proteins.41,42 The activity of ALP and Osx in cells can be specifically increased with a proper content of Zn2+.43,44 Thus, this study demonstrated that with the incorporated ZnO, TNTs can be improved to obtain osteogenic properties.
Antibacterial activity
The reduction rates of S. mutans and P. gingivalis colonization on the ZnO samples are shown in Tables 3 and 4. Different samples have different Zn ion release rates, which led to different antibacterial trends. In general, all the ZnO-decorated samples possessed much higher antibacterial effects for both bacteria than plain Ti. The antibacterial effect on S. mutans was more significant than on P. gingivalis.
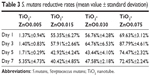  | Table 3 S. mutans reductive rates (mean value ± standard deviation) |
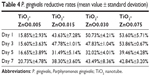  | Table 4 P. gingivalis reductive rates (mean value ± standard deviation) |
Modeling
In all cases, the cubic surface equation fits the experimental data better than the quadratic equation. The other models are given in the Supplementary material. Other modeling results (Figures S6 and 7) are reported below concerning optimizing stem cell responses while minimizing bacteria functions using ZnO.
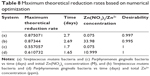
A cubic response surface model was fitted to the cell count (OD) value data with respect to time (days), and initial Zn(NO3)2 concentration (M) and total Zn2+ concentration (ppm). Figure 7 describes that there was only a 2.20% and 1.05% chance that the cubic model would occur due to noise, while R2 values of 0.8873–0.9138 suggested a very good fit of the predicted values to the experimental results, respectively. The cubic model equations are listed in Table 5. Numerical optimization was used to determine the maximum cell counts of the system described above, and the results are summarized in Table 6. The maximum cell proliferation OD value was about 1.5 when the initial Zn(NO3)2/Zn2+ concentration was 0.015 M or 2.08 ppm at day 7. The desirabilities of all the configurations tested resulted in values of at least 0.849, indicating a high probability that the solution parameters provided the maximum stem cell count for the system chosen.
A cubic response surface model was fitted to the reduction rates of S. mutans and P. gingivalis with respect to time (days) and initial Zn(NO3)2 concentration (M) and total Zn2+ concentration (ppm) (Figure 8). The analyses of variance showed that there was only a 0.01%–0.16% chance that the cubic models, given in Table 7, would occur due to noise, while R2 values ranged from 0.9596 to 0.9941, suggesting a very good fit of the predicted values to the experimental results. Adequate precision measures the signal to noise ratio with a ratio greater than four desirable values. The values found here ranged from 12.653 to 33.201, indicating that each model has an adequate signal and the models can be used to navigate the design space.
Numerical optimization was used to determine the maximum reduction rates of the systems described above (Table 8). For S. mutans, the maximum reduction rate was about 0.87 around 2.7 days when the Zn(NO3)2 and Zn2+ concentrations were 0.075 M or 33.98 ppm. For P. gingivalis, the maximum reduction rates were 0.557 and 0.611 around 1.7 days when the Zn(NO3)2 and Zn2+ were 0.075 M or 33.98 ppm. The desirabilities of all the configurations tested resulted in values of at least 0.995, indicating a high probability when the solution parameters provided the maximum.
From the modeling results for cell counts with respect to time (days), initial Zn(NO3)2 concentration (M), and total Zn2+ concentration (ppm), the highest or lowest cell proliferation OD value can be found according to time, Zn(NO3)2 concentration, and Zn2+ concentration; this can be used to predict the proliferation trend of the stem cells on the ZnO-decorated TNT. The modeling of bacteria reduction rate also can be used for predicting the antibacterial effect according to time, Zn(NO3)2 concentration, and Zn2+ concentration. Combining the models of both cell counts and bacterial reductive rates, the optimum concentration of Zn2+ and initial Zn(NO3)2 can be found at different intervals. The best Zn(NO3)2 and Zn2+ concentrations for cell proliferation were found to be 0.015 M and 2.08 ppm. The optimal bacterial reduction rate was found when Zn(NO3)2 was 0.075. When the Zn2+ concentrations were 33.98 ppm and 10.99 ppm, S. mutans and P. gingivalis were inhibited the most, respectively. Based on the above discussion, these first time models can provide an efficient way to calculate the Zn2+ concentration and initial Zn(NO3)2 concentration that will promote stem cell functions and inhibit bacteria functions on ZnO-decorated TNT samples.
Conclusion
In summary, using a simple hydrothermal method, different concentrations of Zn nanoparticles were incorporated into TNTs. The size and the quantity of ZnO nanoparticles can be adjusted by varying the Zn(NO3)2 precursor concentration. In all, 20–50 nm ZnO nanoparticles were distributed homogeneously along the entire length of the nanotube. Owing to the nanotopography of the Ti and proper Zn release, TNT-Zn0.015 generated improved cell compatibility. The results also showed that MSCs promoted more filopodia extension and vinculin on cells on the TNT-Zn0.015, which enhanced osteogenic differentiation of MSCs both at the genetic and protein levels. For the modeling results, the optimal concentration of Zn2+ and Zn(NO3)2 can be found for both stem cell proliferation and bacterial reduction. Thus, this study showed that ZnO-incorporated TNTs materials have a great potential to improve dental applications due to their osteogenic and antibacterial properties, and thus, should be further studied for numerous applications. The model developed in this study can be used to advance Zn-containing medical devices in the future.
Acknowledgments
This work was financially supported by the Beijing Natural Science Foundation (7132059), the National Outstanding Young Investigator Grant of China (51225402), and the Natural Science Foundation of China (51002004 and 81300916). The Importation and Development of High-Caliber Talents Project of Beijing Municipal Institutions (CIT&TCD201404038) and Rixin Talent authorized by Beijing University of Technology.
Disclosure
The authors report no conflicts of interest in this work.
References
Neoh KG, Hu X, Zheng D, Kang ET. Balancing osteoblast functions and bacterial adhesion on functionalized titanium surfaces. Biomaterials. 2012;33:2813–2822. | ||
Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284:1318–1322. | ||
Mei SL, Zhao LZ, Wang W, Ma QL, Zhang YM. Biomimetic titanium alloy with sparsely distributed nanotubes could enhance osteoblast functions. Adv Eng Mater. 2012;14:B166–B174. | ||
Ryoo SR, Kim YK, Kim MH, Min DH. Behaviors of NIH-3T3 fibroblasts on graphene/carbon nanotubes: proliferation, focal adhesion, and gene transfection studies. ACS Nano. 2010;4:6587–6598. | ||
Wang N, Li H, Wang J, Chen S, Ma Y, Zhang Z. Study on the anticorrosion, biocompatibility, and osteoinductivity of tantalum decorated with tantalum oxide nanotube array films. ACS Appl Mater Interfaces. 2012;4:4516–4523. | ||
Harris LG, Tosatti S, Wieland M, Textor M, Richards RG. Staphylococcus aureus adhesion to titanium oxide surfaces coated with non-functionalized and peptide-functionalized poly(L-lysine)-grafted-poly(ethylene glycol) copolymers. Biomaterials. 2004;25:4135–4148. | ||
Park J, Bauer S, Pittrof A, Killian MS, Schmuki P, von der Mark K. Synergistic control of mesenchymal stem cell differentiation by nanoscale surface geometry and immobilized growth factors on TiO2 nanotubes. Small. 2012;8:98–107. | ||
Cao X, Yu WQ, Qiu J, Zhao YF, Zhang YL, Zhang FQ. RGD peptide immobilized on TiO2 nanotubes for increased bone marrow stromal cells adhesion and osteogenic gene expression. J Mater Sci Mater Med. 2012;23:527–536. | ||
Arakawa T, Philo JS, Kita Y. Kinetic and thermodynamic analysis of thermal unfolding of recombinant erythropoietin. Biosci Biotechnol Biochem. 2001;65:1321–1327. | ||
Xin Y, Jiang J, Huo K, Hu T, Chu PK. Bioactive SrTiO(3) nanotube arrays: strontium delivery platform on Ti-based osteoporotic bone implants. ACS Nano. 2009;3:3228–3234. | ||
Ni GX, Lu WW, Xu B, et al. Interfacial behaviour of strontium-containing hydroxyapatite cement with cancellous and cortical bone. Biomaterials. 2006;27:5127–5133. | ||
Applerot G, Lipovsky A, Dror R, et al. Enhanced antibacterial activity of nanocrystalline ZnO due to increased ROS-mediated cell injury. Adv Funct Mater. 2009;19:10. | ||
Tang Y, Chappell HF, Dove MT, Reeder RJ, Lee YJ. Zinc incorporation into hydroxylapatite. Biomaterials. 2009;30:2864–2872. | ||
Hu H, Zhang W, Qiao Y, Jiang X, Liu X, Ding C. Antibacterial activity and increased bone marrow stem cell functions of Zn-incorporated TiO2 coatings on titanium. Acta Biomater. 2012;8:904–915. | ||
Yin H, Casey PS, McCall MJ, Fenech M. Effects of surface chemistry on cytotoxicity, genotoxicity, and the generation of reactive oxygen species induced by ZnO nanoparticles. Langmuir. 2010;26:15399–15408. | ||
Zhang SL, Li J, Lykotrafitis G, Bao G, Suresh S. Size-dependent endocytosis of nanoparticles. Adv Mater. 2009;21:419–420. | ||
Chang YN, Zhang MY, Xia L, Zhang J, Xing GM. The toxic effects and mechanisms of CuO and ZnO nanoparticles. Materials. 2012;5:2850–2871. | ||
Liu W, Su P, Chen S, et al. Synthesis of TiO2 nanotubes with ZnO nanoparticles to achieve antibacterial properties and stem cell compatibility. Nanoscale. 2014;6:9050–9062. | ||
Vanoss CJ, Giese RF, Good RJ. Reevaluation of the surface-tension components and parameters of polyacetylene from contact angles of liquids. Langmuir. 1990;6:1711–1713. | ||
Sauer GR, Wuthier RE. Influence of trace metal ions on matrix vesicle calcification. Bone Miner. 1992;17:284–289. | ||
Lee H, Rho J, Messersmith PB. Facile conjugation of biomolecules onto surfaces via mussel adhesive protein inspired coatings. Adv Mater. 2009;21:431–434. | ||
Grigoriou V, Shapiro IM, Cavalcanti-Adam EA, Composto RJ, Ducheyne P, Adams CS. Apoptosis and survival of osteoblast-like cells are regulated by surface attachment. J Biol Chem. 2005;280:1733–1739. | ||
Lai M, Cai K, Zhao L, Chen X, Hou Y, Yang Z. Surface functionalization of TiO2 nanotubes with bone morphogenetic protein 2 and its synergistic effect on the differentiation of mesenchymal stem cells. Biomacromolecules. 2011;12:1097–1105. | ||
Dolatshahi-Pirouz A, Jensen T, Kraft DC, et al. Fibronectin adsorption, cell adhesion, and proliferation on nanostructured tantalum surfaces. ACS Nano. 2010;4:2874–2882. | ||
Saretzki G, Walter T, Atkinson S, et al. Downregulation of multiple stress defense mechanisms during differentiation of human embryonic stem cells. Stem Cells. 2008;26:455–464. | ||
Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–689. | ||
Benoit DS, Durney AR, Anseth KS. The effect of heparin-functionalized PEG hydrogels on three-dimensional human mesenchymal stem cell osteogenic differentiation. Biomaterials. 2007;28:66–77. | ||
Mirmalek-Sani SH, Tare RS, Morgan SM, et al. Characterization and multipotentiality of human fetal femur-derived cells: implications for skeletal tissue regeneration. Stem Cells. 2006;24:1042–1053. | ||
Olivares-Navarrete R, Hyzy SL, Hutton DL, et al. Direct and indirect effects of microstructured titanium substrates on the induction of mesenchymal stem cell differentiation towards the osteoblast lineage. Biomaterials. 2010;31:2728–2735. | ||
Baglìo SR, Devescovi V, Granchi D, Baldini N. MicroRNA expression profiling of human bone marrow mesenchymal stem cells during osteogenic differentiation reveals Osterix regulation by miR-31. Gene. 2013;527:321–331. | ||
Stiehler M, Lind M, Mygind T, et al. Morphology, proliferation, and osteogenic differentiation of mesenchymal stem cells cultured on titanium, tantalum, and chromium surfaces. J Biomed Mater Res A. 2008; 86:448–458. | ||
Yu WQ, Jiang XQ, Zhang FQ, Xu L. The effect of anatase TiO2 nanotube layers on MC3T3-E1 preosteoblast adhesion, proliferation, and differentiation. J Biomed Mater Res A. 2010;94:1012–1022. | ||
Bjursten LM, Rasmusson L, Oh S, Smith GC, Brammer KS, Jin S. Titanium dioxide nanotubes enhance bone bonding in vivo. J Biomed Mater Res A. 2010;92:1218–1224. | ||
Nakashima K, Zhou X, Kunkel G, et al. The novel zinc finger-containing transcription factor Osterix is required for osteoblast differentiation and bone formation. Cell. 2002;108:17–29. | ||
Lu J, Zhou S, Siech M, Habisch H, Seufferlein T, Bachem MG. Pancreatic stellate cells promote hapto-migration of cancer cells through collagen I-mediated signalling pathway. Br J Cancer. 2013;110(2):409–420. | ||
Lee NK, Sowa H, Hinoi E, et al. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130:456–469. | ||
Hirano T, Murakami M, Fukada T, Nishida K, Yamasaki S, Suzuki T. Roles of zinc and zinc signaling in immunity: zinc as an intracellular signaling molecule. Adv Immunol. 2008;97:149–176. | ||
Fukada T, Hojyo S, Furuichi T. Zinc signal: a new player in osteobiology. J Bone Miner Metab. 2013;31:129–135. | ||
Kawakubo A, Matsunaga T, Ishizaki H, Yamada S, Hayashi Y. Zinc as an essential trace element in the acceleration of matrix vesicles-mediated mineral deposition. Microsc Res Techniq. 2011;74:1161–1165. | ||
MacDonald RS. The role of zinc in growth and cell proliferation. J Nutr. 2000;130:1500S–1508S. | ||
Hie M, Iitsuka N, Otsuka T, Nakanishi A, Tsukamoto I. Zinc deficiency decreases osteoblasts and osteoclasts associated with the reduced expression of Runx2 and RANK. Bone. 2011;49:1152–1159. | ||
Yamaguchi M, Uchiyama S. Receptor activator of NF-kappaB ligand-stimulated osteoclastogenesis in mouse marrow culture is suppressed by zinc in vitro. Int J Mol Med. 2004;14:81–85. | ||
Wu C, Ramaswamy Y, Chang J, Woods J, Chen Y, Zreiqat H. The effect of Zn contents on phase composition, chemical stability and cellular bioactivity in Zn-Ca-Si system ceramics. J Biomed Mater Res B. 2008;87:346–353. | ||
Kawamura H, Ito A, Muramatsu T, Miyakawa S, Ochiai N, Tateishi T. Long-term implantation of zinc-releasing calcium phosphate ceramics in rabbit femora. J Biomed Mater Res A. 2003;65:468–474. |
Supplementary materials
Response surface methodology is a mathematical model, which can be described as a collection of statistical tools and techniques for constructing and exploring an approximate functional relationship between a response variable and a set of design factors.1 Identifying and fitting experimental data to an appropriate response surface model requires the use of statistical experimental design fundamentals, regression modeling techniques, and optimization methods. All three of these are combined into what is called response surface methodology.1
Experimental design matrix
  | Table S1 Response – Zn release (ppm) |
  | Table S2 Response – Zn release (ppm) |
  | Table S3 Response – cell count (OD value) |
  | Table S4 Response – cell count (OD value) |
  | Table S5 Response – Streptococcus mutans bacteria reduction rate |
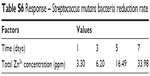  | Table S6 Response – Streptococcus mutans bacteria reduction rate |
  | Table S7 Response – Porphyromonas gingivalis bacteria reduction rate |
  | Table S8 Response – Porphyromonas gingivalis bacteria reduction rate |
Cell count
A quadratic response surface model was fitted to the cell count (optical density [OD]) value data with respect to time (days) and initial Zn(NO3)2 concentration (M) and total Zn+ concentration (ppm). Analyses of variances show that there is only a 1.90% and 0.78% chance that the quadratic model could occur due to noise. While R2 values of 0.7325 and 0.7842 do not suggest a very good fit of the predicted values to the experimental results, the adequate precision values of 6.988 and 8.139 suggest adequate signal. The quadratic model equations are given in Table S9. Numerical optimization was used to determine the maximum reduction rates of the systems described above, and the results are given in Table S10. The desirabilities of all the configurations tested resulted in values of at least 0.824, indicating a high probability that the solution parameters provide the maximum cell count for the system and model chosen.
Bacteria reduction rate
A quadratic response surface model was fitted to the reduction rates of SM and PG bacteria with respect to time (days) and initial Zn(NO3)2 concentration (molar) and total Zn2+ concentration (ppm). Analyses of variances show that there is only between 0.09% and 2.81% chance that the cubic models, given in Table S11, could occur due to noise. While R2 values ranging from 0.6706 to 0.8450 do not suggest a very good fit of the predicted values to the experimental results, the adequate precision measures the signal to noise ratio and is adequate. A ratio greater than 4 is desirable. The values found here ranging from 5.890 to 9.131 indicate that each model has an adequate signal; thus, the models can be used to navigate the design space.
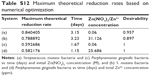
Numerical optimization was used to determine the maximum reduction rates of the systems described above, and the results are given in Table S12. The desirabilities of all the configurations tested resulted in values of at least 0.897, indicating a high probability that the solution parameters provide the maximum.
Reference
Bezerra MA, Santelli RE, Oliveira EP, Villar LS, Escaleira LA. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta. 2008;76:965–977. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.