Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 17
Obstructive Sleep Apnea is Associated with an Increased Prevalence of Polycythemia in Patients with Chronic Obstructive Pulmonary Disease
Authors Zeng Z, Song Y, He X , Yang H, Yue F, Xiong M, Hu K
Received 11 September 2021
Accepted for publication 24 December 2021
Published 15 January 2022 Volume 2022:17 Pages 195—204
DOI https://doi.org/10.2147/COPD.S338824
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Min Zhang
Zhaofu Zeng, Yan Song, Xin He, Haizhen Yang, Fang Yue, Mengqing Xiong, Ke Hu
Department of Respiratory and Critical Care Medicine, Renmin Hospital of Wuhan University, Wuhan, People’s Republic of China
Correspondence: Ke Hu
Department of Respiratory and Critical Care Medicine, Renmin Hospital of Wuhan University, Wuhan, 430060, People’s Republic of China
Tel +86 1897 103 5988
Email [email protected]
Purpose: Chronic obstructive pulmonary disease (COPD) and obstructive sleep apnea (OSA) are associated with polycythemia. However, there still remain unanswered questions about the relationship between overlap syndrome (OVS), where OSA and COPD coexist, and polycythemia. Here, we aimed to establish the prevalence of polycythemia in OVS patients and to explore the impact of OSA on polycythemia.
Patients and Methods: Patients with COPD underwent overnight polysomnography (PSG), pulmonary function tests, echocardiography, and complete blood counts. All patients were ethnic Han Chinese and free of prolonged oral corticosteroid use, hematological system disease, severe systemic disease, and other sleep-disordered breathing. OVS was defined as COPD patients with an apnea–hypopnea index ≥ 15 events/h, and polycythemia was defined as an Hb > 165 g/L in men and > 160 g/L in women.
Results: Eight-hundred and eighty-six patients with COPD were included in the analysis. The prevalence of polycythemia was significantly higher in OVS patients than COPD-alone patients (6.4% vs 2.9%, p < 0.05). The prevalence of polycythemia increased with OSA severity (χ2 = 7.885, p = 0.007), but not in GOLD grade 3– 4 COPD patients (χ2 = 0.190, p = 0.663). After adjusting for confounders, percentage of total sleep time with SaO2 < 90% (TS90) remained independently associated with an increased odds of polycythemia (OR 1.030, 95% CI 1.015– 1.046) and, with an increase in TS90, the hemoglobin increased, especially in GOLD grade 1– 2 patients (p < 0.05).
Conclusion: Patients with OVS have a higher prevalence of polycythemia than those with COPD alone, and TS90 is an independent factor for polycythemia, especially in GOLD1-2 COPD patients.
Keywords: chronic obstructive pulmonary disease, obstructive sleep apnea, overlap syndrome, polycythemia
Introduction
Chronic obstructive pulmonary disease (COPD) is a common and chronic respiratory disease that affects over 380 million people worldwide. COPD has a prevalence of ~10%, and its healthcare burden is expected to rise over the next forty years due to increasing numbers of deaths attributable to the disease.1 COPD is characterized by progressive and persistent airflow limitation resulting in chronic sustained hypoxia and acquired polycythemia, and the latter is closely associated with an increased risk of stroke, venous thromboembolism (VTE), pulmonary hypertension (PH), and mortality.2,3 Furthermore, polycythemia remains present in 2–10% of COPD patients even after long-term oxygen therapy.4–6
Obstructive sleep apnea (OSA) is characterized by recurrent episodes of partial or complete collapse of the upper airway during sleep, and it can lead to chronic intermittent hypoxia related to red blood cell (RBC) proliferation.7,8 OSA severity is significantly associated with hemoglobin (Hb) and RBC counts,7 and the prevalence of polycythemia in OSA patients is reported to be between 1.6% and 10%.9,10 Furthermore, RBC count and hemoglobin significantly decreased in OSA patients after continuous positive airway pressure (CPAP) therapy.11
Overlap syndrome (OVS) refers to the occurrence of COPD and OSA in the same individual. The pathophysiological changes of COPD and OSA are often superimposed or synergistic in OVS patients, the most significant of which is chronic hypoxia. However, there is considerable complexity to the underlying biology of sustained versus intermittent hypoxia in stimulating erythroid hyperplasia,12,13 although it is widely recognized that OVS patients may be particularly vulnerable to oxyhemoglobin desaturation during sleep.14
We previously showed that OVS patients have higher RBC counts than COPD patients;15 however, whether OSA increases the prevalence of polycythemia in COPD patients remains unillustrated. It is also important to firmly establish whether polycythemia is associated with other complications in COPD patients to understand the possible adverse effects of polycythemia on prognosis. Therefore, in this cross-sectional study, we assessed the prevalence of polycythemia in OVS patients to explore the role and mechanism of OSA in the disease and to understand the relationship between polycythemia and other comorbidities.
Methods
Subjects
From December 2016 to August 2020, 1048 participants diagnosed with COPD were enrolled in the study and underwent polysomnography (PSG). Patients in the primary cohort were recruited from four Chinese tertiary hospitals under the auspices of a national COPD Research and Development Program (Clinical Trials ID: NCT 03182309). Eight hundred and eighty-six patients met the following inclusion criteria and were included in the study (Figure 1): (1) age over 40 years; (2) post-bronchodilator forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) <0.7 without any exacerbations in the preceding three months; and (3) willingness to participate in the study and provide written informed consent. The following exclusion criteria were applied: (1) non-Han ethnicity; (2) long-term history of high-altitude exposure; (3) prolonged use of high-dose glucocorticoids or other drugs that affect blood cell production; (4) severe systemic disease (eg, severe infection, malignancy, severe liver and renal dysfunction); (5) hematological disease (eg, leukemia, lymphoma, anemia, polycythemia vera); and (6) known sleep-disordered breathing other than OSA. The study was approved by the Scientific Research and Technology Ethics Committee of Renmin Hospital of Wuhan University and conducted in accordance with the Declaration of Helsinki.
 |
Figure 1 Study flow diagram. Abbreviations: OVS, overlap syndrome; COPD, chronic obstructive pulmonary disease. |
Clinical Assessment and Anthropometric Measurements
Current or former smoking and drinking habits and treatments for COPD were recorded, as were a past medical history of hypertension, stroke, coronary heart disease (CHD), thyroid dysfunction, diabetes, gastritis, and venous thromboembolism (VTE; including pulmonary embolism or deep vein thrombosis). Body mass index (BMI) was obtained by dividing body weight by height squared.
Pulmonary Function Tests
The FVC and the FEV1 were measured with a spirometer (Jaeger MasterScreen Body, Germany). The FEV1/FVC and predicted percentage of FEV1 (FEV1%) and FVC (FVC%) were calculated. According to Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines, patients were divided into those with GOLD grade 1–2 (FEV1% ≥ 50%) and GOLD grade 3–4 COPD (FEV1% < 50%).16
PSG Monitoring
All patients underwent overnight PSG (SOMNOscreen Plus Tele PSG, SOMNOmedics GmbH, Randersacker, Germany) in the hospital’s sleep-monitoring room. The parameters investigated included airflow, electroencephalography, electrooculography, electrocardiography, electromyography, thoracic and abdominal respiratory effort, snoring, body position, and pulse oxygen saturation (SaO2). The apnea–hypopnea index (AHI), average SaO2 (mean SaO2), lowest SaO2 (min SaO2), oxygen desaturation index (ODI), and percentage of total sleep time spent with SaO2 <90% (TS90) were recorded by PSG. Standard PSG was performed by trained sleep technicians and the recordings were analyzed according to the criteria outlined in the 2016 American Academy of Sleep Medicine (AASM) Manual v.2.3.17 All patients were diagnosed with OVS if they had an AHI ≥ 15 events/h, and OVS was further dichotomized into those with an AHI of 15 to <30 events/h and those with an AHI ≥ 30 events/h. All OSA patients are newly diagnosed patients and have not been treated with CPAP.
Echocardiography
All subjects received standard transthoracic Doppler-echocardiography (LOGIQE9, General Electric Company, Boston, MA, USA) by an experienced ultrasound examiner, and the results were confirmed by at least two echocardiography specialists. PH was diagnosed by echocardiography.18
Laboratory Measurements
Fasting blood samples were taken the next morning immediately after PSG. Complete blood cell counts were assessed with an automated count analyzer. The following hematological indices were obtained: white blood cell (WBC) count, RBC count, Hb, and platelets (PLT). Polycythemia was defined as an Hb >165 g/L in men and >160 g/L in women.19
Statistical Analysis
SPSS v25.0 (IBM Statistics, Armonk, NY) statistical software was used for data analysis. Normally distributed data were expressed with means ± standard deviations (SD) and compared using one-way analysis of variance (ANOVA) or t-tests, while non-normally distributed data were expressed as medians and interquartile ranges (IQR) and compared using the Kruskal–Wallis test or U-test. Count data were presented as rates or composition ratios and were tested using the chi-squared test or Fisher’s exact test. Analysis of covariance (ANCOVA) was used to compare linear regression slopes. After univariate analysis, variance inflation factor (VIF) was used to evaluate the collinearity of variables, and binary multivariate logistic regression was applied to further analyze variables with a VIF <3. A p-value <0.05 was considered significant.
Results
Baseline Data and Clinical Characteristics
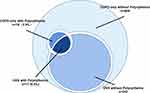
Eighteen patients had polycythemia among 622 (2.9%) patients with COPD alone, and 17 patients had polycythemia among 264 (6.4%) patients with OVS (p<0.05; Figure 2). Subgroup analyses were conducted according to OSA severity, and the baseline characteristics of the subgroups are shown in Table 1. There were no significant differences in age, tobacco use, and FVC% between groups (all p>0.05), but there were significant differences in gender, BMI, neck circumference, alcohol use, FVC, FEV1, FEV1%, and FEV1/FVC between groups (all p<0.05). There were no significant differences in the prevalence of PH, VTE, CHD, hypertension, stroke, thyroid dysfunction, diabetes, or gastritis between groups (all p>0.05), neither were there statistical differences between groups with respect to COPD treatments (all p>0.05).
 |
Table 1 Baseline Characteristics, Comorbidities, Sleep and Laboratory Data of the COPD Only and the OVS Groups |
The Relationship Between the Severity of OSA and Prevalence of Polycythemia
Hb and RBC counts significantly increased with the severity of OSA (p<0.05) in all patients with GOLD1-2 COPD (Figure 3). Consistent with this, as OSA severity increased, the prevalence of polycythemia gradually increased (χ2=7.885, p=0.007). This increasing trend was observed for GOLD1-2 (χ2=10.796, p=0.001) but not GOLD3-4 COPD patients (χ2=0.190, p=0.663) (Figure 4). Interestingly, there was no significant increase in the prevalence of polycythemia with an increase in the severity of airflow limitation.
Independent Factors Associated with Polycythemia
Patients with polycythemia were younger, heavier, and had a larger neck circumference (all p<0.01). The TS90 and ODI of patients with polycythemia were higher and the min SaO2 and mean SaO2 were lower (p<0.05) in patients with polycythemia (Table 2). Increased TS90 was associated with an increased odds of polycythemia (OR 1.029, 95% CI 1.019–1.039) in binary logistic regression analysis. When adjusted for gender, age, BMI, neck circumference, OSA, min SaO2, mean SaO2, tobacco use, alcohol use, FEV1%, and FEV1/FVC, increased TS90 still remained associated with an increased odds of polycythemia (OR 1.029, 95% CI 1.014–1.044) (Table 3). After adjusting for the above factors, TS90 was still associated with polycythemia in both GOLD1-2 and GOLD3-4 COPD patients (OR 1.031, 95% CI 1.007–1.056; OR 1.028, 95% CI 1.006–1.050, respectively) (Table 4). Figure 5 shows that with an increase in TS90, Hb levels and RBC counts gradually increased, especially in GOLD1-2 COPD patients (p<0.05).
 |
Table 2 Demographic and Clinical Characteristics of Patients with or without Polycythemia |
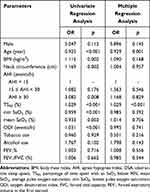 |
Table 3 Results of Logistic Regression in Analyzing the Effect of OSA Severity and Other Parameters on Polycythemia on All of Patients with COPD |
 |
Table 4 Results of Logistic Regression in Analyzing the Effect of OSA Severity and Other Parameters on Polycythemia According to GOLD Grade of COPD Patients |
Discussion
The reported incidence of polycythemia varies with the use of different diagnostic criteria. The prevalence of polycythemia in COPD patients is reported to range from 5.9% to 10.2% when defined as an Hb ≥170 g/L in males and ≥150 g/L in females, but Frank et al reported that only 2% of COPD patients had polycythemia.4 Zhang et al found that 6.6% of COPD patients met the diagnostic criteria for polycythemia with a cutoff of Hb >165 g/L in males and >160 g/L in females and suggested that race and altitude have an impact on the prevalence of polycythemia.20 In our study, the prevalence of polycythemia was only 3.9%, which might be because all our patients lived at high altitudes and ethnic groups other than Han were excluded.
We found that the prevalence of polycythemia in OVS patients was 6.4% and that the severity of disease was positively associated with the prevalence of polycythemia. In a nationwide study in the USA, Pathak et al screened 77,518,944 patients through a medical system and, after excluding other possible causes of secondary erythrocytosis, diagnosed 2,765,267 OSA patients, of whom 13,016 had polycythemia. Furthermore, the presence of OSA was associated with an increased odds of polycythemia (OR 5.9, 95% CI 5.65–6.17).21 In another study, OSA was an independent predictor for polycythemia in patients receiving androgen supplementation.22 Moreover, in the report by Li et al.10 OSA severity was related to the prevalence of polycythemia and mean SaO2 was an independent predictor of polycythemia. We found that the presence of polycythemia was related to age, BMI, neck circumference, severe OSA, min SaO2, mean SaO2, ODI, and TS90 in binary logistic regression analysis, and multivariate analysis showed that age and TS90 were independent predictors after adjusting for other confounders. On this basis, we further compared the diagnostic value of min SaO2, mean SaO2, and TS90 for polycythemia (Supplementary Figure 1). It is currently accepted that intermittent hypoxia can inhibit the degradation of hypoxia-inducible factor (HIF), thereby promoting the secretion of erythropoietin (EPO) and leading to RBC proliferation.23,24 Furthermore, hypoxia stimulation has been shown to increase atrial natriuretic peptide, which can lead to relative polycythemia.25
Unexpectedly, the prevalence of polycythemia did not differ between OVS patients and COPD-only patients with GOLD3-4 disease. We believe that this might be for several reasons: first, the effects of hypoxic stimulation and inflammation on the hematopoietic system were balanced, since hypoxic stimulation is thought to induce quantitative changes while inflammation results in qualitative changes26,27 ie, more severe inflammation results in obvious EPO resistance or hematopoietic dysfunction in severe COPD.5 Therefore, the effect of hypoxia on RBC proliferation was significantly reduced. Second, although COPD and OSA have similar pathophysiology such as chronic hypoxia, chronic sustained hypoxia and chronic intermittent hypoxia have different effects on RBC production. To support this, Song et al24 observed the destruction of new RBCs after reoxygenation in patients with chronic sustained hypoxia (neocytolysis), but this phenomenon was rare in OSA patients. Therefore, since patients with severe COPD may experience more serious neocytolysis, the increase in RBC counts due to OSA was not significant. Ryan et al13 also postulated that the impact of chronic hypoxia on the hematopoietic system involves two main pathways: adaptive and inflammatory. The adaptive pathway places HIF at the center and stimulates erythropoiesis, while the inflammatory pathway is mediated by NF-κB and causes inflammation. This difference in hypoxia pathway activation leads to different effects on erythropoiesis. Finally, severe COPD patients are often older, more malnourished, and receive long-term oxygen therapy, which might also influence the hematopoietic process.
After adjusting for gender, age, BMI and other factors, we found that polycythemia was associated with pulmonary hypertension (Supplementary Table 1), which is consistent with previous studies.14,28 Increased circulating RBCs increase pulmonary vascular resistance and pulmonary arterial pressure through an increase in blood viscosity. Unfortunately, poor vessel compliance in COPD patients may exacerbate this change.29 Interestingly, there were only six cases of VTE in our cohort, of whom none had polycythemia. A case–control study revealed no difference in the number of VTE events in COPD patients with and without secondary polycythemia, although there was a significant difference in PH.30 In this regard, we speculate that polycythemia in COPD patients may have a greater impact on the pulmonary arteries. We also compared the prevalence of other comorbidities including hypertension, diabetes, and stroke between OVS and COPD only groups. Interestingly, although there was no statistically significant difference between the two groups for hypertension, the OVS group had a trend towards more frequent hypertension (40.8% vs 47.7%, p=0.058), and, as the severity of OSA increased, the prevalence of hypertension increased (χ2=4.483, p=0.034).
This study has several limitations. First, the study was cross-sectional, which precludes any conclusions regarding causality of the associations between OSA and polycythemia in OVS patients. Second, this study represents a preliminary study without further exploration of the underlying mechanisms that might explain the different effects of chronic sustained hypoxia and chronic intermittent hypoxia on erythropoiesis. Third, no long-term follow-up was available to study the impact of polycythemia on survival.
Regardless, considering the close association between polycythemia and OSA and its poor prognosis, PSG should be conducted in COPD patients with polycythemia in order to not miss the diagnosis and to initiate appropriate treatment.
Conclusion
Here, we found that patients with OVS had a higher prevalence of polycythemia than those with COPD alone and that TS90 is an independent factor for polycythemia, especially in GOLD1-2 COPD patients.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 81970082) and the National Key Research and Development Program of China [Project Number: 2016YFC1304403].
Disclosure
The authors declare that they have no conflicts of interest.
References
1. D’cruz RF, Murphy PB, Kaltsakas G. Sleep disordered breathing and chronic obstructive pulmonary disease: a narrative review on classification, pathophysiology and clinical outcomes. J Thorac Dis. 2020;12(S2):S202–16. doi:10.21037/jtd-cus-2020-006
2. Siegel FP, Petrides PE. Congenital and acquired polycythemias. Dtsch Arztebl Int. 2008;105(4):62–68. doi:10.3238/arztebl.2008.0062
3. Xu L, Chen Y, Xie Z, et al. High hemoglobin is associated with increased in-hospital death in patients with chronic obstructive pulmonary disease and chronic kidney disease: a retrospective multicenter population-based study. BMC Pulm Med. 2019;19(1):174–182. doi:10.1186/s12890-019-0933-4
4. Frank Y, Regev A, Novack V, et al. Incidence of hospitalizations among chronic obstructive pulmonary disease (COPD) patients with anemia. Harefuah. 2012;151(4):211–215. https://pubmed.ncbi.nlm.nih.gov/22616148/
5. Cote C, Zilberberg MD, Mody SH, et al. Haemoglobin level and its clinical impact in a cohort of patients with COPD. Eur Respir J. 2007;29(5):923–929. doi:10.1183/09031936.00137106
6. Boutou AK, Karrar S, Hopkinson NS, et al. Anemia and survival in chronic obstructive pulmonary disease: a dichotomous rather than a continuous predictor. Respiration. 2013;85(2):126–131. doi:10.1159/000338792
7. Fan Z, Lu X, Long H, et al. The association of hemocyte profile and obstructive sleep apnea. J Clin Lab Anal. 2019;33(2):e22680. doi:10.1002/jcla.22680
8. Gottlieb DJ, Punjabi NM. Diagnosis and management of obstructive sleep apnea: a review. JAMA. 2020;323(14):1389–1400. doi:10.1001/jama.2020.3514
9. Nguyen CD, Holty JC. Does untreated obstructive sleep apnea cause secondary erythrocytosis? Respir Med. 2017;130:27–34. doi:10.1016/j.rmed.2017.07.003
10. Li N, Li HP, Wang P, et al. Nocturnal mean oxygen saturation is associated with secondary polycythemia in young adults with obstructive sleep apnea, especially in men. Nat Sci Sleep. 2019;11:377–386. doi:10.2147/NSS.S226143
11. Feliciano A, Linhas R, Marçôa R, et al. Hematological evaluation in males with obstructive sleep apnea before and after positive airway pressure. Rev Port Pneumol. 2017;23(2):71–78. doi:10.1016/j.rppnen.2016.12.001
12. Malhotra A, Schwartz AR, Schneider H, et al. Research priorities in pathophysiology for sleep-disordered breathing in patients with chronic obstructive pulmonary disease. An Official American Thoracic Society Research Statement. Am J Respir Crit Care Med. 2018;197(3):289–299. doi:10.1164/rccm.201712-2510ST
13. Ryan S, Taylor CT, Mcnicholas WT. Selective activation of inflammatory pathways by intermittent hypoxia in obstructive sleep apnea syndrome. Circulation. 2005;112(17):2660–2667. doi:10.1161/CIRCULATIONAHA.105.556746
14. Kent BD, Mitchell PD, Mcnicholas WT. Hypoxemia in patients with COPD: cause, effects, and disease progression. Int J Chron Obstruct Pulmon Dis. 2011;6:199–208. doi:10.2147/COPD.S10611
15. Zhu J, Zhao Z, Nie Q, et al. Effect of lung function on the apnea-hypopnea index in patients with overlap syndrome: a multicenter cross-sectional study. Sleep Breath. 2020;24(3):1059–1066. doi:10.1007/s11325-019-01961-w
16. Singh D, Agusti A, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease: the GOLD science committee report 2019. Eur Respir J. 2019;53(5):164–176. doi:10.1183/13993003.00164-2019
17. Berry RB, Brooks R, Gamaldo C, et al. The American Academy of Sleep Medicine. In: Berry RB, Brooks R, Gamaldo CE, eds. The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications 2.3 ed. Darien; 2016. Available from: URL: https://aasm.org/clinical-resources/scoring-manual/. Accessed January 08, 2021.
18. Frost A, Badesch D, Gibbs JSR, et al. Diagnosis of pulmonary hypertension. Eur Respir J. 2019;53(1):1801904. doi:10.1183/13993003.01904-2018
19. Mithoowani S, Laureano M, Crowther MA, et al. Investigation and management of erythrocytosis. CMAJ. 2020;192(32):913–918. doi:10.1503/cmaj.191587
20. Zhang J, Demeo DL, Silverman EK, et al. Secondary polycythemia in chronic obstructive pulmonary disease: prevalence and risk factors. BMC Pulm Med. 2021;21(1):235–247. doi:10.1186/s12890-021-01585-5
21. Pathak R, Giri S, Karmacharya P, et al. Obstructive sleep apnea syndrome and secondary polycythemia: analysis of the nationwide inpatient sample. Sleep Med. 2015;16(1):205–206. doi:10.1016/j.sleep.2014.09.012
22. Lundy SD, Parekh NV, Shoskes DA. Obstructive sleep apnea is associated with polycythemia in hypogonadal men on testosterone replacement therapy. J Sex Med. 2020;17(7):1297–1303. doi:10.1016/j.jsxm.2020.03.006
23. Watts D, Gaete D, Rodriguez D, et al. Hypoxia pathway proteins are master regulators of erythropoiesis. Int J Mol Sci. 2020;21(21):8138–8156. doi:10.3390/ijms21218131
24. Song J, Sundar K, Gangaraju R, et al. Regulation of erythropoiesis after normoxic return from chronic sustained and intermittent hypoxia. J Appl Physiol. 2017;123(6):1671–1675. doi:10.1152/japplphysiol.00119.2017
25. Krieger J, Sforza E, Barthelmebs M, et al. Overnight decrease in hematocrit after nasal CPAP treatment in patients with OSA. Chest. 1990;97(3):729–730. doi:10.1378/chest.97.3.729
26. Similowski T, Agustí A, Macnee W, et al. The potential impact of anaemia of chronic disease in COPD. Eur Respir J. 2006;27(2):390–396. doi:10.1183/09031936.06.00143704
27. Khan AM, Ashizawa S, Hlebowicz V, et al. Anemia of aging and obstructive sleep apnea. Sleep Breath. 2011;15(1):29–34. doi:10.1007/s11325-010-0326-7
28. Samareh Fekri M, Torabi M, Azizi Shoul S, et al. Prevalence and predictors associated with severe pulmonary hypertension in COPD. Am J Emerg Med. 2018;36(2):277–280. doi:10.1016/j.ajem.2017.08.014
29. Nakamura A, Kasamatsu N, Hashizume I, et al. Effects of hemoglobin on pulmonary arterial pressure and pulmonary vascular resistance in patients with chronic emphysema. Respiration. 2000;67(5):502–506. doi:10.1159/000067463
30. Nadeem O, Gui J, Ornstein DL. Prevalence of venous thromboembolism in patients with secondary polycythemia. Clin Appl Thromb Hemost. 2013;19(4):363–366. doi:10.1177/1076029612460425
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.