Back to Journals » Journal of Pain Research » Volume 12
Novel Characterization Of Thermal Temporal Summation Response By Analysis Of Continuous Pain Vs Time Curves And Exploratory Modeling
Authors Kong JT , Bagarinao E, Olshen RA, Mackey S
Received 13 June 2019
Accepted for publication 31 October 2019
Published 2 December 2019 Volume 2019:12 Pages 3231—3244
DOI https://doi.org/10.2147/JPR.S212137
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor E Alfonso Romero-Sandoval
Jiang-Ti Kong,1 Epifanio Bagarinao,2 Richard A Olshen,3 Sean Mackey1
1Department Of Anesthesiology, Perioperative And Pain Medicine, Stanford University School of Medicine, Stanford, CA, USA; 2Brain And Mind Research Center, Nagoya University, Nagoya, Japan; 3Department of Biomedical Data Science, School of Medicine, Stanford University, Stanford, CA, USA
Correspondence: Jiang-Ti Kong
Department of Anesthesiology, Perioperative and Pain Medicine, Stanford University School Of Medicine, 1070 Arastradero Road, Suite 200, Palo Alto, CA 94304, USA
Tel +1 650-723-1235
Fax +1 650-725-9642
Email [email protected]
Background: Temporal summation (TS) refers to the increased perception of pain with repetitive noxious stimuli. While thermal TS is generally considered a behavioral correlate of spinal windup, noxious heat pulses also trigger additional sensory processes which were modeled in this study.
Methods: Nineteen healthy volunteers (9 females, mean age 29.2, SD 10.5) underwent two identical TS experiments, spaced a week apart. The TS paradigm consisted of 10 identical heat pulses with individualized temperatures at the thenar eminence (0.5Hz). We extracted 3 features from continuous TS response curves: Lag, time to first feel pain; Slope, the rate of pain increase between the first and most painful heat pulse; and Delta, the maximum drop in pain after peak pain is reached. We then examined the within-individual stability of these features, followed by the Pearson’s correlations among these features and between the features and negative affect.
Results: All 3 features were stable over 1 week. Lag and Delta were negatively correlated (r = −0.5, p = 0.042). Slope did not correlate with Lag or Delta, but strongly correlated with a traditional TS measure, first pulse pain and peak pain difference (r = 0.91, p < 0.0001). Negative affects such as trait and state anxiety were negatively correlated with baseline (r = −0.49, p = 0.031) and peak stimulating temperature (r = −0.48, p = 0.039), respectively, suggesting an association between anxiety and greater pain sensitivity.
Conclusion: We were able to decouple spinal windup from other perceptual processes generated by phasic thermal TS paradigms and demonstrate temporal stability of these curve features. These curve features may help better characterize the complex sensory response to noxious heat pulses and serve as biomarkers to profile patients with chronic pain.
Keywords: thermal, temporal summation, windup, anxiety, depression, test–retest stability
Plain Language Summary
Temporal summation refers to the increased perception of pain in response to a train of identical painful stimuli, such as heat pulses, at frequencies ≥ 0.3Hz. Temporal summation has been traditionally considered a behavioral correlate to windup of spinal secondary neurons.
However, in addition to windup, noxious heat pulses also trigger other processes such as A-delta activation, habituation and/or descending inhibition. These additional processes may be responsible for the large between-individual variability in TS but have not been extensively studied.
In this exploratory study, we generated a continuous pain response curve and selected key features from the curve to model windup and possibly peripheral activation and habituation/inhibition. Our features were stable over time. Furthermore, the feature reflecting windup was highly correlated with traditional windup measures, independent from the other features and from heat pulse temperatures. We believe that our method of quantifying TS provides a more precise measure of windup by removing and characterizing influence from other sensory processes. Together, these curve features may be used as biomarkers of pain sensitivity.
Introduction
Temporal summation (TS) refers to the increased perception of pain with repeated delivery of identical noxious stimuli.1,2 It is often considered a behavioral correlate of windup, where the spinal secondary neuron increases its output in response to recurring C-fiber stimulation at frequencies above 0.3Hz.3,4 Many noxious stimuli generate TS, including thermal, electrical, and punctate pressure.5 The current study focuses on thermal TS, which typically involves the delivery of a set of identical, noxious heat pulses to the body via a thermode. Thermal TS can be delivered in a standardized fashion, is generally well tolerated, and has been widely studied.4,6–12
Although thermal TS is often used to approximate central pain facilitation, problems with methodology have limited its application: one of the most significant is the lack of standardization in quantifying the magnitude of TS.13,14 Most current methods acquire data by using the subject to provide a single pain rating at the end of each heat pulse, but there are many ways to compute TS using these ratings: taking the difference, the slope, or the area under the “curve,” between the first and various later pain ratings,6,7,11 or simply using the pain rating of the 5th pulse.15
Two problems exist with the traditional, varied approaches to describing TS. First, taking a single pain rating by a verbal cue at the end of each pulse artificially subjects the timing of the pain report to the timing of the verbal cue, resulting in less than optimal sampling resolution. Second, the various calculations of TS magnitude are inconsistent. For example, different relationships to factors such as anxiety7,15 and catastrophizing16 versus heat pain threshold17 were observed, depending on which measure was selected. While anxiety and catastrophizing are central, heat pain threshold is generally considered a peripheral property.18–20 Therefore, TS calculated by the various current methods likely reflects various contributions from both peripheral and central sensory processes.
Noxious heat pulses generate a multitude of sensory processes in addition to spinal windup, and there has been little effort to elucidate and isolate these processes from the characterization of thermal windup. To address this gap, we conducted our study in the following 3 steps. First, we expanded TS data collection from single pain ratings to a continuous curve by asking our participants to rate their pain continuously. Second, based on existing pain theories21,22 which broadly categorize pain perception into peripheral nociceptor activation and central pain modulation that include both ascending (which TS is a part of) and descending processes,23,24 we isolated 3 features of our continuous TS curves to represent peripheral nociceptor activation (i),19,25–27 windup (ii), and general inhibitory processes such as habituation28,29 and conditioned pain modulation (iii),24,30 respective. Finally, recognizing our hypothesis, though based on current pain research, might be an over-simplified interpretation of reality, we examined the validity of our model by evaluating (a) the temporal stability of the features; (b) the relationship among the features and between these features and participant characteristics including demographics and psychological profiles, as well as classical measures of TS such as the difference in the pain rating between the first and most painful heat pulse.
Methods
Overview
This report presents a secondary analysis of data from a previously published study.14 The original study was approved by the Stanford University institutional review board and was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent prior to participation in the study. The 2 objectives of the previous study were: (A) to improve the response to thermal TS paradigms by individually adjusting the baseline and peak temperatures of the heat pulses to achieve a moderate amount of TS (50 ± 20 VAS between the pain ratings of the first pulse and peak pain); (B) to assess the test–retest stability of thermal TS within the same day and over 1 week. Participants generated continuous TS response curves by providing a real-time rating of the second pain, although the magnitude of TS in that study was computed using a conventional method (maximal pain minus pain rating from the first pulse).11
In the current study, we used the same continuous TS curves, but extracted curve features representing complex processes triggered by noxious heat pulses, evaluated the within-individual temporal stability of these curve features, and explored the potential physiologic meaning of these features.
Participants And Behavioral Questionnaires
Healthy adults, free of major psychiatric or medical conditions, between the ages of 18 and 50, and did not have ongoing acute or chronic pain, were recruited. Mean age was 29.2 (SD 10.5) and 47% were female. After providing informed consent, each participant filled out a battery of questionnaires including: demographics, Beck’s Depression Inventory (BDI),31 Fear of Pain Questionnaire (FPQ),32 anxiety sensitivity index (ASI),33 Spielberger's state-trait anxiety inventory (STAI)34 including y1s (state anxiety) and y2t (trait anxiety), and sensitivity hypersensitivity scale (SHS).35
Study Timeline
After completion of the questionnaires, each participant underwent individual optimization of baseline and peak temperatures of the heat pulses to achieve an estimated TS (calculated by subtracting pain from the first heat pulse from that of the peak pain) between 30 and 70 on a VAS scale of 0–100, on day 1.14 Then, on day 4 and day 11, five identical TS trials using the optimized temperatures from day 1 were administered between 3 and 120min apart. The five trials from day 4 and five from day 11 were used for the analyses in this manuscript (Figure 1A).
Summary Of Thermal TS Paradigm
Except for the individualized temperatures, heat pulses from all TS trials have the following identical parameters: number of pulses = 10; peak-to-peak duration = 2s (frequency = 0.5Hz); duration of peak temperature = 0.5s; ramp up and down rate = 40°C/s. (Figure 1B) Heat pulses were first administered to the thenar eminence of the non-dominant hand, then alternating hand at each subsequent trial. We used a constant contact thermode, contact heat-evoked potential stimulator (CHEPS) (diameter = 2.9cm) from the Pathway machine (Medoc, Ramat Ishay, Israel) was used for all TS trials.
Training For Continuous Pain Rating During TS Paradigms
Each participant was asked to rate their pain continuously using a continuous output VAS (COVAS) box, which is a standard accessory to the Pathway machine. The COVAS allows real-time rating of pain by the visual analog scale (0 to 100, where 0 means “no pain” and 100 means “worst pain imaginable”).
We explained to each participant that second pain was often felt 1–2s after the delivery of the heat pulse and, that rather than being a sharp sensation, it tended to be dull, throbbing, and burning. The participants were instructed to only rate this pain and to ignore the first pain—the sharp sensation occurring immediately with each pulse, and the ramping up and down between the pulses. Finally, we instructed the participants that second pain may gradually build up and increase or decrease, and that it may linger for a short while before disappearing. The participants were instructed to rate their experience of the second pain continuously, including the build-up, lingering, and end point throughout each TS trial.
After the participants were shown the CHEPS thermode and COVAS and educated about the nature of second pain, they then underwent at least 2 training trials to become comfortable both with experiencing the heat pulses and rating the second pain continuously.14,36
Rationale And Extraction Of Key Curve Features
First, based on existing knowledge on the mechanisms of pain perception37,38 and windup,3,4,39 we hypothesized that noxious heat pulses trigger 3 general processes: (i) peripheral nociception, i.e., activation of A-delta and C fibers,18,25,27 (ii) windup taking place at the spinal dorsal horn,4,39 (iii) inhibitory processes such as peripheral habituation29 and/or conditioned pain modulation30,40 in response to peripheral activation. Second, based on the above hypothesis, we selected curve features that may potentially reflect these 3 processes. For example, the time it takes to first perceive pain is likely related to peripheral sensitivity (i);18,27 the slope of increase in pain perception between the first and most painful pulse most likely represents windup (ii);7,17 and the decrease in pain perception after maximum pain likely is related to some kind of inhibitory process (iii).13,28,29 Lastly, recognizing (a) many features can be extracted from the TS curve and (b) more than one feature can relate to each of the proposed sensory processes, we selected three features which best describe the between-individual variability of the curve shapes, and are relatively easy to compute, as described below.
As Figure 2 shows, the first feature is Lag, representing the time it takes to first perceive pain from the heat pulses. To minimize the influence of noise, we selected a threshold of 5 VAS (over 100) rather than zero. Because Lag represents the beginning of pain perception to noxious heat, we hypothesize that Lag would be a surrogate of peripheral nociception, i.e., c-fiber activation.
The second feature is Slope, defined as the maximum pain rating (Pmax) subtracted by the pain rating from the first heat pulse (PP1) divided by time. Previous investigators have used slope (fitted to discrete pain ratings of each pulse) to define TS because it closely approximates the sensitization to identical series of stimuli.7,11,13,17,41 However, the choices of the starting and ending points for the slope vary. For the starting point of Slope, we picked a point of around 4s on the x-axis, which corresponds to the approximate time when the second pain from the first pulse is perceived.8,14 Evidence suggests that peripheral factors strongly influence the rating of the first pulse;4,42–44 therefore, we minimized the peripheral contribution to our calculation of Slope by subtracting the pain rating of the first pulse (PP1) from the maximum pain rating. We selected the maximum pain rating as the endpoint in our Slope calculation because it represents the maximum augmentation in pain perception in response to repetitive heat stimuli.6,45
Finally, the third feature is Delta, defined as the maximum drop in pain rating after reaching peak pain. Previous authors have suggested the decrease of pain perception may relate to descending inhibitory processes triggered by noxious heat pulses.13,46
Once we determined the choice of the key curve features, we extracted these features from all 10 TS trials from day 4 and day 11, using MATLAB, version 8.2 (Mathworks, Natick, MA, USA). The mean and standard deviation of these features were then calculated.
Analysis Of Temporal Stability Within Individual
We were specifically interested in the stability of the curve features over the course of 1 week. For each participant, we used a permutation test to assess the temporal stability of the extracted features without making distributional assumptions of the features. Our null hypothesis is that the mean value of the features on day 4 is equal to that on day 11. To do test this hypothesis, we randomly divide then 10 observations into two groups: 5 labeled day 4 and 5 labeled day 11, giving 252 possible ways of division (10 choose 5). For each division, we computed the mean values for each day and estimated the mean difference between day 4 and day 11 for each feature. We hypothesized that if there were no difference between days, then it should not matter which 5 of the 10 measurements we selected and designated as “day 4 values.” If the “un-permuted” (that is to say, real) data lead to a difference in absolute value between days that is kth largest among the 252, then an estimate of the “p-value” for the null hypothesis is given by k/252 (by “p-value” we mean “attained significance level,” that is, the chance of seeing a difference as large or larger than what was observed by random chance only, given there is no difference).
Assessment Of Curve Features Via Correlation Analysis
We selected Lag, Slope, and Delta to approximate peripheral nociception, spinal windup, and descending inhibition/habituation, respectively. We explored the relationship among these curve features, and between them and individual characteristics such as demographics and psychological traits. Because we individualized the baseline and peak stimulating temperatures of the heat pulses to generate moderate TS in all individuals, we also included these individualized temperatures in the correlation analysis.
First, we computed the correlations outlined above using the mean features from all 10 observations for each participant. Second, because the distributions of the features, as well as the participant characteristics are not Gaussian, we used permutation statistics to compute the p-value of the correlations, in a fashion similar to the permutation method described earlier to assess the temporal stability of the curve features. The details of our methods are outlined below.
Step 1: Compute p-values by permuting the features among the 19 participants. Specifically, there are a total of (19!),3 approximately 1.8 × 1051 ways to shuffle (permute) the 3 features among the 19 representative curves. For each shuffle, a Pearson’s correlation coefficient, r, was computed. The correlation of the non-permuted features was also computed and ranked as the kth highest among the permuted correlations. The null hypothesis is that there is no correlation, i.e., r = 0. The significance value of the null hypothesis is then k/1051 However, given the large number of possible shuffles possible, we chose to estimate the p-value using Matlab to simulate 10,000 random possible shuffles and then ranked the non-permutated correlation as k’th highest among the 10,000 permuted correlations. This gave us an estimated p-value of k’/10,000.
Readers will note that it would have been enough to fix a randomly chosen feature and to permute the other 2. However (19!)2 is also an enormous number. We would then have had to subsample permutations. Our permuting all features has obvious appeal, anyway.
Step 2: Practical restraints on permutation statistics. Not all random permutations are realizable. For example, if a participant takes a long time to first feel pain (long Lag) and reports a slow pain rise (low Slope), then a high Delta would not be possible. We used the following formulas to screen the permutations generated in step 2 above and retained only those that are physically possible. We needed to reject ~30% of our permutations based on the rules below.
Maximum remaining time = 22 – (time of PP1) = 22–3.9 = 18.1s
Achievable max pain = PP1 + slope × 18.1
Reject permutation if: Delta > Achievable max pain; or (Lag ≥ 22s and Slope > 0).
We also repeated the above analyses after removing outliers. As our primary interest was in windup, we identified subjects whose average Slope from all 10 TS trials exceeded twice the group mean.
Results
Participant Characteristics And Individualized Stimulating Temperatures
A total of 19 participants completed the experiment. Of these, 10 were male (average age 29.7, SD = 10.9) and 9 were female (average age 28.2, SD = 10.9). The demographics of the study cohort and the results of the behavioral questionnaires are summarized in Table S1 in part 1 of the Supplemental Material. The individualized baseline (b-temp) and peak (p-temp) stimulating temperatures are summarized in Table 1. Note that the goal of the optimization is to achieve an estimated TS (calculated as maximum pain reported subtracted by pain from first heat pulse) between 30 and 70 points on a 0–100 VAS scale.14
 |
Table 1 Individualized Baseline (B-temp) And Peak (P-temp) Stimulating Temperatures |
Continuous TS Response Curves And Variability In Curve Shape
We were able to obtain continuous TS response curves for each of the 10 trials for all 19 participants. All 190 curves are available as individual plots and as daily means for each participant in Part 2 and Part 3 of the Supplemental Material, respectively. Figure 3 contains examples of the daily mean TS curves (computed by averaging 5 curves from each day) from 3 representative participants. Figure 3 demonstrates that there is a large variation in the shape of the mean TS curves, but the curve shapes largely preserved between day 4 and day 11.
Summary Of Mean Curve Features
Table 2 summarizes the mean curve features. Because we found these features were temporally stable (detailed in section 3 below), we chose to represent the features as the mean value over all 10 TS trials from both day 4 and day 11. Figure 4 shows the distribution of the curve features before and after taking out two outliers who had >2 times the mean in Slope. We chose to focus on Slope because based on our hypothesis, it was the most relevant feature to windup.
 |
Table 2 Curve Features From Each Participant (Average Of 10 trials) |
Temporal Stability Of Curve Features
Table 3 contains the within-individual, between-day stability of Lag, Slope, and Delta. Bold highlights indicate a significant difference with p < 0.05, which suggests that for these few individuals, the highlighted features may not be identical between days. Overall, the features remain stable for the majority of the participants.
 |
Table 3 Temporal Stability Of Curve Features |
Correlation Analyses Of Curve Features
Correlation Between Slope And Classical TS Measures, And Among Curve Features
First, we found strong correlation between Slope and traditional measures of TS, including the difference between PP1 (pain from first pulse) and Pmax (maximum pain), and between PP1 and pain from the last pulse (r = 0.91, p < 0.0001; and r = 0.72, p =0.0007, respectively). Second, while Lag and Delta were negatively correlated (r = −0.50, p = 0.042), there were no statistically significant correlations between Slope and these two features.
Correlation Between Curve Features And Individualized Stimulating Temperatures
As shown in Table 4, while Lag and Delta demonstrate various correlations with the peak and baseline stimulating temperatures, Slope showed no statistically significant correlation with either temperatures.
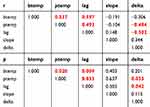 |
Table 4 Correlation Among Curve Features, And Between Curve Features And Stimulating Temperatures |
Correlation Between Curve Features (Including Temperatures) And Participant Characteristics
We found no significant correlations between the curve features and any of the demographic or psychological measures, including age, sex, physical sensitivity (SHS), depression (BDI), fear (FPQ), or anxiety (ASI and STAI).
However, as shown in Table 5, the baseline stimulating temperature (b-temp) was negatively correlated with trait anxiety (STAI-Y2T, r = −0.49, p = 0.031) and the peak stimulating temperature (p-temp) was negative correlated state anxiety (STAI-Y1S, r = −0.48, p = 0.039).
 |
Table 5 Correlation Between TS Curve Features (Including Stimulating temperatures) And Participant Characteristics |
Sensitivity Analysis On Correlations After Removing Outliers In Slope (Table 6)
As shown in Figure 4 and Table 2, ID #7 and #15 had Slope values more than twice that of the group mean. We therefore repeated the correlation analyses above with these subjects removed. Table 6A demonstrates that the relationships among the curve features and between them and the stimulating temperatures remained unchanged compared to Table 4. However, Table 6B shows that new relationship between Slope and BDI (r = 0.49, p = 0.049) appeared with removal of these outliers (compared to Table 5). Furthermore, there is an almost significant correlation between Slope and trait anxiety, STAI-y2t (r= 0.48, p = 0.052).
 |
Table 6 Correlation Analyses AFTER Removal Of Outliers In Slope (ID #7 And ID #15) |
Discussion
In this exploratory study, we have demonstrated the feasibility of recording thermal TS responses using a continuous response vs time curve of pain ratings. We selected 3 curve features to approximate 3 physiological processes triggered by noxious heat pulses: peripheral nociception, windup, and habituation (and/or inhibition). After demonstrating within-individual temporal stability of these features, our preliminary correlation analysis suggested that we have successfully separated windup from other sensory processes triggered by noxious heat pulses.
Advantages Of Recording Thermal TS Response Using A Continuous Curve
Compared with traditional methods in which only discrete pain ratings are reported at the cue of the experimenter, using continuous rating of second pain offers more information and is simpler to perform. It allows us to obtain more precise estimates of parameters such as time to pain perception (Lag) and duration of after sensations. Our method also frees the experimenter from using and recording the subject’s pain level after each pulse. Finally, traditional measures of thermal TS (such as absolute difference between the first and most painful pulse) can easily be derived from this continuous curve.
The major barriers to recording continuous TS response curves are: (1) a device must be available that allows continuous pain recording and is synchronized to the temperature curve; (2) it is more difficult to train the participant to rate the second pain continuously than it is to provide a single rating at a time prompted by the experimenter. Despite these barriers, our results demonstrated strong correlation between our windup measure (Slope) and other traditional measures of windup (e.g., change between pain from the first pulse and that of the most painful pulse), confirming success of our data collection method.
Ultimately, the ability to capture continuous pain ratings in response to thermal TS stimuli would allow the collection of data with higher temporal resolution than previously possible. Consequently, it allows advanced modeling to differentiate the various physiological processes triggered by noxious heat pulses as well as explore the correlation of these features with participant demographic and psychological attributes.
Selection Of Curve Features: Separating Windup From Other Sensory Processes
We hypothesized that Lag, Slope, and Delta may approximate peripheral nociception, spinal windup, and habituation and/or descending inhibition, respectively. Although it is not possible to directly prove our hypothesis, our data support the association between the chose curve features and the proposed underline physiological processes.
First, 3 lines of evidence suggest that we have separated windup (Slope) from other sensory processes triggered by noxious heat pulses. First, our windup measure, Slope, correlated strongly with traditionally TS measures from the literature.7,13,41 Second, while Lag and Delta were negatively correlated, we did not find statistically significant correlation between these features and Slope. Third, while Lag and Delta correlated well with the stimulating temperatures which are related to peripheral sensitivity,13,14 we did not find statistically significant correlations between Slope and the stimulating temperatures. Therefore, the above evidence suggests that Slope is strongly related to windup, a central phenomenon and less related to peripheral sensitivity as represented by the individualized stimulating temperatures.
Second, Lag is defined as the time it takes to first perceive heat pain which has been shown to relate to peripheral nociceptor sensitivity.47 Our data show Lag is correlated to both baseline and peak stimulating temperatures. During optimization,14 we ensured that the baseline stimulating temperature (b-temp) generates a warm (maybe slightly painful) sensation, and is limited between 38°C and 42°C, which is in the range of cutaneous c-fiber threshold in primates.18,26,48 In contrast, the peak stimulus temperature (p-temp) is delivered rapidly and is supra-threshold (47–51°C).14 This range of peak stimulating temperature overlaps with the typical range of A-delta thresholds in primates,19,48 and in C fibers from other studies.49,50 Furthermore, the mean Lag from this group of pain-free individuals is approximately 3s, i.e., 1s after the heat pulse. This reaction time is consistent with reaction time from peripheral c-fiber activation, whose conduction speed is approximately 0.9m/s.25,27 As such, both its correlation with stimulating temperatures and the similarity between Lag and reaction time associated with c-fiber activation support the association between Lag and c-fiber mediated peripheral nociception. Finally, despite our best efforts, some participants may still be reporting A–delta-mediated first pain, especially during the first few pulses. Other researchers have reported this phenomenon.15,39,42
Delta is defined as the maximum decrease in pain ratings after the peak pain is reached. It may relate to peripheral A-delta and/or C fiber activities, as shown by a correlation of −0.48 (p = 0.033) with the peak stimulating temperature.19,48–50 Delta might represent several processes. For example, Bosma et al identified BOLD activities in the RVM and PAG regions of the brain in response to thermal TS paradigm.46 These regions are implicated in endorphin-mediated descending pain inhibition.51 In addition, strong A-delta stimulation, particularly of the type II A-delta mechano-heat (AMH) fibers, leads to fatigue and adaption of peripheral A-delta response.27,52 Anderson et al13 hypothesized that such “peripheral saturation” may explain the lack of TS, or temporal decrease of second pain (TDSP), observed in more than half of the healthy individuals they tested. We have advanced Anderson’s hypothesis by (a) identifying a possible surrogate measure of A-delta sensitivity (peak stimulus temperature); (b) estimating degree of “peripheral contribution” via Delta. In summary, Delta might represent a combination of central inhibition and peripheral adaptation in response to noxious heat pulses, though clearly more studies are needed to characterize the relationship between these processes and Delta.
Between-Day Stability Of Key Curve Features
Our results indicate the general stability of all three chosen curve features. Slope, which closely represents windup, demonstrated identical values for both testing days in 16 of the 19 participants. This result is consistent with prior reports of the general stability of TS as measured by other methods.14,41 The results on Lag and Delta suggest that sensory processes represented by these features are also grossly stable.
Relationship Between Curve Features And Participant Characteristics
In the full sample, while we did not find statistically significant correlations between curve features and participant characteristics, we found inverse correlations between the baseline and peak stimulating temperatures to trait and state anxiety, respectively. Considering we had individually adjusted the temperature settings of the heat pulses to generate moderate amount of TS in our participants, it is possible that these individualized temperature settings reduced the variability in our curve features. Furthermore, the relationship between TS parameters and anxiety found in our study is consistent with literature.7,15 In these other examples, the authors did not individualize the thermal stimulation and found correlation between magnitude of TS (measured as difference between first and most painful pulse) and anxiety.
After removing ID #7 and #15, Slope demonstrated positive correlation with depression and likely anxiety. While these make intuitive sense, given our limited sample size, we are careful not to draw definitive conclusions on our outlier analysis. A greater sample size is clearly needed in our next, validation study.
Limitations And Future Studies
The current study has several limitations.
First, as an exploratory study, we had made several assumptions interpreting the correlation analyses: (i) we only examined linear relationships between curve features and other parameters of interest; (ii) we did not account for multiple comparisons; (iii) in cases where we did not find statistically significant correlations, it is conceivable to observe some correlations with much larger sample size. However, our practice of examining linear relationships in an exploratory study does not deviate significantly from the literature.7,15 Furthermore, looking at Tables 4 and 5, few of the p values are in the gray zone thus making our other points less relevant.
Second, like many other TS methods, our methodology requires extensive participant training to ensure that participants (a) understand the anchors for 0 and 100 on the VAS; (b) able to differentiate first and second pain; (c) able to rate second pain in a continuous fashion. Therefore, a prerequisite for successful capture of continuous thermal TS response is adequate and standardized participant training. While it may be time-consuming, it does allow capture of more information with higher temporal resolution.
Third, we built a simple model of only 3 curve features that may not completely capture the complex between-individual variability in TS response. Furthermore, while our selected curve feature (Slope) demonstrated strong correlations to existing measures of TS, we do not have enough evidence to confirm that Lag represents only peripheral nociceptor activation or Delta in fact measures habituation ± descending pain modulation. However, to the best of our knowledge, our attempt is the first to define features in a phasic thermal TS response curve AND to isolate what appears to be an excellent measure of windup from other processes triggered by noxious heat pulses. The competition and balance between these sensory processes may offer answers to the great between-individual variability seen in thermal TS responses.7,13,53
In the future, we propose to extend our study and make it clinically relevant by: (1) validate our results in larger cohorts and contrasting curve features between those with vs without chronic pain; (2) stratify patients based curve profiles and evaluate relationship between curve profiles and risks of chronic pain as well as response to treatment; (3) further characterize Lag and Delta where TS and other QST modalities will be simultaneously tested. Additionally, we may include other relevant patient characteristics beyond those analyzed here (e.g., catastrophizing, history of childhood trauma, etc.) in studying these curve features.
Conclusions And Clinical Utilities
We have demonstrated feasibility of recording thermal TS responses using a continuous curve of pain ratings. Furthermore, we were successful in isolating a windup measure consistent with previous studies from other sensory processes, and in quantifying these processes using curve feature modeling. Compared to traditional methods of quantifying thermal TS where a single feature is derived based on discrete pain ratings, our method affords higher resolution and the opportunity of better characterizing the complex sensory processes triggered by heat pulse administration. Future studies are clearly needed to further characterize thermal TS response and to understand the physiologic significance of Lag and Delta. Ultimately, these curve features may be used together as biomarkers to differentiate patients with chronic pain from health controls, and to stratify patients into diagnostic and prognostic groups to facilitate personalized delivery of pain relief strategies.
 |
Figure 1 Experimental Design. (A) Study timeline. (B) Phasic thermal temporal summation paradigm. |
 |
Figure 2 Selection of Key Features from an example TS response curve. |
Acknowledgment
We acknowledge Raymond Balise, PhD, for his advice regarding alternative methods to analyze our data, Kevin Johnson, PhD, for discussion on different methods of administering heat pulses, and Sophia You, PhD, RN, for feedback to the composition this manuscript.
Disclosure
The authors report no conflicts of interest with this manuscript.
References
1. Arendt-Nielsen L, Petersen-Felix S. Wind-up and neuroplasticity: is there a correlation to clinical pain? Eur J Anaesthesiol Suppl. 1995;10:1–7.
2. Eide PK. Wind-up and the NMDA receptor complex from a clinical perspective. Eur J Pain. 2000;4(1):5–15. doi:10.1053/eujp.1999.0154
3. Price DD. Characteristics of second pain and flexion reflexes indicative of prolonged central summation. Exp Neurol. 1972;37(2):371–387. doi:10.1016/0014-4886(72)90081-7
4. Price DD, Hu JW, Dubner R, Gracely RH. Peripheral suppression of first pain and central summation of second pain evoked by noxious heat pulses. Pain. 1977;3(1):57–68. doi:10.1016/0304-3959(77)90035-5
5. Arendt-Nielsen L, Yarnitsky D. Experimental and clinical applications of quantitative sensory testing applied to skin, muscles and viscera. J Pain. 2009;10(6):556–572. doi:10.1016/j.jpain.2009.02.002
6. Edwards RR, Fillingim RB. Effects of age on temporal summation and habituation of thermal pain: clinical relevance in healthy older and younger adults. J Pain. 2001;2(6):307–317. doi:10.1054/jpai.2001.25525
7. Granot M, Granovsky Y, Sprecher E, Nir RR, Yarnitsky D. Contact heat-evoked temporal summation: tonic versus repetitive-phasic stimulation. Pain. 2006;122(3):295–305. doi:10.1016/j.pain.2006.02.003
8. Mauderli AP, Vierck CJ
9. Vierck CJ
10. Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley JL
11. Greenspan JD, Slade GD, Bair E, et al. Pain sensitivity risk factors for chronic TMD: descriptive data and empirically identified domains from the OPPERA case control study. J Pain. 2011;12(11 Suppl):T61–74. doi:10.1016/j.jpain.2011.08.006
12. Reynolds WS, Kowalik C, Cohn J, et al. Women undergoing third line overactive bladder treatment demonstrate elevated thermal temporal summation. J Urol. 2018;200(4):856–861. doi:10.1016/j.juro.2018.05.007
13. Anderson RJ, Craggs JG, Bialosky JE, et al. Temporal summation of second pain: variability in responses to a fixed protocol. Eur J Pain. 2013;17(1):67–74. doi:10.1002/ejp.2013.17.issue-1
14. Kong JT, Johnson KA, Balise RR, Mackey S. Test-retest reliability of thermal temporal summation using an individualized protocol. J Pain. 2013;14(1):79–88. doi:10.1016/j.jpain.2012.10.010
15. Robinson ME, Bialosky JE, Bishop MD, Price DD, George SZ. Supra-threshold scaling, temporal summation, and after-sensation: relationships to each other and anxiety/fear. J Pain Res. 2010;3:25–32. doi:10.2147/JPR.S9462
16. George SZ, Wittmer VT, Fillingim RB, Robinson ME. Sex and pain-related psychological variables are associated with thermal pain sensitivity for patients with chronic low back pain. J Pain. 2007;8(1):2–10. doi:10.1016/j.jpain.2006.05.009
17. Bhalang K, Sigurdsson A, Slade GD, Maixner W. Associations among four modalities of experimental pain in women. J Pain. 2005;6(9):604–611. doi:10.1016/j.jpain.2005.04.006
18. Tillman DB, Treede RD, Meyer RA, Campbell JN. Response of C fibre nociceptors in the anaesthetized monkey to heat stimuli: correlation with pain threshold in humans. J Physiol. 1995;485(Pt 3):767–774. doi:10.1113/jphysiol.1995.sp020767
19. Treede RD, Meyer RA, Raja SN, Campbell JN. Evidence for two different heat transduction mechanisms in nociceptive primary afferents innervating monkey skin. J Physiol. 1995;483(Pt 3):747–758. doi:10.1113/jphysiol.1995.sp020619
20. Van Hees J, Gybels J. C nociceptor activity in human nerve during painful and non painful skin stimulation. J Neurol Neurosurg Psychiatry. 1981;44(7):600–607. doi:10.1136/jnnp.44.7.600
21. Melzack R. Gate control theory: on the evolution of pain concepts. Pain Forum. 1996;5(2):128–138. doi:10.1016/S1082-3174(96)80050-X
22. Melzack R, Wall PD. Pain mechanisms: a new theory. Science. 1965;150(3699):971–979. doi:10.1126/science.150.3699.971
23. Phillips K, Clauw DJ. Central pain mechanisms in chronic pain states–maybe it is all in their head. Best Pract Res Clin Rheumatol. 2011;25(2):141–154. doi:10.1016/j.berh.2011.02.005
24. Yarnitsky D, Granot M, Granovsky Y. Pain modulation profile and pain therapy: between pro- and anti-nociception. PAIN®. 2014;155(4):663–5.
25. Treede RD. Peripheral acute pain mechanisms. Ann Med. 1995;27(2):213–216. doi:10.3109/07853899509031961
26. Treede RD, Meyer RA, Campbell JN. Comparison of heat and mechanical receptive fields of cutaneous C-fiber nociceptors in monkey. J Neurophysiol. 1990;64(5):1502–1513. doi:10.1152/jn.1990.64.5.1502
27. Treede RD, Meyer RA, Campbell JN. Myelinated mechanically insensitive afferents from monkey hairy skin: heat-response properties. J Neurophysiol. 1998;80(3):1082–1093. doi:10.1152/jn.1998.80.3.1082
28. Agostinho CMS, Scherens A, Richter H, et al. Habituation and short-term repeatability of thermal testing in healthy human subjects and patients with chronic non-neuropathic pain. Eur J Pain. 2009;13(8):779–785. doi:10.1016/j.ejpain.2008.10.002
29. Greffrath W, Baumgartner U, Treede RD. Peripheral and central components of habituation of heat pain perception and evoked potentials in humans. Pain. 2007;132(3):301–311.
30. Le Bars D. The whole body receptive field of dorsal horn multireceptive neurones. Brain Res Rev. 2002;40(1–3):29–44. doi:10.1016/S0165-0173(02)00186-8
31. Beck AT, Ward CH, Mendelson MM, Mock JJ, Erbaugh JJ. AN inventory for measuring depression. Arch Gen Psychiatry. 1961;4(6):561–571. doi:10.1001/archpsyc.1961.01710120031004
32. McNeil DW, Rainwater AJ
33. Reiss S, Peterson RA, Gursky DM, McNally RJ. Anxiety sensitivity, anxiety frequency and the prediction of fearfulness. Behav Res Ther. 1986;24(1):1–8. doi:10.1016/0005-7967(86)90143-9
34. Spielberger CD. Manual for the State-Trait Anxiety Inventory STAI (Form Y).
35. Benham G The sensory sensitivity scale: sex and population differences.
36. Mackey IG, Dixon EA, Johnson K, Kong JT. Dynamic quantitative sensory testing to characterize central pain processing. J Vis Exp. 2017;120.
37. Garland EL. Pain processing in the human nervous system: a selective review of nociceptive and biobehavioral pathways. Prim Care. 2012;39(3):561–571. doi:10.1016/j.pop.2012.06.013
38. Loeser JD, Melzack R. Pain: an overview. Lancet. 1999;353(9164):1607–1609. doi:10.1016/S0140-6736(99)01311-2
39. Price DD, Dubner R. Mechanisms of first and second pain in the peripheral and central nervous systems. J Invest Dermatol. 1977;69(1):167–171. doi:10.1111/1523-1747.ep12497942
40. Yarnitsky D, Arendt-Nielsen L, Bouhassira D, et al. Recommendations on terminology and practice of psychophysical DNIC testing. Eur J Pain. 2010;14(4):339. doi:10.1016/j.ejpain.2010.02.004
41. Alappattu MJ, Bishop MD, Bialosky JE, George SZ, Robinson ME. Stability of behavioral estimates of activity-dependent modulation of pain. J Pain Res. 2011;4:151–157. doi:10.2147/JPR.S18105
42. Fillingim RB, Ness TJ, Glover TL, Campbell CM, Price DD, Staud R. Experimental pain models reveal no sex differences in pentazocine analgesia in humans. Anesthesiology. 2004;100(5):1263–1270. doi:10.1097/00000542-200405000-00031
43. Staud R, Bovee CE, Robinson ME, Price DD. Cutaneous C-fiber pain abnormalities of fibromyalgia patients are specifically related to temporal summation. Pain. 2008;139(2):315–323. doi:10.1016/j.pain.2008.04.024
44. Valencia C, Fillingim RB, George SZ. Suprathreshold heat pain response is associated with clinical pain intensity for patients with shoulder pain. J Pain. 2011;12(1):133–140. doi:10.1016/j.jpain.2010.06.002
45. Edwards RR, Ness TJ, Weigent DA, Fillingim RB. Individual differences in diffuse noxious inhibitory controls (DNIC): association with clinical variables. Pain. 2003;106(3):427–437. doi:10.1016/j.pain.2003.09.005
46. Bosma RL, Ameli Mojarad E, Leung L, Pukall C, Staud R, Stroman PW. Neural correlates of temporal summation of second pain in the human brainstem and spinal cord. Hum Brain Mapp. 2015;36(12):5038–5050. doi:10.1002/hbm.v36.12
47. Schepers RJ, Ringkamp M. Thermoreceptors and thermosensitive afferents. Neurosci Biobehav Rev. 2010;34(2):177–184. doi:10.1016/j.neubiorev.2009.10.003
48. Churyukanov M, Plaghki L, Legrain V, Mouraux A. Thermal detection thresholds of Aδ- and C-fibre afferents activated by brief CO2 laser pulses applied onto the human hairy skin. PLoS One. 2012;7(4):e35817–e35817. doi:10.1371/journal.pone.0035817
49. Beitel RE, Dubner R. Response of unmyelinated (C) polymodal nociceptors to thermal stimuli applied to monkey’s face. J Neurophysiol. 1976;39(6):1160–1175. doi:10.1152/jn.1976.39.6.1160
50. Lynn B, Carpenter SE. Primary afferent units from the hairy skin of the rat hind limb. Brain Res. 1982;238(1):29–43. doi:10.1016/0006-8993(82)90768-5
51. Fields H. State-dependent opioid control of pain. Nat Rev Neurosci. 2004;5(7):565–575. doi:10.1038/nrn1431
52. LaMotte RH, Campbell JN. Comparison of responses of warm and nociceptive C-fiber afferents in monkey with human judgments of thermal pain. J Neurophysiol. 1978;41(2):509–528. doi:10.1152/jn.1978.41.2.509
53. Raphael KG, Janal MN, Anathan S, Cook DB, Staud R. Temporal summation of heat pain in temporomandibular disorder patients. J Orofac Pain. 2009;23(1):54–64.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


