Back to Journals » Clinical Epidemiology » Volume 8
Mesothelioma in the United States: a Surveillance, Epidemiology, and End Results (SEER)–Medicare investigation of treatment patterns and overall survival
Authors Beebe-Dimmer JL, Fryzek JP, Yee CL, Dalvi TB, Garabrant DH, Schwartz AG, Gadgeel S
Received 30 January 2016
Accepted for publication 26 May 2016
Published 26 October 2016 Volume 2016:8 Pages 743—750
DOI https://doi.org/10.2147/CLEP.S105396
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Vera Ehrenstein
Jennifer L Beebe-Dimmer,1,2 Jon P Fryzek,3 Cecilia L Yee,1,2 Tapashi B Dalvi,4 David H Garabrant,3 Ann G Schwartz,1,2 Shirish Gadgeel1,2
1Department of Oncology, Wayne State University School of Medicine, 2Barbara Ann Karmanos Cancer Institute, Detroit, 3EpidStat Institute, Ann Arbor, MI, 4AstraZeneca, Gaithersburg, MD, USA
Introduction: Mesothelioma is a rare malignancy typically associated with exposure to asbestos and poor survival. The purpose of this investigation was to describe mesothelioma patient characteristics, treatment patterns, and overall survival (OS) utilizing the National Cancer Institute’s Surveillance, Epidemiology, and End Results–Medicare database.
Materials and methods: Patients in this study were diagnosed with malignant mesothelioma of the pleura or peritoneum between January 1, 2005 and December 31, 2009 with follow-up for survival through December 31, 2010. We examined both patient and tumor characteristics at time of diagnosis and subsequent treatment patterns (surgery, radiation, and chemotherapy). Among patients treated with chemotherapy, we determined chemotherapy regimen and OS by line of therapy.
Results: Of the 1,625 patients considered eligible for this investigation, the median age at diagnosis was 78 years. Nearly a third of patients (30%) had surgery as part of their treatment and 45% were given chemotherapy. The median OS was 8 months (range 1–69 months). Among chemotherapy patients, the most commonly (67%) prescribed regimen for first-line therapy was cisplatin or carboplatin (Ca/Ci) combined with pemetrexed (Pe). Among those prescribed Ca/Ci + Pe as first-line therapy, retreatment with Ca/Ci + Pe (28%) or treatment with gemcitabine (30%) were the most common second-line therapies. Median OS for those receiving first-line chemotherapy was 7 months, and among those receiving second-line therapy median OS was extended an additional 5 months.
Conclusion: Irrespective of surgical resection, mesothelioma patients receiving some form of chemotherapy survived longer than patients who did not, with an additional survival benefit among those patients receiving multimodal treatment.
Keywords: mesothelioma, chemotherapy, epidemiology, outcomes, mortality, morbidity, survival
Introduction
Malignant mesothelioma is a rare malignancy of the membrane that covers many internal organs, but affects mainly the pleura of the lung. It may also arise in the peritoneum, and in extremely rare instances the testes or pericardium.1 Mesothelioma is often difficult to diagnose, which contributes to its poor prognosis; however, once the disease is diagnosed, it is uniformly lethal. Patients with malignant mesothelioma typically present with dyspnea, chest pain, and/or pleural effusion. While patients with suspected malignant mesothelioma routinely undergo pleural biopsy to establish diagnosis, cytological examination of the pleural fluid is often nonconfirmatory, even in the presence of an occupational asbestos workplace history, the primary cause of mesothelioma.2 The “gold standard” for diagnosis of malignant mesothelioma is thoracoscopy.1 Similarly, endoscopic biopsies of other areas of the body where mesotheliomas arise are used for confirmatory diagnoses.
About 3,000 new cases of mesothelioma are diagnosed in the US each year, more often in men, those aged 65 years and older, and whites.3 While the rate of mesothelioma increased between the 1970s through the 1990s, it is currently leveling off or even decreasing, likely due to changes in workplace exposure to asbestos beginning in the 1970s.4
Malignant pleural mesothelioma is the only mesothelioma with a formal staging system, with the American Joint Committee on Cancer (AJCC) TNM (tumor, nodes, metastasis) staging system most often used.5 While other staging systems have also been proposed,6 staging for mesothelioma is not a useful prognostic indicator. Cell subtypes, including epithelial, sarcomatoid, and biphasic, are more useful. The epithelial subtype is the most common (50% of tumors) and has the most favorable prognosis.1 Other characteristics associated with prognosis include performance status, sex, age, white blood cell count, and platelet count.7
While the recommended treatment for malignant mesothelioma is based on clinical stage and cell subtype,1 there is a lack of published population-based data describing routine treatment patterns for mesothelioma patients. Therefore, we assembled data from the linked Surveillance, Epidemiology, and End Results (SEER)–Medicare data files to report on initial therapies used for treatment of newly diagnosed mesothelioma and related overall survival (OS) among patients aged 66 years and older.
Materials and methods
The SEER–Medicare (http://healthservices.cancer.gov/seermedicare/obtain/use.html) data set is a unique resource linking two population-based sources of data used to provide information about the experience of elderly patients with cancer in the US. The SEER program, sponsored by the National Cancer Institute, is a network of population-based cancer registries that routinely collects information on patients with a new diagnosis of invasive cancer, residing within one of the registry catchment areas. SEER is composed of 18 statewide or regional cancer registries, collecting data on patient demographics, tumor histology and pathology, first course of treatment, and OS. Through linking SEER registry data to Medicare enrollment and claims information for patients diagnosed in the SEER regions, the SEER–Medicare database provides additional information on treatment and outcomes of approximately 25% of elderly patients diagnosed with cancer in the US.8
Patients eligible for inclusion in this study were aged 66 years and older at the time of diagnosis of primary malignant mesothelioma of the pleura (C38.4), lung (C34–34.9), or peritoneum, including omentum and mesentery (C48–48.9) (International Classification of Diseases from Oncology, third edition) between January 1, 2005 and December 31, 2009, captured among one of 16 SEER registries (Connecticut, Hawaii, Iowa, New Mexico, Utah, rural Georgia, Detroit, Seattle–Puget Sounds, Los Angeles, San Jose–Monterey, San Francisco–Oakland, greater California, Kentucky, Louisiana, and New Jersey). Patients captured in the specialized population registries (Alaska Native Tumor Registry and Arizona Indians) are not included in SEER–Medicare. Patients aged 66 years and older were selected in an effort to document the presence of any comorbid conditions (summarized using a modified Charlson Comorbidity Index) in the 12 months prior to diagnosis. Patients not continuously enrolled in both Part A and Part B Medicare from 12 months prior to diagnosis until death or December 31, 2010 were excluded, in an effort both to minimize misclassification of treatment regimen and to estimate a comorbidity index at the time of diagnosis. Patients who were members of a health maintenance organization at any point during this same period were also excluded, to avoid the potential for missing information due to claims not processed through Medicare. Additional exclusion criteria were 1) diagnosis from death certificate or autopsy only (n=63), 2) diagnosis not microscopically confirmed (n=189), 3) eligibility for Medicare not age related (n=203), 4) year of birth or death from SEER and Medicare not well matched or missing month of diagnosis (n=39), 5) unknown or unconfirmed surgery (n=60), and 6) chemotherapy given, but agent not documented (n=47). Based upon these criteria, a total of 1,625 patients were eligible for inclusion in the study.
All treatments within the first month of diagnosis until death or end of the study were captured using Medicare claim data using relevant Healthcare Common Procedure Coding System, International Classification of Diseases (ICD)-9 diagnosis, and procedure and surgical codes. The most commonly administered chemotherapy agents were cisplatin (Ci), carboplatin (Ca), pemetrexed (Pe), and gemcitabine, with vinorelbine less frequently used. These different agents were either combined or administered singularly and were summarized according to line of therapy. We combined use of either Ci or Ca into one line of therapy (as both are platinum-based chemotherapies), and these agents may or may not have been combined with Pe. A second-line therapy was identified, based on the presence of additional claim data indicating chemotherapy beyond the first prescribed regimen with a gap of at least 3 months postconclusion of first-line therapy. Surgery was considered therapeutic if it was performed within 9 months of diagnosis and palliative when performed more than 9 months after diagnosis. For the purposes of this study, only therapeutic surgery was considered in estimating differences in OS among different patient groups. The study was reviewed by the Wayne State University institutional review board and was granted a concurrence of exemption. Individual patient consent is not deemed neccessary by the institutional review board as only grouped data with more than 10 individuals represented in the group is presented.
Statistical methods
All statistical analyses were performed using SAS version 9.3 (SAS Institute, Cary, NC, USA). The distribution of patient demographic (age at diagnosis, race, sex, SEER region, marital status, census-tract level of education and income relative to the poverty line) and clinical (primary site, histology, AJCC stage, and comorbidities at time of diagnosis) characteristics were determined among all patients and then stratified by receipt of chemotherapy. Among chemotherapy users, we determined the proportion of patients receiving the most common regimens (Ca or Ci) combined with Pe, and Pe only, and all other chemotherapy regimens were combined to form the last group. As Ca or Ci with Pe was by far the most common first-line therapy administered, we also determined the distribution of second-line chemotherapy regimens among patients first receiving any platinum-based chemotherapy with Pe.
The primary outcome was OS. Median OS time and the 95% confidence interval (CI) of these patients were calculated for two follow-up periods with two respective starting dates. The first follow-up was from the date of diagnosis to the date of death (using death information provided in Medicare records) or censored on December 31, 2010, the end of follow-up, whichever came first. The second follow-up was from the date of the start of the specific chemotherapy treatment line (either first-line therapy or second-line therapy) to the date of death (using death information provided in Medicare records) or censored on December 31, 2010, or the end of follow-up, whichever came first. Median OS was determined from the start of first or second lines of chemotherapy, in order to provide context to survival rates reported in clinical trials for therapies targeted for either first- or second-line chemotherapy. In calculating 1-year survival, patients were censored at death, at 12 months postdiagnosis, or December 31, 2010, whichever came first. The proportion of patients surviving 1 year, as well as the associated 95% CI, was calculated, using Kaplan–Meier models, by elected treatment.
Results
Select clinical and demographic characteristics of the 1,625 malignant-mesothelioma patients included in this study are summarized in Table 1. The median age at time of diagnosis among patients was 78 years (range 66–103 years). The majority of patients were white (91.9%), male (78%), and married at the time of diagnosis (62.4%). The greatest proportion of cases lived within the greater California and New Jersey SEER registries. Some form of chemotherapy was administered to 727 (44.7%) of patients, and those receiving chemotherapy were significantly younger at time of diagnosis, white, married, and living in higher-education and -income census tracts. Approximately 45% of patients had one or more comorbidities identified in the 12 months prior to diagnosis. Expectedly, the majority of patients were diagnosed with malignant pleural mesothelioma (94.2%), with an epithelial histology (30.6%), and AJCC stage IV disease (29.1%). Patients receiving chemotherapy were more likely than those not receiving chemotherapy to have been diagnosed with AJCC stage II, III, or IV disease (71.3%), and less likely to have comorbid conditions compared to patients not receiving any chemotherapy. Mesothelioma clinical characteristics were also strongly predictive of multimodal treatment (chemotherapy and therapeutic surgery).
  | Table 1 Frequency distributions of patient/tumor/treatment characteristics in SEER–Medicare study of malignant mesothelioma diagnosed 2005–2009 Notes: aPercentages may not sum 100%, due to rounding; breferring to the census tract in which the patient resided at time of diagnosis and the proportion of residents within the tract with less than 12 years of education or household income below the poverty line; cModified Charlson Comorbidity Index, determined from Medicare claims within 12 months before diagnosis; dany patient with pleural effusion; eincluded single, separated, divorced, and widowed; *cells merged to mask small cell size; **cell size ≤10 suppressed. This study used the linked SEER-Medicare database. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Office of Research, Development and Information, CMS; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-Medicare database.24 Abbreviations: SEER, Surveillance, Epidemiology, and End Results; NOS, not otherwise specified; AJCC, American Joint Committee on Cancer; NA, not applicable. |
Sixty-six percent of patients received one line of chemotherapy, 24% two lines of therapy, and 11% more than two lines (Table 2). The most common first-line chemotherapy administered to malignant-mesothelioma patients in this study was a platinum-based agent coupled with Pe (67.4%), and among these patients, approximately 18% received maintenance treatment with Pe. The second most common first-line chemotherapy was Pe only (22%), followed by 10.5% of patients receiving treatment with another agent. Among those receiving second-line therapy, almost 30% were treated with gemcitabine, while 28.1% were retreated with a platinum-based agent with Pe. Thirty-four percent of patients receiving chemotherapy also had surgery, and this proportion was similar for patients receiving first-line or second-line therapy regimens.
  | Table 2 Frequency distributions of chemotherapy lines and regimens in SEER–Medicare study of malignant mesothelioma diagnosed 2005–2009 (n=1,625) Notes: Percentages may not sum to 100%, due to rounding. aIncludes all patients who had chemotherapy while surgery vs no surgery groups, excludes patients with surgery more than 9 months after diagnosis; bchemotherapy lines administered for 1 month only combined with subsequent or previous chemotherapy line(s) (with common agent) of multiple-month duration; cpemetrexed, when administered within 3 months of the combination cisplatin/carboplatin+pemetrexed, not considered a new line, but combined with cisplatin/carboplatin+pemetrexed; dany chemotherapy with ≥3-month gap considered a new line; eincludes third-line, fourth-line, fifth-line, and additional lines of therapy. fData is not presented for “No surgery” and “Surgery” to preserve patient anonymity. This study used the linked SEER-Medicare database. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Office of Research, Development and Information, CMS; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-Medicare database.24 Abbreviation: SEER, Surveillance, Epidemiology, and End Results. |
Median OS among patients receiving any chemotherapy was 12 months (range 1–69 months) and 4 months (range 1–69 months) among patients who did not receive chemotherapy (Table 3). Median OS was 16 months (range 4–56 months) among patients having both surgery and receiving more than one line of chemotherapy. The median OS after administration of first-line therapy was 7 months overall (range 1–68 months) and was extended an additional 5 months (range 1–33 months) postadministration of second-line therapy. Median OS among patients considered on maintenance therapy with Pe after treatment with a platinum-based agent plus Pe was 12 months postdiagnosis. Patients retreated with Pe only or Pe coupled with Ca or Ci survived an additional median 9 months posttreatment.
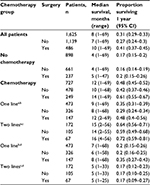  | Table 3 Overall survival statistics of patients in SEER–Medicare study of malignant mesothelioma diagnosed 2005–2009 Notes: aSurvival months calculated from date of diagnosis for each patient; bexcluded 5 patients with surgery later than 9 months after diagnosis; cexcluded 1 patient with surgery later than 9 months after diagnosis; dsurvival months calculated from date of administration of chemotherapy agent (second line for two lines). This study used the linked SEER-Medicare database. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Office of Research, Development and Information, CMS; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-Medicare database.24 Abbreviations: SEER, Surveillance, Epidemiology, and End Results; CI, confidence interval. |
One-year OS was 48% (95% CI 45%–52%) among patients who received chemotherapy and 17% (95% CI 15%–20%) among those who did not receive chemotherapy. One-year OS was 61% (95% CI 55%–67%) among patients receiving surgery and chemotherapy and 42% (37%–46%) among those receiving chemotherapy without surgery. Median OS was similar for men and women (7 months in men vs 8 months in women).
Discussion
This study provides a current view of the descriptive epidemiology of malignant mesothelioma in a population-based sample of US males and females over age 65 years, and provides a description of the patterns of treatment and OS. The most common chemotherapy used was a platinum-based agent (Ci or Ca) coupled with Pe, which is consistent with National Comprehensive Cancer Network guidelines.9 OS was greater for people who got chemotherapy than for people who were not given chemotherapy, and greatest among those who got chemotherapy and surgery. However, we are cautious in the interpretation of this finding, because the observational nature of this study makes it difficult to attribute differences in survival to regimen alone and may be explained by other patient-related factors. This is particularly apparent if comparing median OS among those patients receiving different lines of chemotherapy. Patients receiving two or more lines of chemotherapy had to have survived longer than patients receiving one line of chemotherapy in order to have received their second line of chemotherapy, thus leading to a survivor bias in these types of comparisons.
To our knowledge, this is one of the first population-based studies of treatment patterns for mesothelioma describing the impact of treatment on OS. It is also one of the largest studies, with over 1,600 patients. Previous clinical trials of mesothelioma have included up to only a few hundred patients,10 typically derived from a single clinical center or a small geographic region. Three prior studies using SEER data only evaluated the influence of surgery on OS in mesothelioma patients, two of which suggested that cancer-directed surgery improved OS for patients with epithelioid histology, but not other histologies.11,12 The third13 examined the impact of cancer-directed surgery by race and observed similar survival for both black and white patients. None of the previous studies considered chemotherapy usage in their analyses. We also established that surgery is associated with improved OS, and that chemotherapy modified the effects of surgery. However, it is difficult to attribute confidently the improvement to the surgery itself, in that healthier patients make better surgical candidates.
In our study, we found that patients who received first-line chemotherapy lived about 7 more months after the receipt of their chemotherapy. Similarly, patients who received second-line chemotherapy lived about 5 additional months. A recent systematic review by Blomberg et al14 of Phase II and Phase III randomized studies between systemic medical treatments or between systemic medical treatments and best supportive care (BSC) identified eight studies and four abstracts with results for first-line therapy. Five studies15–19 demonstrated survival lasting slightly more than 1 year after first-line therapy. The longest survival was seen by Kindler et al,16 who found that patients treated with gemcitabine/Ci plus bevacizumab survived on average for 15.6 months after therapy compared to 14.7 months for patients treated with only gemcitabine/Ci. Only one study20 of cyclophosphamide, imidazole carboxamide, and adriamycin versus cyclophosphamide and adriamycin reported survival as low as ours (6.7 months after therapy versus 5.5 months).
Two randomized trials evaluated survival in second-line therapy for mesothelioma.21,22 Buikhuisen et al21 saw that patients administered thalidomide and BSC (10.6 months) survived no longer than patients receiving only BSC (12.9 months) after second-line therapy. Similar results were seen in a trial that looked at survival among patients receiving Pe and BSC (median survival 8.4 months) versus patients receiving BSC (median survival 9.7 months).22 Recently, results from the Mesothelioma Avastin Plus Pemetrexed-cisplatin Study (MAPS) study, a randomized trial of treatment with bevacizumab in addition to Pe and Ci for patients who were not good surgical candidates, found that the addition of bevacizumab resulted in a significant improvement in survival, despite an increase cardiovascular-related side effects or events.23 It is not surprising that these trials demonstrated better survival than our real-world study. We were limited to studying only patients enrolled in Medicare whose average age was higher than the clinical trial patients. In addition, the clinical trials only included patients with a good performance status, whereas we included all available patients in our study.
The data were derived from the SEER–Medicare linkage. SEER has a highly reliable system for identifying incident malignant tumors, and all diagnoses are histologically verified. The 18 SEER registries included in this analysis represent a wide range of regional registries, and are not limited to one location or to the patient population served by any single medical center. The patterns of mesothelioma incidence are believed to be representative of the patterns in the US population. In addition, this study derives from recent clinical data (diagnosis in 2005–2009, with follow-up through 2010). This is probably reflective of current treatment patterns, because no new therapies for mesothelioma have been introduced since 2010. In fact, our results closely follow current treatment guidelines.9 The linkage between SEER and Medicare data allowed us to ascertain comorbidity in the 12 months prior to diagnosis of mesothelioma, which is an advantage over other published studies. Comorbidity is not only a predictor of which treatment is given to patients diagnosed with mesothelioma, but it is also a predictor of survival as well. No other study of mesothelioma survival that we are aware of has investigated prediagnostic comorbidity. Unfortunately, one of the limitations of the SEER–Medicare database is that chemotherapy dose is not completely captured from claims, so determining dose escalation for any of the chemotherapy regimens would be difficult. Likewise, retreatment with the same agent as opposed to maintenance treatment can only be established by looking at patterns of therapy, particularly examining duration of time between administrations of chemotherapy agents and comparing these patterns to current evidence-based guidelines on the standard of care of patients diagnosed with mesothelioma. In conclusion, mesothelioma patients receiving some form of chemotherapy survived longer than patients who did not, regardless of surgery, with an additional survival benefit among those patients receiving multimodal treatment.
Author contributions
JBD is the guarantor of the content of the manuscript, including the data and analysis. JPF, CY, DHG, TD, AS, and SG contributed substantially to the study design, data analysis and interpretation, and the writing of the manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Acknowledgment
This study was funded by AstraZeneca. Details of this study were presented at the 2015 ASCO Annual Meeting, and appeared in abstract form in the Journal of Clinical Oncology (volume 33, 2015).
Disclosure
JPF was employed by AstraZeneca. JPF, CY, AS, DHG, and JBD received funding to support this work from AstraZeneca Inc, Gaithersburg, MD 20878. TD is currently employed by AstraZeneca. The authors report no other conflicts of interest in this work.
References
Ray M, Kindler HL. Malignant pleural mesothelioma: an update on biomarkers and treatment. Chest. 2009;136(3):888–896. | ||
Teta MJ, Mink PJ, Lau E, Sceurman BK, Foster ED. US mesothelioma patterns 1973-2002: indicators of change and insights into background rates. Eur J Cancer Prev. 2008;17(6):525–534. | ||
National Cancer Institute Surveillance, Epidemiology, and End Results. SEER*Stat software. Available from: www.seer.cancer.gov/seerstat. Accessed June 16, 2016. | ||
Moolgavkar SH, Meza R, Turim J. Pleural and peritoneal mesotheliomas in SEER: age effects and temporal trends, 1973–2005. Cancer Causes Control. 2009;20(6):935–944. | ||
Rusch VW. A proposed new international TNM staging system for malignant pleural mesothelioma from the International Mesothelioma Interest Group. Lung Cancer. 1996;14(1):1–12. | ||
Van Schil P. Malignant pleural mesothelioma: staging systems. Lung Cancer. 2005;49 Suppl 1:S45–S48. | ||
Curran D, Sahmoud T, Therasse P, van Meerbeeck J, Postmus PE, Giaccone G. Prognostic factors in patients with pleural mesothelioma: the European Organization for Research and Treatment of Cancer experience. J Clin Oncol. 1998;16(1):145–152. | ||
Potosky AL, Riley GF, Lubitz JD, Mentnech RM, Kessler LG. Potential for cancer related health services research using a linked Medicare-tumor registry database. Med Care. 1993;31(8):732–748. | ||
National Comprehensive Cancer Network. NCCN guidelines for treatment of cancer by site: malignant pleural mesothelioma. 2015. Available from: https://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed June 16, 2016. | ||
Jänne PA. First-line chemotherapy for malignant pleural mesothelioma. In: Pass HI, Vogelzang NJ, Carbone M, editors. Malignant Mesothelioma: Advances in Pathogenesis, Diagnosis, and Translational Therapies. New York: Springer; 2005:593–598. | ||
Milano MT, Zhang H. Malignant pleural mesothelioma: a population-based study of survival. J Thorac Oncol. 2010;5(11):1841–1848. | ||
Meyerhoff RR, Yang CF, Speicher PJ, et al. Impact of mesothelioma histologic subtype on outcomes in the Surveillance, Epidemiology, and End Results database. J Surg Res. 2015;196(1):23–32. | ||
Taioli E, Wolf AS, Moline JM, Camacho-Rivera M, Flores RM. Frequency of surgery in black patients with malignant pleural mesothelioma. Dis Markers. 2015;2015:282145. | ||
Blomberg C, Nilsson J, Holgersson G, et al. Randomized trials of systemic medically-treated malignant mesothelioma: a systematic review. Anticancer Res. 2015;35(5):2493–2501. | ||
Krug LM, Wozniak AJ, Kindler HL, et al. Randomized phase II trial of pemetrexed/cisplatin with or without CBP501 in patients with advanced malignant pleural mesothelioma. Lung Cancer. 2014;85(3):429–434. | ||
Kindler HL, Karrison TG, Gandara DR, et al. Multicenter, double-blind, placebo-controlled, randomized phase II trial of gemcitabine/cisplatin plus bevacizumab or placebo in patients with malignant mesothelioma. J Clin Oncol. 2012;30(20):2509–2515. | ||
Vogelzang NJ, Rusthoven JJ, Symanowski J, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol. 2003;21(14):2636–2644. | ||
Millenson MM, Lee J, Hanna NH, et al. Pemetrexed (Pem) plus gemcitabine (Gem) or carboplatin (Carbo) in patients (pts) with advanced malignant mesothelioma (MPM): a randomized phase II trial, E1B03. J Clin Oncol. 2010;28(15 Suppl):e18053. | ||
Szlosarek PW, Steele JP, Nolan L, et al. Randomized trial of arginine deprivation with pegylated arginine deiminase in patients with malignant pleural mesothelioma. J Clin Oncol. 2014;32(5 Suppl):7507. | ||
Samson MK, Wasser LP, Borden EC, et al. Randomized comparison of cyclophosphamide, imidazole carboxamide, and adriamycin versus cyclophosphamide and adriamycin in patients with advanced stage malignant mesothelioma: a Sarcoma Intergroup study. J Clin Oncol. 1987;5(1):86–91. | ||
Buikhuisen WA, Burgers JA, Vincent AD, et al. Thalidomide versus active supportive care for maintenance in patients with malignant mesothelioma after first-line chemotherapy (NVALT 5): an open-label, multicentre, randomised phase 3 study. Lancet Oncol. 2013;14(6):543–551. | ||
Jassem J, Ramlau R, Santoro A, et al. Phase III trial of pemetrexed plus best supportive care compared with best supportive care in previously treated patients with advanced malignant pleural mesothelioma. J Clin Oncol. 2008;26(10):1698–1704. | ||
Zalcman G, Mazieres J, Margery J, et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed (MAPS): a randomized, controlled, open-label, phase 3 trial. Lancet. 2016;387(10026):1405–1414. | ||
Healthcaredelivery.cancer.gov/ [database on the Internet]. Rockville, MD: National National Cancer Institute, Division of Cancer Control and Population Sciences. Available from: http://healthcaredelivery.cancer.gov/seermedicare/obtain/use.html. Accessed June 16, 2016. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
