Back to Journals » Journal of Hepatocellular Carcinoma » Volume 11
Machine Learning-Based Nomogram for Predicting Overall Survival in Elderly Patients with Cirrhotic Hepatocellular Carcinoma Undergoing Ablation Therapy
Authors Qiao W ![]() , Sheng S, Li J
, Sheng S, Li J ![]() , Jin R, Hu C
, Jin R, Hu C
Received 3 December 2023
Accepted for publication 27 February 2024
Published 7 March 2024 Volume 2024:11 Pages 509—523
DOI https://doi.org/10.2147/JHC.S450825
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr David Gerber
Wenying Qiao,1– 5,* Shugui Sheng,1– 3,* Junnan Li,1– 3,* Ronghua Jin,1– 4 Caixia Hu5
1Beijing Key Laboratory of Emerging Infectious Diseases, Beijing Ditan Hospital, Capital Medical University, Beijing, People’s Republic of China; 2Beijing Institute of Infectious Diseases, Beijing Ditan Hospital, Capital Medical University, Beijing, People’s Republic of China; 3National Center for Infectious Diseases, Beijing Ditan Hospital, Capital Medical University, Beijing, People’s Republic of China; 4Changping Laboratory, Beijing, People’s Republic of China; 5Interventional Therapy Center for Oncology, Beijing You’an Hospital, Capital Medical University, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Ronghua Jin, Beijing Key Laboratory of Emerging Infectious Diseases, BeijingDitan Hospital, Capital Medical University, Beijing, People’s Republic of China, Email [email protected] Caixia Hu, Interventional Therapy Center for Oncology, Beijing You’an Hospital, Capital Medical University, Beijing, People’s Republic of China, Email [email protected]
Purpose: The aim of the study is to identify and evaluate multifaceted factors impacting the survival of elderly cirrhotic HCC patients following ablation therapy, with the goal of constructing a nomogram to predict their 3-, 5-, and 8-year overall survival (OS).
Patients and Methods: A retrospective analysis was conducted on 736 elderly cirrhotic HCC patients who underwent ablation therapy between 2014 and 2022. LASSO regression, random survival forest (RSF), and multivariate Cox analyses were employed to identify independent prognostic factors for OS, followed by the development and validation of a predictive nomogram. Harrell’s concordance index (C-index), calibration plot and decision curve analysis (DCA) were used to assess the performance of the nomogram. The nomogram was finally utilized to stratify patients into low-, intermediate-, and high-risk groups, aiming to assess its efficacy in precisely discerning individuals with diverse overall survival outcomes.
Results: Alcohol drinking, tumor number, globulin (Glob) and prealbumin (Palb) were identified and integrated to establish a novel prognostic nomogram. The nomogram exhibited strong discriminative ability with C-indices of 0.723 (training cohort) and 0.693 (validation cohort), along with significant Area Under the Curve (AUC) values for 3-year, 5-year, and 8-year OS in both cohorts (0.758, 0.770, and 0.811 for training cohort; 0.744, 0.699 and 0.737 for validation cohort). Calibration plots substantiated its consistency, while DCA curves corroborated its clinical utility. The nomogram further demonstrated exceptional effectiveness in discerning distinct risk populations, highlighting its robust applicability for prognostic stratification.
Conclusion: Our study successfully developed and validated a robust nomogram model based on four key clinical parameters for predicting 3-, 5- and 8-year OS among elderly cirrhotic HCC patients following ablation therapy. The nomogram exhibited a remarkable capability in identifying high-risk patients, furnishing clinicians with invaluable insights for postoperative surveillance and tailored therapeutic interventions.
Keywords: hepatocellular carcinoma, HCC, nomogram, LASSO regression, random survival forest, RSF, overall survival, OS
Introduction
Liver cancer poses a significant challenge to global health, occupying the sixth position in terms of its incidence and ranking third in mortality rates.1 Hepatocellular carcinoma (HCC) is the predominant type of liver cancer, comprising 75–85% of all cases.2 With the advent of an aging society, the increasing prevalence of HCC with cirrhosis among elderly individuals has garnered extensive attention.3,4 Surgical resection is indeed the most common method used for the treatment of HCC, particularly in cases where the tumor is localized and the patient’s liver function is adequate. Treatment options for HCC also include transarterial chemoembolization (TACE), radiation therapy, chemotherapy, targeted therapy, and immunotherapy. Over recent years, there has been a gradual rise in the proportion of HCC patients opting for ablation therapy.5–7 Elderly patients with cirrhotic HCC often face challenges in tolerating more invasive treatments like surgery or liver transplantation due to age-related ailments and reduced physiological reserves. Ablation therapy offers a less invasive alternative for this population. It reduces surgical burden, preserves liver function, lowers complication risks, shortens recovery, and provides repeatable options.8,9 Hence, ablation therapy is better suited for elderly patients with cirrhotic HCC, considering its minimally invasive nature and potential benefits in managing their condition. Despite this trend, there is a notable gap in current research, as there is a lack of studies specifically predicting the overall survival (OS) for the particular group (elderly patients with cirrhotic HCC) following ablation treatment. This study is precisely founded on the recognition and understanding of the gap in this academic field, with the goal of addressing this significant research vacuum. It strives to provide an accurate model for predicting the overall survival of elderly cirrhotic HCC patients after undergoing ablation therapy.
Meanwhile, our attention is not solely directed toward bridging the gaps in this academic field; we are equally committed to pioneering innovative strides in research methodologies. Nomograms, as graphical representations of predictive models, have emerged as valuable tools in clinical decision-making by providing individualized risk assessment for patients. By incorporating multiple variables into a single predictive score, nomograms offer clinicians a user-friendly way to estimate patient outcomes. In the field of liver disease research, especially in the establishment of nomograms related to HCC prognosis, traditional statistical methods are still predominantly used.10–14 Although these methods can achieve some success in certain situations, their limitations in handling high-dimensional and non-linear data are gradually becoming apparent with the continuous increase in data volume and a deeper understanding of disease mechanisms. This study introduced the Least absolute shrinkage and selection operator (LASSO) regression and random survival forest (RSF) from machine learning and combined them with traditional statistical methods (multivariate Cox regression), forming a comprehensive research framework. The utilization of advanced data analysis techniques, such as machine learning, offers the advantage of handling high-dimensional data and discerning intricate relationships between variables, enabling a more comprehensive exploration of the factors influencing OS in elderly patients with cirrhotic HCC. By integrating a diverse range of demographic, clinical, and pathological features, machine learning - based nomograms can provide more precise predictions tailored to individual patient characteristics.
In the present study, our primary objective was to evaluate the OS of elderly cirrhotic HCC patients after receiving ablation therapy. This evaluation was conducted through the development of predictive models and the construction of the nomogram, which holistically incorporated a spectrum of demographic and clinical variables. Furthermore, our study extended to the comparison of survival times among different risk groups derived from the established nomogram, aiming to facilitate the identification of high-risk populations and implement timely and effective interventions.
Materials and Methods
This retrospective study was carried out at Beijing You’an Hospital, affiliated with Capital Medical University, with approval obtained from the ethics committee and in compliance with the ethical standards outlined in the Declaration of Helsinki. The committee granted a waiver for the necessity of informed consent, considering the retrospective nature of the study and the use of anonymized patient data.
Patients
Initially, we procured data from 982 elderly cirrhotic HCC patients who received ablation therapy at Beijing You’an Hospital, affiliated with Capital Medical University, between January 2014 and December 2022. Subsequently, 246 patients were excluded from the study due to the lack of cirrhosis (n= 43), the presence of distant metastases (n=51) and concurrent malignancies (n=45), and incomplete clinical or follow-up information (n=107). Therefore, a total of 736 patients were finally enrolled in this study in accordance with the stipulated inclusion criteria. The patient enrollment process is shown in Figure S1. The inclusion criteria were: (1) age≥60 years old; (2) HCC with cirrhosis; (3) no pre-operative surgical resection, immunotherapy, chemotherapy and so on. The exclusion criteria were: (1) lack of liver cirrhosis; (2) with distant metastases; (3) with concurrent malignancies; (4) missing clinical or follow-up data. Then, the 736 patients were randomly allocated into a training cohort (n=515) and a validation cohort (n=221) at a 7:3 ratio.
Data Collection and Follow-Up
The demographic and clinical parameters of the patients were retrieved from their medical records prior to ablation treatment. The gathered information spanned diverse categories, ensuring a thorough assessment of the individuals. (1) Demographic information: age and gender; (2) medical history: smoking, alcohol drinking, antiviral treatment, hypertension, diabetes, family history, alcoholic liver disease (ALD); (3) disease features: tumor number, tumor size, Child-Pugh class and Barcelona Clinic Liver Cancer (BCLC) stage; (4) liver function tests: alanine aminotransferase (ALT), aspartate aminotransferase (AST), ALT/AST, alkaline phosphatase (ALP), total bilirubin (TBIL), direct bilirubin (DBIL), bile acid, gamma glutamyl transpeptidase (GGT), thrombin time (TT), prothrombin time (PT), prothrombin time activity (PTA), activated partial thromboplastin time (APTT) and international normalized ratio (INR); (5) other laboratory results: white blood cells (WBCs), neutrophils (Neus), lymphocytes (Lyms), monocytes (Mons), red blood cells (RBCs), hemoglobin (Hb), platelets (PLTs), neutrophils to lymphocytes ratio (NLR), monocytes to lymphocytes ratio (MLR), platelets to lymphocytes ratio (PLR), albumin (Alb), globulin (Glob), prealbumin (Palb), uric acid, BUN, creatinine, fibrinogen (Fib) and alpha-fetoprotein (AFP).
Our hospital recommends that all patients undergo regular follow-up examinations after ablation therapy. Initial follow-ups were generally conducted around one month after treatment to assess the treatment’s effectiveness. Subsequent evaluations were recommended at intervals of three months during the first year followed by semiannual evaluations thereafter until death or loss of follow-up. During each follow-up, the physician could conduct imaging assessments, such as CT or MRI, to thoroughly examine the healing status of the treatment area and whether there were any new lesions. Liver function indicators such as ALT, AST, and ALP would be monitored to ensure stable liver function. The levels of tumor markers in the blood, especially AFP, might be tested to assess tumor activity. By regularly monitoring symptoms and signs, any potential complications could be promptly identified and addressed. Overall survival (OS), as the main research topic of this study, was defined as the interval from the initiation of ablation to either the occurrence of death or the last follow-up. The final follow-up date for this study was July 1, 2023, with a median follow-up duration of 44.0 months.
Ablation Procedure
Radiofrequency ablation (RFA) procedure was performed by a team of interventionists with a minimum of 5 years of experience. A comprehensive assessment of the patient’s overall condition, liver function indicators, as well as factors such as the size, number, and location of liver cancer, and its relationship with adjacent organs, was conducted to select a rational ablation treatment plan. Before the treatment, patients fasted for 8 hours and received local anesthesia at the puncture site. Under ultrasound guidance, the team determined the entry point, angle, and path of the electrode needle. Once the needle insertion accuracy was confirmed, they activated the RFA system, initiating the treatment. If the tumor was relatively large, multiple ablations were performed. In the case of multiple tumors, each was ablated individually. The ablation range included the tumor in the liver and surrounding 0.5 to 1.0 cm of peritumoral tissue. Upon completion, the needle was withdrawn while simultaneously applying electrocoagulation to prevent bleeding along the needle tract and the seeding or metastasis of tumor cells. Following the ablation, dynamic contrast-enhanced Computed Tomography (CT), Magnetic Resonance Imaging (MRI), or ultrasound imaging was performed. If no enhancement was observed in the arterial phase imaging of the ablated area, it indicated complete tumor ablation. However, if there was still localized arterial phase enhancement within the lesion, suggesting incomplete tumor ablation, a repeat ablation was necessary to achieve the therapeutic goal of complete ablation.
Statistical Analysis
Continuous variables were presented in the form of mean ± standard deviation, while categorical variables were elucidated using frequency and percentage. The Student’s t-test was employed to discern differences between groups in continuous variables. Concurrently, the Chi-square test was applied to assess differences in categorical variables. The training cohort was exclusively used for constructing the nomogram, whereas the validation cohort was specifically employed to verify its performance. All statistical analyses in this study were executed through R version 4.3.2. In this study, we systematically applied advanced statistical techniques, including LASSO regression, RSF, and multivariate Cox regression, to identify independent prognostic variables. After the multivariate Cox regression analysis, factors exhibiting p-values below the threshold of 0.05 were incorporated into the construction of a prognostic nomogram for 3-year, 5-year, and 8-year OS. We employed a variety of complementary methods to assess different aspects of model performance, including discriminative ability, model calibration, and clinical utility. For evaluating discriminative ability, we utilized Harrell’s concordance index (C-index) and the area under the receiver operating characteristic curve (AUC). Model calibration was measured through calibration plots. As for clinical utility, it was assessed through decision curve analysis (DCA). Furthermore, individuals were stratified into three risk groups—low, intermediate, and high—according to the scores derived from the nomogram. Subsequently, Kaplan-Meier curves were employed to predict the survival for each group. By delving into the risk stratification and survival rates of patients, it is possible to better identify high-risk individuals, laying the groundwork for personalized medicine and more effective health management. All statistical analyses were conducted using two-tailed tests, and statistical significance was defined as a P value less than 0.05.
Results
Baseline Characteristics of the Patients
In this study, we included 736 elderly (age≥60) patients who underwent ablation therapy for HCC combined with liver cirrhosis at Beijing You’an Hospital, affiliated with Capital Medical University from January 2014 to December 2022. The 736 patients were randomly divided into two distinct groups for model development and validation, with 515 subjects constituting the training cohort and 221 subjects comprising the validation cohort. The comparative analysis of the baseline characteristic data from the training and validation cohorts revealed no significant differences between the two groups (P>0.05), affirming their comparability and suitability for ensuing studies (Table 1).
 |
Table 1 Comparison of Baseline Characteristics Between the Training and Validation Cohorts |
Within both the training and validation cohorts, about 77% were male and approximately 23% were female. Notably, 44% of cases in the cohorts had a documented history of smoking, while 34% had a history of alcohol drinking. In the training cohort, 368 (71.5%) patients were Child-Pugh class A and 147 (28.5%) patients were Child-Pugh class B; in the validation cohort, 154 (69.7%) patients were Child-Pugh class A and 67 (30.3%) patients were Child-Pugh class B. Regarding BCLC stage, in both cohorts, about 30% of patients were BCLC stage 0 and 70% were BCLC stage A. Tumor characteristics within the cohorts revealed that, in the training cohort, 359 (69.7%) of cases manifested as solitary tumors, contrasting with 156 (30.3%) cases that presented with multiple tumors. In the validation cohort, 150 (67.9%) patients displayed solitary tumors, while 71 (32.1%) exhibited multiple tumors.
Determination of independent Risk Factors for OS
For the identification of independent risk factors affecting the OS of elderly cirrhotic HCC patients after undergoing ablative therapy, we employed a combination of machine learning techniques (LASSO regression and RSF) and traditional statistical methods (multivariate Cox regression). First of all, we made the LASSO regression analysis for OS (Figure 1). LASSO regression is a regularization technique in linear regression that, by introducing an L1 regularization term into the loss function, automatically shrinks the coefficients of features contributing less to the target variable towards zero, thereby achieving feature selection in the model.15 Lambda (λ) serves as the regularization parameter, governing the strength of regularization and influencing the sparsity of the model. A larger λ results in a stronger regularization effect, potentially leading to a simpler and sparser model. Conversely, a smaller λ allows for a more flexible model but may increase the risk of overfitting. This study employed 10-fold cross-validation to select the optimal λ value, striking a balance between the model’s fitting performance and feature selection. The optimal value of λ was determined to be 0.0230244 (log λ=−3.771), concomitantly pinpointing 15 variables as independent risk factors influencing OS, including gender, antiviral treatment, alcohol drinking, BCLC, tumor number, tumor size, MLR, WBCs, Alb, Glob, GGT, Palb, bile acid, uric acid and PTA.
 |
Figure 1 Screening independent risk factors for OS using LASSO regression analysis. Abbreviations: OS, overall survival; LASSO, least absolute shrinkage and selection operator. |
Then, we employed an RSF model to screen factors influencing the OS of elderly cirrhotic HCC patients after undergoing ablative therapy (Figure 2). The RSF model is a machine learning model based on the random forest algorithm, designed for handling right-censored survival data.16 This model utilizes multiple decision trees to predict survival time, and the final prediction is obtained by averaging the predictions of each individual tree. In the RSF model, each decision tree is constructed based on randomly selected features and samples, which helps mitigate the risk of overfitting. By employing the RSF model, researchers can identify factors that significantly impact OS. In this study, it was found that alcohol drinking, bile acid, Palb, Alb, Hb, Glob, tumor number, RBC, GGT, gender, PTA, INR, AST, Child-Pugh class, chlorine, ALD, uric acid, antiviral treatment, total protein, WBCs, diabetes, NLR, creatinine, kalium and MLR were independent risk factors for OS through the RSF model.
Subsequently, we identified the common variables from the results of LASSO regression and RSF model analyses, including alcohol drinking, gender, antiviral treatment, tumor number, MLR, WBCs, Alb, Glob, GGT, Palb, uric acid, bile acid and PTA. These common variables were further incorporated into a multivariable Cox regression analysis, revealing that four factors, including alcohol drinking (HR: 1.556; 95% CI: 1.054–2.298; P=0.026), tumor number (HR: 1.51; 95% CI: 1.06–2.151; P=0.022), Glob (HR: 1.544; 95% CI: 1.012–2.077; P=0.006) and Palb (HR: 0.892; 95% CI: 0.788–0.998; P=0.003), remain significant independent risk factors for OS, as shown in Figure 3. Specifically, the HR value of alcohol drinking, tumor number and Glob were all greater than 1, suggesting an elevated risk. Conversely, the HR value for Palb was below 1, signifying a potentially protective effect.
Development of the OS-Predicting Nomogram
Utilizing the four specified variables identified above, we developed a nomogram to predict the 3-year, 5-year, and 8-year OS of elderly cirrhotic HCC patients following ablation treatment (Figure 4). Within the nomogram, each value of the predictive variable was meticulously assigned a corresponding score. By aggregating these scores across the four predictive variables, a total score could be computed for each patient, which corresponded to the 3-year, 5-year, and 8-year survival probabilities of the patient. We proceeded to the evaluation of the nomogram’s performance after the nomogram had been developed. This evaluation involved the discriminative ability, calibration, and clinical utility of the nomogram model.
 |
Figure 4 Nomogram developed for predicting 3-, 5-, and 8-year OS of elderly cirrhotic HCC patients undergoing ablation therapy. Abbreviations: OS, overall survival; Glob, globulin; Palb, prealbumin. |
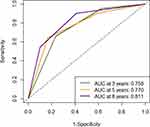
Harrell’s C-index serves as a robust statistical metric for evaluating the discriminative efficacy of a model, specifically its capacity to accurately differentiate between patients exhibiting distinct clinical outcomes and those devoid of such outcomes. The C-index ranges from 0.5 to 1.0, where 0.5 indicates a model with no discriminative ability (similar to random chance) and 1.0 indicates perfect discrimination. The C-index of the nomogram model in the training cohort was 0.723 (95% CI: 0.684–0.762), demonstrating a proficient capacity for distinguishing between survival and non-survival individuals. Additionally, AUC is another widely adopted metric for evaluating the discriminative ability of classification models. Its range of values is comparable to that of the C-index. Our nomogram model yielded AUC values of 0.758, 0.770, and 0.811 for 3-year, 5-year, and 8-year OS, respectively, further confirming its excellent discriminative ability (Figure 5). Moreover, the consistent increase in AUC values with longer follow-up periods suggested the nomogram’s ability to capture the evolving dynamics of survival probabilities over time. This may have important implications for clinical decision-making and patient counseling, providing more nuanced insights into long-term prognosis.
For the evaluation of nomogram model calibration, we drew the calibration plots, in order to explore the degree of consistency between the model’s predictions and actual outcomes. In the calibration plot, the abscissa denotes the patient survival probabilities predicted by the model, while the ordinate represents the actual observed patient survival probabilities. The calibration plot evaluates the consistency of the model by comparing the proximity of the solid line to the dashed line. The greater the proximity between the solid line and the dashed line, the more accurately the model’s predictions correspond to the observed outcomes. In our calibration plot, it can be observed that there was a close match between the solid line and the dashed line, indicating a strong consistency between the observed survival probability of patients and the predicted probability of the 3-, 5-, and 8-year OS by the model (Figure 6). This suggested that the model can accurately capture the survival status of patients, providing a reliable foundation for future predictions and decision-making.
 |
Figure 6 Calibration plots of the nomogram for 3-year (A), 5-year (B), and 8-year (C) OS prediction in the training cohort. Abbreviation: OS, overall survival. |
In examining the clinical utility of the model, DCA curve analysis provides a comprehensive perspective, which displays the net benefit of a predictive model across a range of probability thresholds for a particular outcome. In essence, DCA helps clinicians assess the clinical usefulness of a model by weighing the benefits of true positives against the harms of false positives, taking into account the clinical context and the trade-off between sensitivity and specificity. In the training cohort, our nomogram model for 3-, 5-, and 8-year OS prediction demonstrated a commendable net benefit across a range of threshold probabilities (Figure 7). This favorable outcome underscored its robustness and potential effectiveness in real-world clinical scenarios.
 |
Figure 7 DCA curve analysis of the nomogram for 3-year (A), 5-year (B), and 8-year (C) OS prediction in the training cohort. Abbreviations: DCA, decision curve analysis; OS, overall survival. |
In summary, the comprehensive application of these evaluation methods allows for a thorough understanding of the model’s performance, providing insights into its feasibility and effectiveness in practical medical applications.
Validation of the Nomogram
To enhance the credibility of our nomogram, we performed validation using the separate validation cohort. The internal validation results of the nomogram underscored its commendable accuracy and dependability in forecasting the OS of elderly cirrhotic HCC patients post-ablation therapy. The C-index in the validation cohort was 0.693 (95% CI: 0.634–0.752), and the AUC values for 3, 5, and 8-year OS prediction were 0.744, 0.699 and 0.737, respectively, reiterated the nomogram’s favorable discriminative ability (Figure S2). The calibration curves for 3-, 5-, and 8-year OS prediction further verified the nomogram’s consistency, showcasing a noteworthy alignment between predictions and actual observations (Figure S3). The DCA curve analysis accentuated the outstanding clinical usefulness of the nomogram, highlighting its ability to guide clinical decisions with a well balance of benefits and risks (Figure S4). Overall, these findings collectively validated the nomogram’s performance and highlighted its potential as a valuable tool for predicting survival outcomes in clinical settings.
Risk Stratification for survival Based on the Nomogram-Derived Scores
Patients were stratified into distinct risk groups (low-, intermediate-, and high-risk) according to the scores derived from the nomogram, with the objective of assessing the nomogram’s efficacy in precisely discerning individuals with varying prognostic outcomes. Subsequently, the Kaplan-Meier method was employed to construct survival curves in the training cohort, assessing differences in survival outcomes among three distinct risk groups. The findings revealed a statistically significant difference in OS among the low-risk, intermediate-risk, and high-risk groups (P<0.05) (Figure 8). Specifically, the high-risk group, characterized by higher nomogram-derived scores, exhibited a notably lower OS compared to the intermediate- and low-risk groups. This substantial differentiation in survival outcomes highlighted the nomogram’s capability to discern patients with a heightened risk of unfavorable prognosis. To affirm the strength of our findings, we conducted a validation through the validation cohort, and also observed a statistically significant discrepancy in OS among the three risk groups (P<0.05) (Figure S5). The consistency in results across the two cohorts further reinforced the applicability and reliability of our nomogram, confirming its ability to effectively identify high-risk populations and enable timely intervention for these patients.
 |
Figure 8 Kaplan-Meier survival curves for OS based on the nomogram-derived low, intermediate, and high-risk groups in the training cohort. Abbreviation: OS, overall survival. |
Discussion
In this study, we finally integrated four key parameters (alcohol drinking, tumor number, Glob and Palb) to construct a prognostic nomogram for predicting 3-, 5- and 8-year OS in elderly cirrhotic HCC patients after ablation therapy. These parameters can be readily acquired preoperatively through clinical examination and laboratory tests. Our study’s large-scale cohort, long-term follow-up, and integration of advanced analytical techniques provided a solid foundation for enhancing prognostic insights in elderly cirrhotic HCC patients.
Although previous studies have developed several nomograms for HCC, their applicability to elderly cirrhotic HCC patients undergoing ablation therapy is uncertain. For example, He et al constructed and validated a nomogram model for predicting early relapse and survival following hepatectomy for HCC.17 Liu et al investigated the prognostic value of CT texture parameters in post-hepatectomy HCC patients and developed a radiomics nomogram by integrating clinicopathological factors with the radiomics signature.18 Kuo et al developed a prognostic nomogram capable of assessing the long-term survival of individuals with very early or early-stage HCC following RFA.19 Chen et al constructed a nomogram utilizing a novel inflammation-based score to evaluate the prognosis of HCC patients within the Milan Criteria after ablation.20 These nomograms may have been derived from cohorts comprising predominantly younger patients or those with different treatment modalities, potentially limiting their generalizability to elderly cirrhotic patients after receiving ablation therapy. Therefore, the nomogram tailored to this specific population in this study are essential for providing accurate prognostic information and guiding treatment decisions.
The inclusion of alcohol drinking history as a pivotal factor in our nomogram acknowledges the significant impact of alcohol consumption on the OS of elderly cirrhotic HCC patients. Numerous prior research findings indicated that alcohol consumption could elevate the risk of HCC occurrence, reduce patient survival, and potentially result in an unfavorable prognosis.21–24 Tumor number is another factor identified in this study that affects the OS of elderly cirrhotic HCC patients. Ramacciato et al found that for multifocal HCC, the insufficient reserve function of the remaining liver greatly increased the difficulty of surgical resection.25 The different lesions of multifocal HCC may originate from different tumor centers or intrahepatic metastasis of the same primary lesion.26 By examining the genetic origins and clonal evolution, substantial heterogeneity was observed among separate lesions.27 At the genetic level, HCC exhibits differences in gene mutations and copy number variations among multiple lesions.28 These genetic variations may impact tumor growth, invasive capabilities, and responsiveness to treatment, thereby influencing prognosis. Furthermore, there are significant differences in the immune microenvironment among separate lesions. Studies have revealed that distinct lesions may show varying immune micro-environmental features, including immune effector cells, immune inhibitory cells, cytokines and so on.29,30
The intricate relationship between high preoperative globulin levels and the poor prognosis of HCC patients can be understood by considering factors such as chronic inflammation and immune system dynamics. Chronic inflammation, frequently initiated by hepatitis, establishes an environment conducive to the development and advancement of HCC. In this context, elevated globulin levels denote sustained immune system activation and ongoing inflammatory processes, potentially playing a role in the unfavorable prognosis linked with HCC.31–33 Moreover, the immune response linked with globulin, plays a paradoxical role—while an activated immune system is essential for combating cancer, it can also be compromised, rendering it ineffective in eradicating cancer cells and thereby facilitating disease progression.34–36 Prealbumin, a carrier protein produced by the liver, whose concentration changes are determined by the function of liver cells, and its low levels may reflect impaired liver function.37,38 Low prealbumin levels are also often associated with malnutrition,39 which is closely related to the poor prognosis of HCC patients. Furthermore, prealbumin also exhibits thymus activity, enhancing the body’s immune response by promoting the maturation of lymphocytes, and it has potential implications in anti-tumor activities.40,41 Hence, the decrease in prealbumin levels may suggest a weakening of the immune system, consequently diminishing the overall survival of the patients.
There is a fact that must be acknowledged, although many studies have developed nomograms for HCC, their translation in clinical practice is still limited. However, the nomogram developed in our study represents a significant departure from previous studies. Its foundation on machine learning techniques ensures a higher degree of accuracy and adaptability, particularly in the context of elderly patients with cirrhotic HCC undergoing ablation therapy. By harnessing the power of advanced algorithms, our nomogram not only enhances predictive capability but also facilitates personalized treatment strategies. Its incorporation of a diverse array of clinical and demographic variables allows for a more comprehensive assessment of individual patient profiles, thereby enabling clinicians to make informed decisions regarding treatment selection and prognosis. Moreover, the easy-obtained variables of our nomogram enhance its feasibility for routine clinical use, potentially bridging the gap between research and practice in the management of elderly patients with cirrhotic HCC. As such, we anticipate that our nomogram holds considerable promise for improving patient outcomes and guiding therapeutic interventions in this patient population.
There are some limitations to our study. Initially, this study was carried out exclusively at a singular medical center, and while internal validation was undertaken, the absence of external validation remains a notable limitation. Moreover, the retrospective design of the study introduces inherent selection bias, given its reliance on historical records and patient information. Thirdly, given the study’s execution in China, where the hepatitis B virus (HBV) predominates, prudence is warranted when extending our findings to populations where HBV is not the predominant cause of HCC. As a result, the imperative for extensive, multi-center studies becomes increasingly apparent to establish a solid foundation for the broader implementation of our findings in clinical practice.
Conclusion
Our study successfully developed and validated a robust nomogram model based on four key clinical parameters for predicting 3-, 5- and 8-year OS among elderly cirrhotic HCC patients following ablation therapy. The nomogram exhibited a remarkable capability in identifying high-risk patients, furnishing clinicians with invaluable insights for postoperative surveillance and tailored therapeutic interventions.
Funding
This work was funded by Beijing Bethune Charitable Foundation (QZHX-21-ZQN-014).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi:10.3322/caac.21660
2. Loy LM, Low HM, Choi JY, Rhee H, Wong CF, Tan CH. Variant hepatocellular carcinoma subtypes according to the 2019 WHO classification: an imaging-focused review. AJR Am J Roentgenol. 2022;219(2):212–223. doi:10.2214/ajr.21.26982
3. Arora SP, Liposits G, Caird S, et al. Hepatocellular carcinoma in older adults: a comprehensive review by young international society of geriatric oncology. J Geriatr Oncol. 2020;11(4):557–565. doi:10.1016/j.jgo.2019.10.007
4. Abu-Freha N, Estis-Deaton A, Aasla M, et al. Liver cirrhosis in elderly patients: clinical characteristics, complications, and survival-analyses from a large retrospective study. Aging Clin Exp Res. 2022;34(9):2217–2223. doi:10.1007/s40520-022-02152-6
5. Minami Y, Aoki T, Hagiwara S, Kudo M. Tips for preparing and practicing thermal ablation therapy of hepatocellular carcinoma. Cancers. 2023;15:19.
6. Rhim H, Lim HK. Radiofrequency ablation of hepatocellular carcinoma: pros and cons. Gut Liver. 2010;4(Suppl 1):S113–8. doi:10.5009/gnl.2010.4.S1.S113
7. Kim YS, Lim HK, Rhim H, Lee MW. Ablation of hepatocellular carcinoma. Best Pract Res Clin Gastroenterol. 2014;28(5):897–908. doi:10.1016/j.bpg.2014.08.011
8. Weinstein JL, Ahmed M. Percutaneous ablation for hepatocellular carcinoma. AJR Am J Roentgenol. 2018;210(6):1368–1375. doi:10.2214/ajr.17.18695
9. Horvat N, de Oliveira AI, Clemente de Oliveira B, et al. Local-regional treatment of hepatocellular carcinoma: a primer for radiologists. Radiographics. 2022;42(6):1670–1689. doi:10.1148/rg.220022
10. Huang J, Cui W, Xie X, et al. A novel prognostic model based on AFP, tumor burden score and Albumin-Bilirubin grade for patients with hepatocellular carcinoma undergoing radiofrequency ablation. Int J Hyperthermia. 2023;40(1):2256498. doi:10.1080/02656736.2023.2256498
11. Kawaguchi Y, Hasegawa K, Hagiwara Y, et al. Effect of diameter and number of hepatocellular carcinomas on survival after resection, transarterial chemoembolization, and ablation. Am J Gastroenterol. 2021;116(8):1698–1708. doi:10.14309/ajg.0000000000001256
12. Zhang SW, Zhang NN, Zhu WW, et al. A novel nomogram model to predict the recurrence-free survival and overall survival of hepatocellular carcinoma. Front Oncol. 2022;12:946531. doi:10.3389/fonc.2022.946531
13. An C, Wu S, Huang Z, et al. A novel nomogram to predict the local tumor progression after microwave ablation in patients with early-stage hepatocellular carcinoma: a tool in prediction of successful ablation. Cancer Med. 2020;9(1):104–115. doi:10.1002/cam4.2606
14. Cha DI, Ahn SH, Lee MW, et al. Risk group stratification for recurrence-free survival and early tumor recurrence after radiofrequency ablation for hepatocellular carcinoma. Cancers. 2023;15:3.
15. Vasquez MM, Hu C, Roe DJ, Chen Z, Halonen M, Guerra S. Least absolute shrinkage and selection operator type methods for the identification of serum biomarkers of overweight and obesity: simulation and application. BMC Med Res Methodol. 2016;16(1):154. doi:10.1186/s12874-016-0254-8
16. Pickett KL, Suresh K, Campbell KR, Davis S, Juarez-Colunga E. Random survival forests for dynamic predictions of a time-to-event outcome using a longitudinal biomarker. BMC Med Res Methodol. 2021;21(1):216. doi:10.1186/s12874-021-01375-x
17. He Y, Luo L, Shan R, et al. Development and validation of a nomogram for predicting postoperative early relapse and survival in hepatocellular carcinoma. J Natl Compr Canc Netw. 2023;22(1d):e237069. doi:10.6004/jnccn.2023.7069
18. Liu Q, Li J, Liu F, et al. A radiomics nomogram for the prediction of overall survival in patients with hepatocellular carcinoma after hepatectomy. Cancer Imaging. 2020;20(1):82. doi:10.1186/s40644-020-00360-9
19. Kuo YH, Huang TH, Yen YH, et al. Nomogram to predict the long-term overall survival of early-stage hepatocellular carcinoma after radiofrequency ablation. Cancers. 2023;15:12.
20. Chen S, Ma W, Cao F, et al. Hepatocellular carcinoma within the Milan criteria: a novel inflammation-based nomogram system to assess the outcomes of ablation. Front Oncol. 2020;10:1764. doi:10.3389/fonc.2020.01764
21. Testino G, Leone S, Borro P. Alcohol and hepatocellular carcinoma: a review and a point of view. World J Gastroenterol. 2014;20(43):15943–15954. doi:10.3748/wjg.v20.i43.15943
22. Cho EJ, Chung GE, Yoo JJ, et al. The association between alcohol consumption and the risk of hepatocellular carcinoma according to glycemic status in Korea: a nationwide population-based study. PLoS Med. 2023;20(6):e1004244. doi:10.1371/journal.pmed.1004244
23. Li M, Sun J, Wang Y, et al. Construction of a hepatocellular carcinoma high-risk population rating scale and independent predictors’ assessment. Am J Med Sci. 2023. doi:10.1016/j.amjms.2023.11.016
24. Yang HI, Sherman M, Su J, et al. Nomograms for risk of hepatocellular carcinoma in patients with chronic hepatitis B virus infection. J Clin Oncol. 2010;28(14):2437–2444. doi:10.1200/jco.2009.27.4456
25. Ramacciato G, Mercantini P, Petrucciani N, et al. Does surgical resection have a role in the treatment of large or multinodular hepatocellular carcinoma? Am Surg. 2010;76(11):1189–1197.
26. Chianchiano P, Pezhouh MK, Kim A, et al. Distinction of intrahepatic metastasis from multicentric carcinogenesis in multifocal hepatocellular carcinoma using molecular alterations. Hum Pathol. 2018;72:127–134. doi:10.1016/j.humpath.2017.11.011
27. Xie DY, Fan HK, Ren ZG, Fan J, Gao Q. Identifying clonal origin of multifocal hepatocellular carcinoma and its clinical implications. Clin Transl Gastroenterol. 2019;10(2):e00006. doi:10.14309/ctg.0000000000000006
28. Dhanasekaran R. Deciphering Tumor Heterogeneity in Hepatocellular Carcinoma (HCC)-multi-omic and singulomic approaches. Semin Liver Dis. 2021;41(1):9–18. doi:10.1055/s-0040-1722261
29. Dong LQ, Peng LH, Ma LJ, et al. Heterogeneous immunogenomic features and distinct escape mechanisms in multifocal hepatocellular carcinoma. J Hepatol. 2020;72(5):896–908. doi:10.1016/j.jhep.2019.12.014
30. Huang M, He M, Guo Y, et al. The influence of immune heterogeneity on the effectiveness of immune checkpoint inhibitors in multifocal hepatocellular carcinomas. Clin Cancer Res. 2020;26(18):4947–4957. doi:10.1158/1078-0432.Ccr-19-3840
31. Zhang W, Zhangyuan G, Wang F, et al. High preoperative serum globulin in hepatocellular carcinoma is a risk factor for poor survival. J Cancer. 2019;10(15):3494–3500. doi:10.7150/jca.29499
32. Deng Y, Pang Q, Miao RC, et al. Prognostic significance of pretreatment albumin/globulin ratio in patients with hepatocellular carcinoma. Onco Targets Ther. 2016;9:5317–5328. doi:10.2147/ott.S109736
33. Li J, Li Z, Hao S, et al. Inversed albumin-to-globulin ratio and underlying liver disease severity as a prognostic factor for survival in hepatocellular carcinoma patients undergoing transarterial chemoembolization. Diagn Interv Radiol. 2023;29(3):520–528. doi:10.5152/dir.2022.211166
34. Gonzalez H, Hagerling C, Werb Z. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 2018;32(19–20):1267–1284. doi:10.1101/gad.314617.118
35. Wang Z, Geng Z, Shao W, et al. Cancer-derived sialylated IgG promotes tumor immune escape by binding to Siglecs on effector T cells. Cell Mol Immunol. 2020;17(11):1148–1162. doi:10.1038/s41423-019-0327-9
36. Cui M, Huang J, Zhang S, Liu Q, Liao Q, Qiu X. Immunoglobulin expression in cancer cells and its critical roles in tumorigenesis. Front Immunol. 2021;12:613530. doi:10.3389/fimmu.2021.613530
37. Zhang H, Yang K, Wang Q, et al. Prealbumin as a predictor of short-term prognosis in patients with HBV-related acute-on-chronic liver failure. Infect Drug Resist. 2023;16:2611–2623. doi:10.2147/idr.S402585
38. Liao YY, Teng CL, Peng NF, et al. Serum prealbumin is negatively associated with survival in hepatocellular carcinoma patients after hepatic resection. J Cancer. 2019;10(13):3006–3011. doi:10.7150/jca.30903
39. Keller U. Nutritional laboratory markers in malnutrition. J Clin Med. 2019;8:6.
40. Rostenberg I, Rico R, Peñaloza R. Gc globulin and prealbumin serum levels in patients with cancer and benign inflammatory diseases and in asymptomatic smokers. J Natl Cancer Inst. 1979;62(2):299–300.
41. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1–22. doi:10.1016/j.amjmed.2015.03.032
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.