Back to Journals » Journal of Pain Research » Volume 13
Intraneural Application of microRNA-1 Mimetic Nucleotides Does Not Resolve Neuropathic Pain After Chronic Constriction Injury in Rats
Authors Kuebart A , Wollborn V, Huhn R, Hermanns H , Werdehausen R , Brandenburger T
Received 18 June 2020
Accepted for publication 14 September 2020
Published 13 November 2020 Volume 2020:13 Pages 2907—2914
DOI https://doi.org/10.2147/JPR.S266937
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Michael A Ueberall
Anne Kuebart,1,* Verena Wollborn,1,* Ragnar Huhn,1 Henning Hermanns,2 Robert Werdehausen,3 Timo Brandenburger1
1Department of Anesthesiology, University Hospital Düsseldorf, Medical Faculty, Düsseldorf 40225, Germany; 2Department of Anesthesiology, Amsterdam UMC, University of Amsterdam, Amsterdam, the Netherlands; 3Department of Anesthesiology and Intensive Care, University of Leipzig, Medical Faculty, Leipzig 04103, Germany
*These authors contributed equally to this work
Correspondence: Timo Brandenburger
Department of Anesthesiology, University Hospital Düsseldorf, Moorenstr. 5, Düsseldorf 40225, Germany
Email [email protected]
Robert Werdehausen
Department of Anesthesiology and Intensive Care, University of Leipzig, Medical Faculty, Leipzig 04103, Germany
Email [email protected]
Background: Alterations of the expression of microRNAs (miRNAs) in chronic pain models seem to play a crucial role in the development of neuropathic pain, with microRNA-1 (miR-1) being of particular interest. Recently, we were able to show that decreased miR-1 levels were associated with increased expression of brain-derived neurotrophic factor (BDNF) and Connexin 43 (Cx43). We hypothesized that miR-1 mimetic nucleotides could alleviate neuropathic pain caused by chronic constriction injury in rats.
Methods: MiR-1 mimetic nucleotides were evaluated for effectiveness, functionality, and intracellular stability by transfecting human glioblastoma cells (U-87 MG). In vivo transfection with miR-1 mimics and corresponding scrambled miRNAs serving as control was performed by repetitive injection (days 0, 3, and 7) into the sciatic nerve following chronic constriction injury (CCI) in rats. Quantitative PCR was used to measure miR-1 content. Cx43 expression was determined by Western blot analysis. Effects on neuropathic pain were assessed by detecting paw withdrawal thresholds using an automated filament application.
Results: Transfection of miR-1 mimics was confirmed in U-87 MG cells, with miR-1 content being increased significantly after 48 h and after 96 h (p< 0.05). Effective downregulation of Cx43 expression was observed 48 and 96 h after transfection (− 44 ± 0.07% and − 40 ± 0.11%; p< 0.05). In vivo, repetitive transfection with miR-1 mimetic nucleotides led to a 17.9-fold (± 14.2) increase of miR-1 in the sciatic nerve. However, the protein expression of Cx43 in sciatic nerves as well as paw withdrawal thresholds for mechanical stimulation was not significantly increased 10 days after chronic constriction injury.
Conclusion: While transfection with miR-1 mimics effective reduces Cx43 expression in vitro and restores miR-1 after CCI, we did neither observe altered levels of Cx43 protein level in nerves nor a beneficial effect on mechanical allodynia in vivo, most likely caused by insufficient Cx43 suppression.
Keywords: chronic pain, neuropathic pain, microRNAs, peripheral nerve
Introduction
Neuropathic pain is defined as a pain condition caused by a lesion or disease of the somatosensory nervous system.1 Up to 10% of the general population is affected by neuropathic pain. As the majority of these patients cannot reach a pain-free state, new approaches are crucial to improve the quality of life in these patients.2,3
In neuropathic pain, multiple alterations in RNA expression, protein synthesis as well as ion channel frequencies have been observed in the peripheral and central nervous system.4–6 Still, the underlying mechanisms of neuropathic pain are poorly understood. However, recent findings suggest that especially alterations in the expression of multiple microRNAs (miRNAs) in chronic pain models seem to play a crucial role in the development and maintenance of neuropathic pain.7 Therefore, the analysis of miRNAs seems to be a promising tool in the investigation of neuropathic pain.
Of the several altered miRNAs, in particular, microRNA-1 (miR-1) is suspected to play a significant role in the pathogenesis of neuropathic pain.8,9 In sciatic nerves, a downregulation of miR-1 has been observed after the induction of a painful peripheral neuropathy via chronic constriction injury (CCI).9 Recently, we were able to show that a decreased miR-1 level was associated with an increased expression of two pain-relevant proteins: Besides the brain-derived neurotrophic factor (BDNF) we were able to detect an upregulation of Connexin 43 (Cx43), a highly discussed protein in neuropathic pain research.9 Connexins are the main components of gap junctions.10 Therefore, an upregulation of Cx43 might contribute to the peripheral and central sensitization observed in neuropathic pain.11 As Cx43/Cx30 knockout was shown to protect mice from developing a status of neuropathic pain whereas solely Cx30 knockout did not show any protective function, the crucial role of Cx43 frequencies in the development of neuropathic pain is evident.12
To date, there is no evidence regarding the therapeutic impact of miR-1 and its ability to decrease Cx43 levels and to ease neuropathic pain in vivo. Therefore, in this study miR-1 was administered to rats after inducing neuropathy via CCI. The potential of miR-1 attenuating neuropathic symptoms was evaluated by intraneural injection of miR-1 in CCI and sham animals followed by the assessment of paw withdrawal thresholds as an index parameter for allodynia and therefore for neuropathic pain. Additionally, Cx43 levels were measured after miR-1 treatment and correlated with clinical parameters to evaluate a potential impact on Cx43 protein expression.
Methods
Animal Model
All experiments involving animals were conducted in accordance with the German legislation on the protection of animals and were approved by local authorities (North Rhine-Westphalia State Agency for Nature, Environment and Consumer Protection, Recklinghausen, Germany) and in accordance with the guidelines of the International Association for the Study of Pain. Male adult Wistar rats (6–7 seven weeks of age, weight 180–240 g) were housed in a controlled environment and had free access to food and water at any time. Beginning the experiments, the withdrawal threshold of the left hindpaw was determined and used as a reference point. Neuropathic pain was induced by using the CCI model and was clinically observable within 6–9 days.13 To determine the impact of miR-1 on severity of developed neuropathic pain, CCI animals were treated with an intraneural application of miRNA1-mimics. Sham animals received instead a scrambled sequence of the miR-1 RNA (scr-miR).
Behavioural Testing
Touch sensitivity of the paw was measured using a Dynamic Plantar Aesthesiometer® (Ugo Basile, Gemonio, Italy) and used as a score to define the development of neuropathic pain after CCI surgery. As already earlier described, animals were placed in a cage with a wire mesh bottom to enable access to the paw of the animal.4,14 In a specified duration of 20 s an increasing force of up to 50 g was applied to the left hindpaw with a filament. Animals were free to withdraw their paw in response to uncomfortable or painful sensation, which was registered automatically.
Nerve Ligation
For CCI procedures,13 animals were anesthetized with i.p. 60 mg/kg pentobarbital (Merial, Hallbergmoos, Germany). The optimal depth of anesthesia was confirmed by the absence of any reaction related to a standardized pain stimulus. For preparation of the sciatic nerve, a skin incision (approx. 1 cm) was made dorsal to the pelvis. Following the preparation of the left common sciatic nerve by bluntly separating the overlaying muscle layer, four equispaced (1 mm) loose ligations (3–0 chromic catgut) were made. Subsequently, muscle tissue was closed by sutures (vicryl). The skin incision was closed with wound clips and infiltrated with ropivacaine (0.2%; 0.3 mL) to achieve postoperative analgesia. Sham animals obtained the same procedure, but no ligatures were placed around the nerve. The following days, the development of neuropathic pain was evaluated by detection of paw withdrawal thresholds. After the last measurement, the animals were sacrificed by i.p. injection of pentobarbital (150 mg/kg).
Synthesis of miR-1-Mimetic Nucleotides
MiR-1 mimetic nucleotides were synthetized by GE Healthcare (Little Chalfont, UK). Nucleotides were constructed based on the sequence of rno-miR-1-3p (MIMAT0003125) and modified with 3ʹ-Cholesterol and 3ʹ-FAM (Rno-miR-1-3p 3ʹ Chol-psngr 3ʹ-FAM-guide). As control, 3ʹ-Chol. and 3ʹ-FAM modified scrambled miRNAs were purchased from GE Healthcare (Cat. NC#1 Ref #CN-001000-01 3ʹ-Chol-psngr 3ʹ-FAM-guide, in vivo).
Transfection of U87 Cells with miR1-Mimics
To evaluate intracellular effectiveness, functionality and intracellular stability of the used miR1-mimic, an in vitro transfection model using U87 cells (human glioblastoma cell line U-87 MG; ATCC HTB-14) was established. A stock solution with a final concentration of 500 µmol/mL miR1-mimic (Dharmacon, Lafayette, USA) was prepared by dissolving the miRNA in RNase free water. As a negative control, a solution containing scrambled miRNA (scr-miR) with comparable chemical properties was applied. Before transfection 4x106 cells were plated per well on a six-well plate obtaining a confluence of the cells ranging between 50 up to 70% the following day. After 24 h, the medium was removed and 1 mL of Dulbecco’s modified eagle medium (DMEM) was added per well. For the transfection protocol, two solutions were prepared. The first one was obtained by mixing 5 µL iFect™ (Neuromics, Edina, USA) and 25 µL DMEM). For the second one, 1 µg of miR-mimics, respectively 1 µL of the scr-miR was added to 25 µL siRNA diluent (Neuromics) and 15 µL DMEM and was incubated for five minutes. After incubation, both solutions were mixed and 70 µL of the obtained mix was added to each well. After 4 h of incubation, 1 mL of DMEM containing 20% fetal calf serum (FCS) and 1% penicillin/streptomycin was added to each well. Cells were then incubated at 37 °C for 48 h or 96 h respectively. After incubation, the medium was discarded, and cells were harvested by scraping. Until used for RNA isolation and Western blots, cells were stored at −80 °C.
Application of Intraneural miR1-Mimics
To determine the therapeutic effect of miR-1 on the severity of neuropathic pain, animals received an intraneural injection with miR-1-mimics. Intraneural injections were performed as described previously.15 In brief, animals were anesthetized, and the left sciatic nerve was exposed as described above. Then, a 30-gauge needle (Becton Dickinson) was introduced parallel to the direction of nerve fibres in the centre of the nerve and 15 µg (10 µL) of miR-1-mimics solved in iFECT were injected intraneurally. The needle was then withdrawn, and the wound closed as described above. Sham animals received an intraneural injection of 10 µL of scr-miR control following the same procedure. The first injection was done at the same time as the CCI operating procedure, the following injections were done on day 3 and day 7 after the initial surgery.
RNA Isolation
Cells and sciatic nerve tissue were homogenized by adding 1 mL of Trizol®.16 After incubation for 5 min at room temperature, 0.2 mL chloroform was added and thoroughly mixed. Incubated for 3 min, samples were centrifuged at 12,000 rcf for 15 min at 4 °C. The visible upper layer was separated into a clean tube and 0.5 mL isopropanol (100%) was added. Following 10 min of incubation, samples were again centrifuged at 7500 rcf for 30 min at 4 °C. The supernatant was discarded, and the obtained RNA pellet was thoroughly resolved in ethanol (75%) and centrifuged at 7500 rcf for 5 min at 4 °C. After another washing cycle with ethanol, the pellet was dried and resuspended in preheated (60 °C) RNase-free water. RNA isolation from dorsal root ganglions (DRGs) was performed the same way, apart from using halved volumes to match reduced tissue amounts. RNA quantity was determined using a UV spectrophotometer (NanoDrop ND-1000; Thermo Fisher Scientific, Waltham, USA). The RNA integrity was evaluated by using Agilent microfluid chips (Agilent, Santa Clara, USA). RNA samples showing high RNA integrity numbers (RIN) ≥ 8 were included in the study.
Real-Time Quantitative PCR
For complementary DNA (cDNA) synthesis, the TaqMan® MicroRNA Reverse Transcription Kit (Applied Biosystems, Foster City, USA) was applied following the manufacturer’s instructions. Quantitative PCR (qPCR) assays were conducted for miR-1 (assay ID: 4,427,975; Applied Biosystems), U6 (assay ID: 001973; Applied Biosystems). Rno-Connexin 43 assay were purchased from TIB MOLBIOL (Berlin, Germany). The sequence of the forward primer is AGGAGTTCCACCAACTTTGGC, reverse primer TGGAGTAGGCTTGGACCTTGTC and FMA-AGCTTCCCCAAGGCACTCCAGTC-BBQ for the reporter probe. GAPDH (Assay ID: Rn_01775763, Applied Biosystems) was used for normalization. Samples were prepared according to the annexed protocol. For qPCR, the AB 7300 Real-Time PCR Cycler (Applied Biosystems) was programmed as follows: 50 °C for 2 min, 95 °C for 10 min, followed by 40 cycles at 95 °C for 15 s and 60 °C for 60 s. The relative expression of miRNAs was analyzed using the relative expression tool as described previously.17
miRNA Quantitative Polymerase Chain Reaction Assay
Reverse transcription of total RNA was performed using the High Capacity RNA-to-cDNA Master Mix (Applied Biosystems). The quantitative polymerase chain reaction (qPCR) assays for rno-miR-1 and BDNF (assay ID: 02531967_s1) were purchased from Applied Biosystems and applied according to the manufacturer’s protocol. Quantitative polymerase chain reaction conditions were as follows: 50 °C for 2 min, 95 °C for 10 min, 40 cycles of 95 °C for 15 s, 60 °C for 60 s using an ABI 7300HT thermocycler (Applied Biosystems).
Western Blot Analysis
Western Blot analysis was used to quantify the expression of Cx43 in neuronal tissue. Therefore, neuronal tissue was homogenized, and RIPA-Buffer was added. After 5 min incubation in an ultrasound bath, samples were incubated another 10 min on ice. Protein levels were measured determined using Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific). For denaturation, sodium dodecyl sulfate buffer (SDS-buffer) and mercaptoethanol (9:1) were added and samples were incubated at 95 °C for 5 min. For gel electrophoresis, 10% SDS gels were prepared and samples were loaded into lanes using 40 µg protein/lane. After adequate separation (85 min at 100 V), proteins were transferred onto a polyvinylidene difluoride membrane applying 220 mA for 1 h. Further, the membrane was blocked with TBS-T for 2 h. After the administration of the primary antibody, the membrane was incubated overnight at 4 °C. As primary antibodies, Connexin 43 (Abcam, 1:10,000) and Glyceraldehyde-3-phosphate-dehydrogenase (GAPDH Abcam, 1:50,000) were used. After elimination of unbound antibodies via washing three times with TBS-T, the secondary antibody was added and incubated for 2 h. After disposing the unbound antibodies, membrane-bound antibodies were detected by using the enhanced chemiluminescent detection method (cool snap HQ2; Photometrics®, Tuscon, USA)
Statistical Analysis
Data are presented as mean (standard deviation) unless indicated otherwise. Differences between groups were tested using Student’s t-Test and Bonferroni-Holms correction when more than one comparison was made. In vitro experiments were performed in triplicates. Calculations were performed using GraphPad Prism Software version 7.0 (GraphPad Software Inc., La Jolla, USA). A resulting p-value < 0.05 was considered indicative of a statistically significant difference.
Results
In a first step, we aimed to analyse uptake and intracellular stability of the miR-1 mimics. Therefore, miR1 levels were assessed 48 h and 96 h after transfection in U87 cells. qPCR analyses displayed an excellent uptake of miR-1-mimetic oligonucleotides into U87 cells (Figure 1). Additionally, the 96-h data of this experiment indicate that miRNA mimics are stable at least for several days after their uptake.
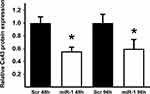
Effect of miR-1 Mimic Transfection on Connexin 43 Protein Levels
Relative Cx43 protein expression in miR-1/scr-miR transfected cells was determined using Western blot analysis. After transfection with miR-1 mimics, Cx43 was found to be significantly downregulated after 48 h, as well as after 96 h (Figure 2).
Intraneural Injection of miR-1 Mimics
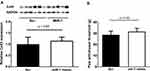
To determine the effect of miR-1 on peripheral neuronal tissue, miR-1 was injected into the sciatic nerve at days 0, 3, and 7 post CCI-surgery (Figure 3A). The injections were assumingly successful as the expression of miR-1 in neuronal tissue was found to be significantly higher in nerves treated with miR-1 injections compared to those treated with scr-miRNA injections (Figure 3B). However, the elevated miR-1 level did not result in a statistically significant downregulation of Cx43 (Figure 4A). This is in line with the results of the allodynia testing which reveal also no significant differences regarding CCI animals treated with miR-1 compared to animals injected with scr-miR serving as a control (Figure 4B).
Discussion
Despite the high number of patients suffering from neuropathic pain and the numerous studies focused on the improvement of neuropathic pain therapies, its treatment remains challenging.3 Therapy with drugs like opioids and/or anticonvulsants can cause severe side effects, and might even aggravate symptoms by provoking opioid-induced hyperalgesia.18 Therefore, the establishment of new therapeutic strategies is the main objective in pain research in these days.
Following this objective, therapeutic options based on molecular genetic mechanisms are a new avenue of research, since alterations in RNA expression, protein expression, and ion channel frequencies have been observed in several neuropathic pain models.4–6 Lately, in particular, miRNAs are seen as promising tools to gain more insight into the underlying mechanisms of the pathological dysregulations in neuropathic pain as a broad range of miRNAs display changed expression levels under this condition.4 The specificity of miRNAs regarding the regulation of their target proteins makes them an interesting candidate not only for the understanding of the crucial, pain-inducing pathways19,20 but also as a therapeutic option themselves.19,21
This project is focusing on miR-1 and aims to evaluate the therapeutic potential of miR-1 attenuating neuropathic pain. Of all differentially regulated miRNAs, miR-1 is one of the most promising determining factors regarding neuropathic pain.8,9,22,23 As we could already show in previous studies, miR-1 targets the proteins Connexin 43 and BDNF.9 Both proteins are considered to be involved in the development and persistence of neuropathic pain. Especially connexins are known for playing a crucial role in the pathogenesis of neuropathic pain.24
As already shown, miR-1 levels are significantly down-regulated in the peripheral nerve after inducing neuropathy via CCI.9 Correspondingly, its targets Cx43 and BDNF were found to be upregulated,9 implicating a negative correlation of miR-1 levels and Cx43 expression levels. Interestingly, in the higher levels of the somatosensory system, like DRGs and dorsal horn of the spinal cord, this downregulation of miR-1 was so far not detected.9 In contrast, Kusuda et al found miR-1 to be upregulated in the DRG but down-regulated in the dorsal horn of the spinal cord.8
So far, changes in the transcription levels of Cx43 are repeatedly described in the status of neuropathic pain.25,26 Increased levels of Cx43 are suspected to impair the physiological structure and frequency of gap junctions, impairing neuromuscular pain transmission, and leading to hypersensitivity.27 In the CNS, it is already taken as evidence that the blockade of gap junctions is alleviating the hypersensitivity.28 This assumption is strengthened by several studies showing attenuated pain levels after blocking Cx43.12,29,30 As a reverse conclusion, it can be assumed that miR-1 shows therapeutic potential via suppressing the transcription of Cx43. The occurrence of Cx43 in the PNS was first described by Yoshimura et al, who detected Cx43 mainly in Schwann cells and in a Schwann cell line.31 Since then, Cx43 is suspected to play a crucial role in the maintenance of neuropathic pain since its involvement in the formation of gap junctions permits the release of various substances including chemokines.32
Here, we firstly tested the estimated therapeutic potential of miR-1. In vitro, in line with the hypothesis, U87 cells transfected with miR-1 showed significantly down-regulated levels of Cx43 remaining stable for at least 96 h. However, these results were not reproducible in CCI animals that were treated with intraneural injections of miR-1. Interestingly, despite successful intraneural injections of miR-1 confirmed by high miR-1 levels of the neuronal tissue post-injection, neither significant downregulation of Cx43 was detected in neuronal tissue nor any significant effect on the paw withdrawal threshold was observable. The diverging results of successful downregulation of Cx43 in vitro could, on the one hand, indicate a more complex pathophysiology in vivo with probably several more regulatory mechanisms of Cx43 than miR-1 itself. On the other hand, it could indicate that via intraneural injection, miR-1 did not reach its target point intracellularly. Unlike cell cultures, nerve tissue is composed of multiple different cell types, as well as extracellular matrixes. Addressing this problem, recently a method was elucidated using lentiviral vectors to gain high expression levels of miRNAs intracellularly.33 Using this, Lee et al were able to induce specific miRNA-expression patterns in the sciatic nerve.34
Conclusions
Taken together, miR-1 remains an interesting candidate regarding therapy of neuropathic pain, especially in peripheral nerves. According to the results of the intraneural injections of miR-1, an alternative method for either application of miR-1 or enhancing neural miR-1 expression will be the subject of future research investigations.
Acknowledgements
The technical assistance of Claudia Dohle is gratefully acknowledged.
Funding
The authors have no sources of funding to declare for this manuscript [or declare funding].
Disclosure
Anne Kuebart and Verena Wollborn are co-first authors for this study. The authors declare no conflicts of interest.
References
1. International Association for the Study of Pain. International Association for the Study of Pain. 2019. Accessed March 15, 2019.
2. van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain. 2014;155(4):654–662. doi:10.1016/j.pain.2013.11.013
3. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162–173. doi:10.1016/S1474-4422(14)70251-0
4. Brandenburger T, Castoldi M, Brendel M, et al. Expression of spinal cord microRNAs in a rat model of chronic neuropathic pain. Neurosci Lett. 2012;506(2):281–286. doi:10.1016/j.neulet.2011.11.023
5. Devor M. Sodium channels and mechanisms of neuropathic pain. J Pain. 2006;7(1, Supplement):3–12. doi:10.1016/j.jpain.2005.09.006
6. Niederberger E, Geisslinger G, Warner D, Warner M. Proteomics in neuropathic pain research. Anesthesiology. 2008;108(2):314–323. doi:10.1097/01.anes.0000299838.13368.6e
7. Andersen HH, Duroux M, Gazerani P. MicroRNAs as modulators and biomarkers of inflammatory and neuropathic pain conditions. Neurobiol Dis. 2014;71:159–168. doi:10.1016/j.nbd.2014.08.003
8. Kusuda R, Cadetti F, Ravanelli MI, et al. Differential expression of microRNAs in mouse pain models. Mol Pain. 2006;7:17. doi:10.1186/1744-8069-7-17
9. Neumann E, Hermanns H, Barthel F, Werdehausen R, Brandenburger T. Expression changes of microRNA-1 and its targets connexin 43 and brain-derived neurotrophic factor in the peripheral nervous system of chronic neuropathic rats. Mol Pain. 2015;11:39. doi:10.1186/s12990-015-0045-y
10. Orthmann-Murphy JL, Abrams CK, Scherer SS. Gap junctions couple astrocytes and oligodendrocytes. J Mol Neurosci. 2008;35(1):101–116. doi:10.1007/s12031-007-9027-5
11. Jeon YH, Youn DH. Spinal gap junction channels in neuropathic pain. Korean J Pain. 2015;28(4):231–235. doi:10.3344/kjp.2015.28.4.231
12. Chen MJ, Kress B, Han X, et al. Astrocytic CX43 hemichannels and gap junctions play a crucial role in development of chronic neuropathic pain following spinal cord injury. Glia. 2012;60(11):1660–1670. doi:10.1002/glia.22384
13. Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 1988;33(1):87–107. doi:10.1016/0304-3959(88)90209-6
14. Muth-Selbach U, Hermanns H, Driehsen C, Lipfert P, Freynhagen R. Racemic intrathecal mirtazapine but not its enantiomers acts anti-neuropathic after chronic constriction injury in rats. Brain Res Bull. 2009;79(1):63–68. doi:10.1016/j.brainresbull.2008.12.015
15. Iohom G, Lan GB, Diarra DP, et al. Long-term evaluation of motor function following intraneural injection of ropivacaine using walking track analysis in rats. Br J Anaesth. 2005;94(4):524–529. doi:10.1093/bja/aei079
16. Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162(1):156–159. doi:10.1016/0003-2697(87)90021-2
17. Pfaffl MW, Horgan GW, Dempfle L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002;30(9):e36–e36. doi:10.1093/nar/30.9.e36
18. Brush DE. Complications of long-term opioid therapy for management of chronic pain: the paradox of opioid-induced hyperalgesia. J Med Toxicol. 2012;8(4):387–392. doi:10.1007/s13181-012-0260-0
19. López-González MJ, Landry M, Favereaux A. MicroRNA and chronic pain: from mechanisms to therapeutic potential. Pharmacol Ther. 2017;180:1–15. doi:10.1016/j.pharmthera.2017.06.001
20. Zhang J, Liu Y, Lu L. Emerging role of MicroRNAs in peripheral nerve system. Life Sci. 2018;207:227–233. doi:10.1016/j.lfs.2018.06.011
21. Kynast KL, Russe OQ, Möser CV, Geisslinger G, Niederberger E. Modulation of central nervous system–specific microRNA-124a alters the inflammatory response in the formalin test in mice. PAIN®. 2013;154(3):368–376. doi:10.1016/j.pain.2012.11.010
22. Strickland ER, Hook MA, Balaraman S, Huie JR, Grau JW, Miranda RC. MicroRNA dysregulation following spinal cord contusion: implications for neural plasticity and repair. Neuroscience. 2011;186:146–160. doi:10.1016/j.neuroscience.2011.03.063
23. Yi S, Yuan Y, Chen Q, et al. Regulation of schwann cell proliferation and migration by miR-1 targeting brain-derived neurotrophic factor after peripheral nerve injury. Sci Rep. 2016;6:29121. doi:10.1038/srep29121
24. Morioka N, Nakamura Y, Zhang FF, Hisaoka-Nakashima K, Nakata Y. Role of connexins in chronic pain and their potential as therapeutic targets for next-generation analgesics. Biol Pharm Bull. 2019;42(6):857–866. doi:10.1248/bpb.b19-00195
25. Yoon S-Y, Robinson CR, Zhang H, Dougherty PM. Spinal astrocyte gap junctions contribute to oxaliplatin-induced mechanical hypersensitivity. J Pain. 2013;14(2):205–214.
26. Kaji K, Shinoda M, Honda K, Unno S, Shimizu N, Iwata K. Connexin 43 contributes to ectopic orofacial pain following inferior alveolar nerve injury. Mol Pain. 2016;12:1744806916633704.
27. Xing L, Yang T, Cui S, Chen G. Connexin hemichannels in astrocytes: role in CNS disorders. Front Mol Neurosci. 2019;12:23. doi:10.3389/fnmol.2019.00023
28. Spray DC, Hanani M. Gap junctions, pannexins and pain. Neurosci Lett. 2019;695:46–52. doi:10.1016/j.neulet.2017.06.035
29. Tonkin RS, Bowles C, Perera CJ, et al. Attenuation of mechanical pain hypersensitivity by treatment with peptide5, a connexin-43 mimetic peptide, involves inhibition of NLRP3 inflammasome in nerve-injured mice. Exp Neurol. 2018;300:1–12. doi:10.1016/j.expneurol.2017.10.016
30. Xu Q, Cheong YK, He SQ, et al. Suppression of spinal connexin 43 expression attenuates mechanical hypersensitivity in rats after an L5 spinal nerve injury. Neurosci Lett. 2014;566:194–199. doi:10.1016/j.neulet.2014.03.004
31. Yoshimura T, Satake M, Kobayashi T. Connexin43 is another gap junction protein in the peripheral nervous system. J Neurochem. 1996;67(3):1252–1258. doi:10.1046/j.1471-4159.1996.67031252.x
32. Chen G, Park CK, Xie RG, Berta T, Nedergaard M, Ji RR. Connexin-43 induces chemokine release from spinal cord astrocytes to maintain late-phase neuropathic pain in mice. Brain. 2014;137(Pt 8):2193–2209. doi:10.1093/brain/awu140
33. Mishima T, Sadovsky E, Gegick ME, Sadovsky Y. Determinants of effective lentivirus-driven microRNA expression in vivo. Sci Rep. 2016;6:33345. doi:10.1038/srep33345
34. Lee J-S, Kwak G, Kim HJ, Park H-T, Choi B-O, Hong YB. miR-381 attenuates peripheral neuropathic phenotype caused by overexpression of PMP22. Exp Neurobiol. 2019;28(2):279–288. doi:10.5607/en.2019.28.2.279
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.