Back to Journals » Nature and Science of Sleep » Volume 10
Inpatient phase-advance therapy for delayed sleep–wake phase disorder: a retrospective study
Authors Takeshima M , Shimizu T , Echizenya M, Ishikawa H, Kanbayashi T
Received 5 July 2018
Accepted for publication 8 September 2018
Published 12 October 2018 Volume 2018:10 Pages 327—333
DOI https://doi.org/10.2147/NSS.S179264
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Steven A Shea
Masahiro Takeshima,1 Tetsuo Shimizu,2 Masaru Echizenya,3 Hiroyasu Ishikawa,1,4 Takashi Kanbayashi1
1Department of Neuropsychiatry, Akita University Graduate School of Medicine, Akita, Japan; 2Mental Health and Welfare Center, Akita, Japan; 3Echizenya Sleep and Mental Clinic, Akita, Japan; 4Department of Neuropsychiatry, Nakadori Rehabilitation Hospital, Akita, Japan
Purpose: The efficacy of inpatient phase-advance therapy among patients with delayed sleep–wake phase disorder (DSWPD) has not been adequately investigated because response rates are considered low. We aimed to examine the efficacy of such treatment in this patient population.
Patients and methods: The present retrospective study included data from 66 patients with DSWPD who had been admitted to Akita University Hospital for inpatient phase-advance therapy between September 1, 2005, and April 30, 2018. DSWPD was diagnosed based on the International Classification of Sleep Disorders, 3rd edition, criteria using electronic medical records. We examined remission rates during inpatient therapy as well as relapse rates at the time of the first outpatient examination following discharge. Univariate analysis was performed to investigate predictive factors for postinpatient therapy relapse.
Results: The rate of DSWPD remission over the course of inpatient phase-advance therapy was 100% (95% CI: 95.6%–100%), with a median duration of 1 day (IQR: 1–2 days; range: 1–9 days) until remission. The rate of relapse following discharge was 45.8% (95% CI: 32.7%–59.2%). Univariate analysis indicated that the rate of relapse was significantly higher for minors (under 18) than adults (18 and over), for those whose age at onset was below 16 years than for those whose age at onset was 16 or above, and for those with relatively low motivation for their occupation (P=0.0339, P=0.0136, and P<0.001, respectively).
Conclusion: The rate of DSWPD remission under inpatient phase-advance therapy was remarkably high (100%), while the rate of relapse after discharge was ~50%. Further studies are required to determine the long-term prognosis of inpatient therapy, risk factors for relapse, and the types of treatment most effective for preventing relapse.
Keywords: delayed sleep–wake phase disorder, chronotherapy, sleep disorders, circadian rhythm sleep disorders
Introduction
The principal symptoms of delayed sleep–wake phase disorder (DSWPD) include difficulty falling asleep and difficulty waking. As such, patients with DSWPD often fail to wake at a socially desirable time, thus affecting their ability to participate in society.1 To date, treatments for DSWPD have included chronotherapy (phase-delay therapy),2 pharmacotherapy (melatonin,3 vitamin B12,4 aripiprazole5), bright light therapy,6 night-time light avoidance,7 and cognitive behavioral therapy.8 As patients with DSWPD exhibit difficulties falling asleep and waking early, the prescribed chronotherapy is “sleep scheduling”,2 in which the patient’s sleep schedule is gradually delayed until the desired sleep–wake rhythm is reached. In our clinical experience, patients with DSWPD are able to easily fall asleep and wake up early within a few days of sleep-study hospitalization. However, the efficacy of inpatient phase-advance therapy has not been adequately systematically investigated among patients with DSWPD. Only one report examined the effect of inpatient phase-advance therapy on DSWPD patients. Iwamitsu et al reported that more than 90% of DSWPD patients with school refusal experienced improved sleep–wake rhythm just after hospitalization and 42.9% of these patients relapsed after discharge.9 In the present retrospective study, we aimed to investigate the efficacy of such treatment in this patient population.
Patients and methods
Study design
The present retrospective epidemiological study was conducted to investigate the efficacy of inpatient phase-advance therapy for DSWPD.
Study setting and population
We included patients meeting the International Classification of Sleep Disorders, 3rd edition (ICSD-3)10 criteria for DSWPD who had been admitted to Akita University Hospital for inpatient phase-advance therapy between September 1, 2005, and April 30, 2018. We excluded patients for whom 7 or more days’ worth of sleep logs or actigraphy data could not be confirmed against the electronic medical records. For patients who were admitted multiple times following the diagnosis of DSWPD, we utilized data from their first hospitalization only. We also excluded patients who had undergone the same treatment at other hospitals.
We examined the rate of remission during inpatient therapy, the number of days until remission during inpatient therapy, the duration of hospitalization, and the number of days until the first outpatient examination after discharge. Sleep logs for the week preceding therapy were also examined to determine mean sleep and wake times. The Clinical Global Impressions Scale-Severity (CGI-S) score measured during hospitalization and at the first outpatient examination was assessed from electronic medical charts. The CGI-S is rated on the following 7-point scale: 1, normal, not at all ill; 2, borderline ill; 3, mildly ill; 4, moderately ill; 5, markedly ill; 6, severely ill; and 7, among the most extremely ill patients. In-hospital remission was defined as “1) being able to wake up at the desired time in the morning with or without the assistance of a physician or hospital staff, successfully adapting to living in the hospital (three complete meals and a 10-minute calisthenics session at 11 am), 2) not meeting the criteria for diagnosis of DSWPD according to ICSD-3, and 3) CGI-S of 6 or 7”. Remission after discharge from the hospital was defined as “1) being able to wake up at the desired time in the morning with or without the assistance of a family member or members, 2) successfully adapting to school or work, 3) not meeting the criteria for diagnosis of DSWPD according to ICSD-3, and 4) CGI-S of 6 or 7”. We also collected data regarding patient sex, age, age at onset, disease duration, number of housemates, comorbid mental disorders (Diagnostic and Statistical Manual of Mental Disorders, 5th edtion [DSM-5]), smoking status, medications during/following hospitalization (melatonin, ramelteon, benzodiazepine hypnotic drugs, vitamin B12, antipsychotics, antidepressants, methylphenidate), and whether bright light therapy was administered during hospitalization. Patients were considered to exhibit or lack motivation for their occupation if their medical records indicated the following, respectively: “Patient wants to go to school/work” or “Patient does not (or does not particularly) want to go to school/work”. When there were no such notes in the medical records, the patients were considered motivated.
Inpatient phase-advance therapy
During hospitalization, 1) lights were turned off at 9 pm and turned on at 6 am; 2) a nurse opened the curtains at 6 am; 3) patients were called to wake up by nurses at 6 am; 4) meals were served at 7 am, 12 pm, and 6 pm; and 5) there was a 10-minute calisthenics session at 11 am. “Waking voluntarily” was defined if the patient was awake when the nurses came to wake up the patients at 6 am. “Waking to a hospital staff” was defined if the patient was asleep when the nurses came to wake up the patients at 6 am. The patients were instructed to follow the rules of the hospital, sleep when the lights went out, and try not to sleep during the day. During their hospitalization, patients were prohibited from consuming alcohol, consuming caffeine after 3 pm, and using smartphones or other light-emitting devices after 9 pm. Nurses checked on the patients 19 times a day (at 6 am, 7 am, 8 am, 10 am, 12 pm, 2 pm, 3 pm, 5 pm, 6 pm, 8 pm, 9 pm, 10 pm, 11 pm, 12 am, 1 am, 2 am, 3 am, 4 am, and 5 am). If a patient was sleeping between 6 am and 8 pm, the nurses attempted to wake up the patient by calling out and/or shaking his or her body for a few minutes. If the patient did not wake up following this intervention, he or she was allowed to remain asleep. Inpatient pharmacotherapy and bright light therapy were administered at the discretion of attending physicians.
Bright light therapy
A portable light box, Bright Light ME Pro (Solartone Incorporated, Tokyo, Japan), was used for bright light therapy. This system generates a fluorescent full-spectrum light with an intensity of 10,000 lx at a distance of 60 cm at eye level. Patients who sat in front of the light box were to be irradiated with light of 10,000 lx for 2 hours between 6 am and 10 am. On the day before starting bright light therapy, the illuminance was checked using a luminometer by a doctor and the patient. The patients were provided with standardized and written instructions.
Data analysis
Continuous variables are expressed as the median and IQR (IQR: 25%–75% percentile). We investigated remission and relapse rates (95% CI) following inpatient phase-advance therapy for DSWPD. Chi-square tests were performed to examine differences in sex, age (ie, <18, ≥18 years), DSWPD onset (ie, <16, ≥16 years), DSWPD duration (ie, <12, ≥12 months), and bright light therapy administration between the remission and relapse groups. Fisher’s exact test was performed to analyze differences in motivation for the occupation and cohabitation.
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R 2.13.0 (R Foundation for Statistical Computing, Vienna, Austria). More precisely, EZR is a modified version of R commander (version 1.6-3) designed to add statistical functions frequently used in biostatistics.11
Ethical approval and consent to participate
This study was approved by the Ethical Committee for Human Research of Akita University (No. 1960), which also exempted the study from the requirement of informed consent because the study involved de-identified data acquired during routine treatment.
Results
Demographics
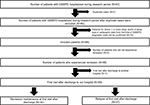
Figure 1 shows the flowchart for participant selection. A total of 87 patients with DSWPD were admitted for inpatient phase-advance therapy during the study period. Fourteen of these patients were excluded because 7 or more days’ worth of their sleep logs or actigraphy data could not be confirmed against their electronic medical records. An additional seven patients with comorbid illnesses were excluded. Prior to their admission to Akita University Hospital, none of the patients had been admitted to other hospitals for inpatient phase-advance therapy for DSWPD. Thus, 66 patients were included in the study. The demographic characteristics of the included patients (32 men, 34 women; median age: 16 years [IQR: 14–21 years]) are presented in Table 1. The median age at onset was 15 years (IQR: 13–17.75 years), while median disease duration was 12 months (IQR: 5.25–28.5 months). Sleep log data for the week preceding hospitalization were available for 41 patients. Mean sleep time was 2:15±0:33, while mean wake time was 12:21±0:31. Sixty-four patients lived with another person, while 2 did not. Patients with comorbid mental disorders included one with intellectual disability, four with autism spectrum disorder (ASD), three with attention-deficit/hyperactivity disorder (ADHD), one with ASD + ADHD, one with bipolar disorder, three with depressive disorder, one with anxiety disorder, and one with somatic symptoms/related disorders. Five patients were smokers.
Table 2 lists the medications initiated during or after hospitalization. Among the 66 patients with DSWPD, 26 were undergoing pharmacotherapy at the time of admission. Twelve patients were on ramelteon at a mean dosage of 5.7 mg (range: 4–8 mg), while ten were on vitamin B12 at a mean dosage of 1,000 µg (range: 500–4,500 µg). Thirteen patients were on benzodiazepine hypnotic drugs as follows: zolpidem 10 mg + triazolam 0.5 mg (n=1), zolpidem 10 mg (n=3), zolpidem 5 mg + triazolam 0.25 mg (n=1), zolpidem 5 mg (n=1), zopiclone 7.5 mg (n=1), rilmazafone 2 mg + flunitrazepam 1 mg (n=1), flunitrazepam 0.5 mg (n=1), brotizolam 0.25 mg (n=2), rilmazafone 2 mg (n=1), and eszopiclone 2 mg (n=1). Five patients were using aripiprazole (APZ) at a mean dosage of 1.6 mg (0.5 mg [n=1], 1 mg [n=3], and 4.5 mg [n=1]). Nine patients were on antidepressants as follows: escitalopram 5 mg (n=1), fluvoxamine 50 mg (n=2), sertraline 50 mg (n=1), milnacipran 25 mg (n=1), trazodone 50 mg (n=1), amoxapine 50 mg (n=1), milnacipran 50 mg + maprotiline 25 mg (n=1), and mianserin 50 mg (n=1). One patient was using methylphenidate at a dosage of 27 mg. Among the 26 patients undergoing pharmacotherapy at the time of admission, 4 were prescribed new drugs during hospitalization. Among the 40 patients who were not on any medication at the time of admission, 22 were prescribed new drugs during hospitalization. New drugs included melatonin 3 mg (n=5), ramelteon 4 mg (n=16), and vitamin B12 (n=6) at a mean dosage of 1,333 µg (range: 1,000–1,500 µg). Three patients were prescribed benzodiazepine hypnotic drugs as follows: rilmazafone 1 mg (n=1), triazolam 0.25 mg + brotizolam 0.25 mg (n=1), and zolpidem 5 mg (n=1). Three patients were on antipsychotic drugs as follows: APZ 0.5 mg (n=1), APZ 1.5 mg (n=1), and olanzapine 1.5 mg (n=1). A total of 27 patients underwent bright light therapy during hospitalization. Light therapy began an average of 7 days following admission (range: 2–12 days) and was continued until discharge. All patients who received bright light therapy did so after remission during inpatient therapy.
All 66 patients experienced remission following inpatient therapy (100% remission rate; 95% CI: 95.6%–100%). It required a median of 1 day (IQR: 1–2 days; range: 1–9 days) for patients to adapt to being awakened by the physician or hospital staff in the hospital and a median of 1 day (IQR: 1–3 days; range: 1–29 days) for patients to wake up on their own in the hospital. A total of 5.1% of patients (3/59) were unable to wake up on their own during the course of hospitalization. The median hospitalization period was 14 days (IQR: 9.25–19). Following discharge, patients continued the medications prescribed during hospitalization, although none continued bright light therapy at home. Among the 66 included patients, 59 underwent examination at Akita University Hospital a median of 14 days after discharge (IQR: 11–19 days). Among these 59 patients, 45.8% (95% CI: 32.7%–59.2%) experienced relapse.
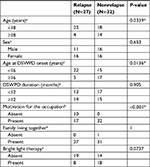
Table 3 shows the clinical characteristics of patients with and without relapse. Relapse rates were significantly higher for minors (under 18) than adults (18 and over), for those whose age at onset was younger than 16 years compared with those whose age at onset was 16 years or older, and for those without motivation relative to those with motivation (P=0.0339, P=0.0136, P<0.001, respectively). No significant differences in the rate of relapse were observed based on sex, disease duration, or cohabitation status.
Discussion
The present study is the second to examine the therapeutic efficacy of inpatient phase-advance therapy for DSWPD and one of the few studies concentrating on the rates of DSWPD remission and relapse. The results of this study are comparable to those of the previous study by Iwamitsu et al, as described above.9
All patients experienced remission for a short period of time following inpatient phase-advance therapy. The strong social time cues in this study may have contributed to the high remission rate, as our patients experienced remission whether or not they were placed on new medications during hospitalization. Given the high remission rates observed over a relatively short period of time, our findings suggest that inpatient phase-advance therapy should be utilized during the early stages of treatment when outpatient therapy has provided unsatisfactory results.
Relapse occurred in approximately half of patients in this study. This result was comparable to that of a previous study where 42.9% of patients relapsed 3 months after discharge.9 The high relapse rate may be related to factors such as 1) low motivation for the occupation (as defined earlier in the manuscript), 2) dearth of social cues following discharge, 3) nighttime use of light-emitting devices, the usage of which was prohibited during hospitalization, 4) inability of a 2-week hospitalization to alleviate the desynchronization between an individual’s sleep–wake rhythm and internal body rhythm, and 5) superficial in-hospital remission may have raised the recurrence rate after discharge.
This study identified that age under 18 years, age at onset under 16 years, and lack of motivation for the occupation were predictive factors for relapse. Further studies should evaluate a greater number of cases in order to identify such predictive factors considering intercorrelations and to develop more effective strategies for preventing relapse in patients with DSWPD.
Limitations
The present study possesses several limitations of note such as 1) a small number of cases, 2) insufficient data from sleep logs or actigraphy, other than that available at the time of diagnosis, 3) lack of core body temperature measurements or other biometric data, 4) lack of data on patient motivation for inpatient phase-advance therapy, 5) inconsistencies of therapies apart from the hospitalization and strict bed schedule within the hospital, which disallowed the identification of effective treatment components and their relation to relapse, 6) long-term prognosis unknown because the observation periods after discharge were short, and 7) cost–benefit ratio unknown because economic loss by DSWPD was not examined.
The definition of in-hospital remission in this study is also controversial. First, this in-hospital remission may be very superficial and only obtainable in the hospital or other very structured environments. Second, with this definition, it cannot be verified whether the patients are sleeping in between checks; therefore, even if they do, it is still regarded as in-hospital remission. Third, even if the patients are not asleep during the day and adapt to life in the hospital, they may not be able to adapt to daily life due to excessive daytime sleepiness. It is hoped that the definition of in-hospital remission should be determined in consideration of sleep–wake rhythm assessment by actigraphy in the future.
Conclusion
The rate of DSWPD remission under inpatient phase-advance therapy was remarkably high (100%), while the rate of relapse after discharge was ~50%. Further studies are required to determine the long-term prognosis of inpatient therapy, risk factors for relapse, and the types of treatment most effective for preventing relapse.
Acknowledgments
The authors would like to thank Dr Yuhei Kayukawa for helpful discussions and Editage (www.editage.jp) for English-language editing.
Author contributions
All authors contributed toward data analysis and revision of the manuscript and agree to be accountable for all aspects of this study.
Disclosure
The authors report no conflicts of interest in this work.
References
Auger RR, Burgess HJ, Emens JS, Deriy LV, Thomas SM, Sharkey KM. Clinical practice guideline for the treatment of intrinsic circadian rhythm sleep–wake disorders: advanced sleep–wake phase disorder (ASWPD), delayed sleep–wake phase disorder (DSWPD), non-24-hour sleep–wake rhythm disorder (N24SWD), and irregular sleep–wake rhythm disorder (ISWRD). An update for 2015: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2015;11(10):1199–1236. | ||
Czeisler CA, Richardson GS, Coleman RM, et al. Chronotherapy: resetting the circadian clocks of patients with delayed sleep phase insomnia. Sleep. 1981;4(1):1–21. | ||
van Geijlswijk IM, Korzilius HP, Smits MG. The use of exogenous melatonin in delayed sleep phase disorder: a meta-analysis. Sleep. 2010;33(12):1605–1614. | ||
Okawa M, Takahashi K, Egashira K, et al. Vitamin B12 treatment for delayed sleep phase syndrome: a multi-center double-blind study. Psychiatry Clin Neurosci. 1997;51(5):275–279. | ||
Omori Y, Kanbayashi T, Sagawa Y, et al. Low dose of aripiprazole advanced sleep rhythm and reduced nocturnal sleep time in the patients with delayed sleep phase syndrome: an open-labeled clinical observation. Neuropsychiatr Dis Treat. 2018;14:1281–1286. | ||
Saxvig IW, Wilhelmsen-Langeland A, Pallesen S, Vedaa O, Nordhus IH, Bjorvatn B. A randomized controlled trial with bright light and melatonin for delayed sleep phase disorder: effects on subjective and objective sleep. Chronobiol Int. 2014;31(1):72–86. | ||
Stothard ER, Mchill AW, Depner CM, et al. Circadian entrainment to the natural light-dark cycle across seasons and the weekend. Curr Biol. 2017;27(4):508–513. | ||
Jansson-Fröjmark M, Danielsson K, Markström A, Broman JE. Developing a cognitive behavioral therapy manual for delayed sleep-wake phase disorder. Cogn Behav Ther. 2016;45(6):518–532. | ||
Iwamitsu Y, Ozeki Y, Konishi M, Murakami J, Kimura S, Okawa M. Psychological characteristics and the efficacy of hospitalization treatment on delayed sleep phase syndrome patients with school refusal. Sleep Biol Rhythms. 2007;5(1):15–22. | ||
American Academy of Sleep Medicine. The International Classification of Sleep Disorders: Diagnostic and Coding Manual. 2nd ed. Dairen, IL: American Academy of Sleep Medicine; 2014:191–197. | ||
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48(3):452–458. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.