Back to Journals » OncoTargets and Therapy » Volume 14
Identification and Targeting of Thomsen–Friedenreich and IL1RAP Antigens on Chronic Myeloid Leukemia Stem Cells Using Bi-Specific Antibodies
Authors Eldesouki RE , Wu C, Saleh FM, Mohammed EAM, Younes S , Hassan NE, Brown TC, Alt EU , Robinson JE, Badr FM, Braun SE
Received 3 April 2020
Accepted for publication 9 November 2020
Published 22 January 2021 Volume 2021:14 Pages 609—621
DOI https://doi.org/10.2147/OTT.S255299
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Federico Perche
Raghda E Eldesouki,1,2 Chengxiang Wu,2 Fayez M Saleh,2,3 Eman Abdel-Moemen Mohammed,1 Soha Younes,4 Naglaa Elsayed Hassan,5 Theresa C Brown,6 Eckhard U Alt,7 James E Robinson,8 Fouad Mohamed Badr,1 Stephen E Braun2,9
1Genetics Unit, Department of Histology and Cell Biology, School of Medicine, Suez Canal University, Ismailia, Egypt; 2Division of Immunology, Tulane National Primate Research Center, Covington, LA, USA; 3Department of Medical Microbiology, Faculty of Medicine, University of Tabuk, Tabuk, Kingdom of Saudi Arabia; 4Department of Clinical pathology, Faculty of Medicine, Suez Canal University, Ismailia, Egypt; 5Women’s Hospital, Hamad Medical Corporation, Doha, Qatar; 6Hayward Genetics Center, Tulane University School of Medicine, New Orleans, LA, USA; 7Applied Stem Cell Laboratory, Departments of Medicine, Tulane University School of Medicine, New Orleans, LA, USA; 8Sections of Infectious Disease, Departments of Pediatrics and Internal Medicine, Tulane University School of Medicine, New Orleans, LA, USA; 9Departments of Pharmacology, Tulane University School of Medicine, New Orleans, LA, USA
Correspondence: Raghda E Eldesouki Email [email protected]
Introduction: Quiescent leukemia stem cells (LSCs) play a major role in therapeutic resistance and disease progression of chronic myeloid leukemia (CML). LSCs belong to the primitive population; CD34+CD38-Lin-, which does not distinguish normal hematopoietic stem cells (HSC) from CML LSCs. Because Thomsen–Friedenreich/CD176 antigen is expressed on CD34+ HSC and IL1RAP is tightly correlated to BCR-ABL expression, we sought to increase the specificity towards LSC by using additional biomarkers.
Methods: We evaluated the co-expression of both antigens on CD34+ peripheral blood mononuclear cells (PBMCs) from both healthy volunteers and CML patients, using flow cytometry. Then, we used site-directed mutagenesis to induce knob-in-hole mutations in the human IgG heavy chain and the human lambda light chain to generate the bi-specific antibody (Bis-Ab) TF/RAP that binds both antigens simultaneously. We measured complement-directed cytotoxicity (CDC) in CML samples with the Bis-Ab by flow cytometry.
Results: In contrast to healthy volunteers, CML samples displayed a highly significant co-expression of CD176 and IL1RAP. When either a double-positive cell line or CML samples were treated with increasing doses of Bis-Ab, increased binding and CDC was observed indicating co-operative binding of the Bis-Ab as compared to monoclonal antibodies.
Discussion: These results show that the bi-specific antibody is capable of targeting IL1RAP+ and CD176+ cell population among CML PBMCs, but not corresponding normal cells in CDC assay. We hereby offer a novel strategy for the depletion of CML stem cells from the bulk population in clinical hematopoietic stem cell transplantation.
Keywords: TF antigen, Thomsen–Friedenreich/CD176 antigen, IL1RAP, chronic myeloid leukemia, bi-specific antibodies, complement-dependent cell cytotoxicity, CDC
Introduction
Chronic myeloid leukemia (CML) is a hematological malignancy that develops when the 9;22 translocation in a single hematopoietic stem cell (HSC) results in the expression of BCR-ABL1 tyrosine kinase fusion protein. If left untreated, CML progresses over approximately 5 years, from relatively benign chronic phase to accelerated phase, and then to fatal blast crisis. The introduction of tyrosine kinase inhibitors (TKIs) specifically targeting the BCR-ABL1 fusion protein was a breakthrough in the management of CML, leading to a significant reduction in mortality and improved 5-year survival rates. However, despite the high annual acquisition costs of all the TKIs; first-, second-, and-third line TKIs1 induce only transient responses in the 10% to 15% of CML patients diagnosed in advanced phase, suboptimal responses in approximately 30% of CML patients during chronic phase (CP) cases that experience disease progression each year during, and only 10–20% chance of successful treatment discontinuation due to disease persistence.2 Among the causes of disease persistence, studies have shown that CML leukemia stem cells (LSC) play a major role in inducing therapeutic resistance and disease progression because they are able to self-renew.3,4 These LSC – a rare subset of immature cells residing in the bone marrow niche – are protected from the action of TKI5 because these cells are normally quiescent and the TKIs are designed to target malignant blast cells that proliferate. That is why current strategies are not able to effectively eliminate the LSC or the disease.3 In CML, LSC are primitive cells expressing CD34+ CD38- with the 9;22 translocations, or the Philadelphia chromosome (Ph).6 However, these markers cannot distinguish the cancer hematopoietic cells from normal ones. Additionally, the BCR-ABL fusion gene encodes for an intracellular tyrosine kinase protein rather than a surface protein, calling for the need to identify unique surface biomarkers for efficient targeting of this cell population with subsequent eradication of the root of the disease.
In 2010, a single biomarker, Interleukin 1 receptor accessory protein (IL1RAP), was found to be up-regulated on the cell surface of BCR-ABL+ LSC. They were able to distinguish Ph+ from Ph- LSCs using IL1RAP.7 A polyclonal anti-human IL1RAP was generated that not only targeted the LSC population but also killed normal peripheral blood mononuclear cells, indicating that this marker was not specific to the LSC.7 Another characteristic cell surface marker has been investigated; Thomsen–Friedenreich antigen (TF, or CD176) a tumor-associated carbohydrate epitope. The CD176 antigen was found to be expressed on the surface of various cancer-initiating cells, such as breast carcinomas,8 colorectal carcinomas,9 several leukemias,10 and other types of cancer, but was absent from almost all normal adult cell types.11 CD176 was also found to be expressed on the surface of CD34+ hematopoietic stem cells of the K562 erythroblastic leukemia cell line; a cell line derived from a CML patient. Being strongly expressed on the surface of cancer cells and virtually absent from normal tissues, CD176 was evaluated as a suitable target for cancer biotherapy8 with the development of an anti-CD176 antibody that induced apoptosis of leukemic cells.12
Using monoclonal antibodies (mAb) as a tool for cancer therapy still has its limitations. Patients who receive mAb therapy may develop drug resistance or fail to respond to treatment owing to the multiple signaling pathways involved in the pathogenesis of cancer and other diseases.13 Targeting more than one molecule has proven to circumvent the regulation of parallel pathways and avoid resistance to the treatment.14 Bi-specific antibodies (Bis-Ab) are antibodies that can recognize two different epitopes. They can redirect specific immune cells to the tumor cells to enhance tumor eradication, enable the simultaneous blocking of two different targets that have common signaling pathways, or interact with two different cell-surface antigens instead of one with subsequent boosting of the binding specificity.13 Thus, the identification of two surface markers specific to the cancer stem cells would be useful in characterizing and targeting CML stem cells, without affecting other blood cells.
In this study, we evaluated co-expression of IL1RAP, linked to BCR-ABL+ expression, and the CD176 antigen, carried on the hematopoietic stem cell marker CD34 molecule, in CML patients. We identified PBMCs co-expressing CD34, IL1RAP, and CD176 antigens using flow cytometry, a finding that allowed for subsequent separation and targeting of such cells from normal HSCs. A bi-specific antibody (TF/RAP), was generated in order to target the IL1RAP+ and CD176+ cell population among PBMCs in patients with CML. We used a flow-cytometry assay as a cell-based assay to measure the antibody binding capability of the TF/RAP Bis-Ab to the cell surface antigens. Our TF/RAP Bis-Ab, increased targeting of the IL1RAP+ and CD176+ cell population among CML PBMCs but not corresponding normal cells, using complement-dependent cytotoxicity assay (CDC). This novel TF/RAP Bis-Ab may provide a novel strategy for the eradication of CML stem cells.
Materials and Methods
Collection of CML Patient Samples
Deidentified samples of peripheral blood from healthy volunteers were obtained from Gulf Coast Regional Blood Bank (Houston, TX, USA) after signing informed consent and used as reference samples. Deidentified samples of peripheral blood mononuclear cells (PBMCs) from consented patients with CML were obtained from Oncology Research Gundersen BioBank (https://www.gundersenhealth.org/research/biobank/, La Crosse, WI, USA). While the samples were de-identified, necessary CML patient characteristics were collected (Table 1). The collection and dissemination protocols for the samples are approved by The Gundersen Human Subjects Committee/Institutional Review Board (IRB) and are in full compliance with National Cancer Institute Best Practices for Biospecimen Resources. Because the de-identified samples were received through Biobanks and not through direct intervention/interaction with a research subject, the Tulane University Human Research Protection Office was notified and this study was classified by the IRB as exempt as the study did not meet the definition of human subjects research according to US Federal policy (HHS regulations, 45 CFR part 46, subpart A, also known as the “Common Rule”). The study was conducted in accordance with the Declaration of Helsinki.
 |
Table 1 CML Patients’ Characteristics |
Cell Lines
HEK 293FT cell line (Invitrogen # R70007) was cultured in DMEM (Life Technologies, Carlsbad, CA, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin, 100 μg/mL streptomycin sulfate, and 4.0 mM L-glutamine (Gibco BRL products, Gaithersburg, MD), at 37°C in a humidified 5% CO2 incubator. The KG1 cell line (ATCC #CCL-246) and transduced derivative cells were cultured in Iscove’s Modified Dulbecco’s Medium (Life technologies) supplemented with 20% FBS at 37°C in a humidified 5% CO2 incubator. K562 cell line (ATCC# CCL-243) was maintained in RPMI-1640 (Life technologies) supplemented with 10% FBS, 100 U/mL penicillin, 100 μg/mL streptomycin sulfate at 37°C in a humidified 5% CO2 incubator.
Lentiviral-Induced IL1RAP Expression in KG1 Cell Line
Construction of pHRST_IL1RAP Lentiviral Vector
The IL1RAP cDNA was PCR amplified from an expression plasmid containing Human IL-1RAcP/IL-1R3 Gene ORF cDNA (Sino biological Inc., HG10121-CM) using Clone Amp HiFi PCR Premix (Takara Bio USA, Inc.), and primers that included either a BamHI or an XhoI site (F-IL1RAP: acgggatccccaccaagcttggtaccatgac; R-IL1RAP: acgctcgagttatacatttttcaaagatg). The PCR fragment was gel extracted as above, sub-cloned into BamHI and XhoI sites in the pHRST-MPSV vector according to standard protocols and confirmed by restriction mapping and sequencing.
Viral Production (HEK Transfection)
Transient production of lentiviral particles in adherent HEK293T was modified from previously described.15 Briefly, HEK293T cells were seeded in a T-75 flask, where we used 4.0 μg of envelope plasmid pMPSV-VSV-G, 10.0 μg packaging plasmid psPAX2, and 26 μg transfer plasmid that has the gene of interest. In our case, the transfer plasmid is either the antibody plasmid or the control. The plasmids were mixed into 500 μL 0.25 M CaCl2 (Sigma Aldrich, St. Louis, MO) and incubated at room temperature for 5 minutes, and then mixed with 500 μL 2xHBS and briefly vortexed. The mixed transfection cocktail was then incubated for 3 minutes at room temperature, and added into the medium of the cells, and mixed gently to make an even distribution. After 16 hours of incubation, the medium was replaced with fresh medium and collected every 24 hours for 3 days. The conditioned medium that contained the vector virus was then pelleted for 10 minutes at 1500 g and passed through a 0.45-μm filter to remove the cell debris, and then frozen at −80°C for long-term storage, or used for the transduction of target cells.
KG1 Lentiviral Transduction with HRST_IL1RAP
Lentiviral transduction was done as previously described.16–18 In brief, lentiviral supernatant was added to KG1 cells cultured in complete IMEM. After overnight incubation, the lentiviral vector was removed, and fresh media was added. After 48 hours, IL1RAP expression was demonstrated by flow cytometry using anti-Human IL-1 RAcP/IL-1 R3 PE-conjugated antibody (#FAB676P, R&D Systems, Minneapolis, MN).
Generation of TF/RAP; the Bi-Specific Anti-Human CD176/IL1RAP Antibody
Generation of Mutated Constant Domains
The CH and CL constant domains in the pLM219 plasmids were amplified with 0.5 nM overlapping mutant primers (Table S1), Deep Vent Polymerase (New England Biolabs), and reaction buffer for forty cycles at 94°C for 10 seconds, 60°C for 45 seconds, and 72°C for 2 minutes. Initial fragments were purified, combined, and used to amplify the entire heavy or light domains (Table S2). The mutated fragments were then gel purified and sub-cloned into their corresponding vectors using restriction enzymes according to standard protocols (Table S2). Sequences were then verified by restriction digestion and sequencing.
Synthesis of Variable Domains
For antibody sequences towards CD176 (TF) and IL1RAP, the VH and VL domains from two clones with the most conserved amino acid sequences (TF Clone 1 and Clone 2 – called TF1 and TF2 for CD176; Clone 4B6 and Clone 4G9 – called RAPa and RAPb for IL1RAP, respectively) were chosen from published sequences.20,21 IL1RAP antibody was designed to target the extracellular membrane anchor-proximal region that comprises an amino acid primary sequence VPAPRYTVELAC within 10 to 15 amino acids of amino acid 361 of human ILR1AP (Gene bank accession Q9NPH3) while the TF antibody was designed to target the same Gal(1–3)GalNAc disaccharide epitope20 as the Bis-Ab. Variable domains (VD) were codon-optimized and synthesized (Gene Art, Invitrogen) to be compatible with 15 base pairs of homologous sequences on both the 3ʹ and 5ʹ ends of pLM2 recipient plasmid flanking the EcoRI restriction enzyme site.
Infusion Cloning of Variable Domains into Mutated Vectors
The pLM2 expression vector was digested with EcoRI to generate a double-stranded break. An In-Fusion HD cloning kit (Clontech, Inc) was used to clone the VD regions of the antibodies between the leader and constant regions of the pLM2 vectors. The correct clones were identified by PCR and restriction mapping and then verified by sequencing.
Transient Transfection and Bi-Specific Antibody Production in HEK293T Cells
Adherent HEK cells were transfected as above. A total of 14 μg high-quality plasmid-DNA, 10% GFP plasmid for assessment of transfection efficiency, while the rest was heavy and light chain plasmid DNA combined at a ratio of 1:1. Six to 8 hours later, cells were gently washed once with PBS and fresh growth medium added. Sixteen hours post-transfection, the medium was replaced with DMEM supplemented with 5% FCS and incubated at 5% CO2 for 24 hours prior to the initial collection of antibody supernatant. A second collection was made after a further 24 hours.
Flow Cytometry-Based Assay
Flow antibodies used were as follows: anti-TF/CD176 mAb mouse IgM (Glycotope, Berlin, Germany) targeting Gal1-3GalNAc epitope; FITC-conjugated anti-mouse IgM secondary antibody (μ-chain specific, #F9259; Sigma); PE-conjugated mouse anti-human IL-1 RAcP/IL-1 R3 monoclonal IgG1 antibody, epitope Ser21-Glu359 (#FAB676P, R&D Systems); APC-conjugated mouse anti-human CD34 monoclonal IgG1 antibody (#QBEnd10, FAB7227A-025, R&D Systems); APC-conjugated mouse antihuman IgG monoclonal antibody (Clone G18-145, mouse IgG1 κ, #550,931, BD Pharmingen™).
LIVE/DEAD™ Fixable Aqua Dead Cell Stain Kit (#L34957, Invitrogen); Vibrio Cholera Neuraminidase (VCN; Sigma Aldrich Inc), an enzyme used to expose the CD176 on the surface of expressing cells. Flow cytometric analyses were performed in a BD LSR Fortessa™ (BD Biosciences, USA) and flow cytometric cell sorting was done in a FACSAriaII (P0010) cell sorter (BD Biosciences, USA). The amount of bi-specific antibody bound to the receptors was calculated from the frequency of total IgG bound receptors.
Fluorescence in-situ Hybridization (FISH) Analysis
Sorted cells were received in RPMI media and then fixed using the standard 3:1 methanol: acetic acid fixative. Standard procedures were used for FISH hybridization and washing.22 The BCR/ABL1 Plus translocation, dual fusion probe set (Cytocell Inc., Tarrytown, NY) was used. Slides were analyzed using Leica Biosystems Cyto Vision©. FISH nomenclature was described according to the ISCN 2016.23
Colony-Forming Unit (CFU) Assay
CD34+CD176+IL1RAP+ and CD34+CD176+IL1RAP- cells were sorted from PBMC samples derived from patients with CML. Cells (1 x 103) were plated in Metho Cult™ Express (#04437, Stem Cell Technologies, Vancouver, Canada) semi-solid media containing recombinant human IL-3, IL-6, G-CSF, GM-CSF, SCF, TPO and cultured for 2 weeks in a humidified atmosphere at 37°C with 5% CO2. Fourteen days after plating, the number of colonies was counted by microscopy.24,25
Complement-Dependent Cytotoxicity (CDC)
The capacity to induce CDC was assessed essentially as has been described.26–28 Briefly, target cells (1×105 cells) were pre-incubated at 37°C for 60 min with diluted antibodies. Human serum from human male AB (Sigma Aldrich) (20% v/v) was added to the cells as a source of complement and incubated at 37°C for an additional 45 min. Cells were then put on ice and viability was determined by staining with LIVE/DEAD™ staining and detected using a FORTESSA flow cytometer (BD Biosciences). CDC activity was expressed as a percentage of lyses as determined from the increase in the percentage of cells stained positive with the LIVE/DEAD™ marker compared to the control samples. Cycloviolacin O2 (CyO2, 0.05nM), a pore-forming peptide, was used as a positive control because it kills cells with the similar mechanisms as CDC by causing pores in the cell membrane.
The capacity to induce CDC was assessed essentially as has been described.26–28 Briefly, target cells (1×105 cells) were pre-incubated at 37°C for 60 min with diluted antibodies. Human serum from human male AB (Sigma Aldrich) (20% (v/v)) was added to the cells as a source of complement and incubated at 37°C for an additional 45 min. Cells were then put on ice and viability was determined by staining with LIVE/DEAD™ staining and detected using a FORTESSA flow cytometer (BD Biosciences). CDC activity was expressed as a percentage of lyses as determined from the increase in the percentage of cells stained positive with the LIVE/DEAD™ marker compared to the control samples. Cycloviolacin O2 (CyO2, 0.05nM), a pore-forming peptide, was used as a positive control because it kills cells with the similar mechanisms as CDC by causing pores in the cell membrane.
Enzyme-Linked Immunosorbent Assay (ELISA)
We measured the production of the Bis-Ab by ELISA. Plates were initially coated with goat anti-Human IgG heavy chain antibody (Axell) and blocked with PBS containing 0.5% Tween 20 (Fisher), 10% FBS (FetalPlex™ Animal Serum Complex, GeminiBio, Cat#100-602), 4% whey protein (BiPRO, AGROPUR). Undiluted or diluted supernatant was added, including the standard curve samples (human IgG MAb 1.7B, kindly provided by Dr. James Robinson), and negative blocking buffer. After incubating at 37°C for 60 min, the plates were washed. Then, goat anti-Human lambda antibody conjugated to HRP (Southern Biotech, Cat# 2070–05) was added at 1:300 in blocking buffer for 60 min and washed five times. A mixture of 0.1M Na Acetate (pH 6), peroxide, and TMB substrate were added. The reaction was terminated by adding 1M phosphoric acid, and the absorbance of each well was measured at 450 nm using a Synergy H1 microplate reader (BioTek).
Statistics
For each experiment, more than three independent replicates were conducted, and the results were expressed as average ± standard deviation. Comparison of multiple groups was conducted using ANOVA-based Test and p< 0.05 (*) represented significances with statistical meaning. Calculation of the Kd was done using the equation % RO = [Ab]/([Ab]+Kd) × 100%, where RO is the receptor occupancy, Ab is the concentration of antibody and Kd is the equilibrium dissociation constant.
Results
CD176 and IL1RAP Antigens are Co-Expressed on CD34+leukemia Cells
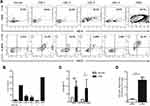
In order to analyze the co-expression of CD176 and IL1RAP antigens on CD34+ cells, peripheral blood mononuclear cells from a normal volunteer (NPBMCs), patients with CML, and K562 cells were isolated and stained with anti-CD34, anti-CD176, and anti-IL1RAP monoclonal antibodies and analyzed by flow cytometry (Figure 1A). It has been previously established that these markers were not expressed on normal PBMCs nor on stem cells7,10 CD34+ cell expression ranged from an average 9–38% in CML samples versus 83.7% in K562 cells (Figure 1A, upper panel). Within the CD34+ cell population, CD176 and IL1RAP antigens were variably expressed in CML samples, ranging from 1.35% in CML-4 to over 50% in CML-1 (Figure 1A, lower panel), while CD176+ IL1RAP+ was detected in 78% of CD34 cells in K562 cells. Surprisingly, surface co-expression of CD176 and IL1RAP was not only detectable on CD34+ cells in patients with BCR-ABL positive CML but was also demonstrable in cells from a treated patient who was BCR-ABL negative (CML-2) (Figure 1B). In Figure 1C, CD34+ cells revealed higher frequency of CD176+ IL1RAP+ in CML group compared to control sample (17.5% versus 3.4%, p<0.001).
CD34+CD176+IL1RAP+ Cell Population are Colony-Forming Cells
In order to analyze the progenitor activity of the various subpopulations, CML-2 and CML-4 were flow-sorted for CD34+CD176+IL1RAP+ and CD34+CD176+IL1RAP- then plated in media t support hematopoietic colony formation. The number of colonies, or colony-forming units (CFU), in CD34+CD176+IL1RAP+ pool represented 6% of the sorted cells with a significant difference between both populations, p<0.01 (Figure 1D and Figure S1).
Generation of the TF/RAP Bis-Ab Mixtures
To facilitate correct interaction of the VH and VL domains, site-directed mutagenesis was used to generate knob-in-hole mutations in the heavy and light chains of the constant domains (Figure 2A) via polymerase chain reaction overlap extension (Figures S2 and 3). Two PCR reactions were performed to generate two amplicons with the specific mutations included in the overlapping primers. The two fragments were then combined in a subsequent “fusion” reaction, in which the overlapping ends anneal, allowing the 3ʹ overlap of each strand to serve as a primer for the 3ʹ extension of the complementary strand. The resulting fusion product served as a template for amplification of the entire constant domain. In order to circumvent the light chain mismatching, an Orthogonal Fab interface was generated. In one Fab, complementary mutation was introduced and verified at the heavy chain constant domain (CH1_H172A_ F174G) and at the light chain constant domain (CL_L135Y_S176W), respectively (Figures S4–6). For the heavy chain heterodimerization, we used the “Knob-in-Hole” strategy, where we inserted the CH3 mutations (S354C and T366W) into different heavy chains (Figures S7 and 8). The VH and VL sequences were synthesized and cloned into the new pLM2-CH and -CL plasmids (Figure 2A) where CD176 was represented by TF1 (VH1 and VL1) and TF2 (VH2 and VL2) while IL1RAP was represented by Clone 4B6 (VHa and VLa) and Clone 4G9 (VHb and VLb). Then, we generated the four different bi-specific antibody mixtures (TF1RAPa, TF1RAPb, TF2RAPa, and TF2RAPb) to evaluate the most effective Bis-Ab (Figure 2B). The bispecific antibody was quantified by ELISA at 283 ng/mL. Since ELISA used the human IgG heavy chain antibody as the primary antibody and a goat anti-human lambda antibody conjugated to HRP as the secondary antibody, these data also confirm the correct association of the heavy and light chains and ensure that monomers are excluded.
IL1RAP is Induced as a Consequence of Lentiviral Expression in KG1 Cells
KG1 cell line is an acute myeloid leukemia cell line that is known to be a positive control for CD176. For optimizing the staining protocol of CD176, KG1 cells were pre-treated with VCN to expose CD176 antigens for better staining (Figure S9). In order to test the binding capability and functional potential of our bi-specific antibody, we generated a dual-positive cell line for expressing both IL1RAP and CD176 through lentiviral transduction (Figure S10A and B). IL1RAP expression was increased by 1.5 folds in KG1/RAP cells as verified by flow cytometry (Figure S10C and D).
Bi-Specific Antibody Validation in a Dual-Positive Cell Line
CD176 antigen is a glycosylated antigen; a protein antigen bound to GAL-NAC moiety which makes the antigen displayed on the cell surface yet not easy to isolate.21 For this reason, a flow-cytometry assay was used to evaluate both the binding capability and toxicity of our Bis-Ab using the gating strategy in Figure S11. KG1 and KG1/RAP cell lines were treated with the various Bis-Ab mixtures. Binding percentage was calculated from the percentage of IgG positive cells, where the secondary IgG antibody is bound to the primary Bis-Ab. The TF1RAPa Bis-Ab showed the highest binding in KG1/RAP cells (Figure 3A) as compared to other mixtures (p<0.001). In contrast, the TF1RAPb antibody revealed slightly reduced binding in KG1/RAP cells. On treating KG1/RAP cells with increasing amounts of TF1RAPa, more binding to the dual-positive KG1/RAP cells was observed (Figure 3B). To demonstrate the specificity of the Bis-Ab, we measured the competition with the CD176 and the IL1RAP monoclonal antibodies. Increasing concentrations of the Bis-Ab specifically inhibited the binding of both the IL1RAP and CD176 mAbs (Figure S12). Then, our KG1/RAP cells were treated with the Bis-Ab TF1RAPa and complement prior to staining with the LIVE/DEAD™ Fixable Aqua Dead Cell Stain Kit, in order to evaluate whether CDC could be achieved using IL1RAP and CD176 as targets. Flow cytometric analysis revealed a significant increase in dead cells in the Bis-Ab treated CD176/IL1RAP dual-positive KG1/RAP population as antibody binding also increased (Figure 3C), p<0.001.
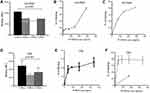 |
Figure 3 Validation of TF-RAP Bi-specific antibody in KG1 cell line and CML samples. (A) MFI for binding of different Bis-Ab mixtures in KG1/RAP (p <0.001). (B) Binding (%) of the Bis-Ab in KG1/RAP cell lines. (C) Shows live/dead (LD) staining (%) in KG1/RAP cell lines after treatment with the Bis-Ab and complement. (D) MFI for binding of different Bis-Ab mixtures p <0.001 in CML cells. (E) Binding of the Bis-Ab (%) in PBMCs from patients with CML. The binding affinity (Kd) of our bispecific antibody was 21 ng/mL, calculated using the % RO = [Ab]/([Ab]+Kd) × 100%, where RO is the receptor occupancy, Ab is the concentration of antibody, and Kd is the equilibrium dissociation constant. This Bis-Ab platform used in this study had the correct molecular weight (95 KDa) and assembled properly (93%) as revealed by SDS-PAGE analysis.38 (F) Live/dead (L/D) staining (%) from patients with CML after treatment with the Bis-Ab and complement. The red square were L/D positive cells treated with CyO2; the percent of L/D staining in normal PBMCs is shown in blue. Each point represents the mean increase in L/D staining ± SEM with three to four replicates. Data from normal samples were low for all doses (data not shown). |
Bi-Specific Antibody Testing in CML Samples
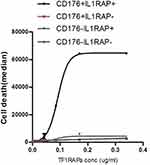
Binding of TF1RAPa, TF2RAPa, and TF2RAPb was also tested in PBMCs from patients with CML. Again, TF1RAPa showed the highest binding relative to other mixtures (p<0.001) (Figure 3D) and with increasing doses (Figure 3E). Based on the CML binding curve, the binding affinity (Kd) of our bispecific antibody was 21 ng/mL. Other therapeutic antibodies, such as ofatumumab directed against CD20, have shown significant CDC against peripheral blood cells obtained from CML patients in chronic phases26 and B cells in CLL,29 respectively. Thus, the TF1RAPa cocktail was used to generate the dose–response curve and to evaluate whether CDC could be achieved using both IL1RAP and CD176 as targets. The ability of the TF1RAPa cocktail was compared to human anti-IL1RAP and anti-CD176 monoclonal antibodies to induce cell death in PBMCs from patients with CML. PBMCs from CML1-4 were tested in CDC assays in parallel to cells from healthy control samples. In CML cells, the binding of TF1RAPa mediated CDC at higher levels than in normal peripheral blood mononuclear control cells, correlating with the expression level of IL1RAP and CD176, particularly at lower antibody concentrations (Figure 3F). More strikingly, among peripheral blood cells, TF1RAPa did not induce CDC of normal cells, whereas a clear dose-dependent CDC effect was observed in CML cells (Figure S13A and B). To address the selectivity of IL1RAP/CD176-targeting antibodies, we also validated the bispecific antibody cytotoxicity on the various subpopulations in peripheral blood. The dual-positive CD176+IL1RAP+ cell populations showed the highest CDC activity as compared to CD176+IL1RAP-, CD176-IL1RAP+, and CD176-IL1RAP- populations (Figure 4 and S13C–F, S14).
Discussion
Targeting molecules involved in multiple pathways is proving to be one of the most reliable strategies for eradicating cancer stem cells. In this report, we present a novel bi-specific antibody, TF/RAP, capable of targeting Thomsen–Friedenreich (TF, CD176) and IL1RAP antigens on CD34+ HSCs in CML and on cell lines. TF is a glycoprotein that has many domains and motifs (eg, LGALS3, Galβ(1,3)GalNAc, LGalS3BP), many related to signaling pathways. It is a known marker for ongoing tumorigenesis and metastasis, as it is expressed on various cancer-initiating cells.8 Interestingly, CD34 and LGALS3 were found to be co-expressed in myeloid cells.30,31 LGALS3 and ABL1 are involved in regulating RUNX1 and the transcription of genes involved in differentiation of hematopoietic stem cells,32 especially myeloid cells33 (Figure S15) IL1RAP, on the other hand, is a member of the Toll-like receptor superfamily and is a well-known co-receptor of IL−1R1.34 IL1RAP plays a role in mediating the effect of the pro-inflammatory cytokine IL-1 and is also involved in activating T cells and mast cells after mediating the signal of IL-1 cytokine.35 It has previously been characterized as a tightly related marker for BCR-ABL positive cells.7 Together, both TF and IL1RAP were related to apoptotic pathways; IL1RAP up-regulation was associated with decreased apoptosis in AML,36 and anti-CD176 antibody induced apoptosis of CD176-positive leukemic cells through multiple pathways.12 Although we did not find a direct link between IL1RAP, CD176 and leukemogenesis, previous studies have shown that each of them is separately expressed on CD34+ cells in leukemia cell lines8,10,12 and patients with CML7
Therefore, we conducted this pilot study, in order to assess the co-expression of IL1RAP and Thomsen–Friedenreich (CD176) antigens on CD34+ HSCs in peripheral blood of patients with CML, using FACS gene expression analyses. Flow-drop FISH and CFU assays were used for the separation of CD34+CD176 BCR-ABL+ and BCR-ABL− CML stem cells, based on IL1RAP expression.7 CFU numbers were significantly lower in CD34+CD176+IL1RAP- cells than in CD34+CD176+IL1RAP+ cells, obtained from CML-2 and CML-4 samples (Figure 1D), particularly CML-2 sample which was obtained from a patient in remission (BCR-ABL-). We found that the frequency of clonogenic hematopoietic progenitor cells was increased in the CD34+ CD176+IL1RAP+ cells in these samples. Testing the stem-cell characteristics of these two cell populations in immune-deficient mice would have been advantageous. Yet, the low numbers of sorted CML cells acquired from the CD34+CD176+ IL1RAP− and IL1RAP+ cell subpopulations, along with the general low engrafting efficiency of chronic phase CML cells in these mice7 prevented us from successfully performing such experiments. Importantly, as IL1RAP expression was correlated with changes from chronic phase (CP) into accelerated phase (AP) and blast phase (BP)37, we also found that the level of IL1RAP/CD176 co-expression was increased , in our patient samples, as the disease progressed, independent of the treatment status (Table S3).
To target both TF and IL1RAP simultaneously, we developed a Bis-Ab specific for both antigens. Because antibodies are normally heterodimers of two heavy and two light chains, we modified the constant domains in the Bis-Ab to maximize the correct interactions of the four immunoglobulin chains within single cells. Here, we used the orthogonal Fab design; CH1_H172A_F174G and CL_L135Y_S176W38 to facilitate selective assembly of the Fab arms for correct dimerization of the antigen-binding domains.39 Therefore, we mutated CH1 and CL binding sites to restrict the assembly of the Fab with the correct VD pairs. The RAP VDs were cloned with the wild type Fab; and the TF VD was linked to the mutant orthogonal Fab design. Published data have shown that the component proteins of this Bis-Ab platform proper assembly were detected at 93% and the complex had a molecular weight of 95 KDa, as revealed by SDS-PAGE analysis.38 Additionally, the CH3 for each Fab was mutated with previously described knob-into-hole mutations40,41 to facilitate hetero-dimerization between the TF and the RAP heavy chains. In our study, we used ELISA to demonstrate that both the VD and Fc were properly paired. Here, because the primary antibody was anti-human VL and the secondary antibody was anti-human IgG, quantifying the Bis-Ab also demonstrated the VD-Fc interactions.
To efficiently validate the specific binding of our Bis-Ab, we generated a dual-positive cell line; KG1/RAP. KG1 cell line expresses CD176+, but IL1RAP is low or absent. Therefore, we induced IL1RAP expression in KG1 cells by lentiviral mediated-gene transfer, as previously used in both immune42 and leukemic cells.43 In the competitive binding assay, increasing concentrations of the Bis-Ab blocked the binding of CD176 and IL1-RAP monoclonal antibodies to the KG1/RAP and KG1 parental cells, demonstrating the specific binding of the Bis-Ab. The level of CD176 expression in KG1 cell line was detected before and after VCN treatment. Increased staining of the KG1/RAP cells compared to the parental KG1 cells indicated that expression of the IL1RAP facilitates the interaction of the Bis-Ab with the target cell. This increased binding of the Bis-Ab to the KG1/RAP cells also increased their susceptibility to complement-dependent cytotoxicity (CDC). We also observed increased binding and increased CDC in the CD176+ IL1RAP+ population of the peripheral blood from patients with CML. As a pilot study and given that on average, 50% of the cells within the CD34+ subpopulation in the patients tested were dual positive for CD176 and IL1RAP antigens, in addition to the almost undetectable CDC in CD34+ cells in normal controls, our data strongly support the idea that the bi-specific antibody (TF/RAP) indeed induces CDC preferentially in CD176+ IL1RAP+ CML CD34+ cells. In generating a bi-specific antibody that targets CD176 and IL1RAP, we are unique in providing proof of concept that CML CD34+CD176+ IL1RAP+ cells can be targeted while preserving corresponding normal cells. The potential to target multiple antigens is supported by studies that demonstrated increased or synergistic CDC activity by non-cross blocking CD20 antibody combinations.44
Therapeutic antibodies are commonly administered intravenously, yet selectivity and specificity are a major concern for reduced toxicity. CD176/IL1RAP co-expression was not present in monocytes unlike the reported weak but present IL1RAP expression in monocytes.7 Both antigens were low or absent in most types of normal bone-marrow progenitor and mature cell types, suggesting that CD176/IL1RAP dual targeting antibodies are expected to show low toxicity on normal hematopoietic cells. Being strongly expressed on the surface of cancer cells and virtually absent from normal tissues, CD176 was evaluated as a potential target for cancer biotherapy with the development of anti-CD176 antibody that induced apoptosis of leukemic cells.8 Added to this, antibodies against IL1RAP were found to be capable of blocking IL-1 signaling as well as inhibiting tumor cells' growth in AML,34 CML,7 breast cancer,45 prostate cancer, breast cancer, lung cancer, colorectal cancer, melanomas, bladder cancer, brain/CNS cancer, cervical cancer, esophageal cancer, gastric cancer, head/neck cancer, kidney cancer, liver cancer, lymphomas, ovarian cancer, pancreatic cancer, and sarcomas46 especially in cancer stem cells, or (CSCs) and progenitor cells, which are responsible, directly or indirectly, for the development of a solid tumor.47 Thus, it may be that our Bis-Ab will not only eradicate the CD176+IL1RAP+ drug-resistant CML stem cells but also may have universal therapeutic potential for preventing relapses in both solid and hematological cancers.Given that the mode of action in CDC is having the antibody direct the complement pathway to target cell killing, we suggest that this therapeutic strategy would be independent of known mechanisms of TKI resistance in CML. Thus, the concept of complement-mediated killing of IL1RAP/CD176 expressing cells may also have the potential to eradicate such cells in patients, either alone or in combination with current regimens, in order to increase their therapeutic effectiveness. And finally, expanded studies need to be performed in order to confirm the co-expression of both markers, especially in resistant and relapsed cancer patients as well as in patient-derived xenografts (PDX).
Acknowledgments
The experimental research was mostly supported by a fellowship to REE from the Egyptian Ministry of Higher Education, Cultural, and Missions Section (JS 3577). The lentiviral vector HRST-cmvGFP and the packaging plasmids were a kind gift from Richard C. Mulligan in the Harvard Gene Therapy Institute. The human IgG heavy and light chain constant genes were provided by JE Robinson (Tulane University). C Wu and SEB were supported by AI110158 and/or OD01104-51; EUA and SEB were supported by the Applied Stem Cell Laboratory.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. All authors have given approval of the final version of the article; and have agreed to be accountable for all aspects of the work.
Disclosure
The abstract of this paper was presented at the AACR annual Meeting 2019; March 29 April3, 2019; Atlanta, GA, as a poster presentation with interim findings. The poster’s abstract was published in ‘Poster Abstracts’ in the AACR meeting proceedings and as a supplement in the AACR Cancer Research Journal [https://cancerres.aacrjournals.org/content/79/13_Supplement/1222A].
Raghda Eldesouki reports grants from Egyptian Ministry of Higher Education. Stephen E Braun reports grants from Egyptian Ministry of Education, Alliance for Cardiovascular Research, NIAID OD01104, and Braun/McGroarty Charitable Fund, during the conduct of the study. In addition, Dr Raghda Eldesouki, Dr Stephen Braun, Dr Fouad Badr and Dr Eman Abdel-Moemen Mohammed have a patent, PCT/EG2019/000014, pending. The authors report no other conflicts of interest in this work.
References
1. Marchetti M. Cost-effectiveness of kinase inhibitors for hematologic malignancies: a systematic and critical review. Expert Rev Pharmacoecon Outcomes Res. 2017;17(5):469–480. doi:10.1080/14737167.2017.1366858
2. Holyoake TL, Helgason GV. Do we need more drugs for chronic myeloid leukemia? Immunol Rev. 2015;263(1):106–123.
3. Zhou H, Xu R. Leukemia stem cells: the root of chronic myeloid leukemia. Protein Cell. 2015;6(6):403–412.
4. Holyoake TL, Vetrie D. The chronic myeloid leukemia stem cell: stemming the tide of persistence. Blood. 2017;129(12):1595–1606. doi:10.1182/blood-2016-09-696013
5. Koptyra M, Falinski R, Nowicki MO, et al. BCR/ABL kinase induces self-mutagenesis via reactive oxygen species to encode imatinib resistance. Blood. 2006;108(1):319–327. doi:10.1182/blood-2005-07-2815
6. Tasian SK, Bornhäuser M, Rutella S. Targeting leukemia stem cells in the bone marrow niche. Biomedicines. 2018;6(1):22. doi:10.3390/biomedicines6010022
7. Järås M, Johnels P, Hansen N, et al. Isolation and killing of candidate chronic myeloid leukemia stem cells by antibody targeting of IL-1 receptor accessory protein. Proc Natl Acad Sci U S A. 2010;107(37):16280–16285. doi:10.1073/pnas.1004408107
8. Goletz S, Cao Y, Danielczyk A, Ravn P, Schoeber U, Karsten U. Thomsen-Friedenreich antigen: the “hidden” tumor antigen. Adv Exp Med Biol. 2003;535:147–162.
9. Kurtenkov O, Innos K, Sergejev B, Klaamas K. The Thomsen-Friedenreich antigen-specific antibody signatures in patients with breast cancer. Bio Med Res Int. 2018;2018:9579828.
10. Sindrewicz P, Lian LY, Yu LG. Interaction of the onco-fetal Thomsen-Friedenreich antigen with galectins in cancer progression and metastasis. Front Oncol. 2016;6:79. doi:10.3389/fonc.2016.00079
11. Lin WM, Karsten U, Goletz S, Cheng RC, Cao Y. Expression of CD176 (Thomsen-Friedenreich antigen) on lung, breast, and liver cancer-initiating cells. Int J Exp Pathol. 2011;92(2):97–105. doi:10.1111/j.1365-2613.2010.00747.x
12. Yi B, Zhang M, Schwartz-Albiez R, Cao Y. Mechanisms of the apoptosis induced by CD176 antibody in human leukemic cells. Int J Oncol. 2011;38:1565–1573.
13. Fan G, Wang Z, Hao M, Li J. Bi-specific antibodies and their applications. J Hematol Oncol. 2015;8:130. doi:10.1186/s13045-015-0227-0
14. Varela MA. Identification of sequences common to more than one therapeutic target to treat complex diseases: simulating the high variance in sequence interactivity evolved to modulate robust phenotypes. BMC Genom. 2015;16(1):530. doi:10.1186/s12864-015-1727-6
15. Wu C, Lu Y. High-titre retroviral vector system for efficient gene delivery into human and mouse cells of hematopoietic and lymphocytic lineages. J Gen Virol. 2010;91(8):1909–1918. doi:10.1099/vir.0.020255-0
16. Ge D, Zhang QS, Zabaleta J, et al. Doublecortin may play a role in defining chondrocyte phenotype. Int J Mol Sci. 2014;15(4):6941–6960. doi:10.3390/ijms15046941
17. Braun SE, Wong FE, Connole M, et al. Inhibition of simian/human immunodeficiency virus replication in CD4+ T cells derived from lentiviral-transduced CD34+ hematopoietic cells. Mol Ther. 2005;12(6):1157–1167. doi:10.1016/j.ymthe.2005.07.698
18. Braun SE, Lu XV, Wong FE, et al. Potent inhibition of simian immunodeficiency virus (SIV) replication by an SIV-based lentiviral vector expressing antisense Env. Hum Gene Ther. 2007;18(7):653–664. doi:10.1089/hum.2007.003
19. Robinson JE, Hastie KM, Cross RW, et al. Most neutralizing human monoclonal antibodies target novel epitopes requiring both Lassa virus glycoprotein subunits. Nat Commun. 2016;7:11544. doi:10.1038/ncomms11544
20. Goltez S, Karasten U Cancer stem cell markers and uses thereof. WIPO, WO2011089004A1
21. Jiang Y, Tso J, Karsunky H. Antibodies that bind membrane-bound IL1rap. European patent EP2935334A1.
22. Keaglen MB, Gersen SL. Basic cytogenetics laboratory procedures. In: Gersen SL, Keagle MB, editors. The Principles of Clinical Cytogenetics. NewYork, NY: Springer NewYork; 2013:53–65.
23. International Standing Committee on Human Cytogenetic Nomenclature. ISCN2016: An International System for Human Cytogenetic Nomenclature. Karger Medical and Scientific Publishers; 2016.
24. Broxmeyer HE, Etienne-Julan M, Gotoh A, et al. Hematopoietic colony formation from human growth factor-dependent TF1 cells and human cord blood myeloid progenitor cells depends on SHP2 phosphatase function. Stem Cells Dev. 2013;22(6):998–1006. doi:10.1089/scd.2012.0478
25. Balduini A, Broxmeyer HE, Braun SE, Cornetta K, Lyman S. Comparative effects of retroviral mediated gene transfer into primary human stromal cells of flt3‐ligand, interleukin 3 and gm‐csf on production of cord blood progenitor cells in long‐term culture. Stem Cells. 1998;16:37–49. doi:10.1002/stem.5530160807
26. Tatake RJ, Maniar HS, Chiplunkar SV, et al. Antibody-dependent cellular cytotoxicity and complement-mediated cytotoxicity on leukemic cells mediated by anti K562 monoclonal antibodies. J Clin Lab Immunol. 1990;31(2):87–91.
27. Lindorfer MA, Beum PV, Taylor RP. CD20 mAb-mediated complement dependent cytotoxicity of tumor cells is enhanced by blocking the action of factor I. Antibodies. 2013;2:598–616. doi:10.3390/antib2040598
28. Gerlach SL, Chander PK, Roy U, et al. The membrane-active phytopeptide. Cycloviolacin O2 simultaneously targets HIV1-infected cells and infectious viral particles to potentiate the efficacy of antiretroviral drugs. Medicines (Basel). 2019;6(1):E33. doi:10.3390/medicines6010033
29. Zen CS, Secreto CR, LaPlant BR, et al. Direct and complement dependent cytotoxicity in CLL cells from patients with high-risk early-intermediate stage chronic lymphocytic leukemia (CLL) treated with alemtuzumab and rituximab. Leuk Res. 2008;32(12):1849–1856. doi:10.1016/j.leukres.2008.05.014
30. Marer N. Galectin‐3 expression in differentiating human myeloid cells. Cell Biol Int. 2000;24:245–251. doi:10.1006/cbir.1999.0501
31. Labbaye C, Testa U. The emerging role of MIR-146A in the control of hematopoiesis, immune function and cancer. J Hematol Oncol. 2012;5:13. doi:10.1186/1756-8722-5-13
32. Huang H, Woo AJ, Waldon Z, et al. A Src family kinase-Shp2 axis controls RUNX1 activity in megakaryocyte and T-lymphocyte differentiation. Genes Dev. 2012;26(14):1587–1601. doi:10.1101/gad.192054.112
33. Zhang HY, Jin L, Stilling GA, et al. RUNX1 and RUNX2 upregulate Galectin-3 expression in human pituitary tumors. Endocrine. 2009;35(1):101–111. doi:10.1007/s12020-008-9129-z
34. Ågerstam H, Karlsson C, Hansen N, et al. Antibodies targeting human IL1RAP (IL1R3) show therapeutic effects in xenograft models of acute myeloid leukemia. Proc Natl Acad Sci U S A. 2015;112(34):10786–10791.
35. McEntee CP, Finlay CM, Lavelle EC. Divergent roles for the IL-1 family in gastrointestinal homeostasis and inflammation. Front Immunol. 2019;10:1266.
36. Barreyro L, Will B, Bartholdy B, et al. Over expression of IL-1 receptor accessory protein in stem and progenitor cells and outcome correlation in AML and MDS. Blood. 2012;120(6):1290–1298. doi:10.1182/blood-2012-01-404699
37. Zhao K, Yin LL, Zhao DM, et al. IL1RAP as a surface marker for leukemia stem cells is related to a clinical phase of chronic myeloid leukemia patients. Int J Clin Exp Med. 2014;7(12):4787–4798.
38. Lewis SM, Wu X, Pustilnik A, et al. Generation of bi-specific IgG antibodies by structure-based design of an orthogonal Fab interface. Nat Biotechnol. 2014;32:191–198. doi:10.1038/nbt.2797
39. Spidel JL, Vaessen B, Chan YY, Grasso LJ, Kline B. Rapid high-throughput cloning and stable expression of antibodies in HEK293 cells. J Immunol Methods. 2016;439:50–58. doi:10.1016/j.jim.2016.09.007
40. Ridgway JB, Presta LG, Carter P. ‘Knobs-into-holes’ engineering of antibody CH3 domains for heavy chain heterodimerization. Protein Eng. 1996;9:617–621. doi:10.1093/protein/9.7.617
41. Atwell S, Ridgway JB, Wells JA, Carter P. Stable heterodimers from remodeling the domain interface of a homodimer using a phage display library. J Mol Biol. 1997;270:26–35. doi:10.1006/jmbi.1997.1116
42. Pan H, Mostoslavsky G, Eruslanov E, Kotton DN, Kramnik I. Dual-promoter lentiviral system allows inducible expression of noxious proteins in macrophages. J Immunol Methods. 2007;329(1–2):31–44. doi:10.1016/j.jim.2007.09.009
43. Biagi E, Bambacioni F, Gaipa G, et al. Efficient lentiviral transduction of primary human acute myelogenous and lymphoblastic leukemia cells. Haematologica. 2001;86(1):13–16.
44. Melis JP, Strumane K, Ruuls SR, Beurskens FJ, Schuurman J, Parren PW. Complement in therapy and disease: regulating the complement system with antibody-based therapeutics. Mol Immunol. 2015;67:117–130. doi:10.1016/j.molimm.2015.01.028
45. Liberg D, Önnervik P, Riva M, Larsson L, Forsberg G, Wachenfeldt K. Antibody Blockade of IL1RAP Signaling Reduces Metastasis in a Breast Cancer Model. Annual Meeting of the American Association for Cancer: McCormick Place North/South Chicago, Illinois, USA; 2018.
46. Fioretos T, Jaras M Method of treatment of a solid tumor with interleukin-1 accessory protein antibody. United States patent US 9403906B2.
47. Fioretos T, Jaras M. Anti - IL1rap antibodies and their use for treating human. European patent EP2665749A1.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.