Back to Journals » Infection and Drug Resistance » Volume 15
High Incidence of Psychiatric Disorders Associated with Cycloserine Treatment of Multidrug-Resistant Tuberculosis Patients: A Cohort Study in Beijing, China
Authors Pang Y, Liu R, Song Y, Lv Z, Gao M, Nie L, Ge Q, Wu X
Received 6 April 2022
Accepted for publication 3 June 2022
Published 13 July 2022 Volume 2022:15 Pages 3725—3732
DOI https://doi.org/10.2147/IDR.S369715
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Héctor Mora-Montes
Yu Pang,1 Rongmei Liu,2 Yanhua Song,2 Zizheng Lv,2 Mengqiu Gao,2 Lihui Nie,2 Qiping Ge,2 Xiaoguang Wu2
1Department of Science and Technology, Beijing Chest Hospital, Capital Medical University/Beijing Tuberculosis and Thoracic Tumor Research Institute, Beijing, 101149, People’s Republic of China; 2Department of Tuberculosis, Beijing Chest Hospital, Capital Medical University/Beijing Tuberculosis and Thoracic Tumor Research Institute, Beijing, 101149, People’s Republic of China
Correspondence: Lihui Nie; Xiaoguang Wu, Department of Tuberculosis, Beijing Chest Hospital, Capital Medical University/Beijing Tuberculosis and Thoracic Tumor Research Institute, No. 9, Beiguan Street, Tongzhou District, Beijing, People’s Republic of China, Tel +86-10-8950 9322 ; +86-10-8950 9122, Email [email protected]; [email protected]
Objective: Cycloserine (CS) is a group B anti-tuberculosis (TB) drug endorsed by the World Health Organization (WHO) for the treatment of drug-resistant (DR)-TB. Despite CS widespread acceptance and known efficacy, the high potential of drug-associated psychiatric disorders is a major concern to multidrug-resistant (MDR)-TB patients. In this study, we investigated CS-associated psychiatric disorders in a cohort of MDR-TB patients in Beijing, China. Our aim was to determine psychiatric disorder prevalence rates and associated risk factors in this population.
Methods: This MDR-TB cohort study was conducted at Beijing Chest Hospital between February 2018 and February 2021. All patients received individualized treatment regimens that included CS at some point during the full treatment course. Patient psychological status was assessed using the Symptom Checklist (SCL-90) questionnaire during the post-treatment follow-up period.
Results: Two hundred and thirty-seven MDR-TB patients were included in the final analysis. Overall, psychiatric disorders were recorded in 22 (9.28%) of the 237 patients in our cohort, with severity grades of 1 or 2 observed for the majority (8.44%) of psychiatric adverse events. As compared to results obtained for control group patients who were ≥ 40 years of age, patients who were < 40 years of age (77.3%, 17/22) were more likely to experience psychiatric disorders [adjusted odds ratio (aOR) = 3.060; 95% CI (1.060– 8.828)]. Additionally, patients with body mass index (BMI)s of < 18.5 kg/m2 [aOR = 3.824; 95% CI (1.502– 9.739)] had significantly higher odds of being afflicted with psychiatric disorders as compared with patients with BMIs that were ≥ 18.5 kg/m2.
Conclusion: Our results demonstrated that approximately one-tenth of Chinese MDR-TB patients experienced psychiatric disorders during CS treatment, with the majority of adverse events of moderate severity. In addition, low BMI and age < 40 years were identified as independent risk factors for psychiatric disorders in MDR-TB patients receiving CS therapy.
Keywords: cycloserine, multidrug-resistant tuberculosis, incidence, risk factor, psychiatric disorder
Introduction
The emergence of drug-resistant tuberculosis (DR-TB), including multidrug-resistant TB (MDR-TB), pre-extensively drug-resistant TB (pre-XDR-TB) and extensively drug-resistant TB (XDR-TB), remains an important obstacle to achieving TB control.1,2 Globally, an estimated 465 000 MDR-TB cases were reported in 2020, of which 69% and 87% were new and previously treated TB cases, respectively.3 Due to intrinsic drug resistance of TB bacilli to the two most powerful anti-TB first-line drugs (FLDs), second-line drugs (SLDs), which are less efficacious, more toxic and costlier than FLD drugs, are frequently used to treat MDR-TB.1,4 Despite widespread administration of SLDs to MDR-TB patients, only 59% of these patients experience successful treatment outcomes.3 This low success rate may be related to the fact that a high proportion of SLDs-treated patients must discontinue treatment after experiencing serious adverse events associated with use of SLDs.5,6 Therefore, successful management of MDR-TB patients is of great importance for improving clinical outcomes and reducing MDR-TB transmission within the community.
Cycloserine (CS), a broad-spectrum antimicrobial agent that inhibits peptidoglycan biosynthesis, possesses moderate in vitro anti-bacterial activity against Mycobacterium tuberculosis (MTB) and clinical efficacy in combating TB,7,8 resulting in the World Health Organization (WHO) endorsement of CS for use as a core group B drug to treat MDR-TB patients.9 However, despite known efficacy and widespread acceptance of CS as a treatment for MDR-TB, potential adverse events associated with CS use are a major concern of MDR-TB patients.10,11 Of the various CS-associated adverse events that have been reported to date, psychiatric disorders noted most frequently have included headaches, depression and mental disturbances.10 Importantly, serious treatment-associated adverse events have generally been reported more frequently in individuals with histories of psychiatric disorders than in those who have never experienced psychiatric illness.12
Notably, the reported association between CS treatment and psychiatric symptoms has been attributed to CS agonistic action on central nervous system glutamate receptors.13 In line with this proposed mechanism, rates of CS-associated adverse events have been found to be positively linked to oral drug dose, with only minimal side effects observed at low CS doses.14 However, most of the available evidence concerning CS-associated adverse events was obtained from retrospective review of medical records.10,15–17 Considering the prolonged duration of MDR-TB patient treatment with CS-containing regimens, retrospective studies have undoubtedly underestimated rates of adverse events, especially of events occurring during outpatient follow-up. To address this concern, we identified psychiatric disorders as they occurred during CS treatment of a cohort of MDR-TB patients in Beijing, China. Our aim was to determine the prevalence rate of psychiatric disorders in this population and identify risk factors associated with CS-associated psychiatric adverse events.
Methods
Patients
This MDR-TB cohort study was conducted at Beijing Chest Hospital between February 2018 and February 2021. All patients received individualized therapeutic regimens during the full course of MDR therapy (MDR-TB: 13–18 months; Pre-XDR- and XDR-TB: 24–30 months).18 Patients enrolled in this study met the following criteria: (i) MDR-TB confirmed by drug susceptibility testing; (ii) patients aged ≥18 years; (iii) patients received regimens containing CS. Patients who reported histories of psychiatric disorders at the first clinic visit were excluded from the study. Patient demographic and clinical characteristics were collected using a standardized form at the time of enrollment.
After inclusion in the study, patients visited the clinic at baseline (prior to treatment), then during treatment at weeks 2 and 4 and every 4 weeks thereafter. At each visit, patients received physical examinations, routine blood counts, biochemical tests, urinalysis testing and psychological assessments based on the Symptom Checklist (SCL-90) questionnaire. This questionnaire was comprised of 90 items, each rated on a 5-point scale based on distress level. These items were clustered within nine dimensions (somatization, obsessive-compulsive, interpersonal sensitivity, depression, anxiety, hostility, phobic anxiety, paranoid ideation and psychoticism). The total SCL-90 score, which was based on the number of items rated positively (PST) and the positive symptom distress index, was calculated by dividing the sum of all items by the PST score and was used to grade all observed psychiatric disorders. Moreover, after all adverse events associated with CS administration were recorded, an expert panel recommended whether therapy should be continued. Psychiatric disorders were classified based on symptoms of insomnia, anxiety, depression, mania, somnolence, metism, tremor, dizzy, headache, etc. Meanwhile, the Division of Acquired Immunodeficiency Syndrome (DAIDS) Table for Grading the Severity of Adult and Pediatric Adverse Events (2017.V2.1) was used to grade adverse events based on severity,19 while treatment outcomes were defined according to WHO criteria as cured, complete, death during treatment, failure and loss to follow-up.20 Cured and complete treatment outcomes were classified as successful outcomes, while death, loss to follow-up and treatment failure were classified as adverse outcomes.
Statistical Analysis
All original data were entered into the database using a double data entry method. After data collection was complete, we conducted crude and adjusted logistic regression analyses to identify and evaluate statistical significance of factors related to CS-associated adverse events. Variables with P values of <0.1 were introduced into multivariate model analysis. All calculations in this study were conducted using SPSS version 20.0 for Microsoft Windows (IBM Corp., Armonk, NY, USA). Differences were considered significant for P < 0.05.
Results
Study Cohort
Two hundred and thirty-seven MDR-TB patients were included in our analysis, of whom 65.8% were male. Baseline demographic and clinical characteristics are shown in Table 1. The median age of patients at time of enrollment was 39.0 years [interquartile range (IQR): 28.0–56.0]. Approximately seven-tenths of patients had histories of anti-TB treatment. Diabetes was noted in 71 (30.0%) patients. Besides CS and bedaquiline (BDQ), the most frequently used TB drugs were linezolid (85.7%, 203/237), clofazimine (60.8%, 144/237) and protionamide (42.6%, 101/237).
 |
Table 1 Demographic and Clinical Characteristics of 237 Multidrug-Resistant Tuberculosis Patients in China |
Psychiatric Disorders Associated with CS Treatment
Overall, psychiatric disorders were recorded for 22 (9.28%) of the 237 patients in our cohort. As summarized in Table 2, the most frequent psychiatric disorder was depression (10/237, 4.22%), followed by anxiety (4/237, 1.69%) and headache (2/239, 0.84%). Severities of most psychiatric-associated adverse events (8.44%) were grade 1 or 2, while grade 3 adverse events were only noted in two patients, one with depression and one with mania. The median time to onset of psychiatric disorders was 8 weeks (range 1–36 weeks). Half of all psychiatric disorders were observed during the 8 weeks of CS dosing, while approximately one-quarter of MDR-TB patients experienced psychiatric symptoms of variable severity during the remainder of the 36-week treatment period (Figure 1). Notably, adverse events eventually ceased after CS discontinuation.
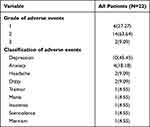 |
Table 2 Safety and Tolerability Profile Among Chinese Patients Enrolled in This Study |
 |
Figure 1 Time to incidence of psychiatric disorders in multidrug-resistant tuberculosis patients treated with cycloserine. |
Further analysis of effects of CS discontinuation on MDR-TB patient clinical outcomes revealed that 210 (88.6%) of 237 MDR-TB patients experienced favorable outcomes after completion of anti-TB treatment. In patients with no history of psychiatric symptoms, the treatment success rate was 87.6% (190/217), a rate comparable to that of patients with histories of psychiatric disorders (20/22, 90.9%), with differences in the favorable outcomes rates between groups found to be statistically insignificant (P = 1.000).
Risk Factors Associated with Psychiatric Disorders
Characteristics of patients with and without psychiatric disorders are summarized in Table 3. Based on the control group of patients (all ≥40 years of age), our results revealed that patients <40 years of age were more likely to suffer from psychiatric disorders [(77.3%, 17/22); AOR = 3.060; 95% CI (1.060–8.828)]. In addition, we found that patients with BMI values <18.5 kg/m2 had significantly higher odds of being diagnosed with psychiatric disorders during CS treatment as compared to patients with BMI values that were ≥18.5 kg/m2 [aOR = 3.824; 95% CI (1.502–9.739)]. Thus, this result demonstrated that underweight status was an independent risk factor for psychiatric disorders in patients receiving CS-containing treatment regimens. Although the psychiatric disorder rate of new cases was slightly lower than that of previously treated cases, this rate difference was not statistically significant [aOR = 3.515; 95% CI (0.970–12.734)]. Moreover, no significant differences were observed between the two groups with regard to baseline results for routine blood counts and biochemical tests (Table S1).
 |
Table 3 Factors Associated with Psychiatric Disorders Among the Patients Enrolled in This Study |
Discussion
When administered together with clofazimine, CS is an important group B treatment option for DR-TB patients.18,21 However, varying incidence rates of psychiatric events have been reported in groups of patients during treatment with CS-containing regimens.10 In this study, our results revealed that 22 of 237 patients (9.28%) experienced psychiatric disorders during the treatment period, a higher rate than reported in a previous meta-analysis conducted by Hwang et al, which revealed an estimated overall CS-associated psychiatric adverse reaction rate of 5.7%.10 Meanwhile, rates of CS-associated psychiatric symptoms have been reported to vary greatly across studies, ranging from 0.0% in Iran to 30.8% in UK.22,23 Such diverse results may stem from heterogeneity of criteria used to define adverse events, as well as from differences related to ethnic differences across study populations. However, in a recent cohort study from China, psychiatric symptoms were recorded in 4.3% of MDR-TB patients receiving CS regimens,15 a rate that was significantly lower than the rate observed in our cohort (9.28%). We hypothesize that this disparity stems from the retrospective design of the previous study that, as an inherent methodological weakness, led to documentation of predominantly serious adverse events (especially severe events), resulting in underestimation of drug-associated adverse events. Thus, our data provide a more accurate estimate of the overall incidence of CS-associated psychiatric adverse events in the Chinese population by reporting less severe adverse events in addition to serious and severe events.
Although approximately one-tenth of MDR-TB patients receiving CS therapy experienced psychiatric symptoms, only a few symptoms (9.1%) were classified as severe adverse events. This rate was dramatically lower than a previously reported rate based on nationwide data in China (29.6%),22 as well as a rate found in a retrospective observational study in Zhejiang whereby 37 (28.0%) of 132 CS group patients experienced severe adverse events.17 There are several potential explanations for the disparity between our results and results of those studies. On the one hand, psychological assessments of MDR-TB patients were routinely conducted in our study prior to initiation of CS therapy, which may have resulted in exclusion of the psychiatrically vulnerable population as the likely explanation for the decreased rate of severe adverse events in our study. On the other hand, our CS pilot study was conducted in a tertiary care hospital with adequate resources and clinicians to monitor and support MDR-TB patients during outpatient follow-up. Such support would have improved patient access to high-quality medical care that may have prevented or mitigated single or multiple adverse events, as demonstrated in previous studies showing that poor adherence to therapy and discontinuation of treatment contributed to poor MDR-TB patient outcomes.24 Nevertheless, in our study outcomes of patients who experienced CS-associated adverse events and associated treatment cessation were comparable to outcomes of patients without CS-associated adverse events, thus indicating vastly different treatment durations between groups did not influence treatment outcomes. We attribute these results to the excellent efficacy of Group A drugs such as bedaquiline and linezolid in combating MDR-TB. Nonetheless, these results highlight the fact that TB treatment should be comprehensive and guided by unique patient characteristics and results of drug susceptibility testing and whole-genome sequencing.25 In the meantime, researchers must continue to strive to meet the relentless demand for novel antibiotics to eliminate drug-resistant tubercle bacilli.
We noted that psychiatric disorders occurred more frequently in patients with low BMI values. This observation may be explained by the lower total tissue volumes of patients with low BMI values that may lead to higher CS blood plasma levels at a given drug dose. Thus, monitoring of plasma drug concentrations of MDR-TB patients throughout the course of drug therapy is extremely important. In addition, half of psychiatric disorders of patients in our cohort were detected after 8 weeks of CS therapy. In view of existing evidence, this result may be explained based on possible CS accumulation to relatively high concentrations in adipose tissue after several weeks of treatment, thereby resulting in increased plasma levels of CS in patients with low BMI values and delayed onset of CS-triggered adverse events. Nonetheless, further pharmacokinetics research is urgently needed to validate our hypothesis.
Importantly, we also found an association between young middle-aged subjects (aged <40 years) and increased incidence of psychiatric disorders. This population, which has played a key role in the economic development of China, has been shown to have experienced technostress in a Chinese cross-sectional study, which demonstrated a positive association between employee age and technostress for employees who were ≤45 years of age.26 Similar results were reported in a Finnish survey which showed that middle-aged employees were most stressed about career planning.27 In addition to work-related stresses, despite their access to health insurance coverage, MDR-TB patients still have to cope with added medical treatment costs and lost wages during treatment. Therefore, we speculate that underlying psychological stress may synergize with CS to increase rates of psychiatric symptoms in patients treated with CS. Thus, more attention should be paid to monitoring of populations of young and middle-aged workers under CS treatment.
We also acknowledge several obvious limitations of the present study. First, this was a single-center study with a relatively small sample size, with both of these limitations weakening the strength of our conclusion. Thus, further studies based on large numbers of study subjects at multiple centers are urgently needed to validate our results. Second, although the results of this study demonstrated an association between low BMI and psychiatric disorders in MDR-TB patients treated with CS-containing regimens, blood plasma concentrations of CS were not measured, thus making it difficult to elucidate the mechanisms underlying this phenomenon. Finally, human–computer interface-based tools have shown promise in preventing psychiatric symptoms in vulnerable populations.28 However, it is unknown whether this novel technology could provide significant relief to patients suffering from CS-associated psychiatric disorders, especially since established psychological interventions are required to best assist these patients. Despite these limitations, our results provide much more accurate estimates of incidence rates of CS-associated psychiatric adverse events in the Chinese population as compared to results of previously reported studies.
In conclusion, our data demonstrate that approximately one-tenth of Chinese MDR-TB patients experienced psychiatric disorders during the CS treatment period, with the majority of adverse events found to be of moderate severity. Importantly, psychiatric disorders occurred more frequently in patients with low BMI and those <40 years of age. These results suggest that close monitoring of patients to detect psychiatric adverse events is important for improving treatment outcomes of patients under treatment for MDR-TB, especially for patients in vulnerable populations.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of Beijing Chest Hospital, Capital Medical University. Study was conducted in compliance with the Declaration of Helsinki. All participants gave informed consent to take part in the study.
Funding
This work was supported by the the Beijing Municipal Science & Technology Commission (Z191100006619077) and the Beijing Hospitals Authority’ Ascent Plan (DFL20191601).
Disclosure
The authors declare that they have no competing interests.
References
1. Gandhi NR, Nunn P, Dheda K, et al. Multidrug-resistant and extensively drug-resistant tuberculosis: a threat to global control of tuberculosis. Lancet. 2010;375(9728):1830–1843. doi:10.1016/S0140-6736(10)60410-2
2. Lange C, Chesov D, Heyckendorf J, Leung CC, Udwadia Z, Dheda K. Drug-resistant tuberculosis: an update on disease burden, diagnosis and treatment. Respirology. 2018;23(7):656–673. doi:10.1111/resp.13304
3. World Health Organization. Global tuberculosis report 2020. Geneva: World Health Organization; 2020. WHO/HTM/TB/2020.
4. Xu C, Pang Y, Li R, et al. Clinical outcome of multidrug-resistant tuberculosis patients receiving standardized second-line treatment regimen in China. J Infect. 2018;76(4):348–353. doi:10.1016/j.jinf.2017.12.017
5. Shean K, Streicher E, Pieterson E, et al. Drug-associated adverse events and their relationship with outcomes in patients receiving treatment for extensively drug-resistant tuberculosis in South Africa. PLoS One. 2013;8(5):e63057. doi:10.1371/journal.pone.0063057
6. Franke MF, Appleton SC, Bayona J, et al. Risk factors and mortality associated with default from multidrug-resistant tuberculosis treatment. Clin Infect Dis. 2008;46(12):1844–1851. doi:10.1086/588292
7. Harris DA, Ruger M, Reagan MA, et al. Discovery, development, and antimicrobial properties of D-4-amino-3-isoxazolidone (oxamycin), a new antibiotic produced by Streptomyces garyphalus n. sp. Antibiot Chemother. 1955;5(4):183–190.
8. Caminero JA, Sotgiu G, Zumla A, Migliori GB. Best drug treatment for multidrug-resistant and extensively drug-resistant tuberculosis. Lancet Infect Dis. 2010;10(9):621–629. doi:10.1016/S1473-3099(10)70139-0
9. World Health Organization. WHO operational handbook on tuberculosis, module 4: treatment - drug-resistant tuberculosis treatment. Geneva: World Health Organization; 2020.
10. Hwang TJ, Wares DF, Jafarov A, Jakubowiak W, Nunn P, Keshavjee S. Safety of cycloserine and terizidone for the treatment of drug-resistant tuberculosis: a meta-analysis. Int J Tuberc Lung Dis. 2013;17(10):1257–1266. doi:10.5588/ijtld.12.0863
11. Court R, Centner CM, Chirehwa M, et al. Neuropsychiatric toxicity and cycloserine concentrations during treatment for multidrug-resistant tuberculosis. Int J Infect Dis. 2021;105:688–694. doi:10.1016/j.ijid.2021.03.001
12. Otto MW, Basden SL, Leyro TM, McHugh RK, Hofmann SG. Clinical perspectives on the combination of D-cycloserine and cognitive-behavioral therapy for the treatment of anxiety disorders. CNS Spectr. 2007;12(1):
13. Hansen KB, Yi F, Perszyk RE, Menniti FS, Traynelis SF. NMDA receptors in the central nervous system. Methods Mol Biol. 2017;1677:1–80. doi:10.1007/978-1-4939-7321-7_1
14. Alghamdi WA, Alsultan A, Al-Shaer MH, et al. Cycloserine population pharmacokinetics and pharmacodynamics in patients with tuberculosis. Antimicrob Agents Chemother. 2019;63(5). doi:10.1128/AAC.00055-19
15. Wang J, Pang Y, Jing W, et al. Efficacy and safety of cycloserine-containing regimens in the treatment of multidrug-resistant tuberculosis: a nationwide retrospective cohort study in China. Infect Drug Resist. 2019;12:763–770. doi:10.2147/IDR.S194484
16. van der Walt ML, Shean K, Becker P, Keddy KH, Lancaster J. Treatment outcomes and adverse drug effects of ethambutol, cycloserine, and terizidone for the treatment of multidrug-resistant tuberculosis in South Africa. Antimicrob Agents Chemother. 2020;65(1). doi:10.1128/AAC.00744-20
17. Li Y, Wang F, Wu L, et al. Cycloserine for treatment of multidrug-resistant tuberculosis: a retrospective cohort study in China. Infect Drug Resist. 2019;12:721–731. doi:10.2147/IDR.S195555
18. Gao M, Gao J, Xie L, et al. Early outcome and safety of bedaquiline-containing regimens for treatment of MDR- and XDR-TB in China: a multicentre study. Clin Microbiol Infect. 2021;27(4):597–602. doi:10.1016/j.cmi.2020.06.004
19. Division of AIDS, National Institute of allergy and infectious diseases National Institutes of health, US department of health and Human services. Division of AIDS (DAIDS) table for grading the severity of adult and pediatric adverse events: corrected version 2.1; 2017. Available from: https://rsc.niaid.nih.gov/sites/default/files/daidsgradingcorrectedv21.pdf.
20. World Health Organization. Meeting report of the WHO expert consultation on drug-resistant tuberculosis treatment outcome definitions, 17–19 November 2020. Geneva: World Health Organization; 2020.
21. Riccardi N, Giacomelli A, Canetti D, et al. Clofazimine: an old drug for never-ending diseases. Future Microbiol. 2020;15(7):557–566. doi:10.2217/fmb-2019-0231
22. Walker WC, Murdoch JM. Cycloserine in the treatment of pulmonary tuberculosis; a report on toxicity. Tubercle. 1957;38(5):297–302. doi:10.1016/S0041-3879(57)80097-X
23. Epstein IG, Nair KG, Boyd LJ, Auspitz P. Cycloserine-isoniazid combination therapy in virgin cases of pulmonary tuberculosis. Dis Chest. 1958;33(4):371–381. doi:10.1378/chest.33.4.371
24. Lan Z, Ahmad N, Baghaei P, et al. Drug-associated adverse events in the treatment of multidrug-resistant tuberculosis: an individual patient data meta-analysis. Lancet Respir Med. 2020;8(4):383–394. doi:10.1016/S2213-2600(20)30047-3
25. Riccardi N, Villa S, Alagna R, et al. Advantages and challenges of tailored regimens for drug-resistant tuberculosis: a StopTB Italia look into the future. Infect Drug Resist. 2020;13:2795–2800. doi:10.2147/IDR.S257480
26. Wu HY, Tian Y, Wang XD, et al. Twenty-four-week interim outcomes of bedaquiline-containing regimens in treatment of adolescents with rifampicin-resistant tuberculosis: a retrospective cohort study in China. J Paediatr Child Health. 2022;58(1):116–121. doi:10.1111/jpc.15672
27. Spiro JE, Brose N, Heinemann SF, Heiligenberg W. Immunolocalization of NMDA receptors in the central nervous system of weakly electric fish: functional implications for the modulation of a neuronal oscillator. J Neurosci. 1994;14(10):6289–6299. doi:10.1523/JNEUROSCI.14-10-06289.1994
28. Price M, Yuen EK, Goetter EM, et al. mHealth: a mechanism to deliver more accessible, more effective mental health care. Clin Psychol Psychother. 2014;21(5):427–436. doi:10.1002/cpp.1855
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
