Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 16
High Blood Eosinophil and YKL-40 Levels, as Well as Low CXCL9 Levels, are Associated with Increased Readmission in Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease
Authors Peng J, Yu Q, Fan S, Chen X, Tang R, Wang D , Qi D
Received 4 December 2020
Accepted for publication 4 March 2021
Published 26 March 2021 Volume 2021:16 Pages 795—806
DOI https://doi.org/10.2147/COPD.S294968
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Junnan Peng, Qian Yu, Shulei Fan, Xingru Chen, Rui Tang, Daoxin Wang, Di Qi
Department of Respiratory Medicine, Second Affiliated Hospital of Chongqing Medical University, Chongqing, People’s Republic of China
Correspondence: Daoxin Wang; Di Qi
Department of Respiratory Medicine, Second Affiliated Hospital of Chongqing Medical University, No. 76 Linjiang Road, Yuzhong District, Chongqing, 400010, People’s Republic of China
Email [email protected]; [email protected]
Background: Readmission after hospital discharge is common among patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD). Predictive biomarkers of readmission would facilitate stratification strategies and individualized prognosis. Therefore, this study aimed to investigate the utility of type 2 biomarkers (eosinophils, periostin, and YKL-40) and a type 1 biomarker (CXCL9) in predicting readmission events in patients with AECOPD.
Methods: This is a prospective observational study design. Blood levels of eosinophils, periostin, YKL-40, and CXCL9 were measured at admission. The clinical outcomes were 12-month COPD-related readmission, time to COPD-related readmission, and number of 12-month COPD-related readmissions. These outcomes were analyzed using logistic and Cox regression models and Spearman’s rank test.
Results: A total of 123 patients were included, of whom 51 had experienced at least one readmission for AECOPD. High levels of eosinophils (≥ 200 cells/μL or 2% of the total white blood cell count, adjusted odds ratio [aOR] =3.138, P=0.009) and YKL-40 (≥ 14.5 ng/mL, aOR =2.840, P=0.015), as well as low CXCL9 levels (≤ 30.1 ng/mL, aOR =2.551, P=0.028), were associated with an increased COPD-related readmission. The highest relative readmission rate was observed in patients with both high eosinophil and YKL-40 levels. Moreover, high eosinophil and YKL-40 levels were associated with a shorter time to first COPD-related readmission and an increased number of 12-month COPD-related readmissions.
Conclusion: High blood eosinophil and YKL-40 levels, as well as low CXCL9 levels, have predictive utility for the 12-month COPD-related readmission rate. Using eosinophils and YKL-40 together allows more precise identification of patients at high risk of COPD-related readmission.
Keywords: COPD, exacerbation, readmission, biomarker, prospective observational
Introduction
Acute exacerbation of chronic obstructive pulmonary disease (AECOPD) with subsequent readmission is considered to be a catastrophic event during the clinical course of COPD.1 In the United States, the COPD readmission rate are 22.6% within 30 days2 and 64.2% within 12 months of discharge,3 while in Europe, approximately 35.1% are readmitted within 90 days,4 and up to 50.0% within 12 months in China.5 This indicates that it is important to identify patients at high risk of readmission after AECOPD.
Airway inflammation is a consistent feature in the pathogenesis and progression of COPD.6 Neutrophilic-associated T helper type 1 (Th1) inflammation is regarded as the predominant characteristic of COPD. However, approximately 30% of patients demonstrate an eosinophilic phenotype with increased T helper type 2 (Th2) immunity,6,7 which is more likely to present with unstable clinical conditions and experience repeated readmissions.7,8 Thus, the biomarker-based strategy for identifying a COPD eosinophilic phenotype and stratifying readmission rates is valuable to improve clinical outcomes of patients with COPD.9
Recent studies have shown a significant association between blood and lung eosinophil counts.7,10 The Global Initiative for Chronic Obstructive Lung Disease (GOLD) has reported blood eosinophils as a potential candidate for Th2-oriented therapies.11 However, there have been inconsistent findings regarding the association of blood eosinophils with clinical outcomes of COPD patients, especially the future risk of readmissions.12–19 Therefore, this association warrants further research.
Given different inflammatory phenotypes of COPD are caused by an imbalance of Th1/Th2 immune response,6 we hypothesized that Th1- and Th2-related biomarkers may be useful predictors of readmission. Periostin is an extracellular matrix protein crucially involved in Th2-associated eosinophilic inflammation,20 and serum concentrations of periostin is reported to parallel with airway eosinophils.21 YKL-40, originally known as an upstream cytokine inducing Th2-type inflammation,22,23 is now thought to be associated with bronchial inflammation and tissue remodeling in COPD.24 CXCL9 is a well-characterized Th1-type chemokine that is primarily induced by IFN-γ,25 and positively correlated with airway neutrophils in COPD.26
Therefore, the primary objective of this study was to investigate the association between these four biomarkers and adverse clinical outcomes following hospitalization for AECOPD. Furthermore, we determined whether any combination of two biomarkers could improve the ability to risk-stratify patients.
Materials and Methods
Study Design and Population
This prospective observational study enrolled patients hospitalized due to AECOPD at the Department of Respiratory Medicine, the 2nd Affiliated Hospital of Chongqing Medical University, between November 2017 and March 2019. The inclusion criteria were being aged above 40 years and having a previous confirmed COPD diagnosis according to the GOLD 2017 classification criteria11 based on smoking history (>10 packs/year) or biomass exposure history (wood, charcoal or grass for cooking and/or heating, exposure ≥100 h/year), typical clinical symptoms, and lung function tests (a post-bronchodilator fixed ratio of FEV1/FVC <0.7). AECOPD was defined as an event in the natural disease course characterized by acute changes in clinical symptoms beyond normal day-to-day variation.11 The exclusion criteria were: 1) having an exacerbation history within the previous 1 year; 2) having radiographic evidence of pneumonia at the time of admission; 3) having coexisting chronic respiratory diseases, including asthma, bronchiectasis, tuberculosis, lung cancer, and interstitial lung disease, as well as immune system diseases such as rheumatoid arthritis, gout, and osteoarthritis; 4) having been treated with systemic corticosteroids before the blood sample collection; 5) having cognitive disorders, liver dysfunction, renal failure, or other severe organ dysfunctions; 6) dying during hospitalization; and 7) refusing to provide voluntary informed consent. During the study period, a total of 419 candidate patients were included. Of these patients, 293 were excluded according to the exclusion criteria and 3 were lost to follow-up. Finally, 123 eligible patients were recruited for this study (Supplementary Figure 1).
Baseline characteristics, including age, sex, body mass index (BMI), smoking status, underlying disease, and exacerbation history, were collected upon hospital admission. Subsequently, the responsible physician assessed each patient to obtain functional data, including lung function measurements; the 6-minute walk distance (6MWD) test; and scores of quality of life tests, including the modified Medical Research Council dyspnea scale and the COPD Assessment Test. According to the American Thoracic Society and European Respiratory Society (ATS/ERS) guidelines,27 spirometry was measured using a computerized spirometer (MasterScreen, Leibnizstrasse, Hochberg, Germany) where parameters, including forced vital capacity (FVC), forced expiratory volume in one second (FEV1), their percent predicted values (%FVC, %FEV1), and FEV1/FVC were recorded. The spirometry was performed just before discharge, at which time the patients had no symptoms of wheezing, and each patient inhaled 400 µg salbutamol at 20 minutes before the test. Chest computed tomography (CT) was conducted within 24 hours after hospital admission to rule out a pneumonia diagnosis. CT images were reconstructed using a standard algorithm (Philips Systems, Netherlands) into a 512 × 512 matrix size with a trans-axial slice thickness of 1.0 mm. Two well-trained radiologists independently read the images with discrepancies being resolved by a third radiologist. Blood samples were collected on the day of admission. At discharge, inpatient laboratory findings, the administration of systemic corticosteroids during hospitalization, home oxygen use, and inhalation therapy were recorded. All patients were followed up at 1, 3, 6, and 12 months (face-to-face visits or telephone calls if unavailable) and assessed for COPD-related readmissions. The study protocol was approved by the Second Affiliated Hospital of Chongqing Medical University (no 2017–08). Written informed consent was obtained from all patients.
Measurements
Blood samples were collected followed by routine analysis of peripheral blood cell counts and C-reactive protein (CRP) at the Department of Clinical Laboratory in our hospital. Cells were classified as neutrophils, lymphocytes, monocytes, eosinophils, and basophils; moreover, the absolute number of each cell type was calculated. The minimum detectable CRP level was 24.9 pg/mL. Additionally, serum samples were collected after centrifugation at 4500 rpm for 15 min and then stored at −80 °C until subsequent analysis. Serum periostin, YKL-40, and CXCL9 levels were determined using commercially available enzyme-linked immunosorbent assay kits (Cloud-Clone, Wuhan, China), according to the manufacturer’s instructions, with their minimum detectable levels being 31.0 pg/mL, 13.5 pg/mL, and 12.2 pg/mL, respectively.
Study Variables and Clinical Outcomes
Regarding blood eosinophil count, the cutoffs for a high and low eosinophil count were (≥200 cells/μL and/or ≥2% of the total white blood cell [WBC] count) and (<200 cells/μL and <2% of the total WBC count), respectively. We selected this threshold since patients with values above this cutoff are more likely to have sputum eosinophilia and are considered to be “eosinophilic patients with AECOPD”.10,16 Most of the observational studies previously done used these cutoff values.16,18 However, there is still controversy regarding the optimum eosinophil cutoff levels and whether to use absolute or relative counts. Therefore, we also reported the results with different cutoff values of eosinophils. Regarding periostin, YKL-40, and CXCL9 levels, cutoffs were arbitrarily defined as high and low levels using the median instead of the mean values, due to their skewed distributions.
The primary outcome was 12-month COPD-related readmission (yes/no). The secondary outcomes were time to first COPD-related readmission and the number of 12-month COPD-related readmissions.
Statistical Analyses
Data were analyzed for normality and descriptive statistics were presented as the numbers (percentages) for categorical variables and mean ± standard deviation (SD) or median (interquartile range; IQR) for continuous variables, as appropriate. Between-group comparisons of normally and non-normally distributed continuous variables were performed using Student’s t-test and Mann–Whitney U-test, respectively. Categorical data were compared using the chi-squared test or Fisher’s exact test when appropriate.
Regarding primary outcome, we used a univariate logistic regression to evaluate the association of the four biomarkers (eosinophils, periostin, YKL-40, and CXCL9) with 12-month COPD-related readmission (yes/no); next, we built a multivariable logistic regression model forcing variables with a P<0.1 in univariate analysis and other potentially related variables, including age, sex, BMI, D’Hoore et al28 version of the Charlson comorbidity index (CCI), smoking status, COPD severity and CRP. Results were presented as odds ratios (ORs) with 95% confidence intervals (CIs). Further, the primary outcome distribution was presented as the cumulative probability for 12-month COPD-related readmission stratified by any pair of the four biomarkers.
Regarding secondary outcomes, Kaplan–Meier survival curves, the Log rank test, and the Cox proportional hazard regression model were used to evaluate the time to first COPD-related readmission. Deaths during the follow-up period were considered censored. Correlations between biomarkers and the number of 12-month COPD-related readmissions were explored using Spearman’s rank test with the results being presented using scatter plots.
Statistical analysis was performed using IBM SPSS 22.0 software (SPSS, Chicago, IL, USA); further, figures were created using GraphPad Prism 8.0 software (San Diego, CA, USA). All tests were two-tailed. A P-value <0.05 was considered significant.
Results
Characteristics of the Patients
Table 1 presents the baseline characteristics of the 123 patients with COPD. There were 90 (73.2%) men and 33 (26.8%) women, with a mean age of 71.1 (SD, 9.6) years. The mean BMI was 22.4 (SD, 3.7) kg/m2. The most common comorbidity was cardiovascular disease (39.0%), followed by diabetes (16.3%); further, the median CCI was 1.0 (IQR, 0.0–1.0). Most patients (66.7%) had a history of smoking with the remaining patients (33.3%) having a history of biomass exposure. Based on the GOLD criteria, 64 (52.0%) patients had severe or very severe airflow obstruction (GOLD grades III–IV). At the time of index admission, the laboratory findings were as follows: median blood eosinophil count: 120.0 cells/μL (IQR, 60.0–240.0 cells/μL); median serum periostin level: 50.6 ng/mL (IQR, 41.9–61.2 ng/mL); median serum YKL-40 level: 14.5 ng/mL (IQR, 11.1–17.7 ng/mL); median serum CXCL9 level: 30.1 ng/mL (IQR, 23.6–40.7 ng/mL).
 |
Table 1 Characteristic Features and Laboratory Findings of the Patients |
Comparison Between the Readmission and Non-Readmission Groups
As shown in Table 2, readmission was more likely to have a higher proportion of severe or very severe airflow obstruction (GOLD grades III–IV) (P=0.018), shorter 6-MWD (P=0.042), and longer length of hospital stay (P=0.022). There were no significant between-group differences in any other clinical variables (all P>0.05). The proportion of patients with high blood eosinophil levels (≥200 cells/μL or 2% of the WBC count, 58.8% vs 36.1%, P=0.013; Figure 1A) and high serum YKL-40 levels (≥14.5 ng/mL, 64.7% vs 40.3%, P=0.008; Figure 1C) were significantly higher in the readmission group than in the non-readmitted group. Further, there was no significant between-group difference in the proportion of patients with high serum periostin levels (≥50.6 ng/mL, 56.9% vs 45.8%, P=0.228; Figure 1B) and low serum CXCL9 levels (≤30.1 ng/mL, 60.8% vs 43.1%, P=0.053; Figure 1D).
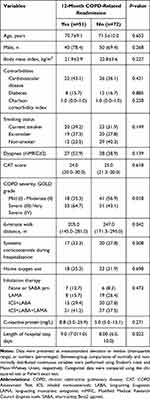 |
Table 2 Comparison Between the Readmission and Non-Readmission Groups |
Association Between Biomarkers and 12-Month COPD-Related Readmission
During the 12-month follow-up period, 51 (41.5%) patients experienced hospital readmission due to COPD exacerbation. Univariate analysis revealed a significant difference in the primary outcome (12-month COPD-related readmission) between the groups stratified according to the blood eosinophil count (high vs low eosinophil count: OR =2.527, 95% CI =1.210–5.278, P=0.014) and serum YKL-40 levels (high vs low YKL-40 levels: OR =2.718, 95% CI =1.293–5.713, P=0.008). Further, multiple logistic regression analysis revealed that a blood eosinophil count ≥200 cells/μL or 2% of the WBC count (aOR =3.138; 95% CI =1.325–7.439; P=0.009), serum YKL-40 levels ≥14.5 ng/mL (aOR =2.840; 95% CI =1.228–6.568; P=0.015), and serum CXCL9 ≤30.1 ng/mL (aOR =2.551; 95% CI =1.108–5.847; P=0.028) were associated with an increased risk of COPD-related hospital readmission. Additionally, the association between higher eosinophils and 12-month COPD-related readmission remained essentially using the different cutoff values of eosinophils. See Table 3 for details.
 |
Table 3 Association Between Biomarkers and 12-Month COPD-Related Readmission |
Combined Assessment of Any Pair of the Four Biomarkers
Upon stratification of event rates by any pair of the four biomarkers, there was an increase in the frequency of 12-month COPD-related readmission events with increased eosinophil, periostin, and YKL-40 levels, as well as decreased CXCL9 levels. The highest relative readmission rate (64.3% group readmission) was observed among patients with high blood eosinophil and serum YKL-40 levels. Further, the lowest relative readmission rate (18.2% group readmission) was observed in patients with low eosinophil and YKL-40 levels (Figure 2).
Secondary Outcome
The Kaplan–Meier curve revealed a significant difference in the first COPD-related readmission rate among patients stratified according to the blood eosinophils, serum YKL-40, and CXCL9 levels (P=0.004, 0.006, and 0.031, respectively; Figure 3A, C and D, respectively). However, when we stratified patients according to the periostin levels, no significant difference was found (P=0.249, Figure 3B). Cox regression analysis revealed that high levels of blood eosinophil (≥200 cells/μL or 2% of the WBC count) and serum YKL-40 (≥14.5 ng/mL), as well as low levels of serum CXCL9 (≤30.1 ng/mL) remained associated with shorter time to first COPD-related readmission (P=0.001, 0.012, and 0.018, respectively). Moreover, there was a significant correlation of the number of COPD-related readmissions with eosinophil (r =0.206, P=0.022; Figure 4A), YKL-40 (r =0.375, P<0.001; Figure 4C), and periostin levels (r =0.213, P=0.018; Figure 4B), but no significant correlation between the number of COPD-related readmissions and CXCL9 levels was observed (r =−0.048, P=0.596; Figure 4D). Figure 4 shows the scatter plots of biomarkers exhibiting correlations.
Discussion
Our study found that levels of blood eosinophil, YKL-40, and CXCL9 were associated with COPD-related readmission during the 12-month follow-up period in patients with AECOPD. Patients with both high serum YKL-40 (≥14.5 ng/mL) and blood eosinophil levels (≥200 cells/μL or 2% of the WBC count) had the highest relative readmission rate. Furthermore, high eosinophil and YKL-40 levels were associated with a shorter time to first COPD-related readmission and an increased number of 12-month COPD-related readmissions.
Hospital readmissions for COPD impart serious burdens on society and patients.1,11 Although the causes of readmission vary widely, respiratory diseases remain the leading cause.29,30 Early recognition of high-risk patients and appropriate interventions can significantly reduce COPD-related readmissions.9 Therefore, we sought to identify useful biomarkers to distinguish patients with high-risk for COPD-related readmission.
A subgroup of patients with COPD presents eosinophilic inflammatory profiles during clinical stability and exacerbation.6 Blood eosinophils is demonstrated to be correspond to eosinophilic airway inflammation of COPD.10 In our study, a high blood eosinophil count (≥ 200 cells/μL or 2% of the WBC count) could predict a more than two-fold increase in the incidence of 12-month readmission for COPD, a shorter time to first COPD-related readmission, and an increased number of 12-month COPD-related readmissions. Consistent findings have been reported in the COPDGene and ECLIPSE cohorts13 as well as the Copenhagen General Population cohort12 of clinically stable COPD, while opposite evidences were reported in the BODE, CHAIN15 and SPIROMICS14 cohorts; moreover, several retrospective and post hoc studies on AECOPD also reported conflicting results.16–19 Couillard et al analyzed these inconsistent findings and found that the relative timing of systemic corticosteroid administration may be a confounding factor.16 Blood eosinophil count is known to decrease by over 50% within the first four hours of corticosteroid administration with a subsequent gradual return to baseline within 24 hours.31 The present study excluded patients who did receive systemic corticosteroid treatment before blood sample collection. Moreover, our results confirmed that COPD patients with higher blood eosinophil counts were more likely to experience repeated readmissions and have a shorter time to rehospitalization. Therefore, future studies should consider systemic corticosteroid use as an important confounding factor for its eosinopenic effect.
YKL-40, also known as Chitinase-3-like protein 1 (CHI3L1), is a crucial regulator in Th2-type inflammation and tissue remodeling.22 Kang et al demonstrated that CHI3L1 contributes to the genesis of Th2 inflammation and pulmonary fibrosis, as well as inhibits Th1 inflammation in mouse models.32 Our present study found that the proportion of patients with high serum YKL-40 levels (≥14.5 ng/mL) was significantly higher in the readmission group than in the non-readmission group. Both univariate and multivariate analyses revealed a high correlation of serum YKL-40 levels with the readmission risk; moreover, YKL-40 levels were negatively correlated with pulmonary function test parameters in our study (data not shown). These findings are similar to those of previous studies,23,24,33 and further support that YKL-40 is useful for predicting COPD-related readmission due to its associations with Th2-type inflammation and critical pathophysiology in COPD. Additionally, the highest and lowest relative readmission rates were observed in patients with high and low levels, respectively, of both eosinophils and YKL-40. This indicates that concurrent measurement of both biomarkers could improve the accuracy of risk stratification in patients with COPD.
CXCL9 is an IFN-γ-inducible chemokine that is released by neutrophils,25,34 and serve as Th1-type inflammatory markers in respiratory diseases.35,36 When compared to healthy controls, CXCL9 concentrations were increased in COPD patients and positively correlated with markers of neutrophilic airway inflammation.26 Fulkerson et al demonstrated that CXCL9 inhibits eosinophil migration to the lungs of allergen-induced mice.37 Collectively, these findings suggest that CXCL9 is associated with suppressed Th2-type inflammation and increased Th1-type inflammation, respectively. In this study, we found that patients with COPD who were readmitted manifested numerically, but not significantly, lower CXCL9 levels than the non-readmitted patients. However, multivariate analysis revealed that low CXCL9 levels were markedly associated with an increased risk of readmission due to AECOPD. In addition, CXCL9 expression was related to the time to the first readmission for exacerbation of COPD, but not to the frequency of hospitalized AECOPD. These findings could be attributed to our sample size being too small to achieve statistical significance in analysis. Therefore, further studies with more participants are needed to clarify the relationship between serum CXCL9 and readmission risk in patients with COPD.
Periostin is an extracellular matrix protein in response to type 2 cytokines IL-4 and IL-13.20,21 Serum periostin is known as an imperfect, surrogate marker of Th2 inflammatory response in patients with asthma.38,39 Although most studies have investigated the role of periostin in asthma, data regarding COPD are scarce.20 In this study, there were no significant differences in serum periostin levels between the readmission and non-readmission groups. Moreover, increased periostin levels were not associated with long-term disease outcomes, including the time to next AECOPD hospitalization and number of COPD-related readmissions. These results are consistent with those of Konstantelou et al, who found that high serum periostin levels could not predict the time of next exacerbation or hospitalization, or the time to death in patients hospitalized for AECOPD.40 Similarly, Carpaij et al reported that serum periostin was not associated with eosinophilic inflammation measured in blood, sputum, and biopsies in clinically stable patients with COPD.41 Collectively, the aforementioned evidence suggests that serum periostin plays a limited role in COPD. Moreover, as the principal risk factor for COPD, cigarette smoke exposure is reported to suppresses periostin expression,42,43 which might attenuate the predictive ability of periostin in COPD-related readmission.
This study has several limitations. First, this was a single-center study with a relatively small sample size, which limits extensive subgroup analyses. Second, all analyses were based on a single measurement and we did not assess longitudinal changes. Third, although we adjusted for some confounding factors, there may be residual confounding factors. Therefore, there is a need for large-scale, multicenter, and well-designed studies to validate the findings.
Conclusions
High eosinophil and YKL-40 levels, as well as low CXCL9 levels, which are assessed in a corticosteroid-free time frame, are associated with an increased risk of 12-month COPD-related readmission rate in hospitalized patients with AECOPD. Higher eosinophil and YKL-40 levels are associated with a shorter time to first COPD-related readmission and an increased number of 12-month COPD-related readmissions. Additionally, concurrent assessment of eosinophil and YKL-40 levels could provide more precise identification of patients at high risk of COPD-related readmission.
Abbreviations
AECOPD, acute exacerbation of chronic obstructive pulmonary disease; CCI, Charlson comorbidity index; CHI3L1, chitinase-3-like protein 1; COPD, chronic obstructive pulmonary disease; CI, confidence interval; CAT, COPD Assessment Test; CRP, C-reactive protein; CT, computed tomography; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; ICS, inhaled corticosteroids; GOLD, Global Initiative for Chronic Obstructive Lung Disease; IQR, interquartile range; LABA: long-acting β-agonist; LAMA, long-acting muscarinic antagonist; mMRC, Modified Medical Research Council dyspnea scale; OR, odds ratio; SABA, short-acting Beta2 agonist; SD, standard deviation; Th1, T helper type 1; Th2, T helper type 2; WBC, white blood cell; 6MWD, 6-minute walk distance.
Data Sharing Statement
Data available on request.
Ethics Approval and Consent to Participate
This study was approved by the Research Ethics Committees of the Second Affiliated Hospital of Chongqing Medical University (no 2017-08) in accordance with the Declaration of Helsinki.
Consent for Publication
Not applicable.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This work was supported by the National Key Research and Development Program of China (2018YFC1313600), National Natural Science Foundation of China (81670071), the National Natural Science Foundation for Yong scholars of China (81800083) and Natural Science Foundation of Chongqing, China(ctsc2019jcyj-zdxmX0031).
Disclosure
The authors declare that they have no competing interests.
References
1. Crisafulli E, Guerrero M, Chetta A, Torres A. Readmission in COPD patients: should we consider it a marker of quality of care or a marker of a more severe disease with a worse prognosis? Eur Respir J. 2016;48(1):279–281. doi:10.1183/13993003.00426-2016
2. Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360(14):1418–1428. doi:10.1056/NEJMsa0803563
3. Lindenauer PK, Dharmarajan K, Qin L, Lin Z, Gershon AS, Krumholz HM. Risk trajectories of readmission and death in the first year after hospitalization for chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;197(8):1009–1017. doi:10.1164/rccm.201709-1852OC
4. Hartl S, Lopez-Campos JL, Pozo-Rodriguez F, et al. Risk of death and readmission of hospital-admitted COPD exacerbations: european COPD Audit. Eur Respir J. 2016;47(1):113–121. doi:10.1183/13993003.01391-2014
5. Ko FW, Dai DL, Ngai J, et al. Effect of early pulmonary rehabilitation on health care utilization and health status in patients hospitalized with acute exacerbations of COPD. Respirology. 2011;16(4):617–624. doi:10.1111/j.1440-1843.2010.01921.x
6. Brightling C, Greening N. Airway inflammation in COPD: progress to precision medicine. Eur Respir J. 2019;54(2):1900651. doi:10.1183/13993003.00651-2019
7. Singh D, Bafadhel M, Brightling CE, et al. Blood eosinophil counts in clinical trials for chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2020;202(5):660–671. doi:10.1164/rccm.201912-2384PP
8. Vashi MT, Willoughby JL, Quinn SJ, Tran BT. Eosinophilic chronic obstructive pulmonary disease: implications for exacerbations, readmissions, and treatment. Am J Respir Crit Care Med. 2019;199(1):110–112. doi:10.1164/rccm.201802-0328RR
9. Kong CW, Wilkinson TMA. Predicting and preventing hospital readmission for exacerbations of COPD. ERJ Open Res. 2020;6(2):00325–02019. doi:10.1183/23120541.00325-2019
10. Bafadhel M, McKenna S, Terry S, et al. Acute exacerbations of chronic obstructive pulmonary disease: identification of biologic clusters and their biomarkers. Am J Respir Crit Care Med. 2011;184(6):662–671. doi:10.1164/rccm.201104-0597OC
11. GOLD. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (Revised 2017). Global Initiative Chron Obstructive Lung Dis. 2017.
12. Vedel-Krogh S, Nielsen SF, Lange P, Vestbo J, Nordestgaard BG. Blood eosinophils and exacerbations in chronic obstructive pulmonary disease. The Copenhagen General Population Study. Am J Respir Crit Care Med. 2016;193(9):965–974. doi:10.1164/rccm.201509-1869OC
13. Yun JH, Lamb A, Chase R, et al. Blood eosinophil count thresholds and exacerbations in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2018;141(6):2037–2047.e2010. doi:10.1016/j.jaci.2018.04.010
14. Hastie AT, Martinez FJ, Curtis JL, et al. Association of sputum and blood eosinophil concentrations with clinical measures of COPD severity: an analysis of the SPIROMICS cohort. Lancet Respir Med. 2017;5(12):956–967. doi:10.1016/S2213-2600(17)30432-0
15. Casanova C, Celli BR, De-torres JP, et al. Prevalence of persistent blood eosinophilia: relation to outcomes in patients with COPD. Eur Respir J. 2017;50(5):1701162. doi:10.1183/13993003.01162-2017
16. Couillard S, Larivée P, Courteau J, Vanasse A. Eosinophils in COPD exacerbations are associated with increased readmissions. Chest. 2017;151(2):366–373. doi:10.1016/j.chest.2016.10.003
17. Håkansson KEJ, Ulrik CS, Godtfredsen NS, et al. High suPAR and low blood eosinophil count are risk factors for hospital readmission and mortality in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2020;15:733–743. doi:10.2147/COPD.S229904
18. Li Q, Larivée P, Courteau J, et al. Greater eosinophil counts at first COPD hospitalization are associated with more readmissions and fewer deaths. Int J Chron Obstruct Pulmon Dis. 2019;14:331–341. doi:10.2147/COPD.S187375
19. Bafadhel M, Greening NJ, Harvey-Dunstan TC, et al. Blood eosinophils and outcomes in severe hospitalized exacerbations of COPD. Chest. 2016;150(2):320–328. doi:10.1016/j.chest.2016.01.026
20. Izuhara K, Conway SJ, Moore BB, et al. Roles of periostin in respiratory disorders. Am J Respir Crit Care Med. 2016;193(9):949–956. doi:10.1164/rccm.201510-2032PP
21. Berry A, Busse WW. Biomarkers in asthmatic patients: has their time come to direct treatment? J Allergy Clin Immunol. 2016;137(5):1317–1324. doi:10.1016/j.jaci.2016.03.009
22. Lee CG, Hartl D, Lee GR, et al. Role of breast regression protein 39 (BRP-39)/chitinase 3-like-1 in Th2 and IL-13-induced tissue responses and apoptosis. J Exp Med. 2009;206(5):1149–1166. doi:10.1084/jem.20081271
23. Zhu Z, Zheng T, Homer RJ, et al. Acidic mammalian chitinase in asthmatic Th2 inflammation and IL-13 pathway activation. Science. 2004;304(5677):1678–1682. doi:10.1126/science.1095336
24. Tong X, Wang D, Liu S, et al. The YKL-40 protein is a potential biomarker for COPD: a meta-analysis and systematic review. Int J Chron Obstruct Pulmon Dis. 2018;13:409–418. doi:10.2147/COPD.S152655
25. Henrot P, Prevel R, Berger P, Dupin I. Chemokines in COPD: from implication to therapeutic use. Int J Mol Sci. 2019;20(11):2785. doi:10.3390/ijms20112785
26. Costa C, Rufino R, Traves SL, Lapa ESJR, Barnes PJ, Donnelly LE. CXCR3 and CCR5 chemokines in induced sputum from patients with COPD. Chest. 2008;133(1):26–33. doi:10.1378/chest.07-0393
27. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. doi:10.1183/09031936.05.00034805
28. D’Hoore W, Bouckaert A, Tilquin C. Practical considerations on the use of the Charlson comorbidity index with administrative data bases. J Clin Epidemiol. 1996;49(12):1429–1433. doi:10.1016/S0895-4356(96)00271-5
29. Alqahtani JS, Njoku CM, Bereznicki B, et al. Risk factors for all-cause hospital readmission following exacerbation of COPD: a systematic review and meta-analysis. Eur Respir Rev. 2020;29(156):190166. doi:10.1183/16000617.0166-2019
30. Nuñez A, Miravitlles M. Preventing readmissions of COPD patients: more prospective studies are needed. Eur Respir Rev. 2020;29(156):200097. doi:10.1183/16000617.0097-2020
31. Thorn GW, Renold AE, Wilson DL, et al. Clinical studies on the activity of orally administered cortisone. N Engl J Med. 1951;245(15):549–555. doi:10.1056/NEJM195110112451501
32. Kang MJ, Yoon CM, Nam M, et al. Role of chitinase 3-like-1 in interleukin-18-induced pulmonary type 1, type 2, and type 17 inflammation; alveolar destruction; and airway fibrosis in the murine lung. Am J Respir Cell Mol Biol. 2015;53(6):863–871. doi:10.1165/rcmb.2014-0366OC
33. Tang H, Fang Z, Sun Y, et al. YKL-40 in asthmatic patients, and its correlations with exacerbation, eosinophils and immunoglobulin E. Eur Respir J. 2010;35(4):757–760. doi:10.1183/09031936.00034409
34. Gasperini S, Marchi M, Calzetti F, et al. Gene expression and production of the monokine induced by IFN-gamma (MIG), IFN-inducible T cell alpha chemoattractant (I-TAC), and IFN-gamma-inducible protein-10 (IP-10) chemokines by human neutrophils. J Immunol. 1999;162(8):4928–4937.
35. Hasegawa T, Okazawa T, Uga H, Kurata H, Mori A. Serum CXCL9 as a potential marker of Type 1 inflammation in the context of eosinophilic asthma. Allergy. 2019;74(12):2515–2518. doi:10.1111/all.13924
36. Nukui Y, Yamana T, Masuo M, et al. Serum CXCL9 and CCL17 as biomarkers of declining pulmonary function in chronic bird-related hypersensitivity pneumonitis. PLoS One. 2019;14(8):e0220462. doi:10.1371/journal.pone.0220462
37. Fulkerson PC, Zimmermann N, Brandt EB, et al. Negative regulation of eosinophil recruitment to the lung by the chemokine monokine induced by IFN-gamma (Mig, CXCL9). Proc Natl Acad Sci U S A. 2004;101(7):1987–1992. doi:10.1073/pnas.0308544100
38. Buhl R, Korn S, Menzies-Gow A, et al. Prospective, single-arm, longitudinal study of biomarkers in real-world patients with severe asthma. J Allergy Clin Immunol Pract. 2020;8(8):2630–2639.e2636. doi:10.1016/j.jaip.2020.03.038
39. Pavord ID, Corren J. Biomarkers of type 2 airway inflammation in airway disease: and then there were two. J Allergy Clin Immunol Pract. 2020;8(8):2640–2642. doi:10.1016/j.jaip.2020.04.047
40. Konstantelou E, Papaioannou AI, Loukides S, et al. Serum periostin in patients hospitalized for COPD exacerbations. Cytokine. 2017;93:51–56. doi:10.1016/j.cyto.2017.05.007
41. Carpaij OA, Muntinghe FOW, Wagenaar MB, et al. Serum periostin does not reflect type 2-driven inflammation in COPD. Respir Res. 2018;19(1):112. doi:10.1186/s12931-018-0818-8
42. Christenson SA, Steiling K, van den Berge M, et al. Asthma-COPD overlap. Clinical relevance of genomic signatures of type 2 inflammation in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;191(7):758–766. doi:10.1164/rccm.201408-1458OC
43. Mertens TCJ, van der Does AM, Kistemaker LE, Ninaber DK, Taube C, Hiemstra PS. Cigarette smoke differentially affects IL-13-induced gene expression in human airway epithelial cells. Physiol Rep. 2017;5(13):e13347. doi:10.14814/phy2.13347
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




