Back to Journals » Drug Design, Development and Therapy » Volume 17
Exploring the Active Compounds of Traditional Mongolian Medicine Baolier Capsule (BLEC) in Patients with Coronary Artery Disease (CAD) Based on Network Pharmacology Analysis, Molecular Docking and Experimental Validation
Authors Wei M, Li F, Guo K, Yang T
Received 8 November 2022
Accepted for publication 28 January 2023
Published 14 February 2023 Volume 2023:17 Pages 459—476
DOI https://doi.org/10.2147/DDDT.S395207
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Tuo Deng
Mengqiu Wei,1,* Fengjin Li,2,* Kai Guo,3,4 Tianxiao Yang5
1Intensive Care Unit, Zhongshan City People’s Hospital, Zhongshan, 528400, People’s Republic of China; 2Department of Gynecology, Longyan First Affiliated Hospital of Fujian Medical University, Longyan, 36400, People’s Republic of China; 3Department of Cardiology, Guangdong Second Provincial General Hospital, Guangzhou, 510000, People’s Republic of China; 4Department of Emergency, Nanfang Hospital, Southern Medical University, Guangzhou, 510000, People’s Republic of China; 5Department of Cardiology, Shandong University Zibo Central Hospital, Zibo, 255000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Kai Guo; Tianxiao Yang, Email [email protected]; [email protected]
Objective: Baolier Capsule (BLEC) is a Traditional Mongolian Medicine comprising fifteen herbs. This study aims to illustrate the synergistic mechanism of BLEC in the treatment of Coronary Artery Disease (CAD) by using network pharmacology method, molecular docking and experimental validation.
Methods: Searching and screening the active ingredients of different herbs in BLEC and target genes related to CAD in multiple databases. Subsequently, Protein–Protein Interactions Network (PPI-Net), gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) enrichment were used to identify the key targets. AutoDock was used to verify the binding ability between the active ingredient and key target through molecular docking. Reverse Transcription-Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR) was used to verify the effect of active ingredient of BLEC on the key target gene. Finally, effect of BLEC on the degree of blood lipids and atherosclerosis was validated by animal experiment.
Results: There are 144 active components and 80 CAD-related targets that are identified in BLEC in the treatment of CAD. What is more, 8 core genes were obtained by clustering and topological analysis of PPI-Net. Further, GO and KEGG analysis showed that fluid shear stress and atherosclerosis are the key pathways for BLEC to treat CAD. These results were validated by molecular docking method. In vitro, active compounds of BLEC (Quercetin, luteolin, kaempferol, naringenin, tanshinone IIA, β-carotene, 7-O-methylisomucronulatol, piperine, isorhamnetin and Xyloidone) can inhibit 8 core gene (AKT1, EGFR, FOS, MAPK1, MAPK14, STAT3, TP53 and VEGFA) expression. Moreover, BLEC not only improve blood lipid levels but also inhibit the development of atherosclerosis in ApoE-knockout mice.
Conclusion: Our research first revealed the basic pharmacological effects and related mechanisms of in the treatment of CAD. The predicted results provide some theoretical support for BLEC or its important active ingredients to treat CAD.
Keywords: coronary artery disease, Baolier capsule, traditional Mongolian medicine, network pharmacology, experimental validation
Introduction
At present, major progress has been made to prevent and treat cardiovascular disease (CVD) through lifestyle changes and drugs, but CVD is still the main cause of death in humans, especially in developed country and developing country.1 It is worth noting that the residual risk of CVD in patients is still high, even though on the basis of the best lifestyle intervention and high-dose statin therapy, new lipid-lowering drugs such as ezetimibe, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors and cholesterol ester transfer protein (CETP) inhibitors, and even kanazumab are used.2–6 Therefore, looking for drugs that can prevent and treat coronary artery disease (CAD) is of great significance to further decrease the incidence of major adverse cardiovascular events (MACEs) in patients who suffer from CAD.
Traditional Chinese medicine has opened up a new path for the treatment of CAD. Among them, Mongolian medicine, as one of the outstanding cultural heritage of the Mongolian nationality, is a summary of the Mongolian experience in fighting against diseases for a long time, and gradually formed by absorbing the experience of traditional Chinese medicine and Tibetan medicine. The curative effect is remarkable and it is increasingly accepted by people. Yogurt, which is a popular Mongolian dairy product, contributed to prevent and manage hyperlipidemia, blood glucose and inflammatory marker.7 Syringa pinnatifolia Hemsl., which is a Mongolian folk medicine in China, may protect against myocardial ischemia.8 Eerdun Wurile (EW) is a well established traditional Mongolian medicine with proven clinical application for treatment of stroke recovery.9,10 Qingganjiuwei powder (QGJWS) is a commonly used Mongolian drug to treat patients with chronic hepatic disease through protecting liver fibrosis.11 The use of Mongolian drugs to treat CAD has certain application prospect.
Baolier capsule (BLEC), one of the Mongolian drugs, comes from the classic Mongolian medicine book “Coral Experience Prescription”, “The Set of Mongolian medicine prescription” and “Zhigao prescription”.12 BLEC consists of fifteen herbs.12 BLEC can protect vascular endothelial function, inhibit inflammation and significantly improve the symptoms of patients with CAD, including number and duration of episodes of angina and number of using for Nitroglycerin.12 However, BLEC is formed of dozens of compounds from Chinese patent medicine and Chinese herbal compound formula, which leads to the difficulties in exploring BLEC mechanism. It is important clinically to discover the target genes and compounds of BLEC in discovering new drugs. Our study innovatively applies network pharmacology method, molecular docking and cellar or animal experiment to analyze key targets, and signal pathways of BLEC in treating CAD, to guide future researches of this formula. The detailed workflow is shown in Figure 1.
 |
Figure 1 Schematic of the network pharmacological analysis used to identify the potential mechanism of BLEC action on CAD. Abbreviations: BLEC, Baolier capsule; CAD, Coronary artery disease. |
Materials and Methods
Network Pharmacology Prediction
Collection and Screening of Candidate Compounds in Baolier Capsule
The herbs in BLEC decoction include Guangzao (Choerospondiatis Fructus), Danshen (Radix Salviae), Roudoukou (Myristicae Semen), Zhizi (Gardeniae Fructus), Qiancao (Rubia Cordifolia), Honghua (Carthami Flos), Sanqi (Panax Notoginseng (Burk). F. H. Chen Ex C. Chow), Muxiang (Aucklandiae Radix), Tanxiang (Santalum Album L)., Niuhuang (Bovis Calculus), Dahuang (Radix Rhei Et Rhizome), Mutong (Caulis Akebiae), Huangqi (Hedysarum Multijugum Maxim). Biba (Piperis Longi Fructus) and Hezi (Chebulae Fructus). The active ingredients in BLEC are gained from the Traditional Chinese Medicine Systems Pharmacology (TCMSP) Database and Analysis Platform (http://tcmspw.com/tcmsp.php), with the keywords Guangzao, Danshen, Roudoukou, Zhizi, Qiancao, Honghua, Sanqi, Muxiang, Tanxiang, Niuhuang, Dahuang, Mutong, Huangqi, Biba and Hezi respectively. TCMSP is a Chinese herbal medicine system pharmacology platform that includes 449 herbs, can catch the relations between diseases, targets and drugs. The database comprises targets, chemicals and corresponding drug-target networks. Oral Bioavailability (OB) is an important parameter in pharmacokinetics, which is directly related to bioavailability. Drug-like (DL) indicates active ingredient’s similarities to present drugs. We selected active ingredients with OB > 30% and DL > 0.18 for later research.13
Identification of Potential Targets of Active Ingredients and CAD
We obtain potential targets of active ingredients in BLEC from the database, retrieve related targets of CAD from Gene cards (https://www.genecards.org), Online Mendelian Inheritance in Man (OMIM, https://www.omim.org/), PharmGKB (http://www.pharmgkb.org), Therapeutic Target Database (TTD, http://db.idrblab.net/ttd/) and DrugBank (https://go.drugbank.com/). In addition, genes related to CAD, which are obtained from Gene cards, were selected according to relevance score greater or equal to 30. UniProt (https://www.uniprot.org) can help people find full name, ID and symbols of related gene.
Construction of Ingredient-Target-Disease Network
To further explore the relations between potential targets and active ingredients, we loaded them into Cytoscape 3.8.0 to form an ingredient-target-disease network.
Construction of Protein–Protein Interactions Network (PPI-Net)
STRING (http://string-db.org) is a convenient online database to build PPI-Net.14 Every interaction between target proteins will be scored. Then, following steps will be performed: selecting “multiple proteins”, inputting target genes that may interact with active ingredients in BLEC and related genes of CAD, searching in “Homo sapiens”, setting minimum required interaction score to high confidence (0.9) and hiding disconnected nodes in the network. Finally, target proteins will be put into Cytoscape to build the interaction network.
Analysis of Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG)
Target genes which are potential ones of active ingredients in BLEC and related ones in CAD are put into the R project for GO and KEGG analysis, in order to discover what role the selected targets play in signal pathways.
Molecular Docking
Construction of Receptor Structure
The ID of core target proteins of PPI-Net was obtained from UniProt. According to the ID, receptor structure file was downloaded from RCSB PDB (http://www1.rcsb.org/) as pdb format. We import the receptor structure file into pyMOL software and save it in pdb format after removing molecules and small molecule ligands.
Construction of Ligand Structure
The 2D structure of the ligand was downloaded from PubChem (https://pubchem.ncbi.nlm.nih.gov/) as SDF format. The ligand structure file is imported into Chem3D software and converted into 3D structure then saved as mol2 format file after using the MM2 position to optimize the energy of all small molecules.
Molecular Docking
Calculation method of molecular docking Autodock vina ADT auto dock tool MGL tools Use ADT 1.5.6 (Auto Dock Tool) to convert the format of receptor and ligand structure files. Import The receptor structure pdb format file was imported into ADT 1.5.6 (Auto Dock Tool) and was saved as pdbqt format after adding hydrogens. The ligand structure pdb format file was converted to pdbqt format. The receptor structure was converted to 2D structure through ADT 1.5.6. set. The number of points in X-dimension, Y-dimension and Z-dimension are set as 40, respectively. Spacing (angstrom) was set as 1. Active pocket parameters is exported to GPF format file. The input file of ligand and receptor structure is modified according to active pocket parameters. The maximum energy difference is 5. The number of models is 20. The outputting the pdbqt file has been imported the pyMOL software together with the receptor file to save the optimal model.
Experimental Verification
Cell Culture
The murine RAW264.7 macrophage cell line was purchased from the China Center for Type Culture Collection (CCTCC, China). The RAW264.7 macrophages were seeded in 60-mm cell culture dishes at 1.0×106 cells per dish in DMEM containing 10% fetal bovine serum and maintained at 37°C in a humidified atmosphere of 5% CO2. The cells were synchronized by replacing the medium with DMEM supplemented with 0.5% BSA for 12 h before the experiments. After 12h, the cells were transfected with PBS or active ingredient of drugs (Final concentration about 25 μmol/L) for 48 h. Quercetin, luteolin, kaempferol, naringenin, tanshinone IIA, β-carotene, 7-O-methylisomucronulatol, piperine, isorhamnetin and Xyloidone were purchased commercially (Solarbio, Beijing, China).
Reverse Transcription-Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR)
Total mRNA was extracted from adenovirus-transfected RAW264.7 cells treated with PBS or active ingredient of drugs using TRIzol reagent (Invitrogen, Gaithersburg, MD) according to the manufacturer’s instructions. The mRNA was reverse transcribed using a PrimeScript™ RT Reagent Kit (TaKaRa, China). Real-time quantitative PCR (qPCR) was performed on a LightCycler 480 II (Roche, Switzerland) using a SYBR Premix Ex Taq Kit (TaKaRa, China). The mRNA expression level of GAPDH was used as an internal control. The quantitative measurements were determined using the ΔΔCt method.15 All samples were assayed in triplicate, and the mean value was considered in the comparative analyses. All qPCR primer sequences were obtained from PrimerBank and are listed in Supplementary Table 1.
Animals Model
ApoE-knockout (ApoE−/−) mice are a classic model of atherosclerosis. All animal experiments were conducted in strict accordance with Southern Medical University guidelines for the Care and Use of Experimental Animals. ApoE−/− mice were approved by the Animal Experiment Committee of Nanfang Hospital at Southern Medical University to be used in this study. The research ethical code for the study is 2020-BSGZ-07-01. The mice were fed with a high-fat diet (HFD, 15% fat and 1.2% cholesterol) and intragastrically administered BLEC once per day for 12 weeks to establish an AS model. According to previous studies and dose of clinical use about BLEC,12 ApoE−/− mice were randomly divided into 4 groups: Control group, Low-Dose BLEC group (37.5mg/kg), Middle-Dose BLEC group (75mg/kg), High-Dose BLEC group (150mg/kg). At the end of the experiment, blood samples and aorta of the mice were collected for further studies. ApoE−/− mice on a C57BL/6 background were obtained from Vital River Laboratory Animal Technology Co. Ltd. (Beijing, China) and HFD was purchased from Guangdong Provincial Animal Medicine Experimental Center. During the experiment, all mice were weighed twice a week, and the drug doses were adjusted accordingly.
The Detection of Lipids
Serum was collected to measure the level of lipids, including triglyceride (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) using assay kits. Whole blood was collected into ethylenediaminetetraacetic acid anticoagulant tubes and stored at 4°C. Samples were centrifuged for 15 minutes at 1000×g at 4°C within 30 minutes of collection and then stored at −80°C until analysis. The quantification assay kits were purchased from Nanjing Jiancheng Biotech Co., Ltd (Nanjing, Jiangsu, China).
Hematoxylin and Eosin (H&E) Staining
When the mice were sacrificed, the tissue of the aortic root was collected, fixed with formalin and made into paraffin sections (5μm) and frozen sections (5μm). The above sections were subjected to H&E. Images are best obtained within 7 days of staining using a Japanese Olympus BX63 microscope.
Statistical Analysis
All statistical analyses were performed using SPSS 25.0 software. The measurement data are expressed as mean±SD. One-way analysis of variance (ANOVA) and Fisher’s least significant difference (LSD) were used. P value <0.05 was considered statistically significant.
Results
Active Ingredients in BLEC
One thousand three hundred and thirty-one bioactive ingredients are selected from TCMSP database consisting of 43 types in Guangzao, 202 types in Danshen, 64 types in Roudoukou, 98 types in Zhizi, 54 types in Qiancao, 189 types in Honghua, 119 types in Sanqi, 106 types in Muxiang, 70 types in Tanxiang, 19 types in Niuhuang, 92 types in Dahuang, 43 types in Mutong, 87 types in Huangqi, 104 types in Biba and 41 types in Hezi. According to the standards OB and DL, 228 active ingredients are selected for further study, including 9 types in Guangzao, 65 types in Danshen, 9 types in Roudoukou, 15 types in Zhizi, 19 types in Qiancao, 22 types in Honghua, 8 types in Sanqi, 6 types in Muxiang, 3 types in Tanxiang, 5 types in Niuhuang, 16 types in Dahuang, 8 types in Mutong, 20 types in Huangqi, 15 types in Biba and 8 types in Hezi (Supplementary Table 2).
Potential Target Genes of Active Ingredients in BLEC
After removal of duplicates, 280 target genes of BLEC are acquired from TCMSP database and stored for further study (Supplementary Table 3).
Related Genes of CAD
As Figure 2 shows, 618 target genes of CAD are attained from Gene cards, OMIM, PharmGKB, TTD and DrugBank. As Figure 3 shows, there are a total of 80 target genes, which are potential ones of BLEC’s active ingredients and CAD’s related genes.
 |
Figure 2 Identification of the CAD-related genes by taking a union of all the results from 5 database. Abbreviation: CAD, Coronary artery disease. |
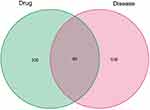 |
Figure 3 Identification of the drug-target disease-related genes by taking an intersection of drug target genes and CAD-related genes. Abbreviation: CAD, Coronary artery disease. |
Construction of Ingredient-Target-Disease Interactive Network
To thoroughly reveal the potential ingredients and targets of BLEC against CAD, the “component-target-disease” interactive network is constructed. Moreover, this research applies network pharmacological analysis to conduct visual analysis with relevant parameters of each target. One hundred and ninety-two nodes and 543 edges with 144 ingredients and 80 targets were displayed in this network. The network shows that the nodes possessing more edges and greater degree value play a more important role in the regulation (Figure 4). Accordingly, this research also establishes the network of ingredients with common targets of drugs and the disease, to directly display the interactions between potential targets and active ingredients. Degree refers to the number of routes that connect to one node and other in the network. The higher the value is, the more significant the corresponding ingredient or target is. After the Network Analyzer plugin in Cytoscape software done its analysis, ten essential active ingredients were listed in Tables 1. The result tells us that active ingredients of BLEC mainly achieved its effect on CAD through acting on the essential targets above.
 |
Table 1 Active Ingredients of BLEC in the Treatment of CAD |
Outcomes of PPI-Net
We use STRING to construct PPI-Net to study the mechanism of target genes of BLEC in treating CAD in accordance with protein-level high confidence (0.9). There are 541 edges and 73 nodes in this network (Figure 5). In order to obtain core target proteins, PPI network was filtered using Cytoscape software. If the Betweenness (BC), Closeness (CC), Degree (DC), Eigenvector (EC), LAC and Network (NC) of the protein is greater than the median, the protein is retained. After two filtration (Filter 1: BC = 21.71263724, CC = 0.1518718315, DC = 4.5, EC = 0.0574660525, LAC = 1.6, NC = 2.7083333335. Filter 2: BC = 8.3583916085, CC = 0.605263158, DC = 8, EC = 0.189531028, LAC = 3.25, NC = 4.3714285715), a total of 8 core target proteins were retained (Figure 6).
 |
Figure 5 Protein–Protein interaction (PPI) network. |
Results of GO and KEGG Enrichment Analysis
This research uses DAVID database for GO analysis of BLEC with potential targets for CAD. Two thousand one hundred and ninety-five GO entries are selected based on P value (P < 0.05). It includes 128 cellular components, 73 molecular function, and 1994 biological processes. In these biological processes, potential targets mainly focus on responding nutrient levels, oxidative stress, oxygen levels and so on. And the targets mainly relate to membrane raft, membrane microdomain, membrane region and so on in the cellular components. For molecular function, potential targets mainly work for phosphatase binding, protein phosphatase binding, cytokine receptor binding and so on (Figure 7 and Supplementary Table 4).
 |
Figure 7 The top 10 of GO enrichment analysis. |
When we use DAVID to do the KEGG enrichment analysis of BLEC for CAD (P < 0.05), 162 pathways are acquired. According to P value, the top 20 pathways are selected (Figure 8A and Supplementary Table 5). The pathway map of the fluid shear stress and atherosclerosis is illustrated in Figure 8B.
Molecular Docking of Active Compounds and Core Target Proteins
This research use the 8 core target proteins from PPI-Net to perform molecular docking. Then, we get active compounds targeting core target proteins from the compound-target interaction network respectively. Subsequently, as it shows in molecular docking, all active compounds can enter and bind the active pocket of the protein easily (Supplementary Figure 1) and the interactions with the biding site with the key amino acid (Figure 9A–H). The scores of docking are displayed in Table 2.
 |
Table 2 Molecular Docking Score |
Effects of Active Compounds of BLEC on Core Target Gene Expression in RAW264.7 Cells
Cellular experiments were conducted to validate the effects of active compounds of BLEC on key genes. The mRNA expressions of the 8 core target gene in active compounds group were significantly reduced relative to control group (Figure 10A–J). In conclusion, active compounds of BLEC inhibits target genes of BLEC in treating CAD expression in macrophages.
BLEC Attenuates the Development of Atherosclerosis and Decreases TC, TG, LDL-C and HDL-C in vivo
Cholesterol metabolic disorder is a key causative factor in atherosclerosis, and thus we further examined the effect of BLEC on serum lipid levels in ApoE−/− mice fed an HFD. As shown in Figure 11A, the mice treated with BLEC showed lower plasma levels of TC, TG, LDL-C and HDL-C than the control mice. The aorta of the above mice in four groups were stained with H&E staining to observe the occurrence of AS and lipid core area. It was suggested that compared with the control mice, mice treatment with BLEC had less lipid core area (Figure 11B). These results show that BLEC inhibits the development of atherosclerosis by modulating plasma lipid levels.
Discussion
CAD is called “heart tingling” in Mongolian medicine. It is due to the dysfunction of the “three roots” and “seven elements”, which hinders the movement of Heyi and the blood. The three roots refer to Heyi Xila and Badagan. Seven elements refer to dietary essence, blood, muscle, fat, bone, bone marrow and semen. Combining modern Western medicine theories, Mongolian medicine currently divides CAD into Heyi heart tingling, bloody heart tingling, adhesive heart tingling and Huyang heart tingling. Mongolian medicine mainly treats CAD by promoting the differentiation of turbidity and improving Heyi and blood circulation.16 BLEC is a prescription confirmed by famous Mongolian medicine experts repeatedly based on the above-mentioned etiology and pathogenesis and combined with Mongolian medicine theory and clinical practice experience.12 It can treat CAD by adjusting functions of the body’s three root and seven element.12
To the best of our knowledge, this is the first study integrating network pharmacology and molecular docking analyses to reveal the pharmacological mechanisms of BLEC for treating CAD. We identified a total of 228 active compounds in BLEC. Furthermore, a total of 80 potential target genes related to the action of BLEC on CAD were identified. PPI analysis revealed that the top-ranking genes, AKT Serine/Threonine Kinase 1 (AKT1), Epidermal Growth Factor Receptor (EGFR), Proto-oncogene c-Fos (FOS), Mitogen-activated protein kinase 1 (MAPK1), Mitogen-activated protein kinase 14 (MAPK14), Signal transducer and activator of transcription 3 (STAT3), Cellular tumor antigen p53 (TP53) and Vascular endothelial growth factor A (VEGFA), may be the crucial targets for BLEC treating CAD. Functional enrichment analysis revealed the over-represented GO terms and their functional domains. KEGG pathway enrichment analysis revealed that the 80 target proteins were significantly enriched in 162 related signal pathways. Among them, fluid shear stress and atherosclerosis appeared to be the most critical pathways involved in the treatment of CAD. Furthermore, molecular docking analysis demonstrated that the representative compounds could bind to the target protein binding site.
In this study, systematic pharmacological method is applied to forecast and explain the molecular mechanisms of BLEC on CAD. In BLEC’s active components-targets network, we get 81 targets affected by 144 bioactive compounds. Several recent studies have found that the active compounds of plants play an important role in the treatment of diseases. Such as, polysaccharides, flavonoids and saponins of genus Polygonatum have function about controlling blood glucose and preventing obese diabetes.17 Xanthohumol of Humulus lupulus L.has strong anti-cancer activity against various types of cancer cells.18 A considerable phenolic content for both varieties in Phaseolus vulgari, and a strong antiobesity activity.19 The 10 top-ranking compounds including quercetin, luteolin, kaempferol, naringin, tanshinone IIA, beta-carotene, 7-O-methylisomucronulatol, piperine, isorhamnetin, Xyloidone are identified as BLEC’s active ingredients. Their biological activities against CAD were mentioned above. Quercetin is a natural bioflavonoid found in vegetables and fruits including onions, grapes, tea, apples, and red wine. It can improve endothelial cell function,20–22 inhibit dendritic cell activation,23 inhibit foam cell formation by inhibiting oxidized low-density lipoprotein-induced apoptosis of macrophages24 and regulates cholesterol metabolism and lipoprotein metabolism by regulating cholesterol metabolism-related gene expression.25–27 Luteolin, is a natural flavone which is abundant in edible plants, including green chilies, broccoli, French beans, onion leaf, white radish, carrots, ragweed pollen and clover blossom.28 Studies have shown that it can inhibit the occurrence of atherosclerosis by inhibiting macrophage inflammation.29,30 Kaempferol, is a common natural yellow flavonoid with a low molecular weight, which exists in a number of traditional Chinese medicine and plant-derived foods. Kaempferol and its glycosides have many pharmacological functions, for example, they can serve as anti-inflammatory, antioxidant, and anticancer, antidiabetic, neuroprotective, analgesic, antimicrobial, and anti-allergic drugs.31 Studies have shown that kaempferol conducts anti-atherosclerotic effect by regulating the expression of gene and protein in inflammatory molecules.32 Naringin is the bitter component in a kind of grapefruit which is often found in its flowering parts, peels, juice and vegetative parts.33 Naringin can improve blood lipid levels,34 metabolic imbalances,35 and inhibit inflammatory reactions,36 and inhibit atherosclerosis.37 Tanshinone IIA is a main bioactive component isolated from the roots of the Chinese herb Salviae miltiorrhiza Bunge (Danshen),38 can regulate the autophagy and polarization of macrophages,39 and even the expression of non-coding RNA related to atherosclerosis and inflammation,40 antioxidant,41 improve endothelial cell function,42 and regulate cholesterol metabolism-related pathways affect cholesterol metabolism.43,44 Epidemiological evidence suggests that beta-carotene helps decrease the risk for atherosclerosis by protecting LDL from oxidation.45,46 Isorhamnetin, a bioactive compound, exists in herbal plants, and possesses multiple biological properties. It can inhibit macrophage apoptosis and inhibit atherosclerosis.47
According to PPI core network analysis and molecular docking results, it is shown that the main components of BLEC can prevent and treat CAD by affecting CAD-related top-ranking genes AKT1, EGFR, FOS, MAPK1, MAPK14, STAT3, TP53 and VEGFA. A large number of studies have confirmed that intervention of the above key genes can achieve the effect of preventing and treating atherosclerosis. The lack of AKT1 can promote the release of pro-inflammatory factors, and enhance the apoptosis of endothelial cells and macrophages, leading to the development of atherosclerosis.48 The expression of FOS in monocytes in the blood of patients with CAD is significantly increased, and its expression level is related to the severity of atherosclerosis.49 Inhibiting the expression of FOS of THP-1 cells can reduce the release of inflammatory factors.49 The activation of STAT3 can promote the occurrence of atherosclerosis by affecting endothelial cell dysfunction, macrophage polarization, inflammation, and immunity. Inhibition of STAT3 can achieve the effect of inhibiting atherosclerosis.50 P53 is a classic tumor suppressor gene. Inhibition of P53 can significantly increase the proportion of cell proliferation and apoptosis, and promote the occurrence of atherosclerosis.51 The MAPK pathway is a classic inflammatory pathway. Inhibiting MAPK can reduce the release of macrophages MCP-1, inhibit the infiltration of macrophages into the plaque, and inhibit the occurrence and development of atherosclerosis.52 The expression of EGFR and its ligands on arterial plaques of patients with CAD is significantly increased. By inhibiting EGFR, the uptake and inflammatory release of lipids by macrophages can be restricted, thereby inhibiting the occurrence and development of atherosclerosis.53 VEGFA plays an important role in the formation of blood vessels, and can induce apolipoprotein to change in the direction of pro-inflammatory, and its expression increases in atherosclerosis model mice. Inhibiting the expression of VEGFA in vascular smooth muscle can reduce the size of atherosclerotic plaques.54
Limitations
However, the study also has its limitations. On the one hand, we ignored the actual dosage and performance of traditional Mongolian medicine, which can be significant in research. Besides, network pharmacology is based on the data in the existing literature. The prediction is limited.
Conclusion
In this pharmacology network–based study, active ingredients, core targets, and key signal pathways of BLEC in treating CAD are systematically investigated. In vitro, active compounds of BLEC (Quercetin, luteolin, kaempferol, naringenin, tanshinone IIA, β-carotene, 7-O-methylisomucronulatol, piperine, isorhamnetin and Xyloidone) can inhibit 8 core target gene (AKT1, EGFR, FOS, MAPK1, MAPK14, STAT3, TP53 and VEGFA) expression. While Animal experiments confirmed that BLEC is a potent prescription to improve the development of atherosclerosis and plasma lipid level.
Abbreviations
CVD, Cardiovascular disease; PCSK9, Proprotein convertase subtilisin/kexin type 9; CETP, Cholesterol ester transfer protein; CAD, Coronary artery disease; MACEs, Major adverse cardiovascular events; EW, Eerdun Wurile; QGJWS, Qingganjiuwei powder; BLEC, Baolier capsule; TCMSP, Traditional Chinese Medicine Systems Pharmacology; OB, Oral Bioavailability; DL, Drug-like; OMIM, Online Mendelian Inheritance in Man; TTD, Therapeutic Target Database; PPI, Protein-protein interaction; GO, gene ontology; KEGG, Kyoto encyclopedia of genes and genomes; RT-qPCR, Quantitative Real-Time-Polymerase-Chain Reaction; HFD, High-fat diet; TG, Triglyceride; TC, Total cholesterol; LDL-C, Low-density lipoprotein cholesterol; HDL-C, High-density lipoprotein cholesterol; ANOVA, One-way analysis of variance; LSD, Fisher’s least significant difference; BC, Betweenness; CC, Closeness; DC, Degree; EC, Eigenvector; NC, Network; AKT1, AKT Serine/Threonine Kinase 1; EGFR, Epidermal Growth Factor Receptor; FOS, Proto-oncogene c-Fos; MAPK1, Mitogen-activated protein kinase 1; MAPK14, Mitogen-activated protein kinase 14; STAT3, Signal transducer and activator of 3; TP53, Cellular tumor antigen p53; VEGFA, Vascular endothelial growth factor A.
Data Sharing Statement
The original contributions presented in the study are included in this article and further inquiries can be directed to the corresponding authors.
Acknowledgment
This study has been presented as preprint in Research Square.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This work was supported by National Natural Science Foundation of China (81900398).
Disclosure
All authors declare no conflicts of interest.
References
1. Moraga P. GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1151–1210. doi:10.1016/S0140-6736(17)32152-9
2. Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376:1670–1681. doi:10.1016/S0140-6736(10)61350-5
3. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387–2397. doi:10.1056/NEJMoa1410489
4. Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713–1722. doi:10.1056/NEJMoa1615664
5. Bowman L, Hopewell JC, Chen F, et al. Effects of anacetrapib in patients with atherosclerotic vascular disease. N Engl J Med. 2017;377:1217–1227. doi:10.1056/NEJMoa1706444
6. Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377:1119–1131. doi:10.1056/NEJMoa1707914
7. Bayat A, Azizi-Soleiman F, Heidari-Beni M, et al. Effect of Cucurbita ficifolia and probiotic yogurt consumption on blood glucose, lipid profile, and inflammatory marker in type 2 diabetes. Int J Prev Med. 2016;7:30. doi:10.4103/2008-7802.175455
8. Feng X, Zhang R, Li J, et al. Syringa pinnatifolia Hemsl. fraction protects against myocardial ischemic injury by targeting the p53-mediated apoptosis pathway. Phytomedicine. 2019;52:136–146. doi:10.1016/j.phymed.2018.09.188
9. Gaowa S, Bao N, Da M, et al. Traditional Mongolian medicine Eerdun Wurile improves stroke recovery through regulation of gene expression in rat brain. J Ethnopharmacol. 2018;222:249–260. doi:10.1016/j.jep.2018.05.011
10. Qiburi Q, Ganbold T, Bao Q, et al. Bioactive components of ethnomedicine Eerdun Wurile regulate the transcription of pro-inflammatory cytokines in microglia. J Ethnopharmacol. 2020;246:112241. doi:10.1016/j.jep.2019.112241
11. Ge H, Wang A, Su Y, et al. Ameliorative effects of Qingganjiuwei powder, a traditional Mongolian medicine, against CCl4-induced liver fibrosis in rats. J Ethnopharmacol. 2021;264:113226. doi:10.1016/j.jep.2020.113226
12. Gang H, Feng H, Huai S. Study on the hypolipidemic effect of Mongolian medicine Baolier capsules. Chin J Tradit Med. 2017;23:10. Chinese. doi:10.16041/j.cnki.cn15-1175.2017.10.042
13. Ahmed SS, Ramakrishnan V. Systems biological approach of molecular descriptors connectivity: optimal descriptors for oral bioavailability prediction. PLoS One. 2012;7:e40654. doi:10.1371/journal.pone.0040654
14. Szklarczyk D, Gable AL, Nastou KC, et al. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021;49:D605–D612. doi:10.1093/nar/gkaa1074
15. Caballero-Solares A, Hall JR. Reverse Transcription-Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR) for gene expression analyses. Methods Mol Biol. 2022;2508:319–340.
16. Na S, Wu R, Chen X. Past present and future of treating angina pectoris with Mongolian medicine dialectics thinking. Chin J Cardiol Med. 2019;24:112–114. Chinese. doi:10.3969/j.issn.1007-5410.2019.02.005
17. Shi Y, Dun S, Chen D, et al. Bioactive compounds from Polygonatum genus as anti-diabetic agents with future perspectives. Food Chem. 2022;408:135183. doi:10.1016/j.foodchem.2022.135183
18. Carbone K, Gervasi F. An updated review of the genus Humulus: a valuable source of bioactive compounds for health and disease prevention. Plants. 2022;11(24):3434. doi:10.3390/plants11243434
19. Stefanucci A, Scioli G, MarinaccioA L, et al. Comparative study on phytochemical fingerprint of two diverse Phaseolus vulgaris var. Tondino del Tavo and Cannellino bio extracts. Antioxidants. 2022;11(8):1474. doi:10.3390/antiox11081474
20. Shen Y, Ward NC, Hodgson JM, et al. Dietary quercetin attenuates oxidant-induced endothelial dysfunction and atherosclerosis in apolipoprotein E knockout mice fed a high-fat diet: a critical role for heme oxygenase-1. Free Radic Biol Med. 2013;65:908–915. doi:10.1016/j.freeradbiomed.2013.08.185
21. Jiang YH, Jiang LY, Wang YC, et al. Quercetin attenuates atherosclerosis via modulating oxidized LDL-induced endothelial cellular senescence. Front Pharmacol. 2020;11:512. doi:10.3389/fphar.2020.00512
22. Luo M, Tian R, Lu N. Quercetin inhibited endothelial dysfunction and atherosclerosis in apolipoprotein E-deficient mice: critical roles for NADPH oxidase and heme oxygenase-1. J Agric Food Chem. 2020;68:10875–10883. doi:10.1021/acs.jafc.0c03907
23. Lin W, Wang W, Wang D. Quercetin protects against atherosclerosis by inhibiting dendritic cell activation. Mol Nutr Food Res. 2017;61. doi:10.1002/mnfr.201700031
24. Cao H, Jia Q, Yan L, et al. Quercetin suppresses the progression of atherosclerosis by regulating MST1-Mediated Autophagy in ox-LDL-Induced RAW264.7 macrophage foam cells. Int J Mol Sci. 2019;20(23):6093. doi:10.3390/ijms20236093
25. Jia Q, Cao H, Shen D, et al. Quercetin protects against atherosclerosis by regulating the expression of PCSK9, CD36, PPARgamma, LXRalpha and ABCA1. Int J Mol Med. 2019;44:893–902. doi:10.3892/ijmm.2019.4263
26. Garelnabi M, Mahini H, Wilson T. Quercetin intake with exercise modulates lipoprotein metabolism and reduces atherosclerosis plaque formation. J Int Soc Sports Nutr. 2014;11:22. doi:10.1186/1550-2783-11-22
27. Li SS, Cao H, Shen DZ, et al. Effect of quercetin on atherosclerosis based on expressions of ABCA1, LXR-alpha and PCSK9 in ApoE(-/-) Mice. Chin J Integr Med. 2020;26:114–121. doi:10.1007/s11655-019-2942-9
28. Lopez-Lazaro M. Distribution and biological activities of the flavonoid luteolin. Mini Rev Med Chem. 2009;9:31–59. doi:10.2174/138955709787001712
29. Li J, Dong JZ, Ren YL, et al. Luteolin decreases atherosclerosis in LDL receptor-deficient mice via a mechanism including decreasing AMPK-SIRT1 signaling in macrophages. Exp Ther Med. 2018;16:2593–2599. doi:10.3892/etm.2018.6499
30. Ding X, Zheng L, Yang B, et al. Luteolin attenuates atherosclerosis via modulating signal transducer and activator of transcription 3-mediated inflammatory response. Drug Des Devel Ther. 2019;13:3899–3911. doi:10.2147/DDDT.S207185
31. Calderon-Montano JM, Burgos-Moron E, Perez-Guerrero C, et al. A review on the dietary flavonoid kaempferol. Mini Rev Med Chem. 2011;11:298–344. doi:10.2174/138955711795305335
32. Kong L, Luo C, Li X, et al. The anti-inflammatory effect of kaempferol on early atherosclerosis in high cholesterol fed rabbits. Lipids Health Dis. 2013;12:115. doi:10.1186/1476-511X-12-115
33. Jourdan PS, McIntosh CA, Mansell RL. Naringin levels in citrus tissues: II. Quantitative distribution of naringin in citrus paradisi MacFad. Plant Physiol. 1985;77(4):903–908. doi:10.1104/pp.77.4.903
34. Mulvihill EE, Assini JM, Sutherland BG, et al. Naringenin decreases progression of atherosclerosis by improving dyslipidemia in high-fat-fed low-density lipoprotein receptor-null mice. Arterioscler Thromb Vasc Biol. 2010;30:742–748. doi:10.1161/ATVBAHA.109.201095
35. Assini JM, Mulvihill EE, Sutherland BG, et al. Naringenin prevents cholesterol-induced systemic inflammation, metabolic dysregulation, and atherosclerosis in Ldlr(-)/(-) mice. J Lipid Res. 2013;54:711–724. doi:10.1194/jlr.M032631
36. Burke AC, Sutherland BG, Telford DE, et al. Naringenin enhances the regression of atherosclerosis induced by a chow diet in Ldlr(-/-) mice. Atherosclerosis. 2019;286:60–70. doi:10.1016/j.atherosclerosis.2019.05.009
37. Orhan IE, Nabavi SF, Daglia M, et al. Naringenin and atherosclerosis: a review of literature. Curr Pharm Biotechnol. 2015;16:245–251. doi:10.2174/1389201015666141202110216
38. Tian XH, Wu JH. Tanshinone derivatives: a patent review (January 2006–September 2012). Expert Opin Ther Pat. 2013;23:19–29. doi:10.1517/13543776.2013.736494
39. Chen W, Li X, Guo S, et al. Tanshinone IIA harmonizes the crosstalk of autophagy and polarization in macrophages via miR-375/KLF4 pathway to attenuate atherosclerosis. Int Immunopharmacol. 2019;70:486–497. doi:10.1016/j.intimp.2019.02.054
40. Chen W, Guo S, Li X, et al. The regulated profile of noncoding RNAs associated with inflammation by tanshinone IIA on atherosclerosis. J Leukoc Biol. 2020;108:243–252. doi:10.1002/JLB.3MA0320-327RRR
41. Zhu J, Xu Y, Ren G, et al. Tanshinone IIA Sodium sulfonate regulates antioxidant system, inflammation, and endothelial dysfunction in atherosclerosis by downregulation of CLIC1. Eur J Pharmacol. 2017;815:427–436. doi:10.1016/j.ejphar.2017.09.047
42. Chen W, Tang F, Xie B, et al. Amelioration of atherosclerosis by tanshinone IIA in hyperlipidemic rabbits through attenuation of oxidative stress. Eur J Pharmacol. 2012;674:359–364. doi:10.1016/j.ejphar.2011.10.040
43. Tan YL, Ou HX, Zhang M, et al. Tanshinone IIA promotes macrophage cholesterol Efflux and attenuates atherosclerosis of apoE-/- mice by omentin-1/ABCA1 pathway. Curr Pharm Biotechnol. 2019;20:422–432. doi:10.2174/1389201020666190404125213
44. Tang FT, Cao Y, Wang TQ, et al. Tanshinone IIA attenuates atherosclerosis in ApoE(-/-) mice through down-regulation of scavenger receptor expression. Eur J Pharmacol. 2011;650:275–284. doi:10.1016/j.ejphar.2010.07.038
45. Omenn GS, Goodman GE, Thornquist MD, et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med. 1996;334:1150–1155. doi:10.1056/NEJM199605023341802
46. Shaish A, Daugherty A, O’Sullivan F, et al. Beta-carotene inhibits atherosclerosis in hypercholesterolemic rabbits. J Clin Invest. 1995;96:2075–2082. doi:10.1172/JCI118256
47. Luo Y, Sun G, Dong X, et al. Isorhamnetin attenuates atherosclerosis by inhibiting macrophage apoptosis via PI3K/AKT activation and HO-1 induction. PLoS One. 2015;10:e120259. doi:10.1371/journal.pone.0120259
48. Fernandez-Hernando C, Ackah E, Yu J, et al. Loss of Akt1 leads to severe atherosclerosis and occlusive coronary artery disease. Cell Metab. 2007;6:446–457. doi:10.1016/j.cmet.2007.10.007
49. Patino WD, Mian OY, Kang JG, et al. Circulating transcriptome reveals markers of atherosclerosis. Proc Natl Acad Sci U S A. 2005;102:3423–3428. doi:10.1073/pnas.0408032102
50. Chen Q, Lv J, Yang W, et al. Targeted inhibition of STAT3 as a potential treatment strategy for atherosclerosis. Theranostics. 2019;9:6424–6442. doi:10.7150/thno.35528
51. Guevara NV, Kim HS, Antonova EI, et al. The absence of p53 accelerates atherosclerosis by increasing cell proliferation in vivo. Nat Med. 1999;5:335–339. doi:10.1038/6585
52. Hopkins PN. Molecular biology of atherosclerosis. Physiol Rev. 2013;93:1317–1542. doi:10.1152/physrev.00004.2012
53. Zeboudj L, Giraud A, Guyonnet L, et al. Selective EGFR (epidermal growth factor receptor) deletion in myeloid cells limits atherosclerosis-brief report. Arterioscler Thromb Vasc Biol. 2018;38:114–119. doi:10.1161/ATVBAHA.117.309927
54. Heinonen SE, Kivela AM, Huusko J, et al. The effects of VEGF-A on atherosclerosis, lipoprotein profile, and lipoprotein lipase in hyperlipidaemic mouse models. Cardiovasc Res. 2013;99:716–723. doi:10.1093/cvr/cvt148
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.