Back to Journals » Journal of Pain Research » Volume 15
Efficacy and Safety of Ketamine in the Treatment of Neuropathic Pain: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Authors Guimarães Pereira JE , Ferreira Gomes Pereira L , Mercante Linhares R , Darcy Alves Bersot C, Aslanidis T , Ashmawi HA
Received 25 January 2022
Accepted for publication 17 March 2022
Published 9 April 2022 Volume 2022:15 Pages 1011—1037
DOI https://doi.org/10.2147/JPR.S358070
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
José Eduardo Guimarães Pereira,1,2 Lucas Ferreira Gomes Pereira,3 Rafael Mercante Linhares,4 Carlos Darcy Alves Bersot,5 Theodoros Aslanidis,6 Hazem Adel Ashmawi1
1Laboratório de Investigação Médica (LIM/08), Department of Anesthesiology, University of São Paulo Medical School - FMUSP, São Paulo, São Paulo, 05403-000, Brazil; 2Department of Anesthesiology at Hospital Unimed Volta Redonda, Volta Redonda, Rio de Janeiro, 27259-000, Brazil; 3Department of Anesthesiology, Centro Médico de Campinas, Campinas, São Paulo, 13083-190, Brazil; 4Department of Anesthesiology, Hospital Municipal Miguel Couto, Rio de Janeiro, Rio de Janeiro, 22430-160, Brazil; 5Department of Anesthesiology, Hospital Federal da Lagoa, Rio de Janeiro, Rio de Janeiro, 22470-050, Brazil; 6Intensive Care Unit, Saint Paul General Hospital, Thessaloniki, Central Macedonia, 54633, Greece
Correspondence: José Eduardo Guimarães Pereira, Laboratório de Investigação Médica (LIM/08), Department of Anesthesiology, University of São Paulo Medical School - FMUSP, Av. Dr. Enéas de Carvalho Aguiar, 155, 2nd Floor, Room 2120, Setor Azul, Prédio dos Ambulatórios, São Paulo, São Paulo, 05403-000, Brazil, Email [email protected]
Purpose: Ketamine is a N-methyl-D-aspartate (NMDA) antagonist with strong analgesic properties. Its addition to the treatment of neuropathic pain may reduce pain intensity and improve overall quality of life. A systematic review and meta-analysis of randomized controlled trials was performed to investigate the addition of ketamine to the treatment of patients with neuropathic pain.
Patients and Methods: GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach was used to rate the overall certainty of the evidence for each outcome. Eighteen (18) randomized controlled trials including 706 participants were included for further analysis.
Results: Ketamine addition to standard treatment of neuropathic pain (NP) resulted in a statistically significant reduction of pain intensity at one week after the end of treatment with ketamine (MD − 2.14, 95% CI − 2.65 to − 1.63; p< 0.00001) and after 30 days after the end of treatment with ketamine (MD − 1.68, 95% CI − 2.25 to − 1.12; p< 0.00001) and a statistically significant increase in discomfort (RR 4.06; 95% CI 1.18 to 13.95; p=0.03), and psychedelic effects (RR 4.94; 95% CI 2.76 to 8.84; p< 0.00001).
Conclusion: There is a statistically significant pain reduction by adding ketamine to the treatment of chronic NP when compared to the standard treatment. However, such pain reduction comes at the expense of adverse outcomes, especially psychedelic effects related to the administration of ketamine. However, the overall quality of certainty of evidence is low due to the clinical heterogeneity among the intervention characteristics of the trials analyzed (different administration routes, dosing regimen, therapy durations, different clinical characteristics of the population investigated). Future large multi-centered trials are necessary to confirm or not the results of the present review.
Keywords: ketamine, chronic pain, neuropathy, neuralgia, treatment
Introduction
Although data on neuropathic pain prevalence in the general population may not be accurate due to different definitions and evaluation methods, estimations indicate that between 6.9 and 10% of the global population are affected by some type of neuropathic pain.1 The World Health Organization (WHO) has estimated that 22% of the world’s primary care patients have chronic debilitating pain making chronic pain a problem to be addressed by all physicians and health professionals.2–4
Neuropathic pain (NP) may develop after a nerve injury or disease, with changes occurring downwards and upwards along the modulating pathways of the injured neuron.
Three factors distinguish NP from others types of pain: a) there is no transduction (conversion of noxious stimulus to electric signal); b) the prognosis is poor: pain from injuries in nervous tissues is more likely to become chronic; c) NP is refractory to therapy with conventional analgesics, such as non-steroidal anti-inflammatory drugs (NSAIDs) and opioids.5 A variety of substances are involved in the development and maintenance of NP, which may explain the high incidence of comorbidities associated with it and why drugs that are effective in other conditions can also be effective in NP.6–9 However, in case of NP etiological therapy is rarely effective. Thus, pain relief is the primary focus.7
N-Methyl-D-aspartate receptors (NMDARs) are ionotropic glutamate receptors that play a role in synaptic transmission, in neuroplasticity, and in learning and memory processes. Alterations on NMDARs functions are involved in some of the nervous system disorders, such as neuropathic pain. Therefore, they have been extensively investigated as possible therapeutic targets for pain management.
In such context ketamine has been receiving new attention and its role has expanded from general anesthesia to depression treatment,10 multimodal analgesia,11 as an anti-hyperalgesic, and in the treatment of NP.12,13
The purpose of the systematic review and meta-analysis is to assess the impact of the addition of ketamine to the treatment of patients suffering from chronic NP.
Materials and Methods
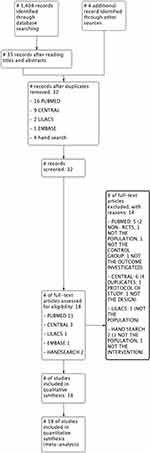
Methodology was followed The Cochrane Handbook for Intervention Reviews.14 This systematic review was registered in PROSPERO International Prospective Register of Systematic Reviews (number CRD42020203060) and is reported in accordance with PRISMA Preferred Reporting Items for Systematic Reviews and Meta-analysis statement (PRISMA) statement.15 (Figure 1 – PRISMA Checklist).
Search Strategy
The search was performed in the following electronic databases: the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE via PubMed (OvidSP), LILACS (Literatura Latino-Americana e do Caribe em Ciências da Saúde), Web of Science and the EMBASE (Excerpta Medica dataBASE). The databases were searched for published RCTs with no restrictions to date, from inception to the last search performed on November 18th, 2021.
The search was conducted using multiple combinations of the following keywords: “neuralgia” and “ketamine” (Appendix 1 – search strategy). No language or publication status restrictions were imposed. In addition, an online search for additional eligible studies was conducted in the Science Research website, and we also hand searched the reference lists of included studies.
Eligibility Criteria and Study Selection
We considered all RCTs evaluating the addition of ketamine, by any route of administration, to the standard treatment compared to standard treatment (ST) plus placebo, or ST alone, in adult patients with NP. (Appendix 2– eligibility criteria). Using standardized screening forms (Appendix 3 – data extraction form), two reviewers (JEGP, LFGP) independently screened all titles and abstracts identified by the literature search, obtained full-text articles of all potentially eligible studies, and evaluated these studies for eligibility. Reviewers resolved the disagreement through discussion, and with third-party adjudication if necessary.
Primary and Secondary Outcomes
The primary outcome of this review was pain measured by numerical rating scale (NRS) or visual analog scale (VAS). Secondary outcomes were the following: worst pain score measured by numerical rating scale, least pain score measured by numerical rating scale, pain interference on life appreciation, pain measured by multidimensional pain scales, quality of life, mood, impact on interpersonal interactions, quality of sleep, impact on general daily activities, impact on work, and adverse outcomes (eg, psychedelic effects, nausea, and vomiting) Eligible studies reported on one or more of the outcomes listed above.
Data Extraction and Quality Assessment
Two reviewers (JEGP, LFGP) independently extracted the following data using a pre-piloted, standardized data extraction form: characteristics of the study design; participants; interventions; outcomes and the length of follow-up. If eligible articles had missing data, we contacted authors for clarification.
Reviewers independently assessed the validity of included studies using the risk of bias approach for Cochrane reviews.14,16,17 Risk of bias was assessed using five separate criteria: adequacy of sequence generation, allocation sequence concealment, blinding (investigators, patients, collectors, statistician, outcome assessors), incomplete outcome data, and selective outcome reporting. For incomplete outcome data, we considered loss to follow-up enough to induce clinically relevant bias as high risk of bias.
Certainty of Evidence
We used the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach to rate the certainty of evidence, in which a body of evidence based on randomized trials begins as high certainty evidence but may be rated down by one or more levels for each of five categories of limitations: risk of bias, inconsistency, indirectness, imprecision and reporting bias.18 Detailed GRADE guidance was used to assess the overall risk of bias, imprecision,19 inconsistency,16 indirectness20 and publication bias,21 and results were summarized in an evidence profile table.
Statistical Analysis
We calculated pooled risk ratios (RRs) for dichotomous outcomes, mean differences (MD) for continuous outcomes and standardized mean difference (SMD) for continuous outcomes measured by different scales, with the corresponding 95% confidence interval (CI). We used a random-effects model with the Mantel-Haenszel statistical method for the dichotomous outcomes and the Inverse Variance for the continuous outcomes. We addressed variability in results across studies using the I2 statistic and the P value (>0.10) obtained from the Cochrane chi-square test.
Risk-ratio does not incorporate zero-event trials, thereby excluding these trials and data from the combined estimate. A random-effect model was chosen because when dealing with a series of studies, subjects differ substantially from one study to another.22
Our primary analyses were based on all randomized patients who had reported outcomes for each study (complete case analysis). We used Review Manager (RevMan®) (version 5.3; Nordic Cochrane Centre, Cochrane Collaboration) for all analyses.23 We planned to perform sensitivity and pre-specified subgroup analyses according to different regimens of administration, but data was insufficient to perform those analyses. Publication biases were assessed via visual inspection of funnel plots for outcomes with 10 or more studies.24
Results
Search results
We identified a total of 1404 studies plus 4 additional studies through hand search. After independent screening by title, and then by abstract, we selected 39 studies, and after duplicate removal, 32 studies were deemed eligible for inclusion in the review. Of those, 14 did not fulfill our eligibility criteria and were excluded (Figure 1. PRISMA flow diagram). We, therefore, included 18 studies Max with a total of 706 participants in this review.25–42
Characteristics of the Included Studies
Among the 18 eligible randomized control studies (RCTs), they took place in a variety of settings (Table 1) including Brazil,29,32,41,42 Canada,36 Denmark,35 Egypt,27,28 France,34 Italy,40 Netherlands,25,30,37 Norway,38 South Korea,31 United Kingdom,33 United States of America.26,39 Sample sizes ranged from eight38 to 21435 participants (Table 1).
 |  |  |  |  |  |
Table 1 Study Characteristics According to Population and Setting |
A total of two trials with 20 participants,29,38 included exclusively patients with post-herpetic neuralgia, while only one trial included 8 patients exclusively suffering from post-traumatic neuralgia;39 two trials with 224 participants, included exclusively patients with cancer related neuropathic pain;33,40 two trials, with 45 participants, included patients with neuralgia from direct damage to either central or peripheral nervous system;35,37 two trials included 79 patients suffering from complex regional pain syndrome (CRPS);25,26 one trial with 92 participants, stated to encompass all types of neuropathic pain36 and eight trials, with 231 participants, did not specify the etiology of neuropathic pain.27,28,30–32,34,41,42 (Table 1).
All except one of our eligible trials included both male and female participants, with one trial including only female patients.39 Males represented 53.82% of the overall population studied (excluding the studies that are not specified). There were two studies that did not specify the gender distribution of the population.31,33 The mean age of the participants ranged from 4039 to 71.938 years (Table 1).
A set of diverse protocols has been adopted for ketamine administration across different trials. Control group in 13 RCTs (558 participants) received placebo.25–30,33,35–40 The duration of treatment with ketamine ranged from one day30 to 12 weeks.25,26,32
Different routes of treatment have been adopted for ketamine administration across the trials, with eleven trials adopting the intravenous route,25,26,28,30,31,33–35,38–40 two trials adopting the epidural route,27,41 two trials administering ketamine through the oral route32,42 and three trials adopting the topical route for treatment administration.29,36,37
A wide range of doses have been utilized, beginning at 0.1 mg/kg/day41 and going up until 0.75 mg/kg/day.39 (Table 2).
 |  |  |  |
Table 2 Study Characteristics Related to Description of Intervention, Control, and Outcomes |
Risk of Bias in Individual Studies
The overall quality of all included studies was considered high. Random sequence generation, allocation concealment, blinding of participants and personnel and selective reporting were all considered of low risk of bias. Publication bias could not be evaluated since no single meta-analysis pooled more than 10 studies. Four studies presented high risk of bias derived from loss to follow-up.26,32,33,36 (Figure 2; Table 3)
 |
Table 3 Risk of Bias |
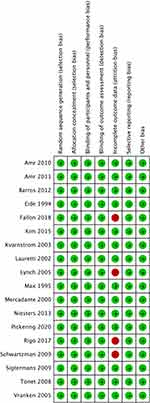 |
Figure 2 Risk of bias according to different domains. |
Effectiveness of Interventions
Primary Outcome: Pain
Overall Pain Reduction Compared to Standard Treatment
Results from six RCTs (212 patients)28–35,31,37 yielded a statistically significant reduction of pain between ketamine and standard treatment (ST) (MD −1.68, 95% CI −2.39 to −0.96; I2= 90%; p < 0.00001) (Figure 3). The certainty of evidence was rated as low because of imprecision (low number of patients (<400) and inconsistency. No publication bias was detected. We were unable to perform sensitivity analysis due to the small number of trials included in this analysis.
 |
Figure 3 Meta-analysis on the overall mean pain reduction. Forest plot is representing the comparison of the overall mean pain between group ketamine and ST. |
Pain Reduction Compared to Standard Treatment at Different Time Periods
Results from five RCTs (182 patients)25–27,29,42 yielded a statistically significant reduction of pain until after one-week post-treatment between ketamine and ST (MD −2.14, 95% CI −2.65 to −1.63; I2= 79%; p < 0.00001) (Figure 4). A mean reduction of 46% compared to baseline pain. The certainty of evidence was rated as low because of imprecision (low number of patients: <400) and inconsistency (Table 4). No publication bias was detected.
 |
Table 4 GRADE Evidence Profile for Clinical Outcomes |
 |
Figure 4 Meta-analysis on the average mean pain reduction. Forest plot is representing the comparison of the mean pain between group ketamine and ST according to time after the end of treatment. |
Results from five RCTs (152 patients)26–28,31,42 yielded a statistically significant reduction in pain between ketamine and ST until up to two weeks post-treatment (MD −1.30, 95% CI −2.04 to −0.57; I2= 84%; p = 0.0005) (Figure 4). A mean reduction of 28% compared to baseline pain The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals) and inconsistency (Table 4). No publication bias was detected.
Results from 4 RCTs (122 patients)26–28,42 yielded a statistically significant reduction on pain after until 30 days post-treatment between ketamine and ST (MD −1.68, 95% CI −2.25 to −1.12; I2= 77%; p < 0.00001) (Figure 4). A mean reduction of 36% compared to baseline pain The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence) and inconsistency (Table 4).
The test for subgroup differences yielded a I2=45% (Figure 4), thus revealing a consistent effect of ketamine compared to ST across the different time points after the treatment. No publication bias was detected.
Pain Reduction Compared to Baseline Pain Levels Over Time
Results from five RCTs (181 patients)25–28,42 yielded a statistically significant reduction of pain between baseline values and after one week of treatment with ketamine (MD −4.12, 95% CI −5.72 to −2.51; I2= 98%; p < 0.00001) (Figure 5). The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals) and inconsistency. No publication bias was detected.
 |
Figure 5 Meta-analysis on the average mean pain reduction over time. Forest plot is representing the comparison of the mean pain in the ketamine at different time points compared to baseline pain. |
Results from other five RCTs (151 patients)26–28,31,42 yielded a statistically significant reduction of pain between baseline values and after two weeks of treatment with ketamine (MD −3.60, 95% CI −4.75 to −2.44; I2= 94%; p < 0.00001) (Figure 5). The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals) and inconsistency. No publication bias was detected.
Results from three RCTs (103 patients)27,28,42 yielded a statistically significant reduction of pain between baseline values and after four weeks of treatment with ketamine (MD −3.86, 95% CI −4.51 to −3.21; I2= 78%; p < 0.00001) (Figure 5). The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals) and inconsistency. No publication bias was detected.
Results from two RCTs (58 patients)26,27 yielded a statistically significant reduction of pain between baseline values and after two months of treatment with ketamine (MD −2.22, 95% CI −4.22 to −0.21; I2= 95%; p = 0.03) (Figure 5). The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals) and inconsistency. No publication bias was detected.
Results from two RCTs (40 patients)26,32 yielded a non-statistically significant difference in pain between baseline values and after three months of treatment with ketamine (MD −3.22, 95% CI −7.66 to 1.22; I2= 98%; p = 0.15) (Figure 5). The certainty of evidence was rated as very low because of imprecision (low number of patients (<400) and wide confidence intervals including both benefit and harm) and inconsistency.
The test for subgroup differences yielded a I2=0%, thus revealing a consistent effect of ketamine across the different time points after the treatment. No publication bias was detected.
Pain Reduction Measured by Multidimensional Scales
Results from five RCTs (356 patients)31,33,34,36,37 yielded a non-statistically significant difference between ketamine and ST on the reduction of pain at different multidimensional pain scales (MD −0.02, 95% CI −0.43 to 0.39; I2= 0%; p = 0.93) (Figure 6). The certainty of evidence was rated as low because of imprecision (low number of patients (<400) and confidence intervals including clinically important benefit and harm) and no publication bias was detected.
Secondary Outcome
Adverse Outcomes
Psychedelic Effects
Results from nine RCTs (266 patients)25,28,30,32,34,35,37,38,40 yielded a statistically significant increase in the number of psychedelic effects when ketamine was used for the treatment of patients with NP compared to ST (RR 4.94, 95% CI 2.76 to 8.84; events (STK:56/132, ST:8/134); I2= 0%; p < 0.00001) (Figure 7). The certainty of evidence was rated as low because of imprecision (low number of events (<400) and wide confidence intervals) (Table 4) and no publication bias was detected (Table 4).
 |
Figure 7 Meta-analyses on the incidence of adverse outcomes. Forest plot is representing the comparison between group ketamine and ST according to different adverse outcomes. |
Discomfort
Results from two RCTs (40 patients)35,38 yielded a statistically significant increase in the number of patients reporting general discomfort when ketamine was used for the treatment of NP compared to ST (RR 4.06, 95% CI 1.18 to 13.95; events (STK:12/20, ST:2/20); I2= 0%; p = 0.03) (Figure 7). The certainty of evidence was rated as low because of imprecision (low number of events (<400) and wide confidence intervals) (Table 4) and no publication bias was detected (Table 4).
Nausea and Vomiting, Fatigue and Dizziness
There was no difference between ketamine and ST regarding nausea and vomiting, fatigue, and dizziness (Figure 7). The certainty of evidence was rated as low for nausea and vomiting, fatigue, dizziness because of imprecision (low number of events (<400) and wide confidence intervals including both clinically important benefit and harm) and no publication bias was detected.
Adverse Outcomes (Composite Analysis)
An overall analysis on adverse outcomes did not detect a significant difference between the two groups. The certainty of evidence was rated as low because of imprecision (low number of events (<400) and wide confidence intervals including both clinically important benefit and harm) and no publication bias was detected according to the characteristic Christmas tree shape of the Funnel Plot (Figure 8).
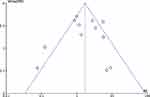 |
Figure 8 Publication bias. Funnel Plot representing the distribution of studies according to their results. |
We were unable to perform the following pre-specified analyses due to the lack of data from the included studies: Worst pain score measured by numerical rating scale; Least pain score measured by numerical rating scale; pain interference on general activities by numerical scale; pain interference on mood activities by numerical scale; pain interference on ability to walk by numerical scale; pain interference on work by numerical scale; pain interference on personal relationships by numerical scale; pain interference on sleep by numerical scale; pain interference on life appreciation by numerical scale.
We tried to contact authors to ask for further information regarding the missing data, but unfortunately no further information has been provided.
Discussion
Main Findings
There is a statistically significant pain reduction by adding ketamine to the treatment of chronic NP when compared to the standard treatment. However, such pain reduction comes at the expense of adverse outcomes, especially psychedelic effects related to the administration of ketamine. This raises questions such as whether these undesirable effects would make patients drop out of treatment and whether (S+) ketamine could provide the same amount of pain relief, but still sparing patients from experiencing such inconvenient side effects.
Strengths and Limitations
To our knowledge this is the most extensive search (no language or publication status restrictions) of the potential use of ketamine as an adjunct to NP treatment in the literature to date. We included studies in which ketamine was administered via different routes, dosing regimen, with different therapy duration. We independently rated the overall certainty of evidence (GRADE approach) for each outcome, extracted and analyzed data on the main outcomes deemed most important for stakeholders. We also assessed publication bias for the outcomes with ten or more studies included and the only funnel plot performed did not suggest the possibility of publication bias (Figure 7).
The primary limitation of our review is the low certainty of evidence due to study limitations, mostly due to inconsistency and imprecision. Thus, revealing the existence of great heterogeneity among ketamine administration protocols and the different clinical presentations and pathophysiology of neuropathic pain conditions. Ketamine was administered through the intravenous route in most of the studies included in this review. Amongst those studies the doses ranged from 0.5 to 1.5 mg/kg/day and duration of treatment lasted up to 10 days, and with pain reduction lasting up to one month.
Despite the moderate number of identified trials,17 the certainty of evidence is low to very low due to the low number of patients/events included in each individual meta-analysis, causing imprecision to be a major limiting factor to the quality of the yielded evidence.
Another limitation of this study is that we were unable to perform sensitivity analyses to explore the causes of high heterogeneity due to the small number of studies included in each meta-analysis.
Finally, due to a variety of outcomes, we could not pool the data of all eligible studies in all the proposed meta-analyses for this review, as this may limit the strength of evidence from yielded results.
Relation to Previous Studies
Six systematic reviews have been published in the recent past relevant to our study objectives.43–48. One review,43 although not performing meta-analysis, evaluated the administration of ketamine along with other NMDA receptor antagonist drugs. Its results suggested that introducing ketamine to the treatment of patients with neuropathic pain brought benefits to patients, but in this review, there were no meta-analysis performed and it also included studies in which patients were evaluated while still on ketamine infusion, therefore turning its conclusions very unlikely to be translated into everyday practice.
Another one limited itself to investigate the use of ketamine strictly through the intravenous route.48 Orhurhu et al48 found, like in our study, significant pain reduction at two weeks after the ketamine infusion in a meta-analysis including only two trials where patients suffered from neuropathic pain and no further benefits at time points thereafter.
Other reviews were less extensive in terms of the population, limiting their scope to only specific clinical conditions presenting neuropathic pain, such as complex regional pain syndrome (CRPS) without a broader and more extensive analysis encompassing several neuropathic pain conditions.43–46
Only one review had a similar approach to ours.47 Nonetheless, at the time (2010) limited itself to RCTs where ketamine was administered through the intravenous route in patients with neuropathic pain post limb amputation. And it is worth noting that neither one of these reviews used the Grading of Recommendations Assessment, and the Development and Evaluation (GRADE) approach to rate the certainty of evidence.
Clinical Implications of the Study
Results showed that ketamine reduced significantly (ranging from 28% to 46%), neuropathic pain compared to the baseline pain in the control group; and its effects can last for up to two months after the end of treatment. Thus, ketamine could be an attractive option to both patients and care providers for the management of breakthrough pain episodes and for cases refractory to standard care.
Although ketamine administration does not seem to cause serious adverse outcomes, our meta-analysis revealed a significant increase in psychedelic effects. Nevertheless, considering that intravenous ketamine administration takes place in Hospital facilities, those side effects would be safely manageable; hence moving the risk-benefit relation in favor of ketamine. In addition, those side effects could be decreased or avoided by choosing (S+) ketamine instead of the racemic ketamine. Yet, only in one study S(+) ketamine was the treatment of choice,37 and its results found a non-significant reduction in pain after the administration of ketamine through intravenous route.
Research Perspectives
Based on the data from this systematic review to determine the potential efficacy of ketamine for neuropathic pain treatment, we found statistically significant results on pain reduction, but we lacked quality of evidence due to imprecision from small sample size and clinical heterogeneity among included studies.
Some insights may be drawn from our results. Although it was not possible to perform the pre-specified meta-analyses based on different characteristics of ketamine therapy regimen, the most prevalent route of administration was the intravenous route, with 11 trials adopting such route doses ranged from 0.15 to 1.5 mg/Kg/day,25,26,28,30,31,33–35,37–40 with one trial using doses from 1.7 up to 10 mg/Kg/day.25 The duration of treatment was under 10 days in 8 out of these 11 trials that opted for the intravenous route.25,26,28,30,31,34,35,38 Plus, all studies from which we could retrieve data used racemic ketamine as the drug of choice for the study. Thus, we believe that more randomized clinical trials should be performed to determine the efficacy of ketamine for the treatment of neuropathic pain.
Conclusion
This comprehensive meta-analysis of 18 RCTs provides current evidence for the addition of ketamine to the standard treatment of patients suffering from neuropathic pain. It confirms previous observations that ketamine can be safely administered to patients, although revealing a significant increase in psychedelic effects amongst these patients. It also demonstrates with very low quality of evidence that the addition of ketamine to the standard treatment of patients with neuropathic pain may be more efficacious than the standard treatment alone on reducing neuropathic pain for as long as two months after the end of the treatment.
Abbreviations
CENTRAL, Central Register of Controlled Trials; CRPS, Complex Regional Pain Syndrome; EMBASE, Excerpta Medica database; FDA, Food and Drug Administration office; GRADE, Grading of Recommendations Assessment, Development and Evaluation; LILACS, Literatura Latino-Americana e do Caribe em Ciências da Saúde; MEDLINE, Medical Literature Analysis and Retrieval System Online; NMDA, N-methyl-D-aspartate; NMDARs, N-methyl-D-aspartate receptors; NP, neuropathic pain; NSAIDs, non-steroidal anti-inflammatory drugs; NRS, numerical rating scale; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-analysis; PROSPERO, International Prospective Register of Systematic Reviews; RCTs, randomized controlled trials; ST, standard of treatment; VAS, visual analog scale; WHO, World Health Organization.
Acknowledgments
We would like to thank Dr. Per Kristian Eide, for replying to our queries.
Consent for Publication
The authors confirm that all the contents in this review can be published.
Funding
Centro de Estudos em Anestesia e Reanimação do Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (FMUSP) (publication fees).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Van Hecke O, Austin SK, Khan RA, et al. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain. 2014;155(4):654–662. doi:10.1016/j.pain.2013.11.013
2. Lépine JP, Briley M. The epidemiology of pain in depression. Hum Psychopharmacol. 2004;19(S1):1. doi:10.1002/hup.618
3. Portenoy RK, Ugarte C, Fuller I, Haas G. Population-based survey of pain in the United States: differences among white, African American, and Hispanic subjects. J Pain. 2004;5(6):317–328. doi:10.1016/j.jpain.2004.05.005
4. Yawn BP, Wollan PC, Weingarten TN, et al. The prevalence of neuropathic pain: clinical evaluation compared with screening tools in a community population. Pain Med. 2009;10(3):586–593. doi:10.1111/j.1526-4637.2009.00588.x
5. Cohen SP, Mao J. Neuropathic pain: mechanisms and their clinical implications. BMJ. 2014;348:f7656. doi:10.1136/bmj.f7656
6. Colloca L, Ludman T, Bouhassira D, et al. Neuropathic pain. Nat Rev Dis Primers. 2017;3(1):17002. doi:10.1038/nrdp.2017.2
7. Cavalli E, Mammana S, Nicoletti F, Bramanti P, Mazzon E. The neuropathic pain: an overview of the current treatment and future therapeutic approaches. Int J Immunopathol Pharmacol. 2019;33:2058738419838383. doi:10.1177/2058738419838383
8. Urits I, Li N, Berardino K, et al. The use of antineuropathic medications for the treatment of chronic pain. Best Prac Res Clin Anaesthesiol. 2020;34(1):493–506. doi:10.1016/j.bpa.2020.08.007
9. Hylands-White N, Duarte RV, Raphael JH. An overview of treatment approaches for chronic pain management. Rheumatol Int. 2016;37(1):29–42. doi:10.1007/s00296-016-3481-8
10. Zarate CA, Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856–864. doi:10.1001/archpsyc.63.8.856
11. Raeder JC, Stenseth LB. Ketamine: a new look at an old drug. Curr Opin Anaesthesiol. 2000;13(4):463–468. doi:10.1097/00001503-200008000-00011
12. Kissin I, Bright CA, Bradley EL. The effect of ketamine on opioid-induced acute tolerance: can it explain reduction of opioid consumption with ketamine-opioid analgesic combinations? Anesth Analg. 2000;91(6):1483–1488. doi:10.1097/00000539-200012000-00035
13. Hocking G, Cousins MJ. Ketamine in chronic pain management: an evidence-based review. Anesth Analg. 2003;97(6):1730–1739. doi:10.1213/01.ANE.0000086618.28845.9B
14. Higgins JPT, Altman DG, Gøtzsche PC, et al. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343(7829):d5928. doi:10.1136/bmj.d5928
15. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–34. doi:10.1016/j.jclinepi.2009.06.006
16. Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines: 7. Rating the quality of evidence - Inconsistency. J Clin Epidemiol. 2011;64(12):1294–1302. doi:10.1016/j.jclinepi.2011.03.017
17. Balshem H, Helfand M, Schünemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64(4):401–406. doi:10.1016/j.jclinepi.2010.07.015
18. Guyatt GH, Oxman AD, Vist G, et al. GRADE guidelines: 4. Rating the quality of evidence - Study limitations (risk of bias). J Clin Epidemiol. 2011;64(4):407–415. doi:10.1016/j.jclinepi.2010.07.017
19. Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines 6. Rating the quality of evidence - Imprecision. J Clin Epidemiol. 2011;64(12):1283–1293. doi:10.1016/j.jclinepi.2011.01.012
20. Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines: 8. Rating the quality of evidence - Indirectness. J Clin Epidemiol. 2011;64(12):1303–1310. doi:10.1016/j.jclinepi.2011.04.014
21. Guyatt GH, Oxman AD, Montori V, et al. GRADE guidelines: 5. Rating the quality of evidence - Publication bias. J Clin Epidemiol. 2011;64(12):1277–1282. doi:10.1016/j.jclinepi.2011.01.011
22. Lane PW. Meta-analysis of incidence of rare events. Stat Methods Med Res. 2013;22(2):117–132. doi:10.1177/0962280211432218
23. Cochrane Handbook for Systematic Reviews of Interventions | Cochrane Training. Available from: https://training.cochrane.org/handbook/current.
24. Sun X, Ioannidis JPA, Agoritsas T, Alba AC, Guyatt G. How to use a subgroup analysis users’ guide to the medical literature. JAMA. 2014;311(4):405–411. doi:10.1001/jama.2013.285063
25. Sigtermans MJ, van Hilten JJ, Bauer MCR, et al. Ketamine produces effective and long-term pain relief in patients with complex regional pain syndrome type 1. Pain. 2009;145(3):304–311. doi:10.1016/j.pain.2009.06.023
26. Schwartzman RJ, Alexander GM, Grothusen JR, Paylor T, Reichenberger E, Perreault M. Outpatient intravenous ketamine for the treatment of complex regional pain syndrome: a double-blind placebo-controlled study. Pain. 2009;147(1–3):107–115. doi:10.1016/j.pain.2009.08.015
27. Amr Y. Epidural ketamine in post spinal cord injury-related chronic pain. Anesth Essays Res. 2011;5(1):83–86. doi:10.4103/0259-1162.84196
28. Amr Y. Multi-day low dose ketamine infusion as adjuvant to oral gabapentin in spinal cord injury related chronic pain: a prospective, randomized, double blind trial. Pain Physician. 2010;13(3):245–249. doi:10.36076/ppj.2010/13/245
29. Antonio Moreira de Barros G, Amante Miot H, Massarico Braz A, Ramos F, Aristoteles Borges M. Topical (S)-ketamine for pain management of postherpetic neuralgia * (S)-cetamina tópica no tratamento da dor da neuralgia pós-herpética. An Bras Dermatol. 2012;87(3):504–505. doi:10.1590/s0365-05962012000300032
30. Niesters M, Aarts L, Sarton E, Dahan A. Influence of ketamine and morphine on descending pain modulation in chronic pain patients: a randomized placebo-controlled cross-over proof-of-concept study. BJA. 2013;110(6):1010–1016. doi:10.1093/bja/aes578
31. Kim YH, Lee PB, Oh TK. Is magnesium sulfate effective for pain in chronic postherpetic neuralgia patients comparing with ketamine infusion therapy? J Clin Anesth. 2015;27(4):296–300. doi:10.1016/j.jclinane.2015.02.006
32. Ferreira J, Karine Rigo F, Trevisan G, et al. Management of neuropathic chronic pain with methadone combined with ketamine: a randomized, double blind, active-controlled clinical trial. Pain Physician. 2017;20(3):207–215. doi:10.36076/ppj.2017.215
33. Fallon MT, Wilcock A, Kelly CA, et al. Oral ketamine vs placebo in patients with cancer-related neuropathic pain: a randomized clinical trial. JAMA Oncol. 2018;4(6):870–872. doi:10.1001/jamaoncol.2018.0131
34. Pickering G, Pickering G, Pereira B, et al. Ketamine and magnesium for refractory neuropathic pain: a randomized, double-blind, crossover trial. Anesthesiology. 2020;133(1):154–164. doi:10.1097/ALN.0000000000003345
35. Kvarnstrom A, Karlsten R, Quiding H, Emanuelsson B-M, Gordh T. The effectiveness of intravenous ketamine and lidocaine on peripheral neuropathic pain. Acta Anaesthesiol Scand. 2003;47(7):868–877. doi:10.1034/j.1399-6576.2003.00187.x
36. Lynch ME, Clark AJ, Sawynok J, Sullivan MJL. Topical 2% amitriptyline and 1% ketamine in neuropathic pain syndromes a randomized, double-blind, placebo-controlled trial. Anesthesiology. 2005;103(1):140–146. doi:10.1097/00000542-200507000-00021
37. Vranken JH, Dijkgraaf MGW, Kruis MR, van Dasselaar NT, van der Vegt MH. Iontophoretic administration of S(+)-ketamine in patients with intractable central pain: a placebo-controlled trial. Pain. 2005;118(1–2):224–231. doi:10.1016/j.pain.2005.08.020
38. Eide P, Jorum E, Stubhaug A, Bremnes J, Brcivik H. Relief of post-herpetic neuralgia with the N-methyl-D-asp&c acid receptor antagonist ketamine: a double-blind, cross-over comparison with morphine and placebo. Pain. 1994;58(3):347–354. doi:10.1016/0304-3959(94)90129-5
39. Max M, Byas-Smith G, Gracely R, Bennett G. Intravenous infusion of the NMDA antagonist, Ketamine, in chronic posttraumatic pain with Allodynia: a double-blind comparison to alfentanil and placebo. Clin Neuropharmacol. 1995;18(4):360–368. doi:10.1097/00002826-199508000-00008
40. Mercadante S, Arcuri E, Tirelli W, Casuccio A. Analgesic effect of intravenous ketamine in cancer patients on morphine therapy: a randomized, controlled, double-blind, crossover, double-dose study. J Pain Symptom Manage. 2000;20(4):246–252. doi:10.1016/s0885-3924(00)00194-9
41. Rocha Lauretti G, de Menezes Rodrigues A, Maria Alves Gomes J, Paulino Dos Reis M. Epidural Ketamine Versus Epidural Clonidine as Therapeutic for Refractory Neuropathic Chronic Pain [Avaliação Clínica Comparativa entre a Cetamina e a Clonidina por Via Peridural no Tratamento da Dor Crônica Neuropática]. Rev Bras Anestesiol. 2002;52(1):34–40. doi:10.1590/S0034-70942002000100005
42. Tonet C, Sakata R, Issy A, Garcia J, Marcelino A. Evaluation of oral ketamine for neuropathic pain [Avaliação da cetamina oral para dor neuropática]. Rev Bras Med. 2008;65(7):214–218.
43. Humble SR, Dalton AJ, Li L. A systematic review of therapeutic interventions to reduce acute and chronic post-surgical pain after amputation, thoracotomy, or mastectomy. Eur J Pain. 2015;19(4):451–465. doi:10.1002/ejp.567
44. Wei Y, Song D, He A, Xu R, Xiu X. Systematic review efficacy of pain relief in different postherpetic neuralgia therapies: a network meta-analysis. Pain Physician. 2018;21:19–32. doi:10.36076/ppj.2018.1.19
45. Zhao J, Wang Y, Wang D. The effect of ketamine infusion in the treatment of complex regional pain syndrome: a systemic review and meta-analysis. Curr Pain Headache Rep. 2018;22:12. doi:10.1007/s11916-018-0664-x.
46. Aiyer R, Mehta N, Gungor S, Gulati A. A systematic review of NMDA receptor antagonists for treatment of neuropathic pain in clinical practice. Clin J Pain. 2018;34(5):450–467. doi:10.1097/AJP.0000000000000547
47. Collins S, Sigtermans MJ, Dahan A, Zuurmond WW, Perez RS. NMDA receptor antagonists for the treatment of neuropathic pain. Pain Med. 2010;11(11):1142–1726. doi:10.1111/j.1526-4637.2010.00981.x
48. Orhurhu V, Orhurhu MS, Bhatia A, Cohen SP. Ketamine infusions for chronic pain: a systematic review and meta-analysis of randomized controlled trials. Anesth Analg. 2019;129(1):241–254. doi:10.1213/ANE.0000000000004185
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.