Back to Journals » Journal of Pain Research » Volume 12
Effectiveness of pregabalin for treatment of chronic cervical radiculopathy with upper limb radiating pain: an 8-week, multicenter prospective observational study in Japanese primary care settings
Authors Taguchi T, Nozawa K, Parsons B , Yoshiyama T, Ebata N , Igarashi A, Fujii K
Received 23 October 2018
Accepted for publication 14 February 2019
Published 3 May 2019 Volume 2019:12 Pages 1411—1424
DOI https://doi.org/10.2147/JPR.S191906
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Michael A Ueberall
Toshihiko Taguchi,1 Kazutaka Nozawa,2 Bruce Parsons,3 Tamotsu Yoshiyama,4 Nozomi Ebata,2 Ataru Igarashi,5 Koichi Fujii2
1Japan Organization of Occupational Health and Safety, Yamaguchi Rosai Hospital, Sanyo-Onoda, Yamaguchi, Japan; 2Medical Affairs, Pfizer Japan Inc, Tokyo, Japan; 3Global Medical Product Evaluation, Pfizer Inc, New York, NY, USA; 4Biometrics and Data Management, Development Japan, Pfizer R&D Japan G.K., Tokyo, Japan; 5Department of Drug Policy and Management, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Tokyo, Japan
Background: Despite high prevalence of chronic neck pain in Japan and the negative impact pain has on patient’s quality of life (QoL), the therapeutic value of pregabalin for chronic neck pain with a neuropathic pain (NeP) component has not been assessed in a typical Japanese health care setting.
Methods: An 8-week, non-interventional, multicenter, observational study of Japanese adults (≥20 years) with chronic refractory cervical pain including a NeP element (for ≥12 weeks) and sleep disturbance on the Pain-Related Sleep-Interference Scale (PRSIS) ≥1 (from 0 “does not interfere with sleep” to 10 “completely interferes”). Patients received either usual care with conventional analgesics or pregabalin (150–600 mg/day) for 8 weeks. “Usual care” with analgesics or other treatment(s) was determined based on physician’s best clinical judgment. Primary endpoint was change from baseline to week 8 in PRSIS. Secondary endpoints included: change from baseline to week 4 in PRSIS, and to week 4 and 8 in pain Numerical Rating Scale (NRS; from 0 “no pain” to 10 “worst possible pain”), and on the Neck Disability Index (NDI). Other assessments of QoL were undertaken. Safety was monitored.
Results: Overall, 369 patients received pregabalin (n=145) or usual care (n=224). The median (range) dose of pregabalin was 49.6 (25.0–251.5) mg/day. Least-squares mean change in PRSIS from baseline to week 8 favored pregabalin (–1.167 vs –0.269; treatment difference –0.898 [95% CI –1.262, –0.535], P<0.001). Similar observations were seen at week 4 in favor of pregabalin versus usual care (P<0.001). Pregabalin significantly improved pain NRS and NDI scores at weeks 4 and 8 (all P<0.001). Improvements in QoL versus usual care were also observed. Pregabalin was generally well tolerated.
Conclusion: In this open-label study, pregabalin improved PRSIS and resulted in clinically meaningful reductions in pain in Japanese patients with NeP associated with chronic cervical pain.
ClinicalTrials.gov identifier: NCT02868359.
Keywords: pregabalin, neck pain, neuropathic pain, Japan, sleep
Introduction
In epidemiologic studies1,2 including those conducted in Japan,3 up to half of adults report that they experience neck or shoulder pain each year. In a cross-sectional postal survey of more than 6,000 Japanese adults, 1,032 respondents reported having chronic pain (pain that lasted for more than 3 months), and 456 respondents reported severe chronic pain (pain that lasted for more than 6 months).4 Of these, 8% (38/456) reported severe chronic pain in their neck region, and pain was found to adversely affect their quality of life (QoL).4 Consistent with these findings, other studies have shown a substantial risk of neck pain symptoms when the condition is chronic, leading to disability, loss of work productivity, and increased health care costs.5–8
Typical causes of neck pain include trauma (eg, whiplash), degenerative changes, as well as physical and psychosocial factors.9–11 Cervical radiculopathy12,13 or cervical spondylotic myelopathy14 is commonly associated with neuropathic pain (NeP). The Japan Society of Pain Clinicians clinical guidelines for pharmacologic treatment of NeP recommend α2δ ligands and tricyclic antidepressants as first-line therapy.15 The α2δ ligand, pregabalin, has been approved in Japan for the treatment of NeP and pain associated with fibromyalgia.16 Moreover, pregabalin is also recommended as a first-line therapy for NeP in the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain.17 The efficacy and safety of pregabalin for improving pain has been demonstrated in clinical trials of NeP conditions.18–22 In addition, pregabalin significantly improves sleep disturbance across multiple NeP conditions through an indirect analgesic effect and a direct effect on sleep.19,20,23,24 The efficacy and safety of pregabalin for treatment of NeP has also been specifically demonstrated in Japanese patients with diabetic peripheral neuropathy, postherpetic neuralgia, central NeP, and lower back pain.21,22,24–27
Despite neck and shoulder pain being a worldwide health concern,2,3 and recommendations for the use of α2δ ligands for the management of NeP,15,28 few studies have specifically investigated the efficacy of pregabalin for reducing chronic neck pain.29–31 One naturalistic study in Spain reported that monotherapy or add-on pregabalin therapy as part of routine medical practice was associated with substantial pain alleviation and other symptomatic improvements in patients with painful cervical or lumbosacral radiculopathy.31 The therapeutic value of pregabalin for chronic neck pain has not, however, been assessed in a typical Japanese health care setting. The aim of this study was, therefore, to evaluate the effectiveness of pregabalin for treatment of patients with chronic cervical pain and accompanying upper limb radiating pain (NeP component) in a primary care setting under routine clinical practice.
Methods
Study design and patients
In this 8-week, non-interventional, multicenter, prospective, observational study conducted in Japan (ClinicalTrials.gov identifier: NCT02868359), patients were enrolled between October 2016 through October 2017. During this period, all patients visiting the study sites who met study criteria were continuously enrolled in order to reduce selection bias. Enrollment took place after the physician prescribed analgesics (see further details below regarding study treatments). The study required three visits: baseline/enrollment, at week 4 and week 8. Patients were required to be able to understand and follow study protocols. All participants gave written informed consent in accordance with the Declaration of Helsinki and the Japanese Act on the Protection of Personal Information (Act No. 57 of May 30, 2003). The present study received approval from the Byoin-Godo Ethical Review Board.
Inclusion criteria for participating in the study required patients ≥20 years of age with a diagnosis of chronic cervical pain with accompanying radiating pain to superior limb(s) with a NeP component of ≥12 weeks in duration at baseline. This pain was required to be refractory to previous analgesics and self-rated as ≥5 on an 11-point Numerical Rating Scale (NRS; 0 to 3 = mild pain, 4 to 6 = moderate pain, and 7 to 10 = severe pain), based on recall over the past week. Patients must also have reported sleep disturbance on the Pain-Related Sleep Interference Scale (PRSIS score ≥1) at baseline (based on recall over the past week on a NRS ranging from 0 (“did not interfere with sleep”) to 10 (“completely interferes with sleep”).32 Assessment scales are detailed further below. Patients were excluded if treated with pregabalin within 12 weeks prior to baseline or regularly treated for cervical pain with nerve blocks (eg, stellate ganglion block, epidural block, radicular block, and trigger point injection).
Treatments
All patients enrolled in the study were treated with at least one analgesic for 8 weeks, either as “usual care” with conventional analgesic care or other treatment(s) based on physician’s best clinical judgment or with pregabalin (monotherapy or in combination with other analgesics they may have been prescribed). Conventional analgesics included paracetamol (acetaminophen) and non-steroidal anti-inflammatory drugs (NSAIDs), opioids, antidepressants, or other antiepileptic drugs (for chronic pain use), among others. As a non-interventional study, therapeutic choice was not related to the decision to take part in the study. The specific treatment was determined by each patient’s physician based on their expert clinical judgment and the local prescribing label of each drug, prior to patient enrollment in the study. For pregabalin, the prescribing information recommends oral administration at a dosage beginning at 75 mg twice daily (maximum 150 mg/day), gradually increasing to 150 mg twice daily (maximum 300 mg/day) over 1 week.16,33 The dose could be adjusted, depending on age, renal function, or symptoms, but the maximum dose could not exceed 300 mg twice daily (total 600 mg/day).
Assessments and endpoints
The primary endpoint was the change from baseline to week 8 in PRSIS score (with a recall of 1 week) between patients prescribed pregabalin (with or without usual care) compared with usual care with conventional analgesics. Physicians each received training in how to complete the questionnaires prior to patient enrollment. The PRSIS has been used previously in clinical studies of pregabalin.20,23,24 Secondary endpoints included changes from baseline to week 4 in PRSIS score and changes from baseline to weeks 4 and 8 in pain NRS scores (based on recall of 1 week)20,23 in patients treated with pregabalin compared with usual care. For assessment of pain, participants were asked to describe their average pain over the past week on the 11-point NRS (as noted earlier). For pain NRS, “responders” were defined as the proportion of patients who achieved or exceeded a percentage reduction (set a 30% and 50% threshold) in pain NRS score by week 8.24
Patients also undertook a number of additional assessments. The Neck Disability Index (NDI) assesses how pain affects a patient’s everyday life.34–36 The NDI includes questions on pain, personal care, work, sleep, and other aspects of daily living, and ranges from 0 to 50, where a lower score indicates better function.35 In the event that a patient had used rescue medication within 12 hrs of the clinic visit, the patient responded to the NDI questions based on their functional ability in the 24-hr period just preceding the use of rescue medication. The EuroQol five dimensions five level (EQ-5D-5L) assessment includes a descriptive system that evaluates QoL using five dimensions (mobility, self-care, usual activities, pain/discomfort, anxiety/depression) using five rating levels (“no problems”, “slight problems”, “moderate problems”, “severe problems”, and “extreme problems”), which combine to calculate a single utility index value.37,38 The EQ-5D-5L also includes a Visual Analog Scale (VAS) to assess health status, on a scale from 0 (“worst imaginable health state”) to 100 (“best imaginable health state”), where a higher score indicates a better health state.37,38 The Patient Global Impression of Change (PGIC) and Clinical Global Impression of Change (CGIC) assessments were collected at week 8/end of study to evaluate perceived change in health status from the perspective of the patient or clinician. Scores on both scales ranged from 1 (“very much improved”) to 7 (“very much worse”), and higher scores on either scale represent a poorer perception of health state.34,36 Each patient-reported outcomes assessment was translated and administered in Japanese.
To evaluate tolerability, adverse events (AEs) were recorded and categorized into three types of events: (1) non-serious AEs (non-SAEs); (2) SAEs; and (3) scenarios involving drug exposure, including exposure during pregnancy or during breastfeeding, medication error, overdose, misuse, extravasation, and occupational exposure. For all AEs, potential relationship to study treatment was determined by expert clinical judgment of the treating investigator.
Statistical methods
This study was powered to detect an assumed standardized effect size of 0.37 (the difference in mean change from baseline, divided by the pooled standard deviation [SD] of the mean change from baseline) in PRSIS score. A target sample size of 360 participants had at least 85% power to detect the treatment difference by a mixed model for repeated measures (MMRM) analysis based on the assumption of withdrawal rate of 25%; correlation of 0.55 for scores between weeks 4 and 8; and a 1:1 ratio of pregabalin compared with usual care with other analgesics. Effectiveness was assessed in the Full Analysis Set, composed of all enrolled patients who received at least one dose of pregabalin or usual care and at least one post-baseline evaluable assessment. The primary analysis compared treatment groups using an MMRM analysis including fixed categorical effects of treatment, visit, and treatment-by-visit interaction, and fixed continuous effect of baseline value. The same MMRM analysis was used for all other analyses of continuous data. Responders of pain NRS were analyzed using a logistic regression model that included treatment and baseline value. A sensitivity analysis of the PRSIS at week 8/endpoint was conducted using the Last Observation Carried Forward (LOCF) approach. Differences between pregabalin and usual care for the full set of categories on the CGIC and the PGIC were analyzed using a chi-square test excluding “not assessed” or “multiple categories selected” using a modified ridit transformation. Statistical significance was evaluated with a 2-tailed test, with a significance level of 0.05. No adjustments were made for multiplicity, since all comparisons, except for a single primary comparison, were considered secondary.
AEs in the pregabalin and usual care treatment arms were summarized descriptively using the Safety Analysis Set, composed of all patients who received at least one dose of pregabalin or usual care, with at least one post-baseline evaluable safety assessment. The statistical analysis plan was developed before any data became available, and all analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient disposition and demographics
In total, 369 patients were enrolled and received analgesic treatment(s). The majority of these completed the study (pregabalin n/N=131/145 [90.3%]; usual care with other analgesics n/N=210/224 [93.8%]) (Figure 1). Subject demographics and clinical characteristics were generally comparable between the pregabalin and usual care groups (Table 1). However, the percentage of male patients in the pregabalin group was higher than in the usual care group (53.1% vs 30.4%, respectively; P<0.001). In addition, the mean age of the pregabalin group was approximately 8 years younger than the usual care group (58.3 years vs 66.4 years, respectively; P<0.001). Baseline PRSIS and pain NRS scores in the pregabalin group (mean [SD]: PRSIS 3.3 [2.4]; pain NRS 6.1 [1.2]) were higher than those receiving usual care (PRSIS 2.8 [2.0]; pain NRS 5.8 [1.1]; P=0.021 and P=0.005, respectively). Patients treated with pregabalin had experienced pain for a shorter duration of time than those treated with usual care (mean duration of 28.8 months vs 37.1 months, respectively; P=0.098).
 | Table 1 Baseline demographic and clinical characteristics |
 | Figure 1 Patient disposition. Abbreviations: AE, adverse event; NSAIDs, non-steroidal anti-inflammatory drugs. |
Over the course of treatment, the mean (SD) daily dose of pregabalin was 59.9 (38.6) mg/day. All patients were using a variety of medications for pain at baseline (Table 1). The most commonly used analgesics prior to the study were NSAIDs for both pregabalin (93.1%) and usual care patients (89.3%). Throughout the study, at least 72% of the pregabalin group and 99% of the usual care group received a concomitant therapy, the majority of which were NSAIDs (Table S1).
Effectiveness of pregabalin compared with usual care
The least-squares (LS) mean (standard error [SE]) change from baseline in primary endpoint of PRSIS scores at week 8 was –1.167 (0.145) with pregabalin (n=121) and –0.269 (0.115) with usual care (n=194). Patients reported an LS mean change from baseline significantly in favor of pregabalin treatment versus usual care, for reduction in pain-related sleep interference (–0.898 [95% CI –1.262, –0.535]; P<0.001; Figure 2).
 | Figure 2 Change from baseline in weekly PRSIS at weeks 4 and 8. Abbreviations: LS, least squares; PRSIS, Pain-Related Sleep Interference Scale; SE, standard error. |
In a supplementary analysis of the change from baseline PRSIS score at week 8 using an LOCF approach, the results were consistent with the primary analysis with LS mean changes from baseline of –1.140 and –0.276 for pregabalin and usual care, respectively (treatment difference: –0.864 [95% CI –1.221, –0.508]; P<0.001). Significant reductions in pain-related sleep interference scores were also seen as early as week 4, with LS mean (SE) change from baseline of –0.852 (0.142) for pregabalin (n=131) and –0.087 (0.113) for usual care (n=207), with a treatment difference significantly favoring pregabalin versus usual care (–0.765 [95% CI –1.123, –0.408]; P<0.001).
Pregabalin also improved pain scores compared with usual care at both weeks 4 and 8, indicated by significantly greater reductions in pain NRS scores at these time points (Figure 3A). When treatment differences were compared (pregabalin vs usual care), the effectiveness for reduction in pain NRS score significantly favored pregabalin at both week 4 (–0.739 [95% CI –1.062, –0.415]) and week 8 (–1.255 [95% CI –1.630, –0.880]; both P<0.001) (Figure 3A). Responder rates showed a higher percentage of patients treated with pregabalin achieving the 30% responder status for pain relief at week 8 compared with patients treated with usual care (Figure 3B). This difference was also statistically significant in favor of pregabalin versus usual care for the 50% responder status (Figure 3B).
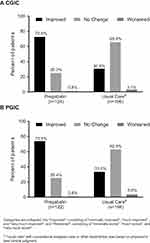
The NDI and EQ-5D-5L (QoL and VAS scores) assessments were statistically significant in favor of pregabalin compared with usual care at weeks 4 and 8 (Table 2). CGIC and PGIC data at week 8 were evaluated with answers of “minimally improved”, “much improved”, or “very much improved” collectively grouped as “improved”. More patients treated with pregabalin versus usual care had some level of improvement in the CGIC (pregabalin 90/124 [72.6%] vs usual care 60/196 [30.6%]) and PGIC (pregabalin 90/122 [73.8%] vs usual care 65/194 [33.5%]) assessments (Figure 4). The differences between treatment groups across all seven change categories were statistically significant for CGIC and PGIC (both P<0.001).
 | Table 2 Neck Disability Index and EuroQoL five dimensions five levels |
Safety and tolerability
The incidence of all-causality, treatment-emergent AEs (TEAEs) in patients receiving pregabalin (n=42/145; 29.0%) was numerically higher than for patients receiving usual care (n=48/224; 21.4%; Table 3). Approximately half of the AEs that occurred with pregabalin were considered treatment-related by the investigators; none of the AEs with other analgesics were classified as treatment-related. The most commonly reported TEAEs (defined as occurring in >2% patients in any treatment group) were dizziness (pregabalin 14 [9.7%]; usual care 1 [0.4%]), nasopharyngitis (pregabalin 6 [4.1%]; usual care 0), somnolence (pregabalin 6 [4.1%]; usual care 0), and bronchitis (pregabalin 0, usual care 5 [2.2%]). Three patients experienced SAEs, which included two patients in the pregabalin group and one in the usual care treatment arm. None of the SAEs were considered to be treatment-related. In patients who received pregabalin, one patient had SAE of pneumonia and acute cardiac failure (recovered), and one patient reported dehydration (recovered) but experienced further aggravated dehydration on the following day (not recovered by the time of last follow-up). This subject completed the study. One patient in the usual care with other analgesics arm had an SAE of malaise (not recovered by time of last follow-up). This subject withdrew from the study due to patient choice.
 | Table 3 Adverse events (Safety Analysis Set)a |
Discussion
In the present observational study of Japanese patients with chronic refractory cervical pain with a NeP component, significant improvements in self-reported sleep interference and pain scores were reported after 8 weeks of treatment with pregabalin (with or without other analgesics) compared with “usual care” with conventional analgesic therapy. In addition to supporting evidence for pregabalin improving aspects of sleep, the data presented here support the efficacy of pregabalin in other patients with neck pain,29–31,39 and in Japanese patients with different types of NeP, based on clinical trial data,21,22,40 as well as non-interventional24 and long-term open-label studies for postherpetic neuralgia, diabetic peripheral neuropathy26,41 and central NeP.27 The reductions in PRSIS (–1.17) and pain NRS (–2.26) in the current study were similar to those reported previously in a similarly designed study of Japanese patients treated with pregabalin for chronic lower back pain associated with NeP.24 A reduction in pain of more than two-points on the 11-point pain NRS (or reduction of approximately 30%) is considered to be the minimal clinically important difference.42 Furthermore, half of the pregabalin patients in the current study had a pain reduction of ≥30% by week 8.
Japanese patients were recruited into the current study who had at least moderate levels of pain (pain NRS ≥5), despite previous use of analgesic therapies, thus reflecting the refractory nature of the chronic cervical pain with a NeP component and the complexity of its treatment.9,12 Mean baseline PRSIS and pain NRS scores were significantly higher in the pregabalin treatment arm compared with the usual care arm, which may suggest a slight bias in physicians reserving use of pregabalin for patients with higher levels of pain, something that has been previously noted in other non-interventional studies involving pregabalin in NeP conditions.24 Supporting this hypothesis, the dose of pregabalin over the course of the present study (median [range] 49.6 [25.0–251.5] mg/day) was well below the recommended starting dose of pregabalin (150 mg/day), prior to titration to the therapeutic dose recommended by guidelines.15 The efficacy of pregabalin has been found to increase in a dose-dependent manner in different patient populations.43 Pain relief and other modalities, including sleep quality, are improved for patients who receive the maximum recommended doses of pregabalin for the given NeP condition.43 Physicians in the present study were instructed to prescribe treatments based on their expert clinical judgment, so the lower-than-recommended doses of pregabalin prescribed by physicians in routine clinical practice could be due to a number of reasons, including safety concerns regarding renal function in Japanese patients.33 If physicians had prescribed the full recommended dose of pregabalin to patients enrolled in this radiculopathy study, the observed treatment differences may have increased in a dose-related manner, as seen with other NeP states.43
Despite the lower-than-recommended prescribed doses, in addition to improving sleep and pain scores, pregabalin treatment reduced the NDI score by 8.5 points by week 8 versus usual care, which was a significant improvement compared with a 2.5-point reduction with usual care. An 8.5-point change on the NDI is the threshold for the “minimal clinically important difference”,44 which suggests that one should be confident (with an uncertainty of 10%) that a patient’s neck pain disability has improved at this level of change.44 Assessment of EQ-5D-5L also favored pregabalin, and both clinicians (CGIC) and patients (PGIC) reported significantly greater improvements in patient health status with pregabalin versus usual care with other analgesics, these latter observations being consistent with a previous study of chronic lower back pain with a NeP component, in patients from Japan.24 Although not a randomized controlled trial (RCT), safety monitoring suggested that pregabalin was generally well tolerated when taken alone or in combination with other analgesic therapies, and no new safety concerns were identified in this population of Japanese patients with chronic cervical pain and radiating NeP. The most commonly occurring AEs of dizziness, nasopharyngitis, and somnolence are consistent with the known safety profile reported in other studies of pregabalin for NeP in Japanese patients.24,25,27
This study should be interpreted in light of some limitations. As an observational, open-label, nonrandomized design in which the treating physician chose the treatment practices based on best clinical judgment, the potential for bias in treatment selection cannot be ruled out. Differences in the baseline characteristics of the two treatment arms, including higher levels of baseline pain in the pregabalin group compared with the usual care arm, may suggest a slight bias in physicians prescribing pregabalin for patients with higher levels of pain. This trend has been previously noted in a non-interventional study involving pregabalin in NeP.24 In addition, patients may have been susceptible to treatment bias if they had prior expectations about the efficacy of pregabalin. The Japanese patients in the current study were required to have moderate-to-severe chronic cervical pain with radiating upper limb NeP to be eligible for enrollment into the study, which limits the generalizability of the results to other patient groups or pain types. Pregabalin has been approved in Japan for the treatment of chronic NeP and pain associated with fibromyalgia,16 and our observations should be considered in light of the population and pain modality under study. One study of acute sciatica in Australian patients suggests pregabalin is not effective at reducing acute pain,45 nor in other types of lumbar radial pain.46 Whereas other studies have demonstrated the efficacy of pregabalin, in combination with other agents, in NeP associated with nerve compression,31,39 including one small study of chronic sciatica in Australian patients.46 These observations collectively highlight the complexity of NeP, and the need for more high-quality studies to confirm the efficacy of specific drug combinations in different patient populations. Finally, this study could not measure or draw conclusions about the pharmacologically mediated effects of pregabalin due to patients being able to carry on receiving concomitant analgesic medications during the study. However, pregabalin was evaluated relative to other analgesic treatments, reflecting a usual-care approach to pain management in Japan. The efficacy/safety outcomes are the simple reflection of the real-world treatment choice physicians made, and additional RCTs, which captures dose information about the comparator arm, would be required if we want to validate the outcome of these two treatment approaches.
Conclusion
This open-label study supports the effectiveness of pregabalin for improving pain-related sleep interference and resulted in clinically meaningful reductions in pain, in patients with chronic cervical pain with a neuropathic component in a usual care setting, reflecting real-world prescribing practices in Japan. In addition, pregabalin was well tolerated. These observations add to previous studies demonstrating the efficacy of pregabalin for the management of NeP in Japanese patients.
Abbreviation list
AE, adverse event; CGIC, Clinical Global Impression of Change; CI, confidence interval; EQ-5D-5L, EuroQol five dimensions five level; JSPC, Japan Society of Pain Clinicians; LOCF, last observation carried forward; LS, least squares; MMRM, mixed model for repeated measures; NDI, Neck Disability Index; NeP, neuropathic pain; NRS, numerical rating scale; NSAIDs, non-steroidal anti-inflammatory drugs; PGIC, Patient Global Impression of Change; PRO, patient-reported outcome; PRSIS, Pain-Related Sleep Interference Scale; QoL, quality of life; SAE, serious adverse event; SD, standard deviation; SE, standard error; TEAE, treatment-emergent adverse event; VAS, visual analog scale.
Ethics approval and informed consent
Patients were required to be able to understand and follow study protocols and all patients gave written informed consent in accordance with the Declaration of Helsinki and the Japanese Act on the Protection of Personal Information (Act No. 57 of May 30, 2003). The present study received approval from the Byoin-Godo Ethical Review Board.
Data sharing statement
Upon request, and subject to certain criteria, conditions and exceptions (see
Acknowledgments
Medical writing support was provided by Ray Beck, Jr, PhD, and Karen Burrows, MPhil, of Engage Scientific Solution, and was funded by Pfizer Inc. This study was funded by Pfizer Inc.
Author contributions
All authors contributed either to the design of the study, data analysis or acquisition, and/or data interpretation. All authors were involved in manuscript development, and take responsibility for the data. All authors approved the final draft of the manuscript for submission, and agree to be accountable for all aspects of the work.
Disclosure
Kazutaka Nozawa, Bruce Parsons, Tamotsu Yoshiyama, Nozomi Ebata, and Koichi Fujii are employees and shareholders of Pfizer, the sponsor of this study. None of the other authors were financially compensated for their collaboration in this project or for the development of this paper. Ataru Igarashi reports personal fees from Abbvie GK, Astellas Pharma Inc., Chugai Pharmaceuticals Inc., Eisai Inc., Nippon Boeringer Ingelheim Inc., Novartis Pharma Japan Inc., Novo Nordisk Japan Inc., Pfizer Inc., Sanofi Japan Inc., Taiho Yakuhin Inc. and Takeda Pharmaceutical Inc., and received grants from Beckton Dickinson and Company, Boston Scientific Japan Inc., CSL Behring Japan Inc., Fuji film Inc., Gilead Sciences KK., Intuitive Surgical GK., Milliman Inc., Pfizer Japan Inc. and Terumo corporation. All authors report no other conflicts of interest related to this work.
References
1. Cote P, Kristman V, Vidmar M, et al. The prevalence and incidence of work absenteeism involving neck pain: a cohort of Ontario lost-time claimants. Spine (Phila Pa 1976). 2008;33(4 Suppl):S192–198. doi:10.1097/BRS.0b013e3181644616
2. Croft PR, Lewis M, Papageorgiou AC, et al. Risk factors for neck pain: a longitudinal study in the general population. Pain. 2001;93(3):317–325.
3. Takasawa E, Yamamoto A, Kobayashi T, et al. Characteristics of neck and shoulder pain in the Japanese general population. J Orthop Sci. 2015;20(2):403–409. doi:10.1007/s00776-014-0676-2
4. Inoue S, Kobayashi F, Nishihara M, et al. Chronic pain in the Japanese community–prevalence, characteristics and impact on quality of life. PLoS One. 2015;10(6):e0129262. doi:10.1371/journal.pone.0129262
5. Holm LW, Carroll LJ, Cassidy JD, et al. The burden and determinants of neck pain in whiplash-associated disorders after traffic collisions: results of the bone and joint decade 2000-2010 task force on neck pain and its associated disorders. Spine (Phila Pa 1976). 2008;33(4 Suppl):S52–59. doi:10.1097/BRS.0b013e3181643ece
6. Schmitt KU, Walz F, Vetter D, Muser M. Whiplash injury: cases with a long period of sick leave need biomechanical assessment. Eur Spine J. 2003;12(3):247–254.
7. Fujiwara H, Oda T, Makino T, Moriguchi Y, Yonenobu K, Kaito T. Impact of cervical sagittal alignment on axial neck pain and health-related quality of life after cervical laminoplasty in patients with cervical spondylotic myelopathy or ossification of the posterior longitudinal ligament: a prospective comparative study. Clin Spine Surg. 2018;31(4):E245–E251. doi: 10.1097/BSD.0000000000000619
8. Iizuka Y, Iizuka H, Mieda T, et al. Association between neck and shoulder pain, back pain, low back pain and body composition parameters among the Japanese general population. BMC Musculoskelet Disord. 2015;16:333.
9. Cote P, van der Velde G, Cassidy JD, et al. The burden and determinants of neck pain in workers: results of the bone and joint decade 2000-2010 task force on neck pain and its associated disorders. Spine (Phila Pa 1976). 2008;33(4 Suppl):S60–74. doi:10.1097/BRS.0b013e3181643ee4
10. Hogg-Johnson S, van der Velde G, Carroll LJ, et al. The burden and determinants of neck pain in the general population: results of the bone and joint decade 2000-2010 task force on neck pain and its associated disorders. Spine (Phila Pa 1976). 2008;33(4 Suppl):S39–S51. doi:10.1097/BRS.0b013e31816454c8
11. Iizuka Y, Shinozaki T, Kobayashi T, et al. Characteristics of neck and shoulder pain (called katakori in Japanese) among members of the nursing staff. J Orthop Sci. 2012;17(1):46–50. doi:10.1007/s00776-011-0177-5
12. Tampin B, Slater H, Briffa NK. Neuropathic pain components are common in patients with painful cervical radiculopathy, but not in patients with nonspecific neck-arm pain. Clin J Pain. 2013;29(10):846–856. doi:10.1097/AJP.0b013e318278d434
13. Tsunoda D, Iizuka Y, Iizuka H, et al. Associations between neck and shoulder pain (called katakori in Japanese) and sagittal spinal alignment parameters among the general population. J Orthop Sci. 2013;18(2):216–219. doi:10.1007/s00776-012-0341-6
14. Yamashita T, Takahashi K, Yonenobu K, Kikuchi S. Prevalence of neuropathic pain in cases with chronic pain related to spinal disorders. J Orthop Sci. 2014;19(1):15–21. doi:10.1007/s00776-013-0496-9
15. Hosokawa T, Okuda Y, Oseto K. Guidelines for the Pharmacologic Management of Pain Conditions.
16.
17. Dworkin RH, O’Connor AB, Audette J, et al. Recommendations for the pharmacological management of neuropathic pain: an overview and literature update. Mayo Clin Proc. 2010;85(3 Suppl):S3–14. doi:10.4065/mcp.2009.0649
18. Cardenas DD, Nieshoff EC, Suda K, et al. A randomized trial of pregabalin in patients with neuropathic pain due to spinal cord injury. Neurology. 2013;80(6):533–539. doi:10.1212/WNL.0b013e318281546b
19. Vinik A, Emir B, Parsons B, Cheung R. Prediction of pregabalin-mediated pain response by severity of sleep disturbance in patients with painful diabetic neuropathy and post-herpetic neuralgia. Pain Med. 2014;15(4):661–670. doi:10.1111/pme.12310
20. Parsons B, Emir B, Knapp L. Examining the time to improvement of sleep interference with pregabalin in patients with painful diabetic peripheral neuropathy and postherpetic neuralgia. Am J Ther. 2015;22(4):257–268. doi:10.1097/MJT.0000000000000100
21. Ogawa S, Suzuki M, Arakawa A. Evaluation of the efficacy and safety of pregabalin in the treatment of postherpetic neuralgia: a randomized, double-blind, multicenter, placebo-controlled study. J Japan Soc Pain Clin. 2010;17:141–152.
22. Satoh J, Yagihashi S, Baba M, et al. Efficacy and safety of pregabalin for treating neuropathic pain associated with diabetic peripheral neuropathy: a 14 week, randomized, double-blind, placebo-controlled trial. Diabet Med. 2011;28(1):109–116. doi:10.1111/j.1464-5491.2010.03152.x
23. Anastassiou E, Iatrou CA, Vlaikidis N, et al. Impact of pregabalin treatment on pain, pain-related sleep interference and general well-being in patients with neuropathic pain: a non-interventional, multicentre, post-marketing study. Clin Drug Investig. 2011;31(6):417–426. doi:10.2165/11589370-000000000-00000
24. Taguchi T, Igarashi A, Watt S, et al. Effectiveness of pregabalin for the treatment of chronic low back pain with accompanying lower limb pain (neuropathic component): a non-interventional study in Japan. J Pain Res. 2015;8:487–497. doi:10.2147/JPR.S88642
25. Ogawa S, Satoh J, Arakawa A, Yoshiyama T, Suzuki M. Pregabalin treatment for peripheral neuropathic pain: a review of safety data from randomized controlled trials conducted in Japan and in the west. Drug Saf. 2012;35(10):793–806. doi:10.2165/11632660-000000000-00000
26. Ogawa S, Suzuki M, Arakawa A, Yoshiyama T. [Long-term efficacy and safety of pregabalin in patients with postherpetic neuralgia: results of a 52-week, open-label, flexible-dose study]. Masui. 2010;59(8):961–970.
27. Onouchi K, Koga H, Yokoyama K, Yoshiyama T. An open-label, long-term study examining the safety and tolerability of pregabalin in Japanese patients with central neuropathic pain. J Pain Res. 2014;7:439–447. doi:10.2147/JPR.S63028
28. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132(3):237–251. doi:10.1016/j.pain.2007.08.033
29. Florez-Garcia M, Ceberio-Balda F, Morera-Dominguez C, Masramon X, Perez M. Effect of pregabalin in the treatment of refractory neck pain: cost and clinical evidence from medical practice in orthopedic surgery and rehabilitation clinics. Pain Pract. 2011;11(4):369–380. doi:10.1111/j.1533-2500.2010.00430.x
30. Nikles J, Keijzers G, Mitchell G, et al. Pregabalin versus placebo in targeting pro-nociceptive mechanisms to prevent chronic pain after whiplash injury in at-risk individuals - a feasibility study for a randomised controlled trial. Trials. 2018;19(1):44. doi:10.1186/s13063-018-2450-9
31. Saldana MT, Navarro A, Perez C, Masramon X, Rejas J. Patient-reported-outcomes in subjects with painful lumbar or cervical radiculopathy treated with pregabalin: evidence from medical practice in primary care settings. Rheumatol Int. 2010;30(8):1005–1015. doi:10.1007/s00296-009-1086-1
32. Cleeland CS, Ryan KM. Pain assessment: global use of the brief pain inventory. Ann Acad Med Singapore. 1994;23(2):129–138.
33.
34. Takeshita K, Hosono N, Kawaguchi Y, et al. Validity, reliability and responsiveness of the Japanese version of the neck disability index. J Orthop Sci. 2013;18(1):14–21. doi:10.1007/s00776-012-0304-y
35. Vernon H, Mior S. The neck disability index: a study of reliability and validity. J Manipulative Physiol Ther. 1991;14(7):409–415.
36. Nakamaru K, Aizawa J, Koyama T, Nitta O. Reliability, validity, and responsiveness of the Japanese version of the patient-specific functional scale in patients with neck pain. Eur Spine J. 2015;24(12):2816–2820. doi:10.1007/s00586-015-4236-z
37.
38. Ikeda S, Shiroiwa T, Igarashi A, et al. [Developing a Japanese version of the EQ-5D-5L value set]. J Natl Inst Public Health. 2015;64:47–55.
39. Lo YL, Cheong PW, George JM, et al. Pregabalin and Radicular Pain Study (PARPS) for cervical spondylosis in a multiracial Asian population. J Clin Med Res. 2014;6(1):66–71. doi:10.4021/jocmr879w
40. Ogawa S, Arakawa A, Hayakawa K, Yoshiyama T. Pregabalin for neuropathic pain: why benefits could be expected for multiple pain conditions. Clin Drug Investig. 2016;36(11):877–888. doi:10.1007/s40261-016-0423-x
41. Satoh J, Yagihashi S, Baba M, Suzuki M, Arakawa A, Yoshiyama T. Efficacy and safety evaluation of pregabalin treatment over 52 weeks in patients with diabetic neuropathic pain extended after a double-blind placebo-controlled trial. J Diabetes Investig. 2011;2(6):457–463. doi:10.1111/j.2040-1124.2011.00122.x
42. Farrar JT, Young JP
43. Arnold LM, McCarberg BH, Clair AG, et al. Dose-response of pregabalin for diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia. Postgrad Med. 2017;129(8):921–933. doi:10.1080/00325481.2017.1384691
44. Young IA, Cleland JA, Michener LA, Brown C. Reliability, construct validity, and responsiveness of the neck disability index, patient-specific functional scale, and numeric pain rating scale in patients with cervical radiculopathy. Am J Phys Med Rehabil. 2010;89(10):831–839. doi:10.1097/PHM.0b013e3181ec98e6
45. Mathieson S, Maher CG, McLachlan AJ, et al. Trial of pregabalin for acute and chronic sciatica. N Engl J Med. 2017;376(12):1111–1120. doi:10.1056/NEJMoa1614292
46. Enke O, New HA, New CH, et al. Anticonvulsants in the treatment of low back pain and lumbar radicular pain: a systematic review and meta-analysis. CMAJ. 2018;190(26):E786–E793. doi:10.1503/cmaj.171333
Supplementary material
 | Table S1 Concomitant medications used through the study period for patients in the pregabalin and usual care treatment arms |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.