Back to Journals » OncoTargets and Therapy » Volume 14
Desmoplastic Melanoma: A Clinicopathological Analysis of Three Cases in the Chinese Population
Authors Fan Y, Xu M, Liang Y, Wu N, Wang F, Du Q, Bai Y, Liu Y
Received 11 December 2020
Accepted for publication 25 March 2021
Published 15 April 2021 Volume 2021:14 Pages 2651—2660
DOI https://doi.org/10.2147/OTT.S295716
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Nicola Silvestris
Yaqi Fan,1 Mingyuan Xu,2 Yulin Liang,2 Nanhui Wu,2 Feiyan Wang,3 Qian Du,2 Yun Bai,1 Yeqiang Liu1
1Department of Pathology, Shanghai Skin Disease Hospital, Tongji University School of Medicine, Shanghai, 200050, People’s Republic of China; 2Shanghai Skin Disease Hospital, Shanghai, 200443, People’s Republic of China; 3Shanghai Skin Disease Clinical College of Anhui Medical University, Shanghai Skin Disease Hospital, Shanghai, 200443, People’s Republic of China
Correspondence: Yeqiang Liu; Yun Bai
Department of Pathology, Shanghai Skin Disease Hospital, Tongji University School of Medicine, 1278 Bao’de Road, Shanghai, 200443, People’s Republic of China
Tel/Fax +86 21 61833180
Email [email protected]; [email protected]
Purpose: To investigate the clinicopathological characteristics and immunophenotype of desmoplastic melanoma (DM) in the Chinese population.
Patients and Methods: We report three cases of DM diagnosed by the Pathology Department of Shanghai Dermatology Hospital. We describe the clinical and pathological characteristics of the three cases and examine molecular markers used in the diagnosis of DM. Finally, we summarize the current literature in the DM field.
Results: Clinically, lesions in the three DM patients were characterized by non-pigmented nodules or papules. Microscopically, we observed an abundance of fibrous interstitium mixed with spindle cells exhibiting various degrees of atypia. Occasionally, these structures exhibited changes in lentigo maligna at the epidermal junction, accompanied by the presence of lymphoid follicular structures and neurophilic behavior. Diagnosis of DM was confirmed by immunohistochemical staining, which revealed high expression levels of S-100 and SOX-10. Melanocyte markers were focally positive or negative. Unlike DMs from other populations, our three patients were negative for WT-1 and P53. All three cases received surgical resection, which is the preferred treatment for DM, and none of the patients experienced recurrence.
Conclusion: DM in these Chinese patients was similar to that observed in other DM populations in terms of immunophenotype and clinical and histological features. A notable absence in p53 staining was observed in the three cases reported here, suggesting that p53 negativity should not exclude the diagnosis of DM in the Chinese population.
Keywords: desmoplastic melanoma, uneven pigmented nodules, histology, Chinese population, immunophenotypes
Introduction
Desmoplastic melanoma (DM) is first described by Conley et al in 1971 as a cutaneous tumor exhibiting similar clinical and histological patterns as invasive fibrous tumors, but with distinct morphological characteristics.1 At present, DM is considered to be a variant of spindle cell melanoma, accounting for less than 4% of all types of melanoma. The annual incidence of DM has grown in the past few decades, and is currently estimated at 2 in 1,000,000.2,3 DM occurs primarily in Caucasian populations aged 60–80 years, and is much rarer in the Asian population, accounting for only 2.2% of the total DM cases.4 DM is generally believed to be related to sun exposure. According to a large, retrospective study, 89% of DM cases occurred in areas such as the head, neck, and limbs that have been over-exposed to ultra-violet light.3,5 Clinically, DM usually presents with hard nodules, papules, or plaques.5 Since over half of DM lesions lack pigmentation and ulceration,3,41 they can be easily confused with fibroproliferative tumors such as dermatofibromas and neurofibromas.
Pathologically, DM is described as the diffuse infiltration of malignant spindle-shaped melanocytes, accompanied by high levels of fibroblastic or myofibroblastic metaplasia and abundant collagen synthesis.6,7 Neoplastic cells exhibit mild-to-moderate atypia with variable cytology and are characterized by either i) long or short spindles, ii) nuclei that are tapered, hyperchromatic or vesicular in shape, iii) an increased nucleoplasmic ratio, and iv) sometimes present with mitotic figures.8 Obvious epidermal melanocytic hyperplasia above the spindle cell bundle is considered to be precursor lesions of DM, and could therefore also serve as a potential diagnostic clue of DM.9,10 In addition, perineural infiltration is commonly observed in DM, accounting in part for the high recurrence rate of the tumor.2,8 Aggregates of lymphocytes, usually presenting as a nodular pattern, are also considered to be a characteristic feature in DM lesions. Based on the distinct morphological heterogeneity, Busam et al divided DMs into two subtypes: pure DM and mixed DM.11 In pure DM, the degree of matrix fibrosis exceeds 90%, with individual tumor cells dispersed in and separated from each other by fibrous tissue, whereas mixed DM is characterized by 10–90% cellular non-fibrosis within the interstitial area and collagen matrix.12,13 Compared with pure DM, mixed DM is more commonly related to lentigo maligna melanoma and superficial spreading melanoma, and it is more clinically recognizable.14 In addition, mixed DM patients suffer a higher locoregional recurrence (12.4–39%) compared to pure DM patients (4.1–26%).7,15–17
Due to its characteristic clinical and pathological features, DM is often confused clinically with diseases such as dermatofibromas, neurofibromas, spindle cell sarcomas, scars, and sclerosing melanocytic nevi. A recent review reported that DM was correctly diagnosed by initial clinical examination in only 27% of cases.18 When suspicion of DM is raised, immunohistochemical examination is required for further confirmation. The most important markers for the diagnosis of DM are S-100 and SOX-10 proteins,19 although Schwann cell tumors (neurofibromas and malignant peripheral nerve sheath tumors) and melanocytic nevi also express S-100 and SOX-10 (Table 1). However, malignant peripheral nerve sheath tumors only display isolated or focal S-100-positive cells, while S-100 expression in DMs is diffuse and strong.13 In addition, lentigo maligna lesions that display strongly stained nuclei and present with focally Melan-A-positive cells are useful markers for diagnosing DM. Furthermore, DMs generally express type IV collagen, vimentin, and P16 (Table 1).16,20 S-100 positive staining together with negative epithelial (p63, CK5/6, MNF116, 34BE12) and muscle (SMA, desmin) stainings can be used to distinguish DMs from epithelial- and muscle-derived tumors. When differentiating between DMs and neurofibromas, Elsensohn suggested that the absence of p53 may not exclude the diagnosis of DM.21 A recent study also revealed that positivity for p75, nerve growth factor receptor, nestin and WT-1 proteins in DMs exceeded 90%, indicating these proteins may be potentially useful biomarkers in the diagnosis of DM.19 Technique of fluorescence in situ hybridisation can also be helpful in diagnosing melanoma. For example, four-color fluorescence in-situ hybridization (FISH) probes specific to the RREB1, CCND1, and MYB genes, and centromere of chromosome 6 exhibited diagnostic value in distinguishing dysplastic nevus and melanoma.22,23
 |
Table 1 Immunohistochemical Stains in the Differential Diagnosis of Desmoplastic Melanoma |
The survival rate of DM patients was at least comparable to that of non-DM patients, who presented with tumors of equivalent thickness.24,25 Compared with non-DMs, DMs have a higher tendency for local recurrence with less frequent metastatic spread to adjacent lymph nodes. The characteristic increased Breslow thickness, higher Clark grade, and increased rate of a positive resection margin might directly contribute to the higher incidence of recurrence. The preferred treatment for DM is surgical resection and wide local excision.26 Radiotherapy can also provide local control for DM.27,28 Whether used alone or as an adjuvant therapy, radiotherapy can significantly reduce the recurrence rate, although the 5-year recurrence-free survival is not affected.27,29 Previous studies have shown that patients with positive sentinel lymph nodes (SLN) had worse disease-free survival and higher melanoma mortality rates than patients with negative SLNs.30
The incidence of DM in the Chinese population is markedly low, and few studies have focused on the specific characteristics of DM in the Chinese population. Here, we report the clinical and pathological characteristics of three cases of DM diagnosed by our center, and examine the different molecular markers used in the diagnosis of DM. We further explore whether the Chinese cases of DM differ from other populations in terms of their clinicopathological features and diagnoses, focusing in particular on the auxiliary diagnosis using molecular markers.
Patients and Methods
The DM cases were obtained from the routine diagnostic workload of the Pathology Department of Shanghai Dermatology Hospital and the consultation practice of one of the authors (L.Y.Q.) between January 2018 and September 2020. All patients gave their written informed consent to have the case details and accompanying images published. To publish the details of the case, we have also reported to Shanghai Dermatology Hospital and obtained their consent. The regulatory requirements of Shanghai Dermatology Hospital were followed. All three cases were retrospectively analyzed.
All specimens were fixed in 4% neutral-buffered formaldehyde for 24 hours, followed by dehydration, paraffin-embedding section, hematoxylin and eosin (H&E) staining, and sealing with gum. Histopathological morphology was observed by light microscopy. Immunohistochemical and FISH examination was performed on the sections from case 1 and case 2 according to standard protocols. In addition, we performed the same immunohistochemical staining on four dermatofibroma cases for further comparison. Three cases of basal cell carcinoma were stained using the p53 antibody as the positive control. Antibodies for routine melanoma markers such as HMB-45, Melan-A, S-100, SOX-10, Ki-67, MITH, Ki67 and P16 were used. We also performed WT-1, P53, neurofibromin, CD34, SMA and vimentin immunohistochemical staining. WT-1 and P53 primary antibodies were purchased from Abcam, and the other antibodies were purchased from Fuzhou Maixin Biotechnology Development Company. EVG staining was performed as described previously.31 FISH examinations including CCND1 gene amplification, MYB gene deletion, MYC gene amplification, CDKN2A gene deletion were performed. CCND1 and MYC gene amplification probe were procured from Shanghai Long Island Antibody Diagnostic Reagent Company. MYB and CDKN2A gene deletion probes were purchased from Wuhan Kangrec Biotechnology Company.
H&E and immunohistochemically stained sections were reviewed by three dermatopathologists (L.Y.Q., W.F., and X.M.Y.) by light microscope. Nuclear staining of P16, SOX-10, MITF, S-100 and P53 was defined as positive; nuclear and cytoplasmic staining of WT-1 was defined as staining; cytoplasmic staining of HMB-45, Melan-A, vimentin, SMA, and neurofibromin was defined as positive; and cell membrane staining of CD34 was defined as positive. The Ki67 proliferation index was calculated as follow: ten areas with the highest nuclear expression density in the high-power field of view were randomly select, and the proportion of positively stained tumor cells were counted to give an average value.
Results
Clinical Data
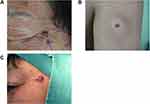
All three patients enrolled in this study were Chinese, aged between 57 to 70 years (mean, 62 years) at the time of diagnosis, and had no family history of skin cancers. None of the patients received physical or drug treatments at the lesion sites. No dysplastic (atypical) nevus were observed on the lesion sites. Long-term sun exposure could be the risk factor for case 1 and case 3, while gender (male) could be the contributing risk factor for case 2.32 All lesions were located in sun-exposed areas with case 1 and case 3 lesions located on the left side of the face, and case 2 located on the left upper limb (Figure 1). All three cases presented with a single nodule or papule with a smooth surface and clear boundaries. Two cases had hyperpigmentation with uneven pigment distribution on the surface, while one case had a hypo-pigmented area around the lesion. No ulceration was observed. Lesions in all cases developed painlessly. The sizes of tumors ranged from 0.5 x 0.5 cm to 1.8 x 1.0 cm at the time of diagnosis (Table 2).
 |
Table 2 (A and B) Clinical and Pathological Findings in Desmoplastic Melanoma |
Microscopical Examination
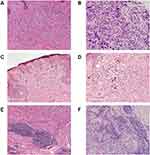
Spindle-shaped melanomas were observed in all three cases. These melanomas were characterized by spindle cell neoplasms with hyperchromatic nuclei and varied heterogeneity, and were found to infiltrate through the tissue with associated thickening of interweaved collagen fibers (Figure 2A). Spindle-shaped tumor cells accounted for 30–80% of the lesions, which was consistent with mixed DM. Tumor cells in two of three cases presented with mild to moderate atypia (Figure 2B). Two cases showed abnormal melanocyte proliferation with moderate atypia at the dermo-epidermal junction (Figure 2C). Melanin deposition was evident in two of three cases (Figure 2D). Two cases had neurophilic characteristics (Figure 2F). In two cases, we observed prominent lymphocytic infiltration, some of which formed lymphoid follicle structures (Figure 2E). The Breslow thickness for the three cases ranged from 2.53 mm to 4.67 mm with a mean of 3.95 mm. The Clark classification was stage IV in one case, and stage V in the other two cases (Table 2).
Immunophenotype
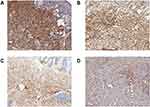
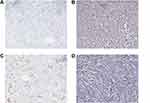
Case 1 and case 2 displayed strong and diffuse positive staining for S-100 (Figure 3A), vimentin (Figure 3B), CD34 (Figure 3C) and P16, with focal expression of SOX-10. Case 2 expressed Melan-A (Figure 3D). Case 1 and case 2 were negative for MITH (Figure 4A), HMB-45 (Figure 4B), SMA (Figure 4C), WT-1, P53 (Figure 4D) and neurofibromin. The Ki-67 positive index in two cases was approximately 5%.
EVG Staining
EVG staining indicated proliferation of the collagen fibers were proliferated, while elastic fibers were not present in the tumor region (Figure 5).
 |
Figure 5 EVG staining (×100) showed that there were fewer elastic fibers in the desmoplastic melanoma lesion (A) than in the para-cancer lesion (B). |
FISH
Case 1 presented with CDKN2A gene deletion (Supplementary Figure 1). The CCND1 gene amplification, MYB gene deletion, and MYC gene amplification were not observed in the patients sections.
Treatment and Follow-Up
All three patients received surgical resection and local radiotherapy (Table 2). The radiotherapy covered the surgical site plus a 1–2 cm margin.The three patients were healthy with no recurrence after 3–19 months’ follow-up.
Discussion
Clinical Features
As a rare type of melanoma, DM usually affects Caucasian populations, while exhibiting a remarkably low incidence rate in the Chinese population. Between January 2018 and September 2020, we diagnosed three cases of DMs compared to 101 cases of melanoma in our center. This 3/101 rate was consistent with the previously reported low incidence rates of DM. Clinically, DM is thought to be related to sun exposure, usually on skin that has been over-exposed to ultra-violet light, including the head, neck, and limbs.5,13 In our study, DM was observed on the face of two patients and on the upper limb of the third patient. DM predominantly manifests as amelanotic nodules or plaques, or scar-like pale lesions.13 The three cases reported here presented with nodule and papule, in the absence of ulcers. The absence of melanin pigmentation and ulceration is probably the main reason why DMs are not recognized clinically as melanoma. None of the cases in our study were diagnosed as malignant tumors at the first consultation. However, case 1 and case 3 exhibited uneven pigment distribution, which differentiated these cases from benign lesions such as dermatofibroma and indicated that further follow up was required. It should be noted that aneurysmal dermatofibroma could also present with uneven pigmentation.33 Case 2 presented with amelanotic papule, similar to dermatofibromas (Figure 1B), accompanied with hypopigmented regions around the lesions presumed to be areas of regression. Our observations indicate that due to the extensive and non-specific clinical manifestations of DM such as those described above, routine histological and immunohistochemical staining of DM are particularly important for DM diagnosis.
Diagnosis
Histopathologically, DM is a variant of spindle cell melanoma, characterized by fibroblastic or myofibroblastic metaplasia and accompanied by abundant collagen synthesis. In the cases examined in our study, the spindle cell neoplasms were disorderly, arranged in a parallel or a mat-like structure that infiltrated the subcutaneous tissue. Tumor cells atypia varied from mild to moderate. We also observed characteristic melanin deposition (2/3 cases), nerve involvement (2/3 cases), atypical melanocytic proliferation (2/3 cases), and patchy lymphoid infiltrates (2/3 cases) in the lesions.
Due to non-specific pathologies being overlooked, histopathological diagnosis of DM is sometimes difficult and represents a significant challenge to the pathologist. In case 2, spindle cells exhibited mild atypia without nuclear division or nuclear heterogeneity, and could therefore have been easily mistaken for a dermatofibroma or neurofibroma lesion. However, the apparent lymphoid follicular-like structures in the reticular layer raised our suspicion of DM. Further immunostaining indicated that the tissue was positive for S-100 and SOX-10 markers, negative for HMB-45, and positive for Melan-A, thereby allowing us to differentiate between dermatofibromas and neurofibromas. These findings suggest that paying particular attention to the presence of focal hyperchromatic spindle cells and possible associated lymphocytic aggregates may be important in the diagnosis of DM. Furthermore, by comparing the staining of four dermatofibroma samples to our three DM cases, we found that immune markers were important for distinguishing DMs from dermatofibromas.19 In particular, S-100 and SOX-10 staining was negative in all four dermatofibroma cases, while 2/4 of the cases revealed focal staining of CD34. In contrast, two DM cases were positive for S-100, CD34 and SOX10. Furthermore, two cases of DM presented with evident lymphoid infiltrates, which were not observed in dermatofibromas.
Case 1 had a “pink”, scar-like appearance that was visible by low-magnification light microscopy. Scrutiny of the lesion revealed several diagnostic clues for DM including i) an overlying epidermis exhibiting atypical melanocytic proliferation pattern of lentigo maligna, which is considered to be a precursor lesion,9,10 ii) proliferating melanophages and iii) melanin deposition in the dermal lesion. Immunohistochemical analysis revealed that staining for melanin markers including HMB-45 and MITF was negative, whereas S-100, SOX-10, and CD34 displayed strong, positive staining. Since the appearance of DM is difficult to determine under low-magnification, caution should be taken when a lesion similar to a neurofibroma or a fibroproliferative disease presents on the sun-exposed skin in the elderly, or when a scar tissue presents in a pigmented nevus lesion. A careful and comprehensive review at high magnification is critical for diagnosing DM. For example, DM is often neurotropic, and lymphocytic follicle-like structures at the periphery and base of the tumor could be suggestive for DM.
In case 3, spindle cells presented with evident atypia, with cells characterized by hyperchromatic nuclei, vesicular shape, increased nucleoplasmic ratio, and sometimes mitotic figures (Figure 2B), indicative of a malignant tumor. Case 3 also presented with characteristic melanin deposition, nerve involvement, atypical melanocytic proliferation, and lymphoid infiltrates. Despite the lack of additional immunostaining, we believed this case was consistent with the diagnosis of DM.
Pathologically, DM with mild cell atypia is often confused with neurofibroma, and S-100 and SOX-10 staining can not distinguish between the lesions. Elsensohn performed P53 staining on 20 neurofibroma tissues and 20 DM tissues, and found that the presence of P53 protein in DM samples (19/20) allowed DMs to be distinguished from neurofibromas which all stained negative for P53 (0/20). In our analysis, neither of the two cases stained positive for P53 (Figure 4D), while the control (basal cell carcinoma) tissue showed strong P53 expression. A previous study consisting of three Chinese DM patients also found that two of three cases were negative for P53 protein.34 Considering the complex role of P53 in the development of melanoma,35 we hypothesize that the positive expression rate for P53 in Chinese patients with DM is lower than the rate reported for other populations (95%). Further verification involving larger sample sizes of Chinese patients is required.
Molecular pathology helps to identify early stage acral and cutaneous melanomas from dysplastic junctional or compound nevus.22 In addition, CDKN2A genes deletion was revealed in DM patients in both protein and gene level.36 In case 1, the deletion of CDKN2A was observed, supporting the diagnosis of DM. CDKN2A is an oncogene in melanoma, and mainly regulates the cell cycle progress. Loss of CDKN2A is a frequent event in invasive melanoma and correlates with sensitivity to the CDK4/6 inhibitor.37
Treatment and Prognosis
DM was initially described as “highly aggressive and locally recurring”.38,39 Previous reports found that the survival rate of DM patients was at least comparable to that of non-DM patients, who presented with tumors of equivalent thickness.25,26 Surgery resection is the most widely used treatment, and has also been identified as an independent prognostic factor of DM in terms of overall survival.40 During resection surgery, the resection is determined by tumor invasion depth. Studies revealed that although the presence of neurophilic features could not predict the survival rate in DM, it did correlate with a higher recurrence rate.38 Although two of three cases in our study presented with nerve involvement, none of the three patients showed evidence of recurrence. This may be because the invasive tumors did not extend beyond the subcutaneous tissue. Alternatively, the lack of recurrence may be because we performed the resection surgeries with margins over 1cm, and applied local radiotherapy afterwards. In addition, it is possible that the follow-up periods in our study were not long enough to determine whether there was recurrence.
This study has several limitations. Firstly, the number of cases involved in our study was very small. The work into diagnosis and treatment of DM would be proceeded in our center, hoping to improve the diagnosis of DM clinically. Secondly, longer follow-up periods are required to fully determine the overall patients survival and recurrence rate. Finally, the hypothesis that Chinese DM patients do not express P53 requires further validation with a larger number of Chinese DM samples.
Conclusion
In conclusion, our findings indicate that DM in the Chinese population exhibit a similar pathology compared with previously reported cases, which is atypical proliferative spindle tumor cells infiltrated in the fibroproliferative matrix. Perineural invasion, lymphocytic infiltrates, and obvious epidermal melanocytic involvement are also present in Chinese DM patients. Clinically, DM is characterized by the absence of pigmentation and lack of specific manifestations of melanoma, which contribute to misdiagnosis. Immunohistochemical staining would be beneficial to differentiate between DM and other fibroblastic proliferative diseases. Furthermore, we propose that the absence of P53 in DM lesions should not exclude the diagnosis of DM in the Chinese population.
Ethics Statement
This study was approved by the Ethics Committee of the Shanghai Skin Disease Hospital.
Acknowledgments
The authors would like to thank the members of the pathology department of Shanghai Dermatology Hospital who generally provided the H&E sections and parts of immunohistochemistry results. This work was supported by the Clinical Research Plan of Shanghai Hospital Development Center (SHDC2020CR4020; SHDC12020117) and Funding from Shanghai Skin Disease Hospital (2018KYQD01).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Conlev J, Lattes R, Orr W. Desmoplastic malignant melanoma (A rare variant of spindle cell melanoma). Cancer. 1971;28(4):914–936. doi:10.1002/1097-0142(1971)28:4<914::AID-CNCR2820280415>3.0.CO;2-E
2. Frydenlund N, Mahalingam M. Desmoplastic melanoma, neurotropism, and neurotrophin receptors—what we know and what we do not. Adv Anat Pathol. 2015;22(4):227–241. doi:10.1097/PAP.0000000000000076
3. Zhuang Feng Z, Wu X, Chen V, Velie E, Zhang Z. Incidence and survival of desmoplastic melanoma in the United States, 1992–2007. J Cutan Pathol. 2011;38(8):616–624. doi:10.1111/j.1600-0560.2011.01704.x
4. Xu Z, Yibulayin F, Shi P, Feng L. Desmoplastic melanoma versus spindle cell melanoma: incidence and survival, 1973 to 2017. Medicine (Baltimore). 2018;97(29):e11563. doi:10.1097/MD.0000000000011563
5. DeWane ME, Kelsey A, Oliviero M, Rabinovitz H, Grant-Kels JM. Melanoma on chronically sun-damaged skin: lentigo maligna and desmoplastic melanoma. J Am Acad Dermatol. 2019;81(3):823–833. doi:10.1016/j.jaad.2019.03.066
6. Labrecque PG, Hu CH, Winkelmann RK. On the nature of desmoplastic melanoma. Cancer. 2015;38(3):1205–1213. doi:10.1002/1097-0142(197609)38:3<1205::AID-CNCR2820380322>3.0.CO;2-K
7. Busam KJ, Mujumdar U, Hummer AJ, et al. Cutaneous desmoplastic melanoma. Am J Surg Pathol. 2004;28(11):1518–1525. doi:10.1097/01.pas.0000141391.91677.a4
8. Lens MB, Newton-Bishop JA, Boon AP. Desmoplastic malignant melanoma: a systematic review. Br J Dermatol. 2015;152(4):673–678. doi:10.1111/j.1365-2133.2005.06462.x
9. Kooper-Johnson S, Mahalingam M, Loo DS. SOX-10 and S100 negative desmoplastic melanoma: apropos a diagnostically challenging case. Am J Dermatopathol. 2020;1.
10. Suo Z, Nesland JM. Electron microscopy in diagnosis of spindle cell tumors. Semin Diagn Pathol. 2003;20(1):5–12.
11. Nicolson NG, Han D. Desmoplastic melanoma. J Surg Oncol. 2019;119(2):208–215. doi:10.1002/jso.25317
12. Busam JK. Cutaneous desmoplastic melanoma. Adv Anat Pathol. 2005;12(2):92. doi:10.1097/01.pap.0000155071.86944.a6
13. Chen LL, Jaimes N, Barker CA, Busam KJ, Marghoob AA. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68(5):825–833. doi:10.1016/j.jaad.2012.10.041
14. Jain S, Allen PW. Desmoplastic malignant melanoma and its variants. A study of 45 cases. Am J Surg Pathol. 1989;13(5):358–373. doi:10.1097/00000478-198905000-00003
15. Rabbie R, Ferguson P, Molina-Aguilar C, Adams DJ, Robles-Espinoza CD. Melanoma subtypes: genomic profiles, prognostic molecular markers and therapeutic possibilities. J Pathol. 2019;247(5):539–551. doi:10.1002/path.5213
16. Ochoa CE, Joseph RW. Desmoplastic melanoma: a brief review and the efficacy of immunotherapy. Expert Rev Anticancer Ther. 2019;19(3):205–207. doi:10.1080/14737140.2019.1574573
17. Han D, Han G, Zhao X, et al. Clinicopathologic predictors of survival in patients with desmoplastic melanoma. PLoS One. 2015;10(3):e0119716. doi:10.1371/journal.pone.0119716
18. De Almeida LS, Requena L, Rütten A, et al. Desmoplastic malignant melanoma: a clinicopathologic analysis of 113 cases. Am J Dermatopathol. 2008;30(3):207–215. doi:10.1097/DAD.0b013e3181716e6b
19. Plaza JA, Bonneau P, Prieto V, et al. Desmoplastic melanoma: an updated immunohistochemical analysis of 40 cases with a proposal for an additional panel of stains for diagnosis. J Cutan Pathol. 2016;43(4):313–323. doi:10.1111/cup.12654
20. Serra S, Chetty R. p16. J Clin Pathol. 2018;71(10):853–858. doi:10.1136/jclinpath-2018-205216
21. Elsensohn A, Shiu J, Grove N, Hosking AM, Barr R, de Feraudy S. Distinguishing neurofibroma from desmoplastic melanoma: the value of p53. Am J Surg Pathol. 2018;42(3):372–375. doi:10.1097/PAS.0000000000000978
22. Morey AL, Murali R, Mccarthy SW, et al. Diagnosis of cutaneous melanocytic tumours by four-colour fluorescence in situ hybridisation. Pathology. 2009;41(4):383–387. doi:10.1080/00313020902915875
23. Lai Y, Wu Y, Liu R, et al. Four-color fluorescence in-situ hybridization is useful to assist to distinguish early stage acral and cutaneous melanomas from dysplastic junctional or compound nevus. Diagn Pathol. 2020;15(1). doi:10.1186/s13000-020-00937-9.
24. Chen JY, Hruby G, Scolyer RA, et al. Desmoplastic neurotropic melanoma: a clinicopathologic analysis of 128 cases. Cancer. 2008;113(10):2770. doi:10.1002/cncr.23895
25. Smithers BM, Mcleod GR, Little JH. Desmoplastic melanoma: patterns of recurrence. World J Surg. 1992;16(2):186–190. doi:10.1007/BF02071519
26. Gershenwald JE, Scolyer RA. Melanoma staging: American Joint Committee on Cancer (AJCC) 8th Edition and beyond. Ann Surg Oncol. 2018;25(8):2105–2110. doi:10.1245/s10434-018-6513-7
27. Oliver DE, Patel KR, Switchenko J. Comparing survival outcomes in early stage desmoplastic melanoma with or without adjuvant radiation. Roles of adjuvant and salvage radiotherapy for desmoplastic melanoma. Melanoma Res. 2016;26(1):35–41. doi:10.1097/CMR.0000000000000201
28. Abbott JL, Qureshi MM, Truong MT, et al. Comparing survival outcomes in early stage desmoplastic melanoma with or without adjuvant radiation. Melanoma Res. 2019;29(4):413–419. doi:10.1097/CMR.0000000000000532
29. Guadagnolo BA, Prieto V, Weber R, Ross MI, Zagars GK. The role of adjuvant radiotherapy in the local management of desmoplastic melanoma. Cancer. 2014;120(9):1361–1368. doi:10.1002/cncr.28415
30. Murali R, Shaw HM, Lai K, et al. Prognostic factors in cutaneous desmoplastic melanoma: a study of 252 patients. Cancer. 2010;116(17):4130–4138. doi:10.1002/cncr.25148
31. Fung MA, Sharon VR, Ratnarathorn M, Konia TH, Barr KL, Mirmirani P. Elastin staining patterns in primary cicatricial alopecia. J Am Acad Dermatol. 2013;69(5):776–782. doi:10.1016/j.jaad.2013.07.018
32. Hay JL, Christian SN. Skin cancer screening. Psycho-Oncology. 2021.
33. Calonje E, Fletcher CDM. Aneurysmal benign fibrous histiocytoma: clinicopathological analysis of 40 cases of a tumour frequently misdiagnosed as a vascular neoplasm. Histopathology. 2010;26(4):323–331. doi:10.1111/j.1365-2559.1995.tb00193.x
34. Yang Y, Yang F, Zhu Y, et al. Desmoplastic melanoma involving head and fingers: diagnostic challenges and clinical characteristics. Int J Clin Exp Pathol. 2019;12(10):3862–3869.
35. Najem A, Krayem M, Sales F, et al. P53 and MITF/Bcl-2 identified as key pathways in the acquired resistance of NRAS-mutant melanoma to MEK inhibition. Eur J Cancer. 2017;83:154–165. doi:10.1016/j.ejca.2017.06.033
36. Shain AH, Garrido M, Botton T, et al. Exome sequencing of desmoplastic melanoma identifies recurrent NFKBIE promoter mutations and diverse activating mutations in the MAPK pathway. Nat Genet. 2015;47(10):1194–1199. doi:10.1038/ng.3382
37. Young RJ, Waldeck K, Martin C, et al. Loss of CDKN2A expression is a frequent event in primary invasive melanoma and correlates with sensitivity to the CDK4/6 inhibitor PD0332991 in melanoma cell lines. Pigment Cell Melanoma Res. 2014;27(4):590–600. doi:10.1111/pcmr.12228
38. Livestro DP, Muzikansky A, Kaine EM. Biology of desmoplastic melanoma: a case-control comparison with other melanomas. J Clin Oncol. 2005;23(27):6739–6746. doi:10.1200/JCO.2005.04.515
39. Han D, Zager JS, Yu D, Zhao X, Walls B. Desmoplastic melanoma: is there a role for sentinel lymph node biopsy? Ann Surg Oncol. 2013;20(7):2345–2351. doi:10.1245/s10434-013-2883-z
40. Shi P, Xu Z, Lei X, et al. Desmoplastic melanoma: demographic and clinicopathological features and disease-specific prognostic factors. Oncol Lett. 2019;17(6):5619–5627. doi:10.3892/ol.2019.10259
41. Debarbieux S, Ronger-Salve S, Dalle S, Balme B, Thomas L. Dermoscopy of desmoplastic melanoma: report of six cases. Br J Dermatol. 2008;159(2):360–363. doi:10.1111/j.1365-2133.2008.08687.x
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.