Back to Journals » Drug Design, Development and Therapy » Volume 13
Delivery of folic acid-modified liposomal curcumin for targeted cervical carcinoma therapy
Authors Wang WY, Cao YX, Zhou X, Wei B
Received 18 February 2019
Accepted for publication 19 April 2019
Published 4 July 2019 Volume 2019:13 Pages 2205—2213
DOI https://doi.org/10.2147/DDDT.S205787
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Cristiana Tanase
Wen-Yan Wang,1,2 Yun-Xia Cao,2 Xiao Zhou,3 Bing Wei1
1Department of Obstetrics and Gynecology, The Second Hospital of Anhui Medical University, Hefei, Anhui, 230601, People’s Republic of China; 2Teaching and Research Group of Obstetrics and Gynecology, Anhui Medical University, Hefei, Anhui, 230032, People’s Republic of China; 3Department of Cardiothoracic Surgery, The Second Hospital of Anhui Medical University, Hefei, Anhui, 230601, People’s Republic of China
Introduction: In this study, novel folic acid (FA)-modified curcumin (CUR) liposomes (LPs) were developed and evaluated for their antitumor activity in vitro and in vivo.
Methods: Characterization of the LPs, including transmission electron microscopy, morphology, particle size, and zeta potential studies, was carried out. Drug entrapment efficiency, drug-loading capacity, and release properties in vitro were tested. The in vitro growth inhibition activity, cellular uptake efficiency, and cell apoptosis of FA-modified CUR LPs were also investigated by a cervical cancer HeLa cell model.
Results: The optimized distearoyl-l-a-phosphatidylethanolamine (DSPE)-PEG2000-FA-LPs/CUR formed spherical vesicles of nanometer sizes and had particle sizes of 112.3±4.6 nm, polydispersity index of 0.19±0.03, and zeta potential of −15.3±1.4 mV. In addition, the EE% and DL% of (DSPE)-PEG2000-FA-LPs/CUR were 87.6% and 7.9%, respectively. Compared with the free drug, FA-modified CUR LPs had sustained-release properties in vitro. In vivo, a strong green fluorescence was observed in the cytoplasmic region after incubation of (DSPE)-PEG2000-FA-LPs/CUR for 2 hrs.
Conclusion: (DSPE)-PEG2000-FA-LPs/CUR showed a superior antiproliferative effect on HeLa cells and had a better antitumor effect in vivo than the non-modified LPs. These results indicated that (DSPE)-PEG2000-FA-LPs/CUR was a promising candidate for antitumor drug delivery.
Keywords: curcumin, folic acid, liposome, release, cell uptake, antiproliferative, antitumor study, HeLa
Introduction
Cervical cancer is the third most commonly diagnosed cancer worldwide and the fourth leading cause of cancer death in women.1 In China, there are about 132,000 new cases of cervical cancer each year, accounting for 28% of the total number of new cases of cervical cancer worldwide.2 At present, the most effective treatment for cervical cancer is achieved through a combination of cisplatin-based chemotherapy with radiation.3 However, the clinical use of this combination treatment is limited because of several serious side effects, such as nephrotoxicity, neurotoxicity, hematological toxicity, chemoresistance, and off-target damage to normal tissues.4 In the early stages of the disease, this treatment regimen would result in a patient survival rate of 90%. However, the figure decreases to 17% among patients at advanced stages5 due to the presence of chemoradioresistant cells.6 Therefore, ongoing efforts are necessary for the development of new, effective therapeutic strategies to enhance chemotherapeutic efficacy and decrease these side effects, maximizing the local response and patient survival.
Curcumin (CUR) is the chief component of the spice turmeric and is derived from the rhizome of the East Indian plant Curcuma longa. Curcuma longa is a member of the Zingiberacae (ginger) family of botanicals and is a perennial plant native to Southeast Asia.7 It has low intrinsic toxicity and a wide range of pharmacological properties, including antitumor, anti-amyloid, antioxidant, and anti-inflammatory effects. Previous studies have shown that free CUR can induce cell cycle arrest and/or apoptosis and inhibit the activation of nuclear factor kappa B in different tumor cell lines.8,9 CUR has been associated with regression of premalignant lesions of the bladder, soft palate, gastrointestinal tract, cervix, and skin, with treatment responses in established malignancy.10,11 The only factor limiting the use of free CUR in the treatment of cancer is its poor solubility in water, which in turn limits the bioavailability of the system when administered orally. The reason for the treatment failure of CUR is most likely that the drug concentration in the body cannot reach the effective antitumor level. In addition, nonspecific binding capacity also decreases CUR concentration in tumor tissues.
The application of liposomal formulations as carriers for anticancer drug modified by tumor-active-targeted ligand folate has been trialed and described in the literature in recent years.12–15 Folate conjugates can bind to the folate receptor (FR) with high affinity and enter the FR-elevated malignant cells by receptor-mediated endocytosis,16,17 and meanwhile avoid being endocytosed by those normal cells that express a low level of FRs.18 Thus, adding folic acid (FA) to the distal ends of liposome (LP)-anchored PEG molecules can further improve tumor cell selectivity and the uptake efficiency of LPs. However, little research has been done on the pharmacodynamics of FA-modified CUR liposomal formulation.
The aim of this study was to develop a novel FA-modified liposomal formulation that could exhibit efficient and stable encapsulation of CUR and therefore mediate its delivery to the solid tumor effectively and specifically. Characterization of the LPs, including transmission electron microscopy (TEM), morphology, particle size, and zeta potential, was carried out. Drug entrapment efficiency, drug-loading capacity, and release properties in vitro were tested. The in vitro growth inhibition activity, cellular uptake efficiency, and cell apoptosis of FA-modified CUR LPs were also investigated by a cervical cancer HeLa cell model.
Materials and methods
Materials
CUR was purchased from Changyue Biological Technology Co., Ltd. (Xi’an, China). Distearoyl-L-a-phosphatidylethanolamine (DSPE)-PEG2000-FA was provided by the HuiJia Biopharma Co., Ltd (Xiamen, China). Soybean phosphatidylcholine (SPC), DSPE-PEG2000, and cholesterol (CHOL) were obtained from Sinopharm Chemical Reagent (Shanghai, China). The human cervical carcinoma HeLa cell line was provided by Second Hospital of Anhui (the Second Hospital of Anhui Medical University) and upon the approval of the ethics committee of the Second Hospital of Anhui Medical University. All other reagents were obtained from Sinopharm Chemical Reagent (Shanghai, China). Methanol and acetonitrile (chromatographic grade) were obtained from Sigma (Aldrich, USA). Deionized water used throughout the research was produced using a Milli-Q System.
Animals
Experiments were carried out on rats weighing 220±20 g and Balb/c mice weighing 20±2 g. The animals were kept in cages in a room at a temperature of 25±2°C, with a 12:12 light–dark cycle. They had free access to food and water. All experiments were carried out in strict accordance with the experimental animal guidelines issued by the National Institutes of Health and approved by the Second Hospital of Anhui Medical University.
Preparation of LPs
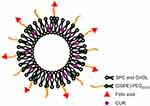
FA-modified LPs containing CUR were prepared by the thin-film hydration method as described previously.19 Briefly, CUR, (DSPE)-PEG2000-FA, SPC, and CHOL (5, 5, 30, and 15, w/w) were dissolved in 5 mL of chloroform to form a mixed solution and then the organic solvent was removed under reduced pressure at 40°C by rotary evaporation to form a thin solid film on the inner walls of the round-bottomed flask. This film was then flushed with nitrogen for 30 mins and stored overnight in a desiccator to remove any traces of chloroform. Glucose and mannitol (1:1, w/w) were dissolved in PBS (pH=7.4). The lipid film was then hydrated with 5 mL of PBS (pH=7.4) at 55°C by rotation (180 rpm×0.5 hr) to form (DSPE)-PEG2000-FA-LPs/CUR (Figure 1). Finally, the (DSPE)-PEG2000-FA-LPs/CUR obtained was freeze-dried into powder. For the preparation of different CUR LPs and DSPE-PEG2000-LPs/CUR, a similar procedure was carried out.
Characterization
In order to determine the optimal parameters relative to the formulation of the (DSPE)-PEG2000-FA-LPs/CUR, drug-loading (DL%), encapsulation efficiency (EE%), particle size, zeta potential, polydispersity index (PDI), and surface morphology of the LPs were measured. Dynamic light scattering (DLS; Zetasizer Nano ZS-90; Malvern Instruments, Malvern, UK) and transmission electron microscope (TEM; Tecnai G2 20; FEI, Memphis, TN, USA) were used for characterizing.
DL% and EE% were calculated as described earlier.20 Firstly, CUR was extracted from LPs (20 mg) with 2 mL 5% glacial acetic acid: acetonitrile (55:45, v/v), and then the extracted solution was properly diluted prior to HPLC analysis. The content of CUR in the LPs was determined by the HPLC method. DL% and EE% were calculated according to Equations (1) and (2), respectively:
(1) (2)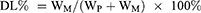
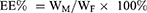
where WP is the weight of initial feeding polymer, WM is the weight of drug incorporated in LPs, and WF is the weight of initial feeding drug.
In vitro drug release
In vitro release of CUR from the LPs was carried out by the dialysis membrane method. (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, CUR LPs, and free CUR (≈5 mg CUR) were suspended in 1 mL aliquots of distilled water and placed in dialysis tubing (8,000–12,000 molecular weight cutoff; Greenbird Inc., Shanghai, China). Dialysis tubes containing (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, CUR LPs, and free CUR were then submerged in 200 mL of PBS (pH 7.4) as the release medium (containing ethanol 20%, v/v) at 37°C and immediately shaken at a speed of 75 rpm. Subsequently, 0.5 mL aliquots of the release medium were sampled at 0.5, 1, 2, 4, 6, 8, 10, 12, and 24 hrs and immediately replaced with equal volumes of fresh release medium. Finally, the samples were analyzed by HPLC. In the release medium, ethanol was used to provide the sink condition because CUR was insoluble in water.
Stability
The stability of (DSPE)-PEG2000-FA-LPs/CUR after storage at 4°C and 25°C for a period of 3 months was analyzed according to the ICH guidelines. The physical appearance, particle size, zeta potential, EE%, and DL% of the samples were analyzed over the 3-month period with an interval of 1 month.
In vitro anticancer activity assay
The in vitro anticancer activity of (DSPE)-PEG2000-FA-LPs/CUR on human cervical carcinoma HeLa cell line was evaluated by MTT assay. Nontoxic control l experiments were carried out using a complete growth culture medium. Briefly, cells were seeded into a 96-well plate at a density of 2×104 cells per well. After 24 hrs, the cells were incubated with (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, CUR LPs, and free CUR (from 0.002 µg/mL to 20 µg/mL) with the same final concentration for 48 hrs. Blank LPs were added at different concentrations as controls. Cells were then incubated with 20 µL MTT solution (5 mg/mL in PBS) for 4 hrs before the medium was removed and cells were dissolved with 200 µL DMSO; the absorbance was 570 and 490 nm as measured by a microplate reader (Thermo Fisher Scientific). Cell viability (%) was calculated according to the following formula:
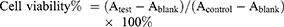
Atest, Acontrol, and Ablank represent the absorbance of cells with different treatments, untreated cells, and blank culture media, respectively. Each experiment was performed six times. Half-maximal inhibitory concentration (IC50) was calculated for all samples by survival analysis using GraphPad Prism 5 software.
Cellular uptake
The cellular internalization of (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, CUR LPs, and free CUR was visualized by confocal microscopy using coumarin-6 as a fluorescent probe. HeLa cells were plated in six-well plates at 37°C under 5% CO2 in folate-free RPMI 1640 medium containing 10% FBS. Hela cells were inoculated in a cell culture dish with 4×105 cells per dish as the initial density. Cells were then incubated with coumarin-6-adsorbed free CUR, (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, and CUR LPs (equivalent to 0.1 µg/mL of coumarin-6). After incubation at 37°C for 2 hrs, the cells were washed three times with cold PBS to remove unbound LPs and then observed under a confocal microscope.
For CUR uptake quantitative estimates, the density of cells inoculated on 24-well plates was 3×104 .When 70–80% confluence was achieved, cells were incubated with coumarin-6-adsorbed free CUR, (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, and CUR LPs (equivalent to 0.1 µg/mL of coumarin-6). After 2 hrs of culture, cells were washed several times with cold PBS. Subsequently, cells were dissolved by addition of Triton X-100 (0.1%). Fluorescence intensities were measured by a multimode microplate reader at an excitation wavelength of 440 nm and an emission wavelength of 520 nm.
Pharmacodynamics
Tumor reduction studies were conducted as reported earlier.22 Female Balb/c mice aged 6–8 weeks were used for the experiment. Tumors were induced by subcutaneous injection of HeLa cells (~5×106 cells in 100 μL PBS) in the lower right flank of mice and were allowed to grow for a period of time (approximately 15 days) to a size of approximately 100–150 mm3 as measured by vernier calipers. The animals were then randomly divided into 5 groups of 8 and blank LPs, free CUR, CUR LPs, (DSPE)-PEG2000-LPs/CUR, and (DSPE)-PEG2000-FA-LPs/CUR (equivalent to 25 mg/kg CUR, on alternate days for three weeks) were injected intraperitonially. The tumor volume was measured every 3 days. Animals were sacrificed after 51 days and the main organs and tumor samples were collected for further analyses. The specimens were fixed with 4% paraformaldehyde and then paraffin embedded for H&E staining and histological examination.
Statistical analysis
All data points were denoted as mean ± SD. A two-tailed t-test or a one-way ANOVA was performed, respectively, when comparing two or more groups. Statistical significance was defined as P<0.05. Data were shown as mean ± SD.
Results and discussion
Preparation and characterization
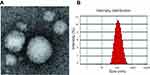
In our current study, we designed FA-mediated stabilized liposomes encapsulating CUR to improve the therapeutic effect of CUR in the treatment of cervical cancer via the combination of active targeting and molecular targeting. A systematic approach was undertaken to develop and characterize preparation methods and liposome formulations in order to control drug leakage and thus maintain optimal drug loading inside the liposomes to increase the delivery of the drug to tumor tissues. CUR was encapsulated into FA-modified and nonmodified LPs using the thin-film hydration method. The obtained physicochemical properties of the formulations are summarized in Table 1. The TEM images and DLS size distribution of the (DSPE)-PEG2000-FA-LPs/CUR are shown in Figure 2. The optimized (DSPE)-PEG2000-FA-LPs/CUR formed spherical vesicles at nanometer sizes and had particle sizes of 112.3±4.6 nm, PDI of 0.19±0.03, and zeta potential of −15.3±1.4 mV. The negative zeta potential might have resulted from the presence of negatively charged lipid DSPE-PEG2000 in the formulation.21 In addition, the EE% and DL% of (DSPE)-PEG2000-FA-LPs/CUR were 87.6% and 7.9%, respectively. Under the same loading conditions, ordinary CUR LPs and nonmodified LPs appeared to have higher EE% and DL% and smaller particle sizes; however, no relevant differences were observed in terms of these parameters among the three CUR LPs. In order to determine the influence of the CUR/lipid incubation ratio on loading, several ratios (1:5, 1:9, 1:18, 1:30, w:w) were tested on (DSPE)-PEG2000-FA-LPs/CUR. While a CUR/lipid incubation ratio of 1:9 or lower provided excellent EE% values (>90%), only the incubation ratio of 1:9 provided the highest DL% among all the formulations.
 |
Table 1 Physicochemical properties of CUR LPs; DSPE-PEG2000-LPs/CUR and (DSPE)-PEG2000-FA-LPs/CUR (n=3) |
Stability
The stability of (DSPE)-PEG2000-FA-LPs/CUR was monitored by changes in physical appearance, particle size, zeta potential, EE%, and DL% during storage at 4°C and 25°C (Table 2). The zeta potential exhibited little change over the 3-month storage, showing the stability of (DSPE)-PEG2000-FA-LPs/CUR. The results also indicated that during storage at 4°C or 25°C, no significant variation in physical appearance, particle size, EE%, and DL% was detected. It appears that the liposomal formulation showed excellent stability during storage, and no drug precipitation or liposome aggregation was observed throughout the 90 days.
 |
Table 2 Stability of (DSPE)-PEG2000-FA-LPs/CUR after storage at 4°C and 25°C for a period of 3 months (n=3) |
In vitro drug release
CUR release in vitro was also investigated using the dialysis method in PBS (pH 7.4) at 37°C for 24 hrs to examine the drug-release properties of LPs (Figure 3). Unlike the rapid release of free CUR, as predicted, three LP types, (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, and CUR LPs, exhibited similar gradual release properties under pH 7.4. Over time, all formulations of CUR LPs were released much more slowly than free drug. After 2 hrs, the cumulative release was about 86% in the free drug (CUR) and 30% in LPs. A biphasic release was observed in this case with a rapid release of about 30% in 2 hrs followed by sustained drug release of about 70% over 12 hrs. It suggested that in comparison with the free drug, (DSPE)-PEG2000-FA-LPs/CUR have sustained release properties. However, there was no significant difference among the three LPs in the release curve.
The (DSPE)-PEG2000-FA-LPs/CUR was surface-coated with the hydrophilic polymer PEG, which formed a hydrophilic barrier to prevent the accumulation of LPs and to improve their physical stability. In addition, phospholipid, the main ingredient used in this experiment, is a corrosive substance. Because of the poor water solubility of the drug, it is difficult for it to penetrate into the matrix of the LPs. The sustained release of the drug might mainly contribute to the corrosion rates of the LPs prepared with these phospholipids. After calculation, the in vitro drug-release kinetic model of (DSPE)-PEG2000-FA-LPs/CUR in release medium was found to fit well with the Higuchi equation: Q=9.523t1/2–1.231 (r=0.993). Thus, it was speculated that the sustained-release property of LPs could assist in decreasing drug leakage before drug-loaded systems arrived at the targeted tumors and cells.
In vitro anticancer activity assay
For anticancer effects in vitro, HeLa cells were treated with either (DSPE)-PEG2000-FA-LPs/CUR, (DSPE)-PEG2000-LPs/CUR, CUR LPs, or free CUR at different concentrations of the drug. Cells were then subjected to an MTT assay. The cell viability profile is presented in Figure 4. As shown, a significant reduction in cell viability was observed in the (DSPE)-PEG2000-FA-LPs/CUR group, while the cytotoxicity of cells was clearly lower in the free CUR and (DSPE)-PEG2000-LPs/CUR groups. The negative control was represented by blank LPs, for which cell survival was 100%. From this result, we found that 1) the carrier material used in this study was safe and nontoxic; 2) as the CUR concentration increased, greater cell death was observed; 3) (DSPE)-PEG2000-FA-LPs/CUR showed a superior antiproliferative effect on HeLa cells, which may be expected to demonstrate obvious antitumor effect in vivo. After calculation, the IC50 value of the (DSPE)-PEG2000-FA-LPs/CUR was found to be 0.45 µg/mL compared with a value of 0.82 µg/mL for (DSPE)-PEG2000-LPs/CUR, 1.47 µg/mL for free CUR, and 1.21 µg/mL for CUR LPs (Figure 4). Blank LPs were tested as controls and did not induce significant cell death (data not shown).
Cellular uptake
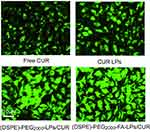
Free CUR, CUR LPs, (DSPE)-PEG2000-LPs/CUR, and (DSPE)-PEG2000-FA-LPs/CUR were labeled by a fluorescent probe. As shown in Figure 5, the nucleus displayed green fluorescence, which was stained by coumarin-6. A strong green fluorescence was observed in the perinuclear region of HeLa cells treated with (DSPE)-PEG2000-FA-LPs/CUR, indicating that a sufficient amount of (DSPE)-PEG2000-FA-LPs/CUR entered the cytoplasm. In addition, the fluorescence intensity of the (DSPE)-PEG2000-LPs/CUR group was weaker than that of the (DSPE)-PEG2000-FA-LPs/CUR group.
Moreover, HeLa cells pretreated with free CUR and CUR LPs also exhibited mild green fluorescence, likely attributable to weak ability to cross cells. The cellular uptake ratio of the LPs was detected by a multimode microplate reader to be 5.7±1.6%, 16.5±4.9%, 38.4±4.4%, and 66.4±6.2% for free CUR, CUR LPs, (DSPE)-PEG2000-LPs/CUR, and (DSPE)-PEG2000-FA-LPs/CUR-treated HeLa cells, respectively. These results demonstrated that the FA-modified strategy can facilitate the high-efficiency uptake of CUR by HeLa cells.
Pharmacodynamics
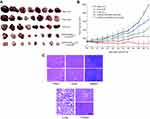
To assess the in vivo antitumor efficacy of (DSPE)-PEG2000-FA-LPs/CUR, human cervical cancer xenograft mice were employed. As shown in Figure 6, all the treatment groups were better able to inhibit tumor growth and decrease tumor size compared with the control group. At the end of the experiment, tumors were found to be more sensitive to (DSPE)-PEG2000-FA-LPs/CUR at a volume of 77.3±56.5 mm3, compared to that of the control group (987.6±89.4 mm3),free CUR (634.3±67.4 mm3), CUR LPs (432.7±52.6 mm3), and (DSPE)-PEG2000-LPs/CUR (232.6±57.2 mm3). The results indicated a significant effect on the reduction rate of tumor volume for (DSPE)-PEG2000-FA-LPs/CUR, which was statistically higher than other groups. The results mainly concerned the following aspects: 1) the effect of EPR (confirmed by another biodistribution study; data not shown); 2) enhancing intratumoral retention of CUR in terms of (DSPE)-PEG2000-FA-LPs/CUR; 3) receptor ligand–mediated targeted delivery. H&E staining was used to determine whether there was organ damage. The results showed that there were no obvious lesions in heart, liver, spleen, lung, and kidney of the two groups, suggesting that (DSPE)-PEG2000-FA-LPs/CUR had no obvious acute toxicity in vivo (Figure 6C).
We prepared a delivery system of FA-mediated liposomes with encapsulated CUR in this study. According to the results, the drug delivery system in our study was in an appropriate nanoscale size range, exhibited specific targets to tumor cells, and induced more cell death and cell apoptosis. These results suggested that (DSPE)-PEG2000-FA-LPs/CUR possessed the basic characteristics of targeted delivery systems and had the potency to improve the antitumor effects of CUR.
Disclosure
The authors report no conflicts of interest in regard to this work.
References
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90. doi:10.3322/caac.20107
2. Di J, Rutherford S, Chu C. Review of the cervical cancer burden and population-based cervical cancer screening in China. Asian Pac J Cancer Prev. 2015;16(17):7401–7407.
3. Ramakrishnan S, Partricia S, Mathan G. Overview of high-riskHPV’s 16 and 18 infected cervical cancer: pathogenesis to prevention. Biomed Pharmacother. 2015;70:103–110. doi:10.1016/j.biopha.2014.12.041
4. Rose PG, Bundy BN, Watkins EB, et al. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med. 1999;340(15):1144–1153. doi:10.1056/NEJM199904153401502
5. Morris M, Eifel PJ, Lu J, et al. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aorticradiation for high-risk cervical cancer. N Engl J Med. 1999;340(15):1137–1143. doi:10.1056/NEJM199904153401501
6. Segovia-Mendoza M, Jurado R, Mir R, et al. Antihormonal agents as a strategy to improve the effect of chemo-radiation in cervical cancer: in vitro and In vivo Study. BMC Cancer. 2015;15:21. doi:10.1186/s12885-015-1584-3
7. Chattopadhyay I, Biswas K, Bandyopadhyay U, et al. Turmeric and curcumin biological actions and medicinal applications. Curr Sci. 2004;87:44–50.
8. Maheshwari RK, Singh AK, Gaddipati J, Srimal RC. Multiple biological activities of 18rcumin: a short review. Life Sci. 2006;78(18):2081–2087. doi:10.1016/j.lfs.2005.12.007
9. Wang Z, Zhang Y, Banerjee S, Li Y, Sarkar FH. Notch-1 down-regulation by curcumin is associated with the inhibition of cell growth and the induction of apoptosis in pancreatic cancer cells. Cancer. 2006;106(11):2503–2513. doi:10.1002/cncr.21904
10. Sa G, Das T. Anticancer effects of curcumin: cycle of life and death. Cell Div. 2017;3:14. doi:10.1186/1747-1028-3-14
11. Adahoun MA, A-Akhras MH, Jaafar MS, Bououdina M. Enhanced anti-cancer and antimicrobial activities of curcumin nanoparticles. Artif Cells Nanomed Biotechnol. 2017;45(1):98–107. doi:10.3109/21691401.2015.1129628
12. Chaudhury A, Tan BJ, Das S, Chiu GNC. Increased ERK activation and cellular drug accumulation in the enhanced cytotoxicity of folate receptor-targeted liposomal carboplatin. Int J Oncol. 2012;40(3):703–710. doi:10.3892/ijo.2011.1262
13. Watanabe K, Kaneko M, Maitani Y. Functional coating of liposomes using a folate-polymer conjugate to target folate receptors. Int J Nanomedicine. 2012;7:3679–3688. doi:10.2147/IJN.S32853
14. Liu Y, Xu S, Teng L, et al. Synthesis and evaluation of a novel lipophilic folate receptor targeting ligand. Anticancer Res. 2011;31(5):1521–1525.
15. Xiong S, Yu B, Wu J, Li H, Lee RJ. Preparation, therapeutic efficacy and intratumoral localization of targeted daunorubicin liposomes conjugating folate-PEG-CHEMS. Biomed Pharmacother. 2011;65(1):2–8. doi:10.1016/j.biopha.2010.10.003
16. Hilgenbrink AR, Low PS. Folate receptor-mediated drug targeting: from therapeutics to diagnostics. J Pharm Sci. 2005;94(10):2135–2146. doi:10.1002/jps.20457
17. Lu Y, Ding N, Yang C, Huang L, Liu J, Xiang G. Preparation and in vitro evaluation of a folate-linked liposomal curcumin formulation. J Liposome Res. 2012;22(2):110–119. doi:10.3109/08982104.2011.627514
18. Sabharanjak S, Mayor S. Folate receptor endocytosis and trafficking. Adv Drug Deliv Rev. 2004;56(8):1099–1109. doi:10.1016/j.addr.2004.01.010
19. Daeihamed M, Dadashzadeh S, Haeri A, et al. Potential of liposomes for enhancement of oral drug absorption. Curr Drug Deliv. 2016;14:289–303.
20. Zhang HX, Wang JX, Zhang ZB, Le Y, Shen ZG, Chen JF. Micronization of atorvastatin calcium by antisolvent precipitation process. Int J Pharm. 2009;374(1–2):106–113. doi:10.1016/j.ijpharm.2009.02.015
21. Hinrichs WL, Mancenido FA, Sanders NN, et al. The choice of a suitable oligosaccharide to prevent aggregation of PEGylated nanoparticles during freeze thawing and freeze drying. Int J Pharm. 2006;311(1–2):237–244. doi:10.1016/j.ijpharm.2005.12.032
22. Sreekanth CN, Bava SV, Sreekumar E, Anto RJ. Molecular evidences for the chemosensitizing efficacy of liposomal curcumin in paclitaxel chemotherapy in mouse models of cervical cancer. Oncogene. 2011;30:3139–3152. doi:10.1038/onc.2011.23
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.