Back to Journals » Cancer Management and Research » Volume 11
Decitabine-Containing Conditioning Regimen for Allogeneic Hematopoietic Stem Cell Transplantation in Patients with Intermediate- and High-Risk Myelodysplastic Syndrome/Acute Myeloid Leukemia: Potential Decrease in the Incidence of Acute Graft versus Host Disease
Authors Wang QY, Li Y, Liang ZY, Yin Y, Liu W, Wang Q , Dong YJ, Sun YH , Xu WL, Ren HY
Received 4 September 2019
Accepted for publication 21 November 2019
Published 4 December 2019 Volume 2019:11 Pages 10195—10203
DOI https://doi.org/10.2147/CMAR.S229768
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Eileen O'Reilly
Qing Ya Wang,* Yuan Li,* Ze Yin Liang, Yue Yin, Wei Liu, Qian Wang, Yu Jun Dong, Yu Hua Sun, Wei Lin Xu, Han Yun Ren
Department of Hematology, Peking University First Hospital, Peking University, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yuan Li; Han Yun Ren
Department of Hematology, Peking University First Hospital, Peking University, No.8 Xi Shi Ku Street, Xi Cheng District, Beijing 100034, People’s Republic of China
Tel +86 1083575746
; +86 1083575680
Email [email protected]; [email protected]
Purpose: To evaluate the role of Decitabine in the allo-HSCT conditioning regimen for intermediate- and high-risk patients with MDS or AML.
Patients and methods: Retrospective analysis of data pertaining to 76 intermediate- and high-risk patients with MDS or AML who underwent allo-HSCT between December 2005 and June 2018 at the Peking University First Hospital. Forty patients received Decitabine-containing conditioning regimen before transplantation, while thirty-six patients received regimen without Decitabine.
Results: Over a median follow-up of 40 months (range, 1 to 155), the cumulative incidence of grade II to IV acute graft versus host disease was 12.4% [95% confidence interval (CI) 4.9–30.9%] in the Decitabine group and 41.5% (95% CI 28.1–61.2%) in the non-Decitabine group (P=0.005). On multivariate analysis, Decitabine-containing conditioning regimen was found to protect against grade II to IV aGVHD (HR=0.279, 95% CI 0.102–0.765, P=0.013). Incidence of respiratory infection in the Decitabine and non-Decitabine groups was 22.5% and 52.78%, respectively (P=0.012). No significant between-group difference was observed with respect to 3-year OS, DFS, or RR (P=0.980, 0.959, and 0.573, respectively), while the median relapse time was longer in the Decitabine group [7 months (range, 2–12) versus 3 months (range, 2–4), P=0.171]. Decitabine-containing conditioning showed a tendency for lower relapse rate among higher risk patients, as assessed by IPSS R; however, the between-group difference was not statistically significant (P=0.085).
Conclusion: Inclusion of Decitabine in the conditioning regimen for allo-HSCT in intermediate- and high-risk patients may lower the incidence of aGVHD and respiratory infections, and contribute to longer median relapse time.
Keywords: myelodysplastic syndrome, acute myeloid leukemia, Decitabine, transplantation conditioning, graft versus host disease, allogeneic hematopoietic stem cell transplantation
Introduction
Myelodysplastic Syndrome (MDS) is a clonal hematopoietic disorder characterized by ineffective hematopoiesis, refractory cytopenia, and hyperproliferative bone marrow. Patients categorized as international prognostic scoring system (IPSS) intermediate-2 and high-risk groups [currently considered as higher risk MDS (HR-MDS)] have aggressive malignancies with a median survival of 1.1 years and 0.4 years, respectively, if treated only with supportive care.1
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only curative treatment for intermediate/high-risk MDS and acute myeloid leukemia (AML). For low- and intermediate-1-risk MDS, delayed hematopoietic stem cell transplantation was shown to achieve maximal life expectancy; however, immediate transplantation conferred the highest survival benefit in patients with intermediate-2- and high-risk disease.2,3 The National Comprehensive Cancer Network (NCCN) guidelines recommend allo-HSCT for MDS of intermediate-2- and high risk as soon as possible. However, relapse and graft versus host disease (GvHD) are two main causes of treatment failure. Recent studies have implicated epigenetic changes such as abnormal DNA methylation, histone modification, and non-coding RNA in the pathogenesis of MDS.4 Decitabine (Dec, 5-aza deoxycytidine), a pyrimidine analogue with significant antileukemic activity, acts as an inhibitor of cytosine methylation, which results in activation of silent genes.5 Some clinical studies have demonstrated the efficacy of Dec in the treatment of refractory leukemia and MDS before or after allo-HSCT. Dec therapy was shown to be associated with reduced incidence of GvHD, lower relapse rate, and increased overall survival with acceptable tolerance.6–9 Thus, Dec may be an ideal candidate for use in combination with standard preparative regimen for patients scheduled to undergo allo-HSCT. In this study, we retrospectively evaluated the efficacy and safety of Dec-containing conditioning regimen in 76 patients with intermediate- or high-risk MDS/AML.
Materials and Methods
Patients and Methods
Seventy-six patients who were diagnosed with intermediate- or high-risk MDS or transformed acute myeloid leukemia (t-AML) at the Peking University First Hospital between December 2005 and June 2017 were analyzed in this study. Of these, 40 patients had received Dec-containing conditioning before allo-HSCT (Dec group). Another 36 patients received non-Decitabine conditioning before allo-HSCT (non-Dec group) were selected as the control group. All patients were classified according to the 2008 WHO MDS Classification and were evaluated by revised international prognostic scoring system (IPSS-R) risk category. We performed allo-HSCT immediately after the diagnosis of MDS of high- and higher-risk categories. As for t-AML patients, standard-dose “3+7” induction chemotherapy (Cytarabine 100–200 mg continuous infusion for 7 days with Idarubicin 12 mg/m2 or Daunorubicin 60–90 mg/m2 for 3 days) or priming chemotherapy was administered before allo-HSCT. In China, CAG priming chemotherapy is widely used in patients with AML. It consists of a small dose of cytarabine (10 mg/m2, every 12 h, subcutaneous injection, × 14 days), plus G-CSF and administered in combination with aclarubicin.10 CR was defined as bone marrow with ≤5% myeloblasts with normal maturation of all cell lines, hemoglobin ≥ 11 g/dL, platelets ≥100×109/L, neutrophils ≥1×109/L, and no circulating blasts. This study was conducted in accordance with the declaration of Helsinki and was approved by the institutional review board at the Peking University First Hospital in June 2018. The requirement for written informed consent was waived off due to the retrospective nature of the study. All data used in this manuscript were anonymized.
Treatment Plan
Conditioning Regimen
The conditioning regimens were modified Bu/Cy and Bu/Flu, which were recommended in China.11 Conditioning protocols were as follows: cytarabine 2 g/m2/d for 3 days; busulfan 3.2 mg/kg/d intravenously for 3 days, combined with cyclophosphamide 1.8g/m2/d for 2 days or fludarabine 50 mg for 3–6 days (total dose: 200 mg/m2).12,13 Dec was administered at a dose of 15 mg/m2 intravenously from day −16 to −12. Rabbit anti-thymocyte globulin (ATG) was used for 3 days before transplantation; patients with haploidentical or unrelated donors were administered 7.5–10 mg/kg ATG as part of conditioning, and patients with HLA identical sibling donors were administered 0 to 5mg/kg ATG.
GvHD Prophylaxis and Treatment
GvHD prophylaxis consisted of cyclosporin A (CsA), mycophenolate mofetil (MMF), and methotrexate (MTX). MMF 50 mg every 12 hrs was administered orally from the start of conditioning up to 30 days after transplantation. Intravenous (i.v.) CsA 5 mg/kg daily was administered from day −6 to the time of bowel function recovery, and then switched to oral CsA. Serum CsA concentration was monitored, and the dosage was adjusted to achieve serum concentrations in the range of 150 to 250 ng/mL. MTX was administered at a dose of 10–15 mg/m2 i.v. on days +1, +3, +5, +11 after transplantation. Grade I aGvHD was controlled by topical treatment and optimization of treatment (e.g. adjustment of CsA dosage). Firstline therapy of grade II–IV aGvHD was methylprednisolone 1 to 2 mg/kg daily. Patients refractory to steroid therapy received secondary therapy, such as basiliximab i.v. 1 mg/kg.
Stem Cell Mobilization, Collection, and Transplantation
Donors received G-CSF 5 µg/kg subcutaneously every 12 hrs starting from 3 days prior to the collection of bone marrow stem cells. Peripheral blood harvesting was carried out on the fourth day after G-CSF treatment.
Supportive Care
Antibiotics were used to prevent microbial infections as appropriate. Co-trimoxazole (SMZ/TMP) was used to prevent pneumocystis carinni pneumonitis (PCP) until 6 months after allo-HSCT. Ganciclovir was used between days −9 to −1 in order to prevent cytomegalovirus (CMV) infection. Acyclovir was used as herpes virus prophylaxis therapy from +1d to 6 months after transplantation. After hematopoietic reconstitution, CMV and Epstein-Barr virus (EBV) polymerase chain reaction (PCR) were monitored every week.
Main Outcome Measures
To assess the treatment response, bone marrow aspiration was performed every month within 6 months after transplantation. Relapse was defined as the presence of >5% blasts in patients with previously normal BM or evidence of extramedullary leukemia. Data pertaining to engraftment, transplantation-related complications, relapse rate (RR), 3-year disease-free survival (DFS), 3-year overall survival (OS), and no relapse mortality (NRM) were retrospectively analyzed. Grading of acute and chronic GvHD (aGvHD, cGvHD) was performed according to the standard criteria. Successful engraftment was defined according to the European Society for Blood and Marrow Transplantation criteria for hematopoietic repopulation; the criteria include 3 consecutive days with leucocytes ≥1.0×109/L, neutrophils ≥0.5×109/L, and thrombocytes ≥50×109/L without any transfusion in the immediately preceding 7 days. Transplantation-related complications include CMV viremia, EBV viremia, de novo pneumonia, hemorrhagic cystitis, aGvHD and cGvHD. DFS was defined as the period from diagnosis to relapse or death. OS was defined as the time from diagnosis to death or the most recent follow-up. NRM was defined as death caused by any reasons before day 28, or death caused by reasons other than relapse after day 28.
Statistical Analysis
Between-group differences were assessed using the Pearson Chi-squared test for categorical variables and Students t-test for continuous variables. The cumulative incidence of acute GVHD, relapse, and NRM was estimated considering the competing risks. Univariate analysis for acute GVHD was performed using Gray’s method. Fine and Gray semiparametric proportional hazards regression model was used for multivariate analysis. OS and DFS were calculated by Kaplan-Meier method and compared using the Log-rank test. P<0.05 was considered indicative of statistical significance. All statistical analyses were performed with SPSS 24.0 (SPSS Inc./IBM, Amonk, NY, USA), R statistical software and the cmprsk package (Comprehensive R Archive Network, TUNA, Tsinghua University, China).
Results
Baseline Characteristics
Baseline characteristics are summarized in Table 1 (Baseline Characteristics). Pre-transplantation therapy, WHO classification, and IPSS-R category were comparable between the two groups. However, there were some differences between the two groups. Compared to the non-Dec group, Dec recipients were older (41 vs 33 years, P=0.028), received smaller amount of MNCs (9.76×108/kg vs 12.48×108/kg, P=0.007) but larger amount of CD34 positive cells (5.77×106/kg vs 3.65×106/kg, P=0.004). Thirteen patients in the Dec group and 7 patients in the non-Dec group were in complete remission (CR) prior to allo-HSCT. A significant between-group difference was also observed with respect to the conditioning regimen; Bu/Cy was used in 5 non-Dec recipients and 0 Dec recipients (P=0.015).
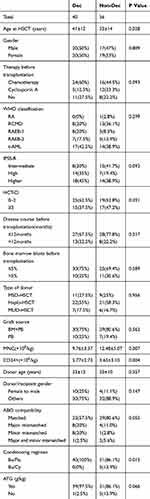 |
Table 1 Between-Group Differences of Baseline Characteristics |
Engraftment
Neutrophil engraftment was successful in all 76 patients. Thirty-nine patients in the Dec group and 35 patients in the non-Dec group had platelet (PLT) repopulated successfully. Median neutrophil engraftment in the Dec group was 13 days, which was similar to that in the non-Dec group (P=0.935). The median PLT engraftment in the Dec and non-Dec groups was 21 (8–86) days and 18 (8–80) days, respectively (P=0.321).
Transplantation-Related Complications
The incidence of CMV viremia, EBV viremia, and hemorrhagic cystitis was comparable in the two groups. However, the de novo incidence of respiratory infection was 22.5% in Dec group as against 53% in the non-Dec group (P=0.006) (shown in Table 2 – Incidence of transplantation-related complications).
 |
Table 2 Between-Group Differences of Transplantation-Related Complications Incidence |
Graft versus Host Disease
Grading of aGVHD was performed according to the International Bone Marrow Transplant Registry (IBMTR), while cGVHD was graded according to the National Institutes of Health (NIH) global severity of cGVHD. Cumulative incidence of grade II to IV aGVHD in the Dec group (12.4%, 95% CI 4.9–30.9%) was significantly lower than that [41.5%, 95% confidence interval (CI) 28.1–61.2%] in the non-Dec group (P=0.005). The incidence rate of cGvHD in the Dec and non-Dec groups was 17.5% and 25%, respectively (P=0.436). Results are shown in Figure 1 [Incidence of (a) aGvHD and (b) cGvHD in the Dec group and the non-Dec group].
Univariate analyses on influencing factors of II to IV grade aGvHD were carried out by Log-rank test of Kaplan-Meier. Results of univariate analyses showed that recipients older than 40 years may be more susceptible to grade II to IV aGvHD (P=0.018) (Table 3 Univariate analysis).
 |
Table 3 Univariate Analysis for II to IV Acute Graft versus Host Disease |
Factors associated with a significance level of P<0.1 on univariate analysis (including age at transplantation and donor type) were further evaluated on multivariate analysis. Risk factor analysis showed that Dec-containing conditioning regimen was a protective factor against grade II–IV aGvHD [hazard ratio (HR): 0.279, 95% CI: 0.102–0.765, P=0.013] (Table 4 – Multivariate analysis).
 |
Table 4 Multivariate Analysis for Acute Graft versus Host Disease |
Outcomes
The median follow-up duration in the entire study cohort was 40 (1 to 155) months. The median follow-up in the Dec and non-Dec groups was 27.5 (range, 1 to 57) months and 70 (range, 1 to 155) months, respectively. No significant between-group difference was observed with respect to the 3-year OS, DFS, or relapse rate (P=0.980, 0.959, and 0.573, respectively). However, median time to relapse was longer in the Dec group [7 months (range, 2 to 12) vs 3 months (range, 2 to 4), P=0.171]. The results are summarized in Figure S1.
Subgroup Analysis
Subgroup analyses were performed according to the IPSS-R risk category. The results suggested that, in the higher risk subgroup, Dec-containing conditioning may potentially decrease the relapse rate (10.6% vs 21.6%, P=0.370), increase disease-free survival (72.2% vs 57.1%, P=0.314), and enhance overall survival (72.2% vs 64.3%, P=0.628). NRM was 10.8% in the Dec group vs 0% in the non-Dec group (P<0.001). However, there were only 2 patients in the Dec group and none of the patients in the non-Dec group developed no relapse death in the competing risk model; therefore, we cannot draw the conclusion that Dec increases NRM in higher-risk patients. The results are summarized in Figure 2 [differences of (a) overall survival, (b) disease-free survival, (c) relapse rate, and (d) no relapse survival between the Dec group and the non-Dec group of IPSS-R higher-risk patients].
Discussion
Decitabine, a hypomethylation agent, can incorporate into deoxyribonucleic acid (DNA) and subsequently bind to DNA methyltransferase (DNMT) covalently. At high concentrations, the predominant effect appears to be cytotoxic effect; however, lower concentrations result in DNMT inhibition which leads to loss of DNA methylation, thus exerting therapeutic epigenetic modulation effect.3 Some researchers have suggested that the effects of Decitabine are mediated by mechanisms other than DNA hypomethylation.14 Though some clinical studies have recommended addition of 15 or 20 mg/m2 before or after stem cell transplantation,6,15 the dose of Decitabine is still not standardized. Therefore, we are currently seeking answers to this question. Based on the previous study, we supposed that adding Decitabine to conditioning regimen may be a rational treatment plan.
In our study, Decitabine was added to conditioning regimen since 2008; after that, our organization started using Decitabine in most high-risk patients, and 20 patients of the non-Dec patients were acquired between 2005 and 2008. Other therapies before or after hematopoietic stem cell transplantation, including prevention and treatment for GVHD, have not been changed. Patients in the Dec group were older and were at a higher risk (as indicated by the IPSS-R risk category) than those in the non-Dec group; however, they achieved similar OS, DFS, RR, and showed longer median time to relapse. These results suggest that Dec may ameliorate the unfavorable prognosis of high-risk patients. Besides, on subgroup analysis, the relapse rate in the Dec group was lower than that in the non-Dec group (10.6% vs 21.6%); however, considering the very small sample, a larger study is required to draw more definitive evidence. In line with our results, previous studies have also demonstrated lower relapse rates with hypomethylation therapy in combination with allo-HSCT. In a study by Field et al, bridge therapy with 5-Aza prior to allo-HSCT achieved a lower relapse rate as compared to that with traditional chemotherapy (20% vs 32%).14 Moreover, Decitabine bridging therapy has been shown to be a safe regimen before allo-HSCT.16 In other studies, post-HSCT maintenance therapy with hypomethylation agents was found to strengthen graft versus tumor effects and decrease the incidence of GVHD at the same time.17–20 Decitabine combined with donor lymphocytes infusion was deemed as an effective therapy for relapsed AML and MDS after allo-HSCT.21
In our study, Dec-containing conditioning regimen was found to protect against grade II to IV aGvHD and relapse. Previous studies have demonstrated that the addition of Decitabine may contribute to better aGVHD control post transplantation; however, in our study, the incidence rate of grade II to IV aGVHD was 17.5%, which is lower than that achieved with hypomethylation agents bridge therapy and maintenance therapy.18,22 The differences may be attributable to the timing of Decitabine therapy. As suggested by previous studies, we believe that the underlying mechanism is related to the immune modulatory effects of Dec. Firstly, Dec induces immunosuppressive effects by increasing the number of regulatory T cells (Treg) with upregulation of the expressions of Foxp3 and HLA-G.17,23−27 Secondly, Dec was shown to exert a protective role after HSCT by modulating the proliferation and differentiation of T cells in a TET2-dependent way.27,28 Finally, Dec alleviates tissue damage by inhibiting the production of proinflammatory cytokines, such as interferon γ and tumor necrosis factor β.29 Moreover, Dec may be a promising epi-immunotherapeutic agent against solid tumors and hematologic cancers.28,30,31
Studies have also demonstrated a critical role of Dec in promoting the graft versus tumor (GvT) effect. The underlying mechanisms may be as follows: firstly, it may upregulate the expressions of antioncogenes that are silenced because of hypermethylation.32 Secondly, Dec remodels specific immune responses by inducing the expression of tumor-associated antigen, restoring the expression of HLA class I antigen, upregulating the expressions of costimulatory molecules expression, inducing heterogeneous expression of killer immunoglobulin-like receptor (KIR) genes, and promoting enrichment or differentiation of tumor-derived APCs.33–42 In summary, Dec exerts its antitumor effects via direct cytotoxicity and strengthened tumor immunity, without sacrificing its GvHD protective effects.
There are some obvious limitations of our research, as this was a small-sample retrospective study; therefore, the treatment allocation algorithm is not clear. Moreover, there were significant between-group differences with respect to some variables; however, these variables were found to have no significant influence on the incidence of aGvHD.
Our study indicates that Dec-containing conditioning regimen probably prevents GvHD whilst maintaining the GvT effect, and may potentially increase the overall survival and disease-free survival with relatively few side effects. However, some limitations of our study should be considered while interpreting our results. These include the small sample size and the retrospective study design, which may have introduced an element of bias.
In conclusion, Dec-containing conditioning regimen in combination with allo-HSCT may have the effect of reducing the incidence of aGvHD and relapse; certainly, a larger prospective study with a longer follow-up period is required to provide more definitive evidence.
Conclusion
In conclusion, Dec-containing conditioning regimen in combination with allo-HSCT may help reduce the incidence of aGvHD and potentially decrease the incidence of relapse. A larger prospective study with a longer follow-up period is required to provide more definitive evidence.
Acknowledgments
We thank all authors for their continuous hard-work in study design, data collection and data analysis. Our study was supported by Youth Clinical Research Project of Peking University First Hospital (2018CR10), the Beijing Medical and Health Fund (YWJKJJHKYJJ-B182202) and the National Science and Technology Major Project of China (2019ZX09201-002).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Zeidan AM, Stahl M, Sekeres MA, Steensma DP, Komrokji RS, Gore SD. A call for action: increasing enrollment of untreated patients with higher-risk myelodysplastic syndromes in first-line clinical trials. Cancer. 2017;123(19):3662–3672. doi:10.1002/cncr.30903
2. Cutler CS, Lee SJ, Greenberg P, et al. A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome. Blood. 2004;104(2):579–585. doi:10.1182/blood-2004-01-0338
3. Koreth J, Pidala J, Perez WS, et al. Role of reduced-intensity conditioning allogeneic hematopoietic stem-cell transplantation in older patients with de novo myelodysplastic syndromes: an international collaborative decision analysis. J Clin Oncol. 2013;31(21):2662–2670. doi:10.1200/JCO.2012.46.8652
4. Wang H, Shao Z. Research progress on treatment of myelodysplastic syndromes with demethylating drugs. Chin J Clin Oncol. 2015;42(18):895–899.
5. Kon Kim T, Gore SD, Zeidan AM. Epigenetic therapy in acute myeloid leukemia: current and future directions. Semin Hematol. 2015;52(3):172–183. doi:10.1053/j.seminhematol.2015.04.003
6. Cruijsen M, Hobo W, van der Velden WJFM, et al. Addition of 10-day decitabine to fludarabine/total body irradiation conditioning is feasible and induces tumor-associated antigen-specific T cell responses. Biol Blood Marrow Transplant. 2016;22(6):1000–1008. doi:10.1016/j.bbmt.2016.02.003
7. Potter VT, Iacobelli S, van Biezen A, et al. Comparison of intensive chemotherapy and hypomethylating agents before allogeneic stem cell transplantation for advanced myelodysplastic syndromes: a study of the myelodysplastic syndrome subcommittee of the chronic malignancies working party of the European Society for Blood and Marrow Transplant Research. Biol Blood Marrow Transplant. 2016;22(9):1615–1620. doi:10.1016/j.bbmt.2016.05.026
8. Pusic I, Choi J, Fiala MA, et al. Maintenance therapy with decitabine after allogeneic stem cell transplantation for acute myelogenous leukemia and myelodysplastic syndrome. Biol Blood Marrow Transplant. 2015;21(10):1761–1769. doi:10.1016/j.bbmt.2015.05.026
9. Zhou J, Wang J, Liu H, et al. Therapeutic efficacies of decitabine application prior to hematopoietic cell transplantation in patients with myelodysplastic syndrome and acute myeloid leukemia. Natl Med J China. 2015;95(12):920–924.
10. Minakata D, Fujiwara S, Ito S, et al. A low-dose cytarabine, aclarubicin and granulocyte colony-stimulating factor priming regimen versus a daunorubicin plus cytarabine regimen as induction therapy for older patients with acute myeloid leukemia: a propensity score analysis. Leuk Res. 2016;42:82–87. doi:10.1016/j.leukres.2015.12.013
11. Xu L, Chen H, Chen J, et al. The consensus on indications, conditioning regimen, and donor selection of allogeneic hematopoietic cell transplantation for hematological diseases in China-recommendations from the Chinese Society of Hematology. J Hematol Oncol. 2018;11(1):33. doi:10.1186/s13045-018-0564-x
12. Mo XD, Kong J, Zhao T, et al. Extramedullary relapse of acute leukemia after haploidentical hematopoietic stem cell transplantation: incidence, risk factors, treatment, and clinical outcomes. Biol Blood Marrow Transplant. 2014;20(12):2023–2028. doi:10.1016/j.bbmt.2014.08.023
13. Lai YR, Chen YH, Hu DM, et al. Multicenter phase II study of a combination of cyclosporine a, methotrexate and mycophenolate mofetil for GVHD prophylaxis: results of the Chinese Bone Marrow Transplant Cooperative Group (CBMTCG). J Hematol Oncol. 2014;7:59. doi:10.1186/s13045-014-0059-3
14. Field T, Perkins J, Huang Y, et al. 5-Azacitidine for myelodysplasia before allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2010;45(2):255–260. doi:10.1038/bmt.2009.134
15. Zhu Y, Zhao H, Zhang X, et al. Decitabine before low-dose cytarabine-based chemotherapy combined with human leukocyte antigen-mismatched stem cell microtransplantation improved outcomes in elderly patients with newly diagnosed acute myeloid leukemia. Biol Blood Marrow Transplant. 2017;23(5):830–835. doi:10.1016/j.bbmt.2017.01.085
16. Fan LY, Hu SY, Xiao PF, et al. [The safety of decitabine as bridging pretreatment regimen before hematopoietic stem cell transplantation in pediatric hematological malignancies]. Zhonghua Nei Ke Za Zhi. 2018;57(9):679–682. doi:10.3760/cma.j.issn.0578-1426.2018.09.011
17. Choi J, Ritchey J, Prior JL, et al. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood. 2010;116(1):129–139. doi:10.1182/blood-2009-12-257253
18. De Lima M, Giralt S, Thall PF, et al. Maintenance therapy with low-dose azacitidine after allogeneic hematopoietic stem cell transplantation for recurrent acute myelogenous leukemia or myelodysplastic syndrome: a dose and schedule finding study. Cancer. 2010;116(23):5420–5431. doi:10.1002/cncr.25500
19. Ma Y, Qu C, Dai H, et al. Maintenance therapy with decitabine after allogeneic hematopoietic stem cell transplantation to prevent relapse of high-risk acute myeloid leukemia. Bone Marrow Transplant. 2019. doi:10.1038/s41409-019-0677-z
20. El-Cheikh J, Massoud R, Fares E, et al. Low-dose 5-azacytidine as preventive therapy for relapse of AML and MDS following allogeneic HCT. Bone Marrow Transplant. 2017;52(6):918–921. doi:10.1038/bmt.2017.31
21. Schroeder T, Rautenberg C, Kruger W, et al. Treatment of relapsed AML and MDS after allogeneic stem cell transplantation with decitabine and DLI – a retrospective multicenter analysis on behalf of the German Cooperative Transplant Study Group. Ann Hematol. 2018;97(2):335–342. doi:10.1007/s00277-017-3185-5
22. Kim DY, Lee JH, Park YH, et al. Feasibility of hypomethylating agents followed by allogeneic hematopoietic cell transplantation in patients with myelodysplastic syndrome. Bone Marrow Transplant. 2012;47(3):374–379. doi:10.1038/bmt.2011.86
23. Fransolet G, Ehx G, Somja J, et al. Azacytidine mitigates experimental sclerodermic chronic graft-versus-host disease. J Hematol Oncol. 2016;9(1). doi:10.1186/s13045-016-0281-2
24. Landman S, Cruijsen M, Urbano PCM, et al. DNA methyltransferase inhibition promotes Th1 polarization in human CD4(+)CD25(high) FOXP3(+) regulatory T cells but does not affect their suppressive capacity. J Immunol Res. 2018;2018:4973964. doi:10.1155/2018/4973964
25. Moon C, Kim SH, Park KS, et al. Use of epigenetic modification to induce FOXP3 expression in naïve T cells. Transplant Proc. 2009;41(5):1848–1854. doi:10.1016/j.transproceed.2009.02.101
26. Stamou P, Marioli D, Patmanidi AL, et al. Simple in vitro generation of human leukocyte antigen-G-expressing T-regulatory cells through pharmacological hypomethylation for adoptive cellular immunotherapy against graft-versus-host disease. Cytotherapy. 2017;19(4):521–530. doi:10.1016/j.jcyt.2017.01.004
27. Wang X, Wang J, Yu Y, et al. Decitabine inhibits T cell proliferation via a novel TET2-dependent mechanism and exerts potent protective effect in mouse auto and allo-immunity models. Oncotarget. 2017;8(34):56802–56815. doi:10.18632/oncotarget.18063
28. Sánchez-Abarca LI, Gutierrez-Cosio S, Santamaría C, et al. Immunomodulatory effect of 5-azacytidine (5-azaC): potential role in the transplantation setting. Blood. 2010;115(1):107–121. doi:10.1182/blood-2009-03-210393
29. Li X, Mei Q, Nie J, Fu X, Decitabine: HW. A promising epi-immunotherapeutic agent in solid tumors. Expert Rev Clin Immunol. 2015;11(3):363–375. doi:10.1586/1744666X.2015.1002397
30. Engel N, Rank A. Epigenomics in hematopoietic transplantation: novel treatment strategies. Epigenomics. 2011;3(5):611–623. doi:10.2217/epi.11.80
31. Ishikawa T, Fujii N, Imada M, et al. Graft-versus-leukemia effect with a WT1-specific T-cell response induced by azacitidine and donor lymphocyte infusions after allogeneic hematopoietic stem cell transplantation. Cytotherapy. 2017;19(4):514–520. doi:10.1016/j.jcyt.2016.12.007
32. Goodyear OC, Dennis M, Jilani NY, et al. Azacitidine augments expansion of regulatory T cells after allogeneic stem cell transplantation in patients with acute myeloid leukemia (AML). Blood. 2012;119(14):3361–3369. doi:10.1182/blood-2011-09-377044
33. Chan HW, Kurago ZB, Stewart CA, et al. DNA methylation maintains allele-specific KIR gene expression in human natural killer cells. J Exp Med. 2003;197(2):245–255. doi:10.1084/jem.20021127
34. Coral S, Sigalotti L, Gasparollo A, et al. Prolonged upregulation of the expression of HLA class I antigens and costimulatory molecules on melanoma cells treated with 5-aza-2ʹ-deoxycytidine (5-AZA-CdR). J Immunother. 1999;22(1):16–24. doi:10.1097/00002371-199901000-00003
35. Hambach L, Ling KW, Pool J, et al. Hypomethylating drugs convert HA-1 negative solid tumors into targets for stem cell based immunotherapy. Blood. 2009;113(12):2715–2722. doi:10.1182/blood-2008-05-158956
36. Krishnadas DK, Bao L, Bai F, Chencheri SC, Lucas K. Decitabine facilitates immune recognition of sarcoma cells by upregulating CT antigens, MHC molecules, and ICAM-1. Tumor Biol. 2014;35(6):5753–5762. doi:10.1007/s13277-014-1764-9
37. Liu Y, Kuick R, Hanash S, Richardson B. DNA methylation inhibition increases T cell KIR expression through effects on both promoter methylation and transcription factors. Clin Immunol. 2009;130(2):213–224. doi:10.1016/j.clim.2008.08.009
38. Santourlidis S, Trompeter HI, Weinhold S, et al. Crucial role of DNA methylation in determination of clonally distributed killer cell Ig-like receptor expression patterns in NK cells. J Immunol. 2002;169(8):4253–4261. doi:10.4049/jimmunol.169.8.4253
39. Stübig T, Badbaran A, Luetkens T, et al. 5-azacytidine promotes an inhibitory T-cell phenotype and impairs immune mediated antileukemic activity. Mediators Inflamm. 2014;2014:1–12. doi:10.1155/2014/418292
40. Wang J, Zhou J, Zheng HF, Fu ZZ. Effect of decitabine on immune regulation in patients with acute myeloid leukemia after allogeneic hematopoietic stem cell transplantation. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2014;22(5):1448–1452.
41. Yang H, Bueso-Ramos C, Dinardo C, et al. Expression of PD-L1, PD-L2, PD-1 and CTLA4 in myelodysplastic syndromes is enhanced by treatment with hypomethylating agents. Leukemia. 2014;28(6):1280–1288. doi:10.1038/leu.2013.355
42. Daurkin I, Eruslanov E, Vieweg J, Kusmartsev S. Generation of antigen-presenting cells from tumor-infiltrated CD11b myeloid cells with DNA demethylating agent 5-aza-2ʹ-deoxycytidine. Cancer Immunol Immunother. 2010;59(5):697–706. doi:10.1007/s00262-009-0786-4
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


