Back to Journals » Nature and Science of Sleep » Volume 11
Could long-term administration of melatonin to prepubertal children affect timing of puberty? A clinician’s perspective
Authors Boafo A ![]() , Greenham S
, Greenham S ![]() , Alenezi S
, Alenezi S ![]() , Robillard R
, Robillard R ![]() , Pajer K, Tavakoli P, De Koninck J
, Pajer K, Tavakoli P, De Koninck J ![]()
Received 25 July 2018
Accepted for publication 10 December 2018
Published 31 January 2019 Volume 2019:11 Pages 1—10
DOI https://doi.org/10.2147/NSS.S181365
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Steven Shea
Addo Boafo,1,2 Stephanie Greenham,1,3 Shuliweeh Alenezi,1,2 Rébecca Robillard,3,4 Kathleen Pajer,1,2 Paniz Tavakoli,1 Joseph De Koninck3,4
1Children’s Hospital of Eastern Ontario, Ottawa, ON, Canada; 2Department of Psychiatry, Faculty of Medicine, University of Ottawa, Ottawa, ON, Canada; 3School of Psychology, University of Ottawa, Ottawa, ON, Canada; 4Sleep Research Unit, Royal’s Institute of Mental Health Research, Ottawa, ON, Canada
Abstract: Exogenous melatonin can be used to treat sleep disturbance in adults, children, and adolescents. While its short-term use is considered safe, there are some concerns that long-term use might delay children’s sexual maturation, possibly by disrupting the decline in nocturnal melatonin levels that occur at the onset of puberty. This narrative review aimed to summarize some of the current knowledge about the potential effects of exogenous melatonin on puberty. We found no clinical studies that experimentally tested the effects of melatonin on pubertal timing in children, but we reviewed the small number of observational studies. We also drew on animal data to try to answer our question. The photoperiod and melatonin-mediated seasonal transitions in sexual activity and breeding in some mammals across the seasons have been used as a model of sexual development in mammals, including humans. The switch from non-sexual activity (in the non-breeding period) to sexual activity (in the breeding period) has been likened to the onset of puberty as there are similarities between the two. We conclude that to investigate an association between melatonin and pubertal timing, it will be important to conduct long-term randomized controlled trials of latency age children and also examine the cellular and systems-level interactions between melatonin and kisspeptin, a recently identified neuropeptide with a locus of action at the gonadotropin releasing hormone neurons that is important in contributing to the timing of puberty onset.
Keywords: melatonin, puberty, hypothalamic-pituitary-gonadal-axis, kisspeptin, sleep disturbance
Overview and purpose
In recent years, there has been an increase in the use of melatonin for the treatment of sleep disturbances with positive results. Human studies have shown that short-term use of melatonin is safe, even in high doses, with only mild adverse effects such as dizziness, headache, nausea, and sleepiness.1 Carefully timed exogenous melatonin effectively reduces sleep latency while restoring chronobiological alignment of sleep with the internal biological clock2,3 without negative alteration of sleep architecture and other side effects observed with most hypnotics. However, melatonin use with adolescents and children is still off-label and concerns have been raised that its long-term use may affect sexual maturation.4 The present review first offers a brief synopsis of the nature and properties of melatonin, the prevalence of sleep disorders in children and adolescents, the current understanding of mechanisms of onset of puberty, and a brief review of kisspeptin’s effects on the gonadotropin releasing hormone (GnRH) neurons as a potential model of puberty. This is followed by a review of some of the existing animal and human studies on melatonin and the onset of puberty. We conclude with a comment about non-pharmacological interventions for sleep disturbance and future direction of research.
Properties and effects of melatonin
The neurohormone, melatonin (N-acetyl-5-methoxytryptamine), is produced in the pineal gland of mammals. It first came to attention in 1958 when Lerner, a dermatologist, found that melatonin could cause the lightning of frog skin.5 Subsequently in 1960, he and his colleagues defined melatonin’s chemical structure.6 Since then, melatonin has been found to affect a wide range of physiological processes such as; circadian rhythms,7 sleep-wake cycles,8 sexual maturation,9 aging,10 and exerts neuroendocrine,11 cardiovascular,12 and oncostatic effects.13 Many laboratory studies conducted under well-controlled conditions have shown that the administration of exogenous melatonin acutely affects sleep and thermoregulation in humans, and that the soporific effect of melatonin is mediated by an increase in distal heat loss through increased skin temperature over the course of the sleep cycle.14–16 In healthy volunteers, a single melatonin dose of 0.3 mg or 1.0 mg orally has been reported to have significant soporific effects.17 Although exogenous melatonin is often referred to as a hypnotic, it has been proposed that soporific is a more accurate term than hypnotic when referring to the sleepiness-inducing characteristics of melatonin. Exogenous melatonin is also considered as a chronobiotic due to its ability to shift the timing of sleep and circadian rhythms.3,18
The photoneuroendocrine system
In mammals, photoperiodic (or relative length of light and dark periods) information is received at the level of the retina and is transmitted through a multi-synaptic neural pathway to the pineal gland to modulate melatonin secretion.19 More specifically, the endogenous production and release of melatonin are suppressed by exposure to bright light. Pineal melatonin secretion is also regulated by the main biological clock in the suprachiasmatic nucleus of the hypothalamus.20 In humans, melatonin release under dim light conditions usually starts in the evening, with some averages ranging between 19:30 and 21:30 in adults, and between 19:00 and 21:00 in children from 6–12 years of age.21 The duration of melatonin secretion varies according to day length and provides an endocrine representation of the photoperiod.22 When administered in the daytime, exogenous melatonin has an elimination half-life of 35–45 minutes.23
Prevalence of sleep disturbances in children and adolescents, and melatonin use
It is estimated that 15%–25% of children and adolescents in the general population have sleep disturbances.24,25 A higher prevalence of sleep disturbances is seen in children and adolescents with medical and psychiatric conditions. For example, the estimated rates of sleep disturbances range from 30%–53% in individuals with autism spectrum disorder,26 and up to 70% in those with attention deficit hyperactivity disorder.27 In individuals with neurodevelopmental disorder, the prevalence of sleep disturbance is estimated to be 13%–86%.28 With respect to anxiety disorders in children and adolescents, 80%–90% report at least one sleep-related problem.29,30 In major depression, the prevalence of sleep disturbances is estimated to be 72.7%.31 In line with the considerable changes affecting the biological clock during this developmental period,32,33 sleep disturbances occurring in late childhood and adolescence often take the form of delayed sleep phase.34,35 If untreated, such sleep disturbances can negatively impact children, adolescents, and their families with respect to physical and mental health, social, academic, and cognitive functioning.36–38
Off-label, over-the-counter exogenous melatonin use in children and youth has grown tremendously in recent years. For example, Hartz et al39 studied recurrent pediatric melatonin use across Norway. From 2004–2012, the prevalence of people using melatonin between the ages of 4 and 17 increased from 3.4–11.0 per 1,000 in boys and 1.5–7.7 per 1,000 in girls. Twenty-nine percent of boys and 23% of girls were recurrent melatonin users, with highest level of recurrent use in young children 4–8 years of age. Most of the children or youth had a diagnosis of a psychiatric or neurologic disorder.39
Melatonin and sexual maturation
In a cross-sectional study, Waldhauser et al40 examined nocturnal serum melatonin levels in 367 individuals ranging from 3 days to 90 years of age. They noted that the highest nighttime serum melatonin levels were found in very young individuals (1–3 years), with mean levels dropping progressively by 80% throughout childhood until adolescence and young adulthood (15–20 years). This is consistent with an earlier study in which they described a 75% drop in nocturnal serum melatonin levels when comparing children aged 1–5 years to young adults.41 There are some indications that the drop in nocturnal melatonin levels during adolescence parallels sexual maturation processes.42 Accordingly, the decline in melatonin levels has been found to relate to the progression of Tanner stages.43 These observations, together with animal studies showing that exogenous melatonin can suppress GnRH secretion,44 contributed to the emergence of concerns about the possibility that exogenous melatonin supplementation may affect children’s sexual maturation.4
In brief, given how common the use of melatonin has become in the treatment of sleep disturbances in children and adolescents, it is imperative to review the evidence from clinical and basic science studies to answer the question of whether melatonin could affect pubertal timing in adolescence.
Physiology of puberty
Puberty, the process by which a child becomes sexually mature and capable of sexual reproduction, is associated with a growth spurt and the development of secondary sexual characteristics. This is the product of a complex series of neuroendocrine steps in which the primary mechanisms are still unclear.45 The start of puberty is marked by a sustained increase in the pulsatile release of GnRH46 by a small population of GnRH neurons which extend axons from the preoptic area and the infundibular nucleus of the hypothalamus.47 The hypothalamus forms part of the hypothalamic-pituitary-gonadal axis which controls the onset and progression of puberty. This axis is active in the embryonic and early postnatal stages of life and is subsequently restrained during childhood only to be reactivated during the initiation of puberty.48 The exact mechanisms which trigger the onset of puberty are unknown. However, it is believed that the augmentation of activating factors leading to GnRH secretion, together with the suppression of inhibitory factors of GnRH secretion result in the initiation of puberty. These activating and inhibitory factors involve neurotransmitters and neuropeptides which originate in the hypothalamus in addition to peripheral or gonadal signals. In humans, the timing of puberty is highly variable due to several factors such as: gender; epigenetic and genetic factors; intrauterine conditions; nutrition; light-dark cycles; climatic conditions; hormones (eg, leptin, ghrelin, IGF-1, sex steroid); and exposure to endocrine-disrupting chemicals in the environment.49 Figure 1, taken from Livadas and Chrosusos,47 illustrates the interaction of hypothalamic factors and peripheral signals for puberty onset.
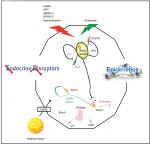  | Figure 1 Interaction of hypothalamic factors and peripheral signals on puberty onset. Notes: Republished with permission of Wolters Kluwer Health, from Control of the onset of puberty, Livadas S, Chrousos GP, volume 28(4), 2016; permission conveyed through Copyright Clearance Center, Inc.47 Schematic presentation of the highly specialized complex comprised of KNDy and GnRH neurons, and pituitary gonadotropes regulating pubertal onset. The integral function of KNDy neurons (with their auto regulatory loops) and GnRH neurons is depicted, as this pertains to gonadotropin secretion. The effects of positive and negative stimuli on this complex are presented with red and green arrows respectively. Energy sensing, hormones, epigenetic processes, and endocrine disruptors participate in the process. |
Kisspeptin signaling, effects on GnRH neurons, and sensitivity to melatonin
As reported by Liu and Herbison,50 the cancer suppressor gene, KISS1, was discovered in 1996 in Hershey, Philadelphia and named KISS1, after the Hershey’s Kisses chocolate.51
It encoded for a group of four neuropeptides, called kisspeptins. Their receptor was found to be the orphan G protein-coupled receptor GPR54 also known as KISS1R. Of the four kisspeptins, kisspeptin 10 has been used the most in research.51
In 2005, kisspeptin was discovered to be the most potent activator of GnRH neuronal excitability52 and the direct effects of kisspeptin are believed to be critical for both the electrophysiological and reproductive actions of kisspeptin.53–55 In rodents (hamsters and rats) and sheep, most GnRH neurons (greater than 90%) express the receptor, Kiss1r. Kiss1r is the non-human ortholog of the human KISS1R. It is believed that the mammalian reproductive system is driven by pulsatile release of GnRH and that kisspeptin’s effects on reproduction are mediated through GnRH neurons. It is noted however that in humans not all GnRH neurons receive kisspeptin neuron input, suggesting that additional molecules regulate the process of human puberty.47
Animal studies have shown that melatonin supplementation reduces kisspeptin under conditions of prolonged photoperiod,58 while endogenous melatonin suppression (following pinealectomy) abolishes the effects of reduced photoperiod on kisspeptin expression.59 In addition, a recent study demonstrated that exogenous melatonin first induces a reduction in kisspeptin gene expression in the acute phase, but that longer effects lead to an increase in kisspeptin gene expression60 which could then actively stimulate the hypothalamic-pituitary-gonadal axis. Overall, this suggests that the effects of melatonin on the reproduction system may be modulated by kisspeptin and that these effects may differ considerably between the acute and longer-term phases of melatonin supplementation.
We therefore conducted a non-systematic review of the literature of studies assessing the effects of melatonin on puberty and the potential modulating role of kisspeptin.
Literature search: strategy and selection of studies
Medline and PubMed were searched for papers published in English up to 2018 using the search terms “melatonin”, “puberty”, “puberty onset”, “hypothalamic-pituitary-gonadal axis”, “kisspeptin”, “children”, and “adolescents”. The relevant articles were read and the reference lists were searched and those identified as relevant were also read. We first report on animal studies, followed by human studies.
Animal studies
Exogenous melatonin and puberty
There are several reasons for including animal studies in this narrative review. The first rests in the fact that experimental studies that would directly test the hypothesis that exogenous administration of melatonin might delay children’s sexual maturation by assigning children to long-term melatonin treatment or placebo, do not currently exist and would be unethical. Second, the answer to the question may be informed by considering models of seasonal breeding in some mammals. For example, the two most commonly employed mammalian experimental models used to explain the endogenous rhythms governing reproduction involve hamsters (Syrian and Siberian) and sheep.61–64 It has previously been suggested the transition from the non-breeding (anestrous) season to the breeding season in these animals could represent a mechanism similar to puberty.65 Numerous similarities can be drawn between puberty and the transition to anestrous season. For example, in both pre-pubertal and seasonally anestrous ewes, preovulatory luteinizing hormone (LH) surges, which cause ovulation, do not occur. Also, in both cases, the neural wiring and innate ability to produce an LH surge are present,66–68 but appear to be suppressed. Prior to puberty, LH secretion is pulsatile, as is the case during the anestrous season, but the frequency of LH pulses is markedly reduced.69 While these similarities are interesting, it is noted that the precise neuroendocrine systems which govern puberty and reproductive functions are not fully understood.28,45,49
In the next sections, we provide a few examples of the effects of exogenous melatonin and pinealectomy on pubertal onset in animals. Subsequently, we present some investigations on the relationship between the photoperiod and changes in kisspeptin expression between breeding and non-breeding systems. Table 1 also summarizes examples of the effects of exogenous melatonin administration on the timing of puberty in lambs, gilts, rats, and male Siberian hamsters.
  | Table 1 Examples of the effects of exogenous melatonin administration on the timing of puberty in lambs, gilts, rats, and male Siberian hamsters Note: Extensive literature exists about changes in sexual behavior in seasonally breeding mammals by manipulation of the photoperiod, which is beyond the scope of this narrative review (additional information can be found elsewhere).61–64 |
In the male Djungarian hamster, which usually starts puberty 25 days after birth,70 a 10-day treatment with short days or melatonin during the pre-pubertal period (day 15 to day 25) arrested the onset of puberty.71 While one study failed to see any significant effect of melatonin-filled implants on the timing of puberty in gilts,72 another study showed that prepubertal ewe lambs treated with melatonin implants had puberty onset delayed by 4 weeks compared to controls.73 Conversely, another study showed that oral administration of melatonin to Suffolk prepubertal ewe lambs, from 10 weeks of age onward, advanced the onset of puberty by 3 weeks compared to control lambs maintained under the same natural photoperiod.74 This may be in line with another study reporting that pinealectomy delayed the onset of puberty by 12 weeks in ewe lambs.75 To summarize, there are some indications that exogenous melatonin can alter the timing of puberty in seasonally breeding mammals, but findings to date seem to be inconsistent.
Kisspeptin, melatonin, and puberty
Investigators considered whether photoperiod could be a primary stimulus for changes in kisspeptin expression between breeding and nonbreeding seasons. It has been demonstrated that, in Soay ewes, the photoperiodic changes in kisspeptin levels were related to changes in the pattern of melatonin secretion.76 The Soay ewe is a short-day breeder, breeding during Autumn and Winter when days are short and nights are long (ie, entailing longer periods of melatonin secretion). Similarly, Simonneaux et al,58 also found that kisspeptin expression was influenced by the photoperiod in male Syrian hamsters, which are long-day breeders. During the long-day period (Spring and Summer) the duration of melatonin secretion is shorter compared to short-day periods (Autumn and Winter). Kisspeptin cells are, however, not known to express melatonin receptors,77 suggesting that there could be intermediary processes (yet to be elucidated) between the photoperiod, melatonin, and kisspeptin expression. Simonneaux, Ancel, Poirel, and Gauer78 proposed that RFRP-3, a member of the RRamide peptide group, known to inhibit GnRH, could play such an intermediary role. Figure 2, from Simmonneaux et al,78 illustrates photo-inhibitory melatonergic message in the male Syrian hamster.
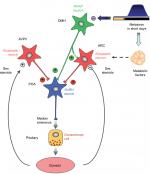  | Figure 2 Working model indicating how the photo-inhibitory melatonergic message in short day conditions is integrated in the hypothalamus to further regulate the gonadotropic axis in the male Syrian hamster. Notes: Photo-inhibitory melatonergic message in male Syrian hamster. Reproduced with permission from Simonneaux et al, 2013.78 |
Another postulated pathway for melatonin’s inhibitory effect in the period of reproductive quiescence in hamsters during short days (Autumn and Winter) is that melatonin may alter and reinforce the negative feedback effect of sex steroids on kisspeptin expression.59,79
On the other hand, there is evidence that even in the seasonally breeding mammals the hypothalamic-pituitary-gonadal axis may escape the driving influence of light and melatonin on the timing of fertility and reproduction at least under experimental conditions. It is known that after 10 weeks of exposure to short days (Autumn and Winter and longer periods of melatonin exposure) some hamsters become sexually inactive and this was associated with a decrease in Kiss1 expression in the hypothalamus (arcuate nucleus). If the short-day exposure continues over 20–30 weeks, the hamsters are known to become refractory to the inhibitory effects of short day lengths and become sexually active spontaneously – implying an escape from the effect of a long period of exposure to melatonin. The usual cycle becomes reset only after prolonged exposure to long-day conditions (Spring and Summer). It was found that the sexual reactivation was associated with an increase and Kiss1 expression similar to findings in the Spring and Summer.59,80
In summary, there is evidence that in some seasonal breeders photoperiodic and melatonin level changes are associated with changes in kisspeptin signaling. However, some studies failed to show any significant association, or suggested that there could be temporary dissociations.
Human studies: pubertal timing in pediatric melatonin use
Only three human studies have tracked pubertal timing along the course of exogenous melatonin use, and all examined pubertal timing as a secondary outcome while the primary focus was on melatonin effectiveness or dosing. Two studies involved the Meldos Trial, which was a randomized controlled trial investigating optimal melatonin dosing for intractable insomnia in children and youth.81
The first report encompasses data on 59 of the 69 6–12-year-old participants who had completed the Meldos Trial at least a year before.81 The children, having used melatonin for 6 months or longer, were asked to fill out questionnaires including self-report Tanner staging with pictures, mother’s age of menarche, and boys’ ages of first ejaculation for them and for their fathers. The ages across Tanner stage were compared to Dutch population data. Only 19 of the participants reached possible pubertal age during this study, and only 62% of the boys and 91% of the girls answered the puberty questions. In this limited sample, there was no evidence of pubertal timing being different from the population norms.
This group was again followed up 9–12 years later, comparing the 33 participants who completed the follow-up phase to publicly available population samples on perceived pubertal timing (early, normal, late).82 In this subsequent study, 31.3% of the participants reported a late perceived pubertal timing, compared to the population sample control rate of 17%.
Carr et al83 conducted a longitudinal study of melatonin treatment in 44 children with neurodevelopmental disorders and treatment-resistant circadian rhythm sleep disorders. The baseline study was a placebo-controlled double-blind cross-over trial of sustained-release melatonin. The follow-up study consisted of a structured caregiver telephone interview every 3 months for up to 3.8 years. The median age of puberty was 11.5 years, with a range of 12–15 years. Precocious puberty was present in five children with severe neurodevelopmental disability, most of whom experienced it prior to the melatonin treatment. In the remaining children, pubertal timing was considered within normal limits, with a mean age of 13.4 years ±1.4 years.
In summary, there are only few studies that have examined pubertal timing in children and youth given melatonin on a long-term basis. Drawing any conclusions from the three that exist is difficult because they have very small samples, incomplete follow-up, and poor measures of pubertal timing.
It is also useful to consider indirect human studies. Magee et al84 suggested the possibility of a role of exposure to light in human puberty, as they noted that blind girls tended to have earlier menarche. However, these reports were not subsequently confirmed.49 In a similar vein, other studies suggested that menarche occurred more frequently in the Winter than in the Summer,85–87 suggesting that light would have an inhibiting effect on the onset of puberty. This is however contrary to the finding that the long Winter months in the Arctic may be associated with reduced pituitary-gonadal function and low conception rates.88
As to the finding that the level of melatonin falls in the lead-up to puberty in humans,89–91 it was observed that the decrease in melatonin secretion occurs usually after the onset of puberty, between Tanner stages II and III.90
Discussion
In this narrative review, we highlighted that there is some evidence that exogenous melatonin may alter pubertal timing in some animals, that photoperiodic and melatonin level changes may be associated with changes in kisspeptin signaling in some seasonal breeders, and that one of the longitudinal studies in humans suggested that long-term melatonin use might be linked to a delay in puberty onset; however, the determination of puberty delay was subjective (perceived), and not objective. These findings are all based on very limited research and the findings are not consistent across studies. Hence, drawing any strong conclusions at this stage is difficult.
To the extent that the transition from the sexually inactive to sexually active phase could be an animal model for the onset of puberty and light, darkness and melatonin are important in this transition, it could be argued that perhaps melatonin plays an important role in the onset of puberty in seasonally breeding mammals. Whether or not melatonin could play an important role in the pubertal onset in humans is still an open question.
Since humans are not seasonal breeders like hamsters and sheep, an argument could also be made that the human hypothalamic-pituitary-gonadal axis may have, during the process of evolution, escaped the driving influence of light and melatonin on the timing of fertility and reproduction.
Future directions
We suggest three future areas of research to advance our knowledge about the effects of long-term use of exogenous melatonin on pubertal timing in children and adolescents. First, established methodologies of longitudinal studies of pubertal timing81,82,92 should be applied to observational studies in children who are spontaneously electing to take melatonin. It would be ideal to start with children who are as young as possible and follow them annually, measuring the somatic manifestations of puberty (eg, age of menarche, Tanner stage development, age at first ejaculation). Because the normal age range of puberty in girls begins at 8 years of age and at 9 in boys, studies should recruit girls and boys at young ages, ideally 5 or 6 years of age. Second, it will be important to re-examine and get a better understanding of the reasons for the inconsistent findings in studies involving exogenous melatonin administration and pubertal onset in animals. Third, further investigations are needed to clarify the potential role of cellular and systems-level interactions between melatonin and kisspeptin, a neuropeptide which acts on GnRN neurons and is considered important in the timing of puberty onset. Such studies can be done at a basic science level.
Conclusion
Our review suggests that the role of melatonin in sexual maturation and the timing of puberty is understudied in humans. The three human studies that have examined the question have done so as an ancillary research question in small samples of children and youth, some of whom had neurodevelopmental disorders. This limits the generalizability to the general population and is insufficient evidence to draw conclusions for patients with mental health and neurological disorders. Further experimental studies on the impact of melatonin on puberty, notably in non-seasonal mammals, and advances in the research about the intermediary processes between melatonin and kisspeptin activation, could ultimately inform us about the potential influence of exogenous melatonin on puberty.
We would be remiss not to mention that, aside from melatonin intake, non-pharmacological interventions can be effective for the treatment of sleep disturbances in children and adolescents. They have been found to be more beneficial than placebo, alternative treatments, and frequently used medications in children, adolescents, and adults.93 Non-pharmacological approaches range from sleep hygiene and psychoeducation, cognitive behavioral therapy for insomnia, physical exercise,94 and mindfulness-based meditation,95 to relaxation-based therapies.96
Acknowledgment
Financial support was provided by funds from the Psychiatry Associates at the Children’s Hospital of Eastern Ontario and from the Institute of Mental Health Research University of Ottawa.
Author contributions
AB, SG, SA, RR, KP, and PT contributed to the rationale and design of the paper. The manuscript was written by AB. AB, SG, SA, RR, KP, PT, and JDK conducted searches of pertinent literature. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Andersen LPH, Gögenur I, Rosenberg J, Reiter RJ. The safety of melatonin in humans. Clin Drug Investig. 2016;36(3):169–175. | ||
van Geijlswijk IM, van der Heijden KB, Egberts AC, Korzilius HP, Smits MG. Dose finding of melatonin for chronic idiopathic childhood sleep onset insomnia: an RCT. Psychopharmacology. 2010;212(3):379–391. | ||
Cardinali DP, Furio AM, Reyes MP, Brusco LI. The use of chronobiotics in the resynchronization of the sleep-wake cycle. Cancer Causes Control. 2006;17(4):601–609. | ||
Kennaway DJ. Potential safety issues in the use of the hormone melatonin in paediatrics. J Paediatr Child Health. 2015;51(6):584–589. | ||
Lerner AB, Case JD, Takahashi Y, Lee TH, Mori W. Isolation of melatonin, the pineal gland factor that lightens melanocytes. J Am Chem Soc. 1958. | ||
Lerner AB, Case JD, Takahashi Y. Isolation of melatonin and 5-methoxyindole-3-acetic acid from bovine pineal glands. J Biol Chem. 1960;235:1992–1997. | ||
Gillette MU, Tischkau SA. Suprachiasmatic nucleus: the brain’s circadian clock. Recent Prog Horm Res. 1999;54:33–58; discussion 58–59. | ||
Reiter RJ. Pineal melatonin: cell biology of its synthesis and of its physiological interactions. Endocr Rev. 1991;12(2):151–180. | ||
Díaz López B, Díaz Rodríguez E, Urquijo C, Fernández Álvarez C. Melatonin influences on the neuroendocrine-reproductive axis. Ann N Y Acad Sci. 2005;1057:337–364. | ||
Pierpaoli W, Regelson W. Pineal control of aging: effect of melatonin and pineal grafting on aging mice. Proc Natl Acad Sci USA. 1994;91(2):787–791. | ||
Cardinali DP, Pévet P. Basic aspects of melatonin action. Sleep Med Rev. 1998;2(3):175–190. | ||
Reiter RJ, Tan DX, Korkmaz A. The circadian melatonin rhythm and its modulation: Possible impact on hypertension. J Hypertens Suppl. 2009;27(6):S17–S20. | ||
Korkmaz A, Tamura H, Manchester LC, Ogden GB, Tan DX, Reiter RJ. Combination of melatonin and a peroxisome proliferator-activated receptor-gamma agonist induces apoptosis in a breast cancer cell line. J Pineal Res. 2009;46(1):115–116. | ||
Cajochen C, Krauchi K, Wirz-Justice A. Role of melatonin in the regulation of human circadian rhythms and sleep. J Neuroendocrinol. 2003;15(4):432–437. | ||
Krauchi K, Cajochen C, Wirz-Justice A. A relationship between heat loss and sleepiness: effects of postural change and melatonin administration. J Appl Physiol. 1997;83(1):134–139. | ||
Dawson D, van den Heuvel CJ. Integrating the actions of melatonin on human physiology. Ann Med. 1998;30(1):95–102. | ||
Attenburrow MEJ, Cowen PJ, Sharpley AL. Low dose melatonin improves sleep in healthy middle-aged subjects. Psychopharmacology. 1996;126(2):179–181. | ||
Wirz-Justice A, Armstrong SM. Melatonin: nature’s soporific? J Sleep Res. 1996;5(2):137–141. | ||
Hastings MH, Maywood ES, Reddy AB. Two decades of circadian time. J Neuroendocrinol. 2008;20(6):812–819. | ||
Simonneaux V, Ribelayga C. Generation of the melatonin endocrine message in mammals: a review of the complex regulation of melatonin synthesis by norepinephrine, peptides, and other pineal transmitters. Pharmacol Rev. 2003;55(2):325–395. | ||
van Geijlswijk IM, Korzilius HP, Smits MG. The use of exogenous melatonin in delayed sleep phase disorder: a meta-analysis. Sleep. 2010;33(12):1605–1614. | ||
Tosini G, Fukuhara C. Photic and circadian regulation of retinal melatonin in mammals. J Neuroendocrinol. 2003;15(4):364–369. | ||
Fourtillan JB, Brisson AM, Gobin P, Ingrand I, Decourt JP, Girault J. Bioavailability of melatonin in humans after day-time administration of D(7) melatonin. Biopharm Drug Dispos. 2000;21(1):15–22. | ||
Shamseer L, Vohra S. Complementary, Holistic, and Integrative Medicine: Melatonin Definition and Description. Available from: www.cfsan.fda.gov/. Accessed November 12, 2018. | ||
Owens J. Classification and epidemiology of childhood sleep disorders. Prim Care. 2008;35(3):533–546. | ||
Sivertsen B, Posserud MB, Gillberg C, Lundervold AJ, Hysing M. Sleep problems in children with autism spectrum problems: a longitudinal population-based study. Autism. 2012;16(2):139–150. | ||
Cortese S, Faraone SV, Konofal E, Lecendreux M. Sleep in children with attention-deficit/hyperactivity disorder: meta-analysis of subjective and objective studies. J Am Acad Child Adolesc Psychiatry. 2009;48(9):894–908. | ||
Sajith SG, Clarke D. Melatonin and sleep disorders associated with intellectual disability: a clinical review. J Intellect Disabil Res. 2007;51(Pt 1):2–13. | ||
Chase RM, Pincus DB. Sleep-related problems in children and adolescents with anxiety disorders. Behav Sleep Med. 2011;9(4):224–236. | ||
Alfano CA, Ginsburg GS, Kingery JN. Sleep-related problems among children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(2):224–232. | ||
Liu X, Buysse DJ, Gentzler AL, et al. Insomnia and hypersomnia associated with depressive phenomenology and comorbidity in childhood depression. Sleep. 2007;30(1):83–90. | ||
Carpenter J, Robillard R, Therapy IH. Variations in the Sleep–Wake Cycle from Childhood to Adulthood: Chronobiological Perspectives. Citeseer; 2015. Available from: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.838.9127&rep=rep1&type=pdf. Accessed November 22, 2018. | ||
Tonetti L, Adan A, Di Milia L, Randler C, Natale V. Measures of circadian preference in childhood and adolescence: A review. Eur Psychiatry. 2015;30(5):576–582. | ||
Crowley SJ, Acebo C, Carskadon MA. Sleep, circadian rhythms, and delayed phase in adolescence. Sleep Med. 2007;8(6):602–612. | ||
Thorpy MJ, Korman E, Spielman AJ, Glovinsky PB. Delayed sleep phase syndrome in adolescents. J Adolesc Heal Care. 1988;9(1):22–27. | ||
Fallone G, Owens JA, Deane J. Sleepiness in children and adolescents: clinical implications. Sleep Med Rev. 2002;6(4):287–306. | ||
Brand S, Kirov R. Sleep and its importance in adolescence and in common adolescent somatic and psychiatric conditions. Int J Gen Med. 2011;4:425. | ||
Sadeh A. Cognitive-behavioral treatment for childhood sleep disorders. Clin Psychol Rev. 2005;25(5):612–628. | ||
Hartz I, Handal M, Tverdal A, Skurtveit S. Paediatric off-label use of melatonin-A register linkage study between the norwegian prescription database and patient register. Basic Clin Pharmacol Toxicol. 2015;117(4):267–273. | ||
Waldhauser F, Weiszenbacher G, Tatzer E, et al. Alterations in nocturnal serum melatonin levels in humans with growth and aging. J Clin Endocrinol Metab. 1988;66(3):648–652. | ||
Waldhauser F, Waldhauser M, Lieberman HR, Deng MH, Lynch HJ, Wurtman RJ. Bioavailability of oral melatonin in humans. Neuroendocrinology. 1984;39(4):307–313. | ||
Waldhauser, F., Weiszenbacher, G., Frisch,H., Zeitlhuber, U., Waldhauser, M., & Wurtman, R. J. Fall in nocturnal serum melatonin during prepuberty and pubescence. Lancet. 1984; 1(8374):362–365. | ||
Crowley SJ, Acebo C, Carskadon MA. Human puberty: salivary melatonin profiles in constant conditions. Dev Psychobiol. 2012;54(4):468–473. | ||
Roy D, Belsham DD. Melatonin receptor activation regulates GnRH gene expression and secretion in GT1-7 GnRH neurons. Signal transduction mechanisms. J Biol Chem. 2002;277(1):251–258. | ||
Terasawa E, Fernandez DL. Neurobiological mechanisms of the onset of puberty in primates. Endocr Rev. 2001;22(1):111–151. | ||
Belchetz PE, Plant TM, Nakai Y, Keogh EJ, Knobil E. Hypophysial responses to continuous and intermittent delivery of hypopthalamic gonadotropin-releasing hormone. Science. 1978;202(4368):631–633. | ||
Livadas S, Chrousos GP. Control of the onset of puberty. Curr Opin Pediatr. 2016;28(4):551–558. | ||
Wennink JM, Delemarre-van de Waal HA, Schoemaker R, Schoemaker H, Schoemaker J. Luiteinizing hormone and follicle stimulatiing hormone scretion patterns in girls throughout puberty measures using highly sensitive immunoradiometric assays. Clin Endocrinol. 1990;33(3):333–344. | ||
Parent AS, Teilmann G, Juul A, Skakkebaek NE, Toppari J, Bourguignon JP. The timing of normal puberty and the age limits of sexual precocity: variations around the world, secular trends, and changes after migration. Endocr Rev. 2003;24(5):668–693. | ||
Liu X, Herbison AE. Kisspeptin regulation of neuronal activity throughout the central nervous system. Endocrinol Metab. 2016;31(2):193–205. | ||
Lee JH, Miele ME, Hicks DJ, et al. KiSS-1, a novel human malignant melanoma metastasis-suppressor gene. J Natl Cancer Inst. 1996;88(23):1731–1737. | ||
Han SK, Gottsch ML, Lee KJ, et al. Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuroendocrine switch for the onset of puberty. J Neurosci. 2005;25(49):11349–11356. | ||
Kirilov M, Clarkson J, Liu X, et al. Dependence of fertility on kisspeptin-Gpr54 signaling at the GnRH neuron. Nat Commun. 2013;4(1):2492. | ||
Novaira HJ, Sonko ML, Hoffman G, et al. Disrupted kisspeptin signaling in GnRH neurons leads to hypogonadotrophic hypogonadism. Mol Endocrinol. 2014;28(2):225–238. | ||
León S, Barroso A, Vázquez MJ, et al. Direct Actions of Kisspeptins on GnRH Neurons Permit Attainment of Fertility but are Insufficient to Fully Preserve Gonadotropic Axis Activity. Sci Rep. 2016;6:19206. | ||
Messager S, Chatzidaki EE, Ma D, et al. Kisspeptin directly stimulates gonadotropin-releasing hormone release via G protein-coupled receptor 54. Proc Natl Acad Sci USA. 2005;102(5):1761–1766. | ||
Irwig MS, Fraley GS, Smith JT, et al. Kisspeptin activation of gonadotropin releasing hormone neurons and regulation of KiSS-1 mRNA in the male rat. Neuroendocrinology. 2004;80(4):264–272. | ||
Simonneaux V, Ansel L, Revel FG, Klosen P, Pévet P, Mikkelsen JD. Kisspeptin and the seasonal control of reproduction in hamsters. Peptides. 2009;30(1):146–153. | ||
Revel FG, Saboureau M, Masson-Pévet M, Pévet P, Mikkelsen JD, Simonneaux V. Kisspeptin mediates the photoperiodic control of reproduction in hamsters. Curr Biol. 2006;16(17):1730–1735. | ||
Gingerich S, Wang X, Lee PK, et al. The generation of an array of clonal, immortalized cell models from the rat hypothalamus: analysis of melatonin effects on kisspeptin and gonadotropin-inhibitory hormone neurons. Neuroscience. 2009;162(4):1134–1140. | ||
Larkin JE, Jones J, Zucker I. Temperature dependence of gonadal regression in Syrian hamsters exposed to short day lengths. Am J Physiol Regul Integr Comp Physiol. 2002;282(3):R744–R752. | ||
Lehman MN, Coolen LM, Goodman RL, Viguie C, Billings HJ, Karsch FJ. Seasonal plasticity in the brain: the use of large animal models for neuroanatomical research. Reprod Suppl. 2002;59:149–165. | ||
Paul MJ, Zucker I, Schwartz WJ. Tracking the seasons: the internal calendars of vertebrates. Philos Trans R Soc B Biol Sci. 2008;363(1490):341–361. | ||
Goodman RL, Jansen HT, Billings HJ, Coolen LM, Lehman MN. Neural systems mediating seasonal breeding in the ewe. J Neuroendocrinol. 2010;22(7):674–681. | ||
Foster DL. Puberty in the female sheep. In: Knobil E, Neill JD. (Eds.), Physiology of Reproduction. Raven Press. 1988. New York. | ||
Foster DL, Karsch FJ. Development of the mechanism regulating the preovulatory surge of luteinizing hormone in sheep. Endocrinology. 1975;97(5):1205–1209. | ||
Tran CT, Edey TN, Findlay JK. Pituitary response of prepuberal lambs to oestradiol-17 beta. Aust J Biol Sci. 1979;32(4–5):463. | ||
Clarke IJ. Gonadotrophin-releasing hormone secretion (GnRH) in anoestrous ewes and the induction of GnRH surges by oestrogen. J Endocrinol. 1988;117(3):355–360. | ||
Foster DL, Lemons JA, Jaffe RB, Niswender GD. Sequential patterns of circulating luteinizing hormone and follicle-stimulating hormone in female sheep from early postnatal life through the first estrous cycles. Endocrinology. 1975;97(4):985–994. | ||
Yellon SM, Goldman BD. Photoperiod control of reproductive development in the male Djungarian hamster (Phodopus sungorus). Endocrinology. 1984;114(2):664–670. | ||
Buchanan KL, Yellon SM. Delayed puberty in the male Djungarian hamster: effect of short photoperiod or melatonin treatment on the GnRH neuronal system. Neuroendocrinology. 1991;54(2):96–102. | ||
Kennaway DJ, Hughes PE, van Wettere WH. Melatonin implants do not alter estrogen feedback or advance puberty in gilts. Anim Reprod Sci. 2015;156:13–22. | ||
Kennaway DJ, Peek JC, Gilmore TA, Royles P. Pituitary response to LHRH, LH pulsatility and plasma melatonin and prolactin changes in ewe lambs treated with melatonin implants to delay puberty. J Reprod Fertil. 1986;78(1):137–148. | ||
Recabarren SE, Lobos A, Henriquez J, Peneipil C, Parilo J. Effect of daily melatonin treatment on the profile of luteinizing hormone secretion in prepubertal ewes. Agrociencia. 1998;14:303–15. | ||
Kennaway DJ, Gilmore TA, Dunstan EA. Pinealectomy delays puberty in ewe lambs. J Reprod Fertil. 1985;74(1):119–125. | ||
Wagner GC, Johnston JD, Clarke IJ, Lincoln GA, Hazlerigg DG. Redefining the limits of day length responsiveness in a seasonal mammal. Endocrinology. 2008;149(1):32–39. | ||
Yellon SM, Foster DL. Melatonin rhythms time photoperiod-induced puberty in the female lamb. Endocrinology. 1986;119(1):44–49. | ||
Simonneaux V, Ancel C, Poirel VJ, Gauer F. Kisspeptins and RFRP-3 act in concert to synchronize rodent reproduction with seasons. Front Neurosci. 2013;7:22. | ||
Malpaux B, Migaud M, Tricoire H, Chemineau P. Biology of mammalian photoperiodism and the critical role of the pineal gland and melatonin. J Biol Rhythms. 2001;16(4):336–347. | ||
Goldman BD. Mammalian photoperiodic system: formal properties and neuroendocrine mechanisms of photoperiodic time measurement. J Biol Rhythms. 2001;16(4):283–301. | ||
van Geijlswijk IM, Mol RH, Egberts TC, Smits MG. Evaluation of sleep, puberty and mental health in children with long-term melatonin treatment for chronic idiopathic childhood sleep onset insomnia. Psychopharmacology. 2011;216(1):111–120. | ||
Zwart T, Smits M, Egberts T, Rademaker C, van Geijlswijk I. Long-Term Melatonin Therapy for Adolescents and Young Adults with Chronic Sleep Onset Insomnia and Late Melatonin Onset: Evaluation of Sleep Quality, Chronotype, and Lifestyle Factors Compared to Age-Related Randomly Selected Population Cohorts. Healthcare. 2018;6(1):23. | ||
Carr R, Wasdell MB, Hamilton D, et al. Long-term effectiveness outcome of melatonin therapy in children with treatment-resistant circadian rhythm sleep disorders. J Pineal Res. 2007;43(4):351–359. | ||
Magee K, Basinska J, Quarrington B, Stancer HC. Blindness and menarche. Life Sci. 1970;9(1):7–12. | ||
Bojlén K, Bentzon MW. Seasonal variation in the occurrence of menarche. Dan Med Bull. 1974;21(5):161–168. | ||
Albright DL, Voda AM, Smolensky MH, Hsi BP, Decker M. Seasonal characteristics of and age at menarche. Chronobiol Int. 1990;7(3):251–258. | ||
Cohen P. Month at menarche: a re-evaluation of the seasonal hypothesis. Ann Hum Biol. 1993;20(2):198–202. | ||
Rojansky N, Brzezinski A, Schenker JG. Seasonality in human reproduction: an update. Hum Reprod. 1992;7(6):735–745. | ||
Silman RE, Leone RM, Hooper RJ, Preece MA. Melatonin, the pineal gland and human puberty. Nature. 1979;282(5736):301–303. | ||
Salti R, Galluzzi F, Bindi G, et al. Nocturnal melatonin patterns in children. J Clin Endocrinol Metab. 2000;85(6):2137–2144. | ||
Cavallo A, Ritschel WA. Pharmacokinetics of melatonin in human sexual maturation. J Clin Endocrinol Metab. 1996;81(5):1882–1886. | ||
Marceau K, Ram N, Houts RM, Grimm KJ, Susman EJ. Individual differences in boys’ and girls’ timing and tempo of puberty: modeling development with nonlinear growth models. Dev Psychol. 2011;47(5):1389–1409. | ||
Taylor DJ, Roane BM. Treatment of insomnia in adults and children: a practice-friendly review of research. J Clin Psychol. 2010;66(11):1137–1147. | ||
Kalak N, Gerber M, Kirov R, et al. Daily morning running for 3 weeks improved sleep and psychological functioning in healthy adolescents compared with controls. J Adolesc Health. 2012;51(6):615–622. | ||
Creswell JD, Irwin MR, Burklund LJ, et al. Mindfulness-Based Stress Reduction training reduces loneliness and pro-inflammatory gene expression in older adults: a small randomized controlled trial. Brain Behav Immun. 2012;26(7):1095–1101. | ||
Morin CM, Bootzin RR, Buysse DJ, Edinger JD, Espie CA, Lichstein KL. Psychological and behavioral treatment of insomnia:update of the recent evidence (1998–2004). Sleep. 2006;29(11):1398–1414. | ||
Kennaway DJ. Light, neurotransmitters and the suprachiasmatic nucleus control of pineal melatonin production in the rat. Neurosignals. 1997;6(4–6):247–254. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
