Back to Journals » Clinical Interventions in Aging » Volume 15
Cervical Interferential Current Transcutaneous Electrical Sensory Stimulation for Patients with Dysphagia and Dementia in Nursing Homes
Authors Hara Y, Nakane A, Tohara H , Kubota K, Nakagawa K , Hara K , Yamaguchi K , Yoshimi K , Minakuchi S
Received 31 July 2020
Accepted for publication 26 November 2020
Published 5 January 2021 Volume 2020:15 Pages 2431—2437
DOI https://doi.org/10.2147/CIA.S274968
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Yoshiko Hara,1 Ayako Nakane,2 Haruka Tohara,2 Kazumasa Kubota,1 Kazuharu Nakagawa,2 Koji Hara,2 Kohei Yamaguchi,2 Kanako Yoshimi,2 Shunsuke Minakuchi1
1Department of Gerodontology and Oral Rehabilitation, Division of Gerontology and Gerodontology, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, Tokyo, Japan; 2Department of Dysphagia Rehabilitation, Division of Gerontology and Gerodontology, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, Tokyo, Japan
Correspondence: Ayako Nakane
Department of Dysphagia Rehabilitation, Division of Gerontology and Gerodontology, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, 1-5-45 Yushima, Bunkyo-ku, Tokyo 113-8549, Japan
Tel/Fax +81-3-5803-5562
Email [email protected]
Purpose: We aimed to determine the efficacy of interferential current transcutaneous electrical sensory stimulation (IFC-TESS) in patients with dementia who were being treated for dysphagia in nursing homes under normal living conditions.
Patients and Methods: This study recruited 54 nursing home residents (13 males, 41 females; mean age, 84.5 ± 10.7 years) with dysphagia and dementia. IFC-TESS was performed with Gentle Stim® (Careido Co. Ltd., Kanagawa, Japan) for 15 minutes. Two sessions per day were administered 5 days per week, for 3 weeks. The outcomes included the improvement of the patients’ cough reflexes and nutritional states across the 3 weeks of intervention; the former was evaluated with the cough test based on the changes in cough latency time and cough frequency, while the latter was assessed based on changes in the functional oral intake scale (FOIS) score and oral calorie intake.
Results: The cough latency times before and after the 3-week intervention were 15.8 (7.0– 60.0) and 6.7 (3.6– 30.7) s/min, respectively; the cough frequencies were 5.0 (0.0– 5.0) and 5.0 (5.0– 5.0) t/min, respectively; the FOIS scores were 5.0 (5.0– 6.0) and 5.0 (5.0– 6.0), respectively; and oral calorie intakes were 1300.0 (862.5– 1420.0) and 1300.0 (1005.0– 1462.5) kcal/day, respectively. All parameters significantly improved across the intervention (p < 0.05). When the results were adjusted for age, sex, body mass index, mini nutritional assessment results, Barthel index, and mini-mental state examination scores, IFC-TESS improved the cough reflex and oral calorie intake in older nursing home residents with dementia.
Conclusion: Cervical IFC-TESS can improve the cough reflexes and nutritional states of patients with dysphagia and dementia in nursing homes. As it has previously been difficult to perform active rehabilitation with dementia patients, IFC-TESS may be effective and practicable for the rehabilitation of this patient population.
Keywords: cough frequency, cough latency time, FOIS, IFC-TESS, oral calorie intake
Introduction
With a global morbidity of 7.7 million people,1 dementia is associated with significant economic and social burdens. The treatment of patients with dementia can be complicated in cases of comorbid dysphagia, as diminished cognition compromises the treatment of dysphagia.2
Rehabilitative electrical stimulation includes therapeutic electrical stimulation to increase muscular strength, functional electrical stimulation to induce compensatory muscular contraction, and transcutaneous electrical nerve stimulation (TENS) to achieve analgesia. The principal targets for these treatments are the nerves and muscles, and many TENS procedures use low frequencies. In dysphagia rehabilitation, mainly low-frequency electricity has been applied to increase muscular strength in the neck.3–7 However, as TENS cannot stimulate structures situated deep in the body, an interferential current has been developed to overcome this disadvantage;8 interference between electricity of two different frequencies generates a new interferential current inside the human body that can affect deeply situated sensory nerves.9
The use of interferential currents to facilitate swallowing training has yielded the following findings concerning interferential currents: (i) in decerebrate and paralyzed animals, the reduction of the latency time in the elicitation of the swallowing reflex and the excitation of swallowing-related neurons in the brain stem;10 (ii) in healthy human volunteers, the reduction of the swallowing threshold;11,12 and (iii) in patients with chronic, pharyngeal-stage dysphagia and mini-mental state exam (MMSE) scores of 21 or more, the improvement of the swallowing reflex.13,14 Furthermore, interferential current transcutaneous electrical sensory stimulation (IFC-TESS) improved cough latency times and increased oral calorie intake in patients with dysphagia and dementia in an acute or recovery medical care facility.15 However, the effects of IFC-TESS on swallowing function in the large population of patients with chronic dysphagia and associated dementia, with whom training is difficult to perform, remain unknown.
The present study aimed to elucidate the effects of IFC-TESS on nursing home residents with dysphagia and dementia. We hypothesized that IFC-TESS would improve cough latency times, cough frequencies, functional oral intake scale (FOIS) scores, and oral calorie intake in these patients. Improvements in cough reflex and oral calorie intake due to IFC-TESS intervention would offer major benefits to dysphagia and dementia treatment, where there is a need for improvement in sustainable nutritional rehabilitation.
Materials and Methods
Subjects
A total of 74 patients residing in three nursing homes were administered a texture-modified diet for their condition. Patients who met one or more of the following criteria were excluded: (i) asthma, (ii) receipt of total parenteral nutrition, (iii) epilepsy, (iv) receipt of pacemakers, (v) MMSE scores of 28 or more, or (vi) abstention from the intervention for 3 or more days (the patients left the study for various reasons: hospitalization, left the facility, refused continuation). Hence, 54 patients were considered in the final analysis (13 males, 41 females; mean age, 84.5 ± 10.7 years). Figure 1 presents the patient flowchart for this study.
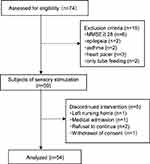 |
Figure 1 Participant flowchart. |
This study was performed from February to September 2019. The subjects led their usual lives, and no intervention for dysphagia apart from the diet and the IFC-TESS was provided.
Study Design and Evaluation
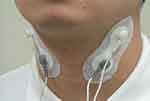
IFC-TESS was performed using Gentle Stim® (Careido Co. Ltd., Kanagawa, Japan, Medical device certification number: 227AHBZX00026000) for 15 minutes in the morning and afternoon, 5 days per week (the weekdays), for 3 weeks. The output current at the time of treatment was set to 2.0 to 3.0 mA. Previous studies have found this current to produce no sensation of pain.12,15 After the necks of the patients were wiped with ethanol for disinfection, the Gentle Stim® electrodes were attached to both the right and left sides of the neck, along the anterior border of the sternocleidomastoid muscle, which is delimited by the lower edge of the mandibular angle and the horizontal level of the thyroid cartilage (Figure 2). The ipsilateral electrodes were preserved and insulated at 4 cm, and two pairs of electrodes with different carrier frequencies were placed diagonally across the thyroid cartilage13,16 (Details are described in the supplementary materials, Figures S1 and S2).
The cough reflex was used to evaluate the efficacy of the intervention.15 To assess the cough reflex, 1% citric acid dissolved in physiological saline solution was administered by inhalation using a nebulizer (NE-U22 Handy Type Mesh Nebulizer, Omron Co., Kyoto, Japan). The participant was instructed to practice deep breathing beforehand to be able to inhale enough mist through the mouth. A nasal plug was used to restrict participants to mouth breathing. The cough latency and frequency were recorded for a maximum period of 1 minute. The cough latency was defined as the time from the initial exposure to the 1% citric acid until the first cough. The number of coughs was used to screen for silent aspiration, with five or more coughs being considered negative.16
The nursing home’s records were referenced to obtain the FOIS score and oral calorie intake. In addition, the age, sex, and cause of the swallowing disorder were obtained from the facility’s records. The body mass index (BMI), which was used to indicate the obesity status, and the results of the mini nutritional assessment (MNA) were used as indicators of nutritional state. The MMSE was used to assess cognitive function.17 In addition, the Barthel index (BI) was used as an index of activities of daily living.18
Sample Size
The requisite sample size was calculated using G*Power (Kiel University, Kiel, Germany). As the necessary effect size of the primary endpoint was determined to be 0.46 in a previous study,15 the effect size, α-value, and detection power were determined in the present study to be 0.46, 0.05, and 0.8, respectively.19,20 It was calculated that 41 subjects were required. A predicted withdrawal rate of 10% informed the decision to enroll at least 45 participants.
Endpoints
Cough latency time was the primary endpoint.15 Cough frequency, FOIS score, and oral calorie intake were the secondary endpoints. Each metric was measured before and after the 3-week intervention, and the results were compared.
Statistical Analysis
The Shapiro–Wilk test was used to assess data normality. The Wilcoxon signed-rank test was used to compare findings across the intervention. Statistical significance was set to p < 0.05. Multiple regression analysis was performed, with the differences in cough latency time, cough frequency, FOIS score, and oral calorie intake before and after intervention as the dependent variables, and age, sex, BMI, MNA, BI, and MMSE as the independent variables. For the post hoc analysis, the effect size was calculated using G*Power. SPSS software (version 25.0; IBM, Tokyo, Japan) was used for statistical analysis.
Results
The participant characteristics are shown in Table 1. Five participants were unable to continue the intervention (Figure 1). Our findings concerning the cough reflex and nutritional status across the 3-week IFC-TESS intervention are presented in Table 2.
 |
Table 1 Baseline Characteristics of the Patients |
 |
Table 2 Outcome Comparisons of Cough Reflex and Nutritional Intake Before and After the 3-Week IFC-TESS |
Cough latency time decreased significantly across the intervention (p < 0.001), while cough frequency increased significantly (p = 0.001). The median of the FOIS score and the oral calorie intake did not change but the interquartile range of the oral calorie intake increased after the intervention; conclusively, a statistically significant change was observed for both variables (FOIS, p = 0.02; oral calorie intake, p < 0.001); this may have been due to progressive improvements in participants with particularly low oral calorie intake at baseline (Figure S3), and FOIS scores improved (Figure S4). The results of multiple regression analysis adjusted for age, sex, BMI, MNA, BI, and MMSE are shown in Table 3.
 |
Table 3 Comparison of Differences in Cough Latency, Cough Frequency, FOIS, and Nutritional Oral Intake Before and After the 3-Week IFC-TESS |
Discussion
Administration of IFC-TESS to Dementia Patients in a Chronic Facility
Improvements in the primary endpoint (cough latency time) and the secondary endpoints (cough frequency, FOIS score, and oral calorie intake) were found among patients with dementia residing in nursing homes. In dysphagia rehabilitation, neuromuscular electrical stimulation (NMES) is initially applied to the surfaces of the pharynx-associated muscles to increase muscular strength; however, as pain develops with the increasing intensity of stimulation,21 patients with dementia have often been excluded from previous NMES studies.22–24 While no consensus has been reached regarding the optimal intensity at which TENS should be applied to alleviate pain,25,26 the stimulation parameters are considered to determine the efficacy of pain alleviation and the duration of the maintenance of analgesia after stimulation. In general, unpleasant sensation at the time of electrical stimulation decreases as the frequency increases, and muscular contraction does not occur at frequencies of 1 kHz or higher.27
IFC-TESS is a form of sensory stimulation and consequent pain attenuation or elimination that delivers an interferential current generated by the difference between two currents of medium frequencies of 2000 and 2050 Hz. Electrical skin resistance can be reduced with medium frequency, enabling conduction to deeper tissues.28 In addition, in the present study, the stimulation intensity of 2–3 mA induced no sensation of pain. Our findings thus indicate that the continued use of IFC-TESS can be effective in dysphagia rehabilitation without inducing unpleasant sensations in dementia patients.
Improvement of the Cough Reflex
The decreased cough latency time and increased cough frequency we observed indicated improvements in the cough reflex. In addition, improvements were found in three of the six subjects with a previous history of aspiration pneumonia. No form of dysphagia rehabilitation apart from IFC-TESS was performed during the intervention period, and the patients continued their usual lives. Hence, the improvement in cough reflex indicates the efficacy of IFC-TESS intervention, which has been attributed to the effect of IFC-TESS on the superior laryngeal nerve.12,29
The inner branch of the superior laryngeal nerve, which innervates the epiglottis, laryngeal vestibule, and hypopharynx, is well known to be the most important sensory nerve for inducing coughing and swallowing.30–32 In addition, the cough center in the solitary tract nucleus of the medulla oblongata is positioned close to and is closely associated with, the swallowing center. Furthermore, once the transient receptor potential vanilloid channels of the C-fibers (which are centripetal sensory nerves in the airway) are activated by chemical and physical stimuli, the cough reflex is elicited through the mediation of the neurotransmitter substance-P, reinforcing the cough and swallowing reflexes.33,34 Substance-P production is reduced in the cervical ganglia of older individuals and those with cerebrovascular disorders. Further, substance-P can become depleted as a result of aspiration pneumonia.35 In particular, damage to the basal ganglia results in diminished dopamine levels and consequently reduces concentrations of substance-P in the glossopharyngeal and vagus nerves.34 However, it has been shown recently that cervical electrical stimulation induces the temporary pharyngeal accumulation of substance-P in healthy volunteers and patients with stroke-induced acute severe dysphagia.36,37 With IFC-TESS, the superior laryngeal nerve in this region is targeted,12,29 and both the swallowing reflex and cough reflex are thus improved. In dysphagic patients, due to improvement in cough sensitivity, and likewise in airway defense function,15 IFC-TESS treatment can contribute to the prevention of aspiration pneumonia.
Effects of IFC-TESS
The adjusted results of the multiple regression analysis showed that IFC-TESS was effective in improving cough latency time, cough frequency, FOIS score, and oral calorie intake. The progression of dementia is accompanied by a decreasing amount of exercise consequent to increasing time spent seated and a diminished ability to walk. As there are few means by which to overcome the reduced amount of exercise, the physical abilities of patients with dementia tend to deteriorate; this is common among residents in chronic facilities, and adversely affects recovery during dysphagia rehabilitation.38 We have shown that IFC-TESS therapy improves the cough reflex and oral calorie intake, irrespective of dementia severity and BI. Therefore, IFC-TESS therapy may be of great benefit to patients with dysphagia, especially those with a compromised nutritional status and impaired cognition.
Limitations
We could have performed more frequent follow-ups or examined maintenance of the effects beyond the 3-week intervention period, but this study was designed to help determine the appropriate stimulation parameters. In addition, this study excluded patients with severe dysphagia for whom oral intake was impossible; hence, the efficacy of IFC-TESS in this population remains unknown. The participants in this study had chronic dysphagia, with little possibility of improvement in the absence of rehabilitation; therefore, a control group was not utilized due to ethical considerations. As such, the possibility of bias due to other confounding variables, which may have affected the outcomes, could not be ruled out. Furthermore, while the participants were provided with a texture-modified diet, video endoscopy or video fluorography were not performed in all cases (unless necessary), due to invasiveness and ethical considerations. Thus, as the functional evaluation was restricted, we evaluated increases in oral calorie intake and FOIS as indicators of improved function.
Conclusion
Our findings suggest that cervical IFC-TESS in patients with dementia and dysphagia in nursing homes improves the cough reflex and increases oral calorie intake. IFC-TESS thus offers another effective mode of dysphagia rehabilitation that can be feasibly administered to dementia patients, with whom active modes of rehabilitation are difficult to complete.
Abbreviations
BI, Barthel index; BMI, body mass index; FOIS, functional oral intake scale; IFC-TESS, interferential current transcutaneous electrical sensory stimulation; MMSE, mini-mental state exam; MNA, mini nutritional assessment; NMES, neuromuscular electrical stimulation; TENS, transcutaneous electrical nerve stimulation.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Ethics Approval and Informed Consent
Written and oral informed consent for participation in this study was obtained from all participants and their family members. In accordance with the Declaration of Helsinki, this study was approved by the Ethics Committee at Tokyo Medical and Dental University’s Faculty of Dentistry (approval no.: D2018-005, UMIN 000032262). IFC-TESS is approved as a medical device in Japan. The study was performed safely without any of the participants complaining of pain or unpleasant sensation, and there were no reports of withdrawal symptoms or dependency.39
Acknowledgments
We would like to thank all the patients who participated in this study.
Funding
Part of this study was supported by JSPS KAKENHI Grant-in-Aid for Scientific Research (JP16K11883), and Research Agency grant 8020, 2019 (12100001201x2911; Ayako Nakane).
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Dementia: A Public Health Priority. Geneva, Switzerland: World Health Organization; 2012.
2. Meguro K. Evidence-based rehabilitation of dementia patents: presentation of problems. High Brain Funct Res. 2012;32:477–484. doi:10.2496/hbfr.32.477
3. Tang Y, Lin X, Lin XJ, et al. Therapeutic efficacy of neuromuscular electrical stimulation and electromyographic biofeedback on Alzheimer’s disease patients with dysphagia. Medicine. 2017;96:e8008. doi:10.1097/MD.0000000000008008
4. Permsirivanich W, Tipchatyotin S, Wongchai M, et al. Comparing the effects of rehabilitation swallowing therapy vs. neuromuscular electrical stimulation therapy among stroke patients with persistent pharyngeal dysphagia: A randomized controlled study. J Med Assoc Thai. 2009;92:259–265.
5. Leelamanit V, Limsakul C, Geater A. Synchronized electrical stimulation in treating pharyngeal dysphagia. Laryngoscope. 2002;112:2204–2210. doi:10.1097/00005537-200212000-00015
6. Freed ML, Freed L, Chatburn RL, Christian M. Electrical stimulation for swallowing disorders caused by stroke. Respir Care. 2001;46:466–474.
7. Carnaby-Mann GD, Crary MA. Adjunctive neuromuscular electrical stimulation for treatment-refractory dysphagia. Ann Otol Rhinol Laryngol. 2008;117:279–287. doi:10.1177/000348940811700407
8. Hamada Y, Amano R. Treatment methods for orthopedic disorders using low-frequency electricity. Biomedical Eng. 1990;4:33–39.
9. Venancio RC, Pelegrini S, Gomes DQ, Nakano EY, Liebano RE. Effects of carrier frequency of interferential current on pressure pain threshold and sensory comfort. Arch Phys Med Rehabil. 2013;94:95–102. doi:10.1016/j.apmr.2012.08.204
10. Umezaki T, Sugiyama Y, Fuse S, Mukudai S, Hirano S. Supportive effect of interferential current stimulation on susceptibility of swallowing in guinea pigs. Exp Brain Res. 2018;236:2661–2676. doi:10.1007/s00221-018-5325-0
11. Tsujita J. Transcutaneous cervical stimulation with interferential currents reduces swallowing threshold. Phys Ther Res. 2012;39:1349.
12. Furuta T. Interferential electric stimulation applied to the neck increases swallowing frequency. Dysphagia. 2012;27:94–100. doi:10.1007/s00455-011-9344-2
13. Sugishita S. Effect of short term interferential current stimulation on swallowing reflex in dysphagic patients. Int J Speech Lang Pathol Audiol. 2015;3:1–8. doi:10.12970/2311-1917.2015.03.01.1
14. Ortega O, Rofes L, Martin A, Arreola V, López I, Clavé P. A comparative study between two sensory stimulation strategies after two weeks treatment on older patients with oropharyngeal dysphagia. Dysphagia. 2016;31(5):706–716. doi:10.1007/s00455-016-9736-4
15. Maeda K. Interferential current sensory stimulation, through the neck skin, improves airway defense and oral intake in patients with dysphagia: a double-blind randomized controlled trial. Clin Interv Aging. 2017;12:1879–1886. doi:10.2147/CIA.S140746
16. Wakasugi Y, Tohara H, Nakane A, et al. Investigation of usefulness of cough tests in screening for silent aspiration. Japanese J Dysphagia Rehabil. 2008;12:109–117.
17. Folstein MF, Folstein SE, McHugh PR. “Mini-Mental State.” A practical method for grading the cognitive state of patients for the clinician. J Psychiat Res. 1975;12:189–198. doi:10.1016/0022-3956(75)90026-6
18. Mahoney FI, Barthel DW. Functional evaluation: the Barthel Index. Md State Med J. 1965;14:61–65.
19. Dupont WD, Plummer WD. Power and sample size calculations for studies involving linear regression. Control Clin Trials. 1998;19:589–601. doi:10.1016/S0197-2456(98)00037-3
20. Cohen J. Statistical Power Analysis for the Behavioral Sciences.
21. Nakajoh K. Relation between incidence of pneumonia and protective reflexes in post-stroke patients with oral or tube feeding. J Intern Med. 2000;247:39–42. doi:10.1046/j.1365-2796.2000.00565.x
22. Huang KL, Liu TY, Huang YC, Leong CP, Lin WC, Pong YP. Functional outcome in acute stroke patients with oropharyngeal dysphagia after swallowing therapy. J Stroke Cerebrovasc Dis. 2014;23:2547–2553. doi:10.1016/j.jstrokecerebrovasdis.2014.05.031
23. Sun SF, Hsu CW, Lin HS, et al. Combined neuromuscular electrical stimulation (NMES) with fiberoptic endoscopic evaluation of swallowing (FEES) and traditional swallowing rehabilitation in the treatment of stroke-related dysphagia. Dysphagia. 2013;28:557–566. doi:10.1007/s00455-013-9466-9
24. Lee KW, Kim SB, Lee LH, Lee SJ, Ri JW, Park JG. The effect of early neuromuscular electrical stimulation therapy in acute/subacute ischemic stroke patients with dysphagia. Ann Rehabil Med. 2014;38:153–159. doi:10.5535/arm.2014.38.2.153
25. Hara K, Yoshida H, Kataishi Y, et al. Investigation of pain-alleviating effects of transcutaneous electrical nerve stimulation (TENS): focus on physiological indices and TENS stimulation intensity. J Phys Ther Sci. 2015;30:63–68.
26. Claydon LS, Chesterton LS, Barlas P, Sim J. Dose-specific effects of transcutaneous electrical nerve stimulation (TENS) on experimental pain: a systematic review. Clin J Pain. 2011;27:635–647. doi:10.1097/AJP.0b013e31821962b4
27. Watanabe K, Nagaoka M. Expansion of electrical stimulation therapy in rehabilitation. Juntendo Medical J. 2010;56:29–36. doi:10.14789/pjmj.56.29
28. Yanagisawa K. Pain, physical therapy, ultrasound therapy, and electrical stimulation therapy. J Phys Ther Sci. 2000;15:105–110.
29. Oku Y, Okada Y. Transcutaneous electrical stimulation lowers swallowing threshold. Ann Japanese Respiratory Society. 2006;44:133.
30. Oku Y, Tanaka I, Ezure K. Activity of bulbar respiratory neurons during fictive coughing and swallowing in the decerebrate cat. J Physiol. 1994;480:309–324. doi:10.1113/jphysiol.1994.sp020361
31. Doty RW. Influence of stimulus pattern on reflex deglutition. Am J Physiol. 1951;166(1):142–158. doi:10.1152/ajplegacy.1951.166.1.142
32. Hiroto I, Toyozumi Y, Yatake Y, et al. Comparative anatomy of laryngeal nerves. J Otolaryngol Jpn. 1967;71:212–216.
33. Shioya T, Sato K, Sano M, Watanabe H. Transient receptor potential (TRP) channel and cough. Folia Pharmacol Jpn. 2008;131:417–422. doi:10.1254/fpj.131.417
34. Takahashi O. Preventive strategies for aspiration pneumonia in elderly disabled persons. Tohoku J Exp Med. 2005;207:3–12. doi:10.1620/tjem.207.3
35. Nakagawa T, Ohrui T, Sekizawa K, Sasaki H. Sputum substance P in aspiration pneumonia. Lancet. 1995;345:1447. doi:10.1016/S0140-6736(95)92638-0
36. Suntrup-Krueger S, Bittner S, Recker S, et al. Electrical pharyngeal stimulation increases substance P level in saliva. Neurogastroenterol Motil. 2016;28(6):855–860. doi:10.1111/nmo.12783
37. Muhle P, Suntrup-Krueger S, Bittner S, et al. Increase of substance P concentration in saliva after pharyngeal electrical stimulation in severely dysphagic stroke patients–an indicator of decannulation success? Neurosignals. 2017;25:74–87. doi:10.1159/000482002
38. Mikogami Y, Maruyama M, Hashimoto N, et al. Analysis of malnutrition and effects of rehabilitation in elderly patients with dysphagia. JPEN J Parenter Enteral Nutr. 2010;25:47
39. Nagami S, Maeda K, Fukunaga S, Ikeno M, Oku Y. Safety of transcutaneous electrical sensory stimulation of the neck in terms of vital parameters in dysphagia rehabilitation. Sci Rep. 2019;9:13481. doi:10.1038/s41598-019-49954-9
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.