Back to Journals » ClinicoEconomics and Outcomes Research » Volume 13
Budget Impact Associated with the Introduction of the Impella 5.0® Mechanical Circulatory Support Device for Cardiogenic Shock in France
Authors Le Guyader A, Pernot M , Delmas C , Roze S, Fau I , Flecher E, Lebreton G
Received 21 August 2020
Accepted for publication 15 December 2020
Published 19 January 2021 Volume 2021:13 Pages 53—63
DOI https://doi.org/10.2147/CEOR.S278269
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Giorgio Colombo
Alexandre Le Guyader,1 Mathieu Pernot,2 Clément Delmas,3 Stéphane Roze,4 Isabelle Fau,5 Erwan Flecher,6 Guillaume Lebreton7
1Department of Thoracic and Cardiovascular Surgery, Dupuytren University Hospital, Limoges, France; 2Department of Cardiology and Cardio‐Vascular Surgery, Haut-Lévèque University Hospital, Bordeaux, France; 3Cardiology Department, Rangueil University Hospital, Toulouse, France; 4Vyoo Agency, Lyon, France; 5SGE Consulting, Paris, France; 6Department of Cardio-Thoracic and Vascular Surgery, Pontchaillou University Hospital, Rennes, France; 7Cardiac Surgery Department, Pitié-Salpétrière Hospital, Sorbonne University, Paris, France
Correspondence: Isabelle Fau Email [email protected]
Aim: Cardiogenic shock (CS), if not diagnosed and treated rapidly, can lead to irreversible multiorgan damage and death. An economic analysis was conducted to determine the budget impact of the introduction of Impella 5.0®, a mechanical circulatory support (MCS) device that directly unloads the left ventricle, into clinical practice in patients with left ventricular CS in France.
Methods: A budget impact model was developed to compare the cost of Impella 5.0 with veno-arterial extracorporeal membrane oxygenation (VA-ECMO) from the perspective of the French national healthcare insurer. Costs associated with Impella 5.0, plus complication-related costs for VA-ECMO or Impella 5.0 from 2019 were included and clinical input data relating to complication rates and time spent on device were sourced from published literature. Extensive scenario and one-way deterministic sensitivity analyses were performed to explore the influence of uncertainty around key input parameters.
Results: Over a time horizon of 5 years, the introduction of Impella 5.0 was associated with cumulative savings of EUR 4.3 million. The results were driven by the lower risk of device-related complications associated with Impella 5.0. Savings were apparent from Year 1 onwards, with savings in excess of EUR 375,000 projected in Year 1 alone. On a per-patient level, in Year 1, estimated savings with the introduction of Impella 5.0 totaled EUR 616 per patient. Sensitivity analyses showed that the findings of the analysis were robust.
Conclusion: The Impella 5.0 device was associated with cumulative cost savings in excess of EUR 4 million over a 5-year period compared with current practice. Projected savings were driven by a lower rate of device-related complications with Impella 5.0 compared with VA-ECMO.
Keywords: cardiogenic shock, left-ventricular assist devices, Impella 5.0®, budget impact, France
Introduction
Cardiogenic shock (CS) is characterized by hypotension (systolic blood pressure [SBP] ≤90 mmHg) despite adequate filling pressures, accompanied by clinical and biochemical signs of systemic hypoperfusion.1 Early recognition and treatment of CS is critical to prevent irreversible organ damage and death.2 The most common underlying etiology of CS is acute myocardial infarction (MI) with left ventricular dysfunction, which accounts for up to 80% of cases,1,3 and in France in 2015 an estimated 3% of acute MIs were complicated by CS.4
There is a high unmet need for effective interventions for patients with CS; it remains the most common cause of in-hospital mortality in patients hospitalized for MI,1 with reported rates of in-hospital mortality in CS patients ranging from 40–50% at 1 month but up to 70% for patients with refractory CS.5 Pharmacologic intervention with inotropes or vasopressors is generally advocated as a first-line treatment option;6 however, 15–20% of patients with CS are refractory to pharmacologic treatment.7
Mechanical circulatory support (MCS) devices represent a critical and increasingly widely used component in the management of patients with refractory CS. Temporary MCS devices serve as a bridge to recovery, the placement of a durable MCS or heart transplantation. One such MCS device is Impella 5.0®, which initially received the CE mark for marketing in Europe in 2003 and which can be used to support left ventricular function and maintain systemic perfusion in patients with CS related to left ventricular failure for up to 10 days.8 The Impella 5.0 device is a microaxial pump mounted on a catheter. Once in position, the device pumps blood from the dysfunctional left ventricle into the ascending aorta at a flow rate of up to 5 L/min at peak level.9 The Impella 5.0 device directly unloads the left ventricle, which reduces workload and myocardial oxygen demand, it also increases mean arterial pressure and cardiac output, which in turn increases both coronary and systemic perfusion.10–12
Large-scale, prospective randomized controlled trials (RCTs) of interventions in CS are relatively scarce, in part due to the challenges associated with conducting RCTs in critically ill patients.13 Available clinical data in CS are therefore largely from retrospective observational analyses. For example, a recently published retrospective analysis by Karami et al examined CS outcomes with Impella 5.0 compared with veno-arterial extracorporeal membrane oxygenation (VA-ECMO) in a total of 128 patients (n=90 for Impella 5.0 and n=38 for VA-ECMO).14 The incidence of device-related complications, some of which can be associated with high direct costs (e.g. access site infection, limb ischemia requiring surgery or removal of the device), were significantly lower with Impella 5.0 than VA-ECMO (17% versus 40%; p<0.01).14
As with many advances in technology, despite the ease of use and clinical benefit in terms of lower incidence of device-related complications, the acquisition costs of new interventions, particularly if higher than costs associated with the standard of care, frequently represent a barrier to widespread uptake, particularly for payers and policy makers faced with budgetary constraints. In France in particular, in 2013, spending on healthcare constituted 11.6% of gross domestic product (GDP), one of the highest proportions in Europe.15 As such, there has been increasing emphasis on the economic considerations associated with new interventions including cost-effectiveness and budget impact analyses. The healthcare system in France is one of universal health coverage through statutory health insurance (SHI) funded through employee and employer contributions and taxation. This is often supplemented with voluntary supplementary insurance that may cover co-payment and areas where SHI coverage is poor.15 However, over three quarters of health care expenditure is publicly funded through SHI.15 Given the increasing importance of economic considerations, a budget impact analysis was conducted to determine the budget impact associated with the introduction of the Impella 5.0 device to the CS care pathways for two indications as follows: 1) treatment of adults aged <65 years with left-ventricular refractory post-cardiotomy CS without refractory respiratory failure requiring extracorporeal ventilatory support and without severe multiorgan failure and 2) patients aged <65 years with refractory CS without refractory respiratory failure requiring extracorporeal ventilatory support and without severe multiorgan failure, awaiting heart transplantation or long-term circulatory support.16
Methods
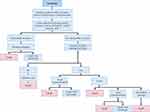
A budget impact model (BIM) was developed in Microsoft Excel (Redmond WA, USA). The model consisted of two distinct components of the care pathway for patients with CS. The first component considered the care pathway for a patient with left ventricular CS (with or without cardiac surgery) prior to the decision to utilize either Impella 5.0 or VA-ECMO (see Figure 1). The initial component of the care pathway was developed in collaboration with six leading clinical experts working within the French healthcare system (including one cardiologist-intensivist, four cardiac surgeons and one pharmacist). The initial component of the care pathway, including all transition probabilities, was identical for all patients and therefore costs incurred during this phase were not captured in the budget impact calculation.
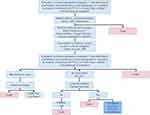 |
Figure 1 Treatment pathway prior to Impella 5.0®. Abbreviations: BNP, brain natriuretic peptide; LVAD, left ventricular assist device; PiCCO, pulse contour cardiac output. |
The second component of the care pathway considered the costs and outcomes after initiation of VA-ECMO or Impella 5.0. This component of the care pathway captured the likelihood of myocardial recovery, and for patients without myocardial recovery, palliative care, heart transplantation or placement of a long-term left-ventricular assist device (LVAD). The likelihood of mortality at each juncture was also captured (Figures 2 and 3). Event probabilities for both the Impella 5.0 and VA-ECMO arms were based on clinical expert opinion, clinical databases, and published literature14,17 (Table 1). Specifically, clinical input was sought from a total of six clinical experts who participated in the data collection and were based at French hospitals, such that the modeled care pathway was an accurate representation of the current situation in clinical practice in France. Two scenarios were considered in the BIM. Scenario 1 represented current clinical practice, wherein Impella 5.0 is not funded and all patients received VA-ECMO. Scenario 2 represented a potential future practice, wherein Impella 5.0 was available for eligible CS patients. The projected market share for Impella 5.0 relative to VA-ECMO throughout the 5-year time horizon of the analysis is shown in Table 2.
 |
Table 1 Clinical Input Data Utilized in the Budget Impact Analysis |
 |
Table 2 Projected Market Share Estimates in Years 1–5 (2019–2024) |
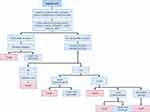 |
Figure 2 Treatment pathway after decision to use Impella 5.0®. Abbreviations: BNP, brain natriuretic peptide; LVAD, left ventricular assist device. |
In terms of patient population, it was assumed that, on a national level, each year a total of 610 patients with refractory left-ventricular CS would be eligible for either Impella 5.0 or VA-ECMO. Details on how this patient population was estimated are provided in the Supplementary Material.
Costs accounted in the base case analysis were derived from published sources18–20 and included those associated with stay in the intensive care unit (per day), cost of Impella 5.0, cost of circulatory support withdrawal strategy and non-diagnosis-related group (DRG) costs for device-related complications; this included the costs of daily supplements (daily supplementary cost for intensive care unit [ICU] stay, supplementary cost for a dialysis session), add-on list costs (LVADs) as well as the costs of complications over the 5-year time horizon (lower limb amputation, management of stroke), all of which represent costs incurred by the insurer (Table 3). As the payer perspective of the analysis was that of mandatory health insurance intra-DRG costs were not captured within the budget impact calculation. Hospital costs were 2019 values and add-on list prices were 2020 values, cost data relating to amputations were sourced from published literature and inflated to 2019 values and finally, costs associated with the management of stroke over the 5-year period were sourced from 2017 data and not adjusted.
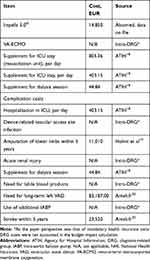 |
Table 3 Unit Cost Data |
A series of scenario and deterministic sensitivity analyses were performed to assess the robustness of findings to changes in key input parameters. Specifically, a scenario analysis was performed in which costs associated with device-related complications were not accounted and scenario analyses were also performed around Impella 5.0 market share and the costs associated with VA-ECMO-related consumables in the VA-ECMO arm. Deterministic sensitivity analyses were also performed around target population size, length of stay with both Impella 5.0 and VA-ECMO, treatment costs, costs of complications and the incidence of complications (including lower limb amputation and vascular site access infections). Here, analyses were performed in which inputs in the base case analyses were increased or decreased by 20% (±10% for the Impella 5.0 tariff sensitivity analysis).
The analysis was performed over a time horizon of 5 years from the perspective of the French healthcare payer (mandatory health insurance) and no discounting was applied to costs, in line with national recommendations and guidelines issued by the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) for budget impact analyses.21,22
Results
In the base case analysis, which included direct costs attributable to device-related complications, over a period of 5 years, total costs for patients with left ventricular refractory CS without Impella 5.0 were projected to be EUR 90.8 million (Table 4). With the introduction of Impella 5.0, total costs were estimated at EUR 86.5 million over a 5-year period. Consequently, the introduction of Impella 5.0 into clinical practice was estimated to result in savings of EUR 4.3 million from the payer perspective over 5 years. Indeed, savings were evident from Year 1 onwards. At an estimated market share of 20% relative to VA-ECMO in Year 1, total annual savings were estimated at EUR 375,600, which increased steadily to EUR 1.3 million per year in Year 5, when the utilization of Impella 5.0 relative to VA-ECMO was estimated at 70%. On a per patient level, the introduction of Impella 5.0 was projected to lead to a cost-saving of EUR 616 per patient in Year 1 and up to EUR 2155 per patient in Year 5 relative to current clinical practice utilizing VA-ECMO alone (Table 4).
 |
Table 4 Budget Impact Associated with the Introduction of Impella 5.0® in France |
Scenario analyses showed that savings were largely driven by the lower incidence of device-related complication costs with Impella 5.0 compared with VA-ECMO (Table 5). The management of complication costs represented a substantial component of the total overall cost of care for patients with left-ventricular refractory CS. In Scenario 1 (without Impella 5.0) total cumulative 5-year costs were EUR 28.6 million when complications were excluded but EUR 90.8 million when complication costs were accounted. In current clinical practice, without Impella 5.0, the cost of VA-ECMO-related complications was estimated at EUR 62.3 million (i.e. 69% of total costs). In Scenario 2, with Impella 5.0, total 5-year costs were projected to be EUR 46.8 million not including complications compared with EUR 86.5 million when complications were included. These results indicate that the use and funding of Impella 5.0 would result in a EUR 22.5 million reduction in direct costs related to complications over a period of 5 years. Thereby suggesting that with these market share predictions, the incremental cost associated with the Impella 5.0 device is entirely offset by savings resulting from the lower incidence of device-related complications with Impella 5.0. Moreover, faster and/or higher adoption rates of Impella 5.0 would be expected to further increase the cost savings to the French health system.
 |
Table 5 Summary Findings of Scenario Analyses |
Scenario analyses were also performed around market share and VA-ECMO-related consumable costs (Table 5). In the base case, as a conservative assumption no costs related to VA-ECMO-related consumables were accounted; however, in an analysis in which a cost of EUR 3000 was assumed for VA-ECMO-related consumables, total savings over the 5-year time horizon increased to EUR 8.5 million, representing a 97% increase in savings compared with the base case analysis.
One-way deterministic sensitivity analyses were performed around length of stay, target population size, treatment costs, complication costs and incidence of complications (Table 6). These showed that the results of the analysis were most sensitive to changes in assumptions around the length of stay for patients receiving VA-ECMO, the cost of the Impella 5.0 device and the proportion of Impella 5.0-treated patients receiving a long-term LVAD. Specifically, if the length of stay for VA-ECMO was decreased by 20% total savings over the 5-year period decreased to EUR 2.5 million, but if the length of stay for VA-ECMO was increased by 20% total savings increased notably to EUR 6.4 million. Results were also sensitive to the cost of Impella 5.0; here a decrease of 10% relative to the base case increased total savings to EUR 6.4 million over the 5-year time horizon.
 |
Table 6 Summary Findings of Deterministic Sensitivity Analyses |
Discussion
Overall, the findings of the budget impact analysis suggest that, from the perspective of the French national health insurer, the use of Impella 5.0 for the treatment of patients with left-ventricular refractory CS requiring temporary MCS is likely to result in total savings of EUR 4.3 million over a period of 5 years compared with current clinical practice. The projected savings associated with the introduction of Impella 5.0 were driven primarily by the lower incidence of device-related complications and shorter length of stay with Impella 5.0 relative to VA-ECMO. Indeed, deterministic sensitivity analyses showed that the overall findings were highly sensitive to assumptions relating to the length of stay with VA-ECMO. Additionally, a recent analysis in temporal trends of CS in the Paris area showed that the proportion of ICU patients with CS increased almost two-fold over the period 1997–2012.23 Consequently, the higher number of patients treated for CS makes both the clinical and economic outcomes associated with different management options increasingly important to both physicians and payers in France.
It is important to note that as the payer perspective of the analysis was that of mandatory national health insurance, intra-DRG costs were not included in the budget impact calculation. Intra-DRG costs included costs associated with consumables related to VA-ECMO as well as treatment costs for device-related vascular access site infection (although costs of VA-ECMO-related consumables were captured in sensitivity analyses). In particular, Karami et al reported that the proportion of patients requiring labile blood products as well as the proportion of patients requiring an additional IABP was significantly higher for VA-ECMO than Impella 5.014 but the costs associated with this resource use were not captured in this analysis. This perspective may represent a conservative picture of the overall costs associated with the management of patients with CS, particularly those in whom VA-ECMO is used, given the extensive medical resource use required for VA-ECMO. In one US-based analysis in CS, patients managed with ECMO had mean total hospital charges of USD 580,066 per patient, compared with USD 156,437 per patient without ECMO.24 The magnitude of the management costs for patients with CS when all direct costs are captured therefore underscores the cost-saving potential associated with the introduction of Impella 5.0 into clinical practice in this patient population.
In the present analysis, the projected incidence of device-related complications was largely based on the findings of a retrospective two-center study in the Netherlands in a total of 128 patients with CS following acute MI. Here, Karami et al reported that the overall incidence of device-related complications (defined as limb ischemia, access site infection or access site bleeding) was 17% with Impella 5.0 compared with 40% with VA-ECMO (p<0.01).14 In particular, the incidence of access site-related infection was 1.1% for Impella 5.0 compared with 15.8% for VA-ECMO.14 Additionally, in their analysis, Karami et al also report a significantly shorter median duration of time spent on device support (3 days versus 6 days; p<0.01) as well as significantly shorter median length of stay in the ICU with Impella 5.0 than with VA-ECMO (6 days versus 16 days; p<0.01), although part of this may be related to the lower incidence of device-related complications.14 A limitation of the use of clinical input data from the analysis by Karami et al is the fact that the study design was that of a retrospective observational analysis rather than an RCT. However, there is general paucity of RCT data in CS, which may be partly due to the logistical complexities of conducting RCTs in critically ill patients. It should also be noted that observational data may also be more representative of the situation in routine clinical practice than RCTs. Further, the findings from other observational studies concur with the findings of Karami et al. For example, in one retrospective analysis of patients with CS following STEMI or post-cardiotomy shock, Impella 5.0 was associated with a significantly lower use of blood products as well as a significantly lower incidence of arterial thromboembolism compared with VA-ECMO.25
The scope of the analysis presented here is limited solely to the budget impact associated with the introduction of Impella 5.0 to clinical practice in France and does not capture other aspects of value to clinicians such as ease of use. Although conversely, this analysis also does not capture any increased time or resource use that may be associated with a learning curve for physicians using devices for the first time. Additionally, the analysis did not capture value associated with factors such as improved mobility levels and rehabilitation, particularly as mobility may be an important prognostic indicator for patients with CS.26,27 Previous investigators have highlighted the ease of use of Impella 5.028 as well as conversely the logistical challenges, complexity and high levels of medical resource use associated with the use of VA-ECMO.14,24 Additionally, when interpreting the findings of the analysis presented here it is important to note that although the use of Impella 5.0 may be associated with cost savings there are instances where the use of VA-ECMO may represent a more appropriate intervention. For example, VA-ECMO is the preferred treatment modality for patients with refractory cardiac arrest, multiorgan failure or respiratory failure.7 Allied to this, there are key differences in the underlying mechanical aspects of Impella 5.0 and VA-ECMO that may also influence clinical decision making. For example, for some patients with CS, a key advantage associated with the use of Impella 5.0 is the direct unloading of the left ventricle. In contrast, VA-ECMO provides circulatory support and blood oxygenation without unloading the left ventricle. This can lead to elevated left ventricular pressure, which can in turn result in myocardial wall stress, delayed ventricular recovery and potentially even pulmonary venous hypertension and pulmonary edema.29 A key downstream effect of the direct unloading of the left ventricle is decreased myocardial oxygen consumption, which in turn creates a setting more conducive to myocardial recovery. In the setting of acute MI decreased myocardial oxygen consumption has been linked to reduced infarct size,30 with infarct size in turn being a key determinant of outcomes including mortality, reinfarction and heart failure.31 Indeed, ventricular unloading has recently emerged as a key strategy for preventing ischemia-reperfusion injury, facilitating myocardial recovery and reducing infarct size in the setting of MI.
As with any economic analysis, the analysis presented here is associated with a number of limitations. In particular, as noted, the clinical input data used in the model were sourced from a combination of expert opinion and observational studies rather than RCTs. However, as noted RCT data comparing Impella 5.0 and VA-ECMO are currently lacking. The analysis was also performed from the perspective of the national health insurer and therefore does not capture all the cost components associated with the management of patients with CS, the omission of costs such as those associated with the use of additional IABPs or labile products, which was higher with ECMO than with Impella 5.0, may therefore present a conservative picture in terms of the overall patient management costs. Further, a simplifying assumption used in the model was that all simulated patients were identical. However, in reality, the patient population is likely to be heterogeneous. For example, some patients may have a greater degree of impairment with regard to renal and hepatic function, the cost implications of which were not considered in the model. Additionally, the comparison is limited to Impella 5.0 and does not consider the use of other temporary MCS devices, nor the utilization of VA-ECMO and Impella 5.0 in the same patient, which has been documented in clinical practice.29,32 Potential differences between settings such as placement in a catheter laboratory versus operating room (e.g. in terms of medical resource use) were also not considered in the analysis; however, this would not have influenced the results of the analysis when considered from the payer (health insurance) perspective. The generalizability of the findings is also limited; these findings are specific to the French setting and the care pathway used in the model. Differences in resource use, direct medical costs and local treatment pathways should be taken into account when applying the findings of the current analysis to different settings.
In conclusion, the findings of this budget impact analysis suggest that in France, the introduction of Impella 5.0 for use in patients with left ventricular refractory CS is likely to be associated with cost savings when compared with current clinical practice.
Funding
This study was supported by funding from SGE Consulting and Abiomed.
Disclosure
IF and SR are current employees of consultancy companies that have received consulting fees from Abiomed. MP has received financial support from Abiomed for teaching programs. GL has received financial support from Abiomed for a research program and reports proctoring (HVAD) from Medtronic. CD has received honoraria from Abiomed, Abbott, AstraZeneca and Boston for consulting or speaker fees. The authors report no other conflicts of interest in this work.
References
1. Van Diepen S, Katz JN, Albert NM, et al., American Heart Association Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; Council on Quality of Care and Outcomes Research; and Mission: Lifeline. Contemporary management of cardiogenic shock: a scientific statement from the American Heart Association. Circulation. 2017;136(16):e232–e268. doi:10.1161/CIR.0000000000000525
2. Ponikowski P, Voors AA, Anker SD, et al., ESC Scientific Document Group. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37(27):2129–2200. doi:10.1093/eurheartj/ehw128
3. Thiele H, Ohman EM, Desch S, Eitel I, de Waha S. Management of cardiogenic shock. Eur Heart J. 2015;36(20):1223–1230. doi:10.1093/eurheartj/ehv051
4. Kolte D, Khera S, Aronow WS, et al. Trends in incidence, management, and outcomes of cardiogenic shock complicating ST-elevation myocardial infarction in the United States. J Am Heart Assoc. 2014;3(1):e000590. doi:10.1161/JAHA.113.000590
5. Ollivier J, Fresselinat A, Seguy B, et al. The use of Impella 5.0 in refractory cardiogenic shock: a medico-economic report. Pharm J Med Devices. 2019;1. Available from: https://www.euro-pharmat.com/la-revue-du-dm. Accessed March 17, 2020.
6. van Diepen S, Thiele H. An overview of international cardiogenic shock guidelines and application in clinical practice. Curr Opin Crit Care. 2019;25(4):365–370. doi:10.1097/MCC.0000000000000624
7. Bonello L, Delmas C, Schurtz G, et al. Mechanical circulatory support in patients with cardiogenic shock in intensive care units: a position paper of the “Unité de Soins Intensifs de Cardiologie” group of the French Society of Cardiology, endorsed by the “Groupe Athérome et Cardiologie Interventionnelle” of the French Society of Cardiology. Arch Cardiovasc Dis. 2018;111(10):601–612. doi:10.1016/j.acvd.2018.03.008
8. Gaudard P, Mourad M, Eliet J, et al. Management and outcome of patients supported with Impella 5.0 for refractory cardiogenic shock. Crit Care. 2015;19:363. doi:10.1186/s13054-015-1073-8
9. Seese L, Hickey G, Keebler ME, et al. Direct bridging to cardiac transplantation with the surgically implanted Impella 5.0 device. Clin Transplant. 2020;34(3):e13818. doi:10.1111/ctr.13818
10. Lemaire A, Anderson MB, Lee LY, et al. The Impella device for acute mechanical circulatory support in patients in cardiogenic shock. Ann Thorac Surg. 2014;97(1):133–138. doi:10.1016/j.athoracsur.2013.07.053
11. Batsides G, Massaro J, Cheung A, Soltesz E, Ramzy D, Anderson MB. Outcomes of Impella 5.0 in cardiogenic shock: a systematic review and meta-analysis. Innovations (Phila). 2018;13(4):254–260. doi:10.1097/IMI.0000000000000535
12. Glazier JJ, Kaki A. The Impella device: historical background, clinical applications and future directions. Int J Angiol. 2019;28(2):118–123. doi:10.1055/s-0038-1676369
13. Bonello L, Delmas C, Gaubert M, Schurtz G, Ouattara A, Roubille F. Trials of mechanical circulatory support with percutaneous axial flow pumps in cardiogenic shock complicating acute myocardial infarction: mission impossible? Arch Cardiovasc Dis. 2020;113(6–7):448–460. doi:10.1016/j.acvd.2020.02.001
14. Karami M, den Uil CA, Ouweneel DM, et al. Mechanical circulatory support in cardiogenic shock from acute myocardial infarction: Impella CP/5.0 versus ECMO. Eur Heart J Acute Cardiovasc Care. 2019;2048872619865891. doi:10.1177/2048872619865891
15. Chevreul K, Berg Brigham K, Durand-Zaleski I, Hernandez-Quevedo C. France: health system review. Health Syst Transit. 2015;17(3):1–21.
16. Haute Autorité de Santé. Impella 5.0 March 2020. Available from: https://www.has-sante.fr/upload/docs/evamed/CNEDIMTS-5865_IMPELLA%205%200_24_mars_2020_(5865)_avis.pdf.
17. Cheng R, Hachamovitch R, Kittleson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: a meta-analysis of 1866 adult patients. Ann Thorac Surg. 2014;97(2):610–616.
18. Agence Technique de l’Information sur l’Hospitalisation. Guide d’utilisation: valorisation de l’activité 2018 par les tariffs 2018 et par les tariffs 2019. Available from: https://www.atih.sante.fr/sites/default/files/public/content/3592/guide_dutilisation_-_valorisation_de_lactivite_2018_par_les_tarifs_2018_et_tarifs_2019.pdf.
19. Halimi S, Benhamou PY, Charras H. Le cout du pied diabetique. Available from: https://www.fmc-tourcoing.org/PROGRAMME_FMC/DIABETE_10_00/DIABETEFMI/cout-pied-3.html.
20. Liste des Produits et Prestations Remboursables. Available from: https://www.ameli.fr/sites/default/files/Documents/615994/document/lpp.pdf.
21. Ghabri S, Autin E, Poullié AI, Josselin JM. The French National Authority for Health (HAS) guidelines for conducting Budget Impact Analyses (BIA). Pharmacoeconomics. 2018;36(4):407–417. doi:10.1007/s40273-017-0602-5
22. Sullivan SD, Mauskopf JA, Augustovski F, et al. Budget impact analysis-principles of good practice: report of the ISPOR 2012 budget impact analysis good practice II task force. Value Health. 2014;17(1):5–14. doi:10.1016/j.jval.2013.08.2291
23. Puymirat E, Fagon JY, Aegerter P, et al; Collège des Utilisateurs de Bases de données en Réanimation (CUB-Réa Group [Intensive Care Database User Group]). Cardiogenic shock in intensive care units: evolution of prevalence, patient profile, management and outcomes, 1997–2012. Eur J Heart Fail. 2017;19(2):192–200. doi:10.1002/ejhf.646
24. El Sibai R, Bachir R, El Sayed M. Outcomes in cardiogenic shock patients with extracorporeal membrane oxygenation use: a matched cohort study in hospitals across the United States. Biomed Res Int. 2018;2018:2428648.
25. Lamarche Y, Cheung A, Ignaszewski A, et al. Comparative outcomes in cardiogenic shock patients managed with Impella microaxial pump or extracorporeal life support. J Thorac Cardiovasc Surg. 2011;142(1):60–65. doi:10.1016/j.jtcvs.2010.07.075
26. Esposito ML, Jablonski J, Kras A, Krasney S, Kapur NK. Maximum level of mobility with axillary deployment of the Impella 5.0 is associated with improved survival. Int J Artif Organs. 2018;41(4):236–239. doi:10.1177/0391398817752575
27. Sassard T, Scalabre A, Bonnefoy E, Sanchez I, Farhat F, Jegaden O. The right axillary artery approach for the Impella recover LP 5.0 microaxial pump. Ann Thorac Surg. 2008;85(4):1468. doi:10.1016/j.athoracsur.2007.09.017
28. Griffith BP, Anderson MB, Samuels LE, Pae WE
29. Moazzami K, Dolmatova EV, Cocke TP, et al. Left ventricular mechanical support with the Impella during extracorporeal membrane oxygenation. J Tehran Heart Cent. 2017;12(1):11–14.
30. Curran J, Burkhoff D, Kloner RA. Beyond reperfusion: acute ventricular unloading and cardioprotection during myocardial infarction. J Cardiovasc Transl Res. 2019;12(2):95–106. doi:10.1007/s12265-019-9863-z
31. Stone GW, Selker HP, Thiele H, et al. Relationship between infarct size and outcomes following primary PCI: patient-level analysis from 10 randomized trials. J Am Coll Cardiol. 2016;67(14):1674–1683. doi:10.1016/j.jacc.2016.01.069
32. Schrage B, Becher PM, Bernhardt A, et al. Left ventricular unloading is associated with lower mortality in patients with cardiogenic shock treated with venoarterial extracorporeal membrane oxygenation: results from an international, Multicenter Cohort Study. Circulation. 2020;142(22):2095–2106. doi:10.1161/CIRCULATIONAHA.120.048792
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.