Back to Journals » Journal of Inflammation Research » Volume 14
Association of Early Inflammation with Age and Asymptomatic Disease in COVID-19
Authors Xie C, Li Q, Li L, Peng X, Ling Z, Xiao B, Feng J, Chen Z, Chang D , Xie L, Dela Cruz CS, Sharma L
Received 29 January 2021
Accepted for publication 11 March 2021
Published 30 March 2021 Volume 2021:14 Pages 1207—1216
DOI https://doi.org/10.2147/JIR.S304190
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Chunmei Xie,1,* Qing Li,1,* Linhai Li,2,3,* Xiaohua Peng,4,5,* Zhijian Ling,1 Bin Xiao,3 Jingjing Feng,4,6 Zhenhong Chen,7 De Chang,2,8 Lixin Xie,2 Charles S Dela Cruz,4 Lokesh Sharma4
1Department of Blood Transfusion, Guangzhou 8th People’s Hospital, Guangzhou Medical University, Guangzhou, 510440, People’s Republic of China; 2College of Pulmonary and Critical Care Medicine, Chinese PLA General Hospital, Beijing, 100083, People’s Republic of China; 3Department of Laboratory Medicine, General Hospital of Southern Theatre Command of PLA, Guangzhou, 510010, People’s Republic of China; 4Section of Pulmonary and Critical Care and Sleep Medicine, Department of Medicine, Yale University School of Medicine, New Haven, CT, 06520, USA; 5Department of Rehabilitation Medicine, The First Affiliated Hospital of Chongqing Medical University, Chongqing, 400016, People’s Republic of China; 6Department of Pulmonary and Critical Care Medicine, Shanghai Fifth People’s Hospital, Fudan University, Shanghai, 200240, People’s Republic of China; 7Department of Pulmonary and Critical Care Medicine, Secondary Medical Center, Chinese PLA General Hospital, Beijing, 100083, People’s Republic of China; 8 Department of Pulmonary and Critical Care Medicine, Third Medical Center, Chinese PLA General Hospital, Beijing, 100039, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lixin Xie; De Chang Email [email protected]; [email protected]
Background: Disease severity in COVID-19 ranges from asymptomatic infection to severe disease and death, especially in older subjects. The risk for severe infection and death has been reported to be 2X in those between 30 and 40 years, 3X in those between 40 and 50 years, and 4X in those between 50 and 65 years, compared to the reference group of 18– 29 years.
Objective: To investigate the early changes in host immune responses that are altered with age and the difference in the early host inflammatory response that dictates a symptomatic versus asymptomatic course of COVID-19.
Patients and Methods: COVID-19 subjects were identified by screening at the airport upon arrival from a foreign destination to China. Patients were either asymptomatic or had a mild disease when the first oro-pharyngeal (OP) swab samples were collected. Patients were quarantined and blood and throat swabs were collected during the course of the disease, allowing identification of the earliest host response to COVID-19. These patients were followed until their OP sample turned COVID-19 negative.
Results: Data were obtained from 126 PCR-confirmed COVID-19 patients. The blood samples were obtained within 48 days of qPCR confirmation of viral infection. Older subjects (> 30 years) had significantly elevated levels of anti-inflammatory cytokine IL-10, a significant decrease in the percentage of CD8+ T cells, and expansion in NKT cell fraction. This was associated with significantly elevated viral load and a delayed humoral response in older subjects. Compared to symptomatic subjects, asymptomatic patients had an early increase in pro-inflammatory cytokine IL-2, while a decrease in both T regulatory cells and anti-inflammatory cytokine IL-10. Further, asymptomatic disease was associated with early humoral response and faster viral clearance.
Conclusion: Early inflammatory response potentially plays a critical role for host-defense in COVID-19. The impaired early inflammatory response was associated with older age while a robust early inflammation was associated with asymptomatic disease.
Keywords: COVID-19, SARS-CoV-2, early inflammation, asymptomatic patient, elder patient
Introduction
COVID-19 pandemic has infected approximately 114 million people with deaths of over 2.5 million people worldwide.1 COVID-19 has especially affected older individuals where the majority of morbidity and mortality were observed in individuals with advanced age,2 while on the other end of the age spectrum, children are largely protected from severe disease. As per the Centers for Disease Control (CDC), the risk of hospitalization and death by COVID-19 is 2X higher in the age group of 30–39 years, 3X times higher in the age group of 40–49 years, and 4X times higher in the age group of 50–64 years when compared to the age group of 18–29 years.3 Currently, it remains unclear what drives the increased susceptibility to the COVID-19 in patients with older age. Many factors such as cytokine storm,4,5 impaired T cell function,6 coagulopathy,7,8 and metabolic disruption9 have been associated with the disease. However, these conclusions are mostly derived from comparing different disease severity, ie mild vs severe disease, where younger people are highly presented in the mild group while the older population presents the majority of the severe patients. It remains unclear if these differences are rather a consequence of the disease severity or the cause, in COVID-19. A multitude of factors, such as existing of multiple co-morbidities, may contribute to the excessive morbidity and mortality in patients with advanced age. However, it is difficult to distinguish age-related changes independent of co-existing morbidities, especially in those older than 65 years where multiple co-morbidities are often present.
The role of inflammation and inflammatory cytokines in mediating the pathogenesis of COVID-19 remains controversial. While some studies have reported cytokine storm as a potential cause of death in COVID-19,10 other studies have shown a lack of elevation of many inflammatory cytokines, even in most critical patients from the baseline.11 Further, it remains unclear how subjects that are at the elevated risk of disease have their initial inflammatory response to COVID-19 that may determine the overall disease course.
Another interesting aspect of the COVID-19 biology that remains incompletely understood is the range of clinical presentation ranging from asymptomatic infection to severe disease and death. Whether the initial immune response is different between those who develop symptoms and those who remain asymptomatic remains to be known.
In this study, we used early samples from a unique population of relatively healthy patients who were screened at the airport upon arrival from a foreign country to China and were mostly asymptomatic or had a mild disease at the time of screening. Many of these patients remained asymptomatic while others developed symptoms that were classified as mild/moderate during the disease course. Using these samples, we retrospectively asked two specific questions: 1. Whether early inflammatory or immune response is different between those at higher risk for COVID-19 (>30 years)? and 2. Whether asymptomatic subjects have a differential early immune response compared to those who eventually develop symptoms?
Our data reveal that older subjects (as defined by >30 years) have increased levels of anti-inflammatory cytokine IL-10, along with the high fraction of NKT cells, while a lower percentage of CD8+ T cells. These early changes translated into a delayed onset on antibody response measured as both IgG and IgM response and increased viral loads in older patients. Analysis of symptomatic and asymptomatic subjects revealed that patients who failed to develop a robust early inflammatory response measured as pro-inflammatory cytokines or had elevated levels of regulatory T cells had delayed antibody response and eventually developed disease symptoms. These data provide novel insight into the initial age-dependent host response and factors that contribute to the symptom onset in COVID-19.
Methods
Source of the Data
Data were retrospectively obtained from 126 patients admitted at Guangzhou Eighth People’s Hospital from March 15, 2020, to April 26, 2020, upon their arrival from a foreign destination to Guangzhou, China. The viral qPCR was performed in the hospital laboratory where the presence of SARS-CoV-2 was confirmed during their admission and the viral tests were performed every other day during their stay in the hospital. All the patients were discharged from the hospital once their symptoms were resolved and they had two consecutive negative viral qPCR on samples collected at least 24 hours apart.
Case Definitions
All the patients in this study were confirmed to have SARS-CoV-2 viral infection, detected by viral qPCR assay. The disease severity was classified as severe and non-severe based on the clinical parameters as described before.12 Data were obtained as part of standard clinical care. This study was approved by the ethics committee of Guangzhou Eighth People’s Hospital and written consent was obtained from each of the study participants. Patients were divided into two groups based on their age. The young group included patients younger than 30 years which pose a relatively low risk of hospitalization and death due to COVID-19. The second group included older patients which included patients with an age of 30 years or older representing a high-risk group.
Laboratory Tests
Routine measurements and laboratory tests were performed in the hospital, including markers of inflammation, such as cytokines, cell differentials, and IgM/IgG antibody tests as described below:
Serum Collection
The whole blood was centrifuged at 3000 rpm for 10 minutes at 4°C. The serum was harvested and subjected to subsequent analysis.
Measurement of Serum Cytokines
The serum cytokines were quantified using the Human Th1/Th2 Cytokine Kit (Guangzhou WeimiBio-Tech, China) following the manufacturer’s manual. This kit contains six distinct microspheres packaged with IL-2, IL-4, IL-6, IL-10, TNFa and INFg specific antibodies. Microspheres precoated with specific antibodies were mixed with the samples or standards provided with the kit and the fluorescent-labeled antibodies are added to the mixture. The levels of cytokines were determined by plugging the sample intensity with the predefined standards.
Flow Cytometry
To quantify the lymphocytes and T cell subsets, flow cytometry was performed using BD Multitest 6-Color TBNK Reagent kit. 100 µL of whole blood from patients was incubated in 900 µL of Tris-NH4Cl buffer at room temperature for 5 minutes to lyse erythrocytes. Then, the samples were washed twice with phosphate-buffered saline (PBS) and incubated with the antibodies in the kit following the manufacturer’s manual. All samples were tested within 6 hours of being obtained. The gating strategy is defined in Supplementary Figure 1. T cells were defined by CD3+, which were either CD4 or CD8+. B cells were defined as CD3- and CD19 + ve. CD16+ and CD56+ cells were defined as NK cells and the presence of CD3 along these markers defined NKT cells.
Antibody Testing
Antibody testing was performed using a diagnostic Kit for IgM/IgG antibody to coronavirus (SARS-Cov-2) Lateral Flow (Livzon, Zhuhai, China, http://www.livzondiagnostics.com/en-us/info/17.html) according to the manufacturer’s protocol. The results were obtained as positive or negative.
Clinical Baseline Standards
Baseline levels of cytokine were determined as per clinical standards (Ref) are as follows: IL-6≤19.6pg/mL, TNFa≤4.9 pg/mL, IL-10≤6.4pg/mL, IL-2≤11pg/mL, IL-4≤12.1pg/mL.
Statistical Analysis
Data were analyzed from 126 patients. The data between the two groups were compared using the chi-square test (for trend) or Fisher’s exact test for categorical variables or Student’s t-test for continuous variables. The relationship between variables was analyzed by linear regression. P≤0.05 was considered statistically significant. Data were analyzed using GraphPad Prism (version 8.4.2) and Stata/PM 16.1.
Results
Patient Population
The basic characteristics of patients are depicted in Table 1. These patients arrived from a foreign destination to Guangzhou Baiyun International Airport, China between March 15, 2020, and April 26, 2020, and were screened for COVID-19 in Guangzhou Eighth People’s Hospital upon their arrival. Most of these patients were asymptomatic at the time of their testing and largely remained mild/moderate disease with only 7 of 126 patients falling in the severe category during the course of the disease where all but one of the severe patients were in the older age group (≥30 years).
 |
Table 1 Demographics and Clinical Characteristics of the Patient Population |
Increased Levels of Anti-Inflammatory Cytokines in the Older Patients
Inflammatory response has been associated with the disease severity in COVID-19, however, the data remains inconsistent.13,14 Some of the studies have shown that the upregulation of certain cytokines in the severe disease, while other cytokines, such as tumor necrosis factor α, IL-1β, and IL-12p70, are not elevated from the normal range even in the most severe patients.11 Also, the kinetics of cytokine response remains critically important as an early cytokine response may be essential to promote the viral clearance and early resolution of inflammation. Here we found that at presentation, cytokine levels including IL-2, IL-4, IL-6, TNFα, and IFNγ did not change with age, where only IL-6 showed a trend towards an increase in older subjects, however, it did not reach statistical significance (Figure 1A–F). In contrast, the levels of anti-inflammatory cytokine IL-10 were significantly elevated in the older population compared to young patients. We also evaluated whether this elevation was mainly contributed by a smaller number of patients who developed severe disease. The elevated trend in IL-10 became even stronger after the removal of severe patients (P = 0.0093), while the trend in IL-6 was diminished after the removal of severe patients (Supplementary Table 1). These data suggest that early response to COVID-19 in older patients is marked by elevated anti-inflammatory cytokine response, which may indicate an impaired early host response to the virus.
Altered Peripheral Cellular Composition in Older Subjects
Next, we sought to determine whether there are age-associated changes in the cellular composition in COVID-19 patients. Lymphocytopenia is almost universally reported as a marker of the disease severity in COVID-19, where both the depletion of CD4+ and CD8+ cells are reported and associated with the disease severity.15,16 At the same time, high neutrophil or monocytes counts are reported in severe cases.17–19 Here we first investigated whether there are any changes in the cellular composition of peripheral blood in an age-dependent manner in the COVID-19 patient population at earlier time points. Our data revealed that there are no age-dependent differences in total WBC counts, neutrophils, lymphocytes, or eosinophils among the two patient groups (Figure 2B–F). However, an elevation in the RBC (Figure 2A) and monocyte counts (Figure 2E) was observed in the older population, compared to the younger patients. In the lymphocytic compartment, no difference was observed in total lymphocyte counts between young and older subjects, including in T cells, B cells, or NK cells (Figure 2G, H, J, K, M and N). However, a significant increase was observed in the NKT cell population (Figure 2I) while a decrease was noted in the CD8+ T cell percentage (Figure 2L). Similar trends were observed in absolute cell counts for the majority of cell populations (Supplementary Figure 2). These data suggest that while lymphocytopenia may be a marker of disease severity in COVID-19 and an outcome of severe disease, an early decrease in CD8+ T cells in older subjects may be a contributing factor to the disease severity.
Impaired Humoral Response and Viral Clearance in Older Patients
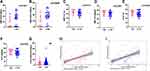
Next, we wanted to understand whether altered cellular composition affects the host’s ability to mount a humoral response. We measured the duration of onset of IgM and IgG antibodies in these patients. Our data reveal that both the IgM and IgG onsets were delayed in the older subjects compared to the young patients (Figure 3A and B). As the humoral response is a critical host antiviral response, we measured the viral load between these two groups.
We measured the viral load in nasopharyngeal cavities by qPCR of either ORF1 or N gene of SARS-CoV-2. We analyzed the Ct values of these patients at the presentation (Figure 3C and E) as well as when Ct values were lowest (highest viral load) during the course of their disease (Figure 3D and F). Our data show a significant increase in the viral load in older patients when measured at the presentation or at the time of the highest viral load in these patients using the ORF-1 gene. Similar trends were observed in the N gene. However, no differences were observed in the duration of viral persistence between the two groups (Figure 3G). To assess whether the onset of antibodies was related to viral persistence, we investigated a correlation between the duration of viral persistence and the duration of antibody onset. Our data show that viral persistence was positively correlated with both the IgM (Figure 3H) and IgG (Figure 3I) onset time, suggesting that a delayed humoral response may contribute to viral persistence. These data suggest that even in the absence of any severe disease, age-associated changes in the host defense are sufficient to impair the viral clearance in COVID-19.
A Robust Inflammatory Response is Associated with Asymptomatic Infection
Finally, we decided to determine if there was a difference in cytokine levels or peripheral cellular immune composition at the earliest samples in patients who remained symptomatic or those who later developed disease symptoms. Our data show that patients who remained asymptomatic had a robust early inflammatory response marked by significantly elevated levels of IL-2 (Figure 4A) and a decreased IL-10 response (Figure 4B). There were no major differences observed in IL-4 and IL-6 between these two groups (Figure 4C and D). This increased inflammatory response in asymptomatic patients was associated with a decreased level of T regulatory cells (Figure 4E). Besides, the asymptomatic patients had a quicker humoral response measured as both IgM and IgG (Figure 4F and G). As expected, the early humoral response was associated with a shorter viral shedding duration in asymptomatic patients compared to those who developed symptoms (Figure 4H). These data suggest that an early robust inflammatory response may facilitate the humoral response leading to asymptomatic infection.
Discussion
COVID-19 continues to cause morbidity and mortality worldwide with no sign of slowing down. Despite extensive studies in the last nine months, many aspects of the COVID-19 disease remain to be known. Older age is one of the most significant risk factors for this disease and a major proportion of the COVID-19 related deaths occurred in the older age population.20 Although the highest rate of mortality is reported in patients >65 years of age, the age-dependent risk of increased disease severity and death starts as early as 30 years.3 Currently, there is a lack of precise understanding of differences in the early host response in older subjects that may lead to increased disease severity in these patients.
Using this unique cohort, where patients were identified at early time points post-COVID-19 and without the severe disease, we demonstrate that various age-associated factors in host immunity are already evident and potentially contribute toward the impaired host immunity in older subjects. Our data reveal that before the manifestation of lymphocytopenia, a decrease specifically in CD8+ T cells is evident in older patients during early disease (Figure 2L). Age-associated decrease in the CD8+ fraction has been reported previously.21,22 This depletion likely puts the older patients at a disadvantage when infected with SARS-CoV-2, leading to an increased risk of severe disease in older populations.
Although the increase in inflammatory cytokines has been reported during the severe disease in COVID-19, it remains unclear what causes a higher inflammatory response in severe patients. It is possible that the patients with advanced age are more immunogenic to the viral PAMPS or are unable to mediate a strong antiviral response, which leads to increased viral burden and damage, which in turn may induce a damage-associated molecular pattern mediated inflammation. Our data show that in the absence of severe disease, the only cytokine that was significantly elevated in older subjects was IL-10 (Figure 1D). A trend was observed in IL-6 levels but partly was driven more by severe patients as removal of severe patients from the analysis diluted the IL-6 trend but not the IL-10 findings (Supplementary Table 1). These data strongly indicate that instead of older patients being hyper-inflammatory, a subdued early inflammation may be a contributing factor towards increased disease severity in older subjects. Our data is supported by a recent analysis demonstrating severe immune suppression and functional impairment in the lymphocytes as a potential cause of death rather than uncontrolled inflammation.23
We observed an increase in the viral load in the older subjects compared to younger patients, measured as the first test and the highest viral load observed in these individuals (Figure 3C–F). The correlation of viral load and disease severity has been controversial, given some studies did not find a significant positive correlation between viral titers and the disease severity24 while others reported higher viral load in severe patients.25 However, these viral loads are often measured in mild to severe cases, which presents technical difficulties in assessing the true viral titers in the host. While the mild disease is often limited to the upper respiratory tract, the severe disease often manifests when the viral infection progresses to the lower lung. The current sampling methods only involve assessing the viral load in the upper respiratory tract by nasopharyngeal or oropharyngeal swabs. It remains technically difficult to assess the lower lung, especially the part of the lung which is actively infected. In our population, viral load measurements may reflect the true viral load patterns between younger and older patients, given all of them had mild disease.26
The increased viral load in older patients may be multifactorial. Various markers of innate and adaptive immunity appeared impaired in older subjects. The innate immunity arm showed increased IL-10 response, which has been shown to impair the antiviral function of T cells and lead to viral persistence.27 At the same time, we observed a striking increase in the NKT cell population (Figure 2I), a finding previously unreported in COVID-19 patients. The precise contributions of NKT cells in the older population are not clear, however, the contribution of NKT cells in producing both pro- and anti-inflammatory cytokine production has been reported.28 A direct role of NKT cells in contributing to the altered inflammatory milieu was not explored in our study.
In addition to impaired inflammatory cytokines, older patients also demonstrated a delayed antibody response, both in IgM and IgG (Figure 3A and B). A delayed antibody response is associated with severe disease.29 Overall the duration of antibody onset positively correlated with the viral persistence for both IgM and IgG, suggesting an important role of humoral response on viral clearance (Figure 3H and I).
When comparing the patients who developed symptoms versus those who remained asymptomatic, we gained novel insights into COVID-19 pathogenesis. Our data show that patients who mount a robust initial inflammatory response while maintaining the anti-inflammatory cytokines low remain asymptomatic (Figure 4A–D). The cellular basis of high inflammatory cytokines, such as IL-2 and lower anti-inflammatory cytokine IL-10, is not clear but may be contributed by T regulatory cells (Figure 4E). Our data further show that asymptomatic patients have an early humoral response (Figure 4F and G), which may contribute to eliminating the virus early. Although we did not find a significant difference in the viral titers between asymptomatic and symptomatic patients (Supplementary Figure 3A–D), symptomatic patients had significantly prolonged viral persistence (Figure 4H). Although the precise mechanism of the asymptotic disease remains unknown, but few theories have been postulated. Shankar et al hypothesized that the potential tolerance of innate immunity to the viral components may protect the animals from the disease while they harbor high levels of viral load,30 we hypothesized that in humans, a potential robust early inflammation can help eradicate the virus faster and prevent a severe disease.31 However, it is possible that a during a severe disease, excessive inflammation is detrimental to the host,32 which is supported by the fact that dexamethasone provides therapeutic benefits during a severe disease.33
Although female gender has been associated with protection against severe and deadly disease,34 we did not observe any sex differences in cytokines and cell components (data not shown), potentially due to milder disease and younger patient population. Further, increased co-morbidities are believed to be a key contributor to severe disease in men,34,35 our population had very limited co-morbidities, potentially explaining lack of differences.
Interestingly, we observed age-dependent differences in our patients despite none of them being >65 years, confirming the notion that age-related decline in host defense against COVID-19 starts earlier. Although a similar early response in older patients (>65 years) can shed further light on host ability to respond to SARS-CoV-2 in this highly vulnerable population, it may be compounded by their multiple co-morbidities often present in advanced age patients. In conclusion, our study suggests that the early defects in host immunity due to age and may render the older subjects at higher risk of disease severity in COVID-19. In addition, the impaired early inflammatory and humoral responses potentially contribute to the symptom onset. Together these data suggest an essential role of an early inflammatory response and potential deleterious effects of anti-inflammatory cytokines in impairing viral control. To the best of our knowledge, this is the first study to demonstrate early inflammatory response as a potential regulator of viral clearance and symptom onset.
Limitations
Our study has few limitations that are worth discussing here. First, as this is an observational study, it remains to be decided if the early robust inflammatory response can prevent a symptomatic or severe disease in prospective studies. Secondly, most of our subjects in the study population were young and had a mild disease, warranting further investigation into the role of the early inflammatory response in the most vulnerable population such as older subjects during the severe disease. Thirdly, our study population included only one race (Asians), which precludes generalizing our findings to other races. Finally, we lacked a healthy control group due to technical difficulty in recruiting such a cohort during the pandemic, thus we relied on standard clinical range prescribed as the normal range. However, despite these limitations, this study sheds important light on early host response during COVID-19 and its association with symptomatic disease, which is allowed by this unique setting of the study where we included asymptomatic patients screened for viral positivity at the airport.
Acknowledgments
This study was supported by funding from Beijing Nova Program Interdisciplinary Cooperation Project (DC; No. Z191100001119021), Chinese PLA General Hospital Youth Project (DC; No. QNF19074), and China 13th Five-year National Key Grant (LXX; No.2018ZX09201013). Lokesh Sharma is supported by Francis B Parker Foundation and American Lung Association Catalyst Award. Chunmei Xie, Qing Li, Linhai Li, and Xiaohua Peng are co-first authors for this study. Charles S Dela Cruz and Lokesh Sharma are senior authors for this study.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that there are no competing interests.
References
1. World Health Organization. COVID-19 weekly epidemiological update; 2021. Available from: https://www.who.int/publications/m/item/weekly-epidemiological-update—2-march-2021.
2. CDC. Older adults; 2020. Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/older-adults.html.
3. CDC. COVID-19 hospitalization and death by age; 2020. Available from: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-age.html.
4. Schwartz M, Emerson S, Punt J, Goff WJA. Decreased Naïve T-cell production leading to cytokine storm as cause of increased COVID-19 severity with comorbidities. Aging Dis. 2020;11(4):742–745. doi:10.14336/AD.2020.0619
5. Mehta P, McAuley D, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet (London, England). 2020;395(10229):1033–1034. doi:10.1016/S0140-6736(20)30628-0
6. Diao B, Wang C, Tan Y, et al. Reduction and functional exhaustion of T Cells in patients with coronavirus disease 2019 (COVID-19). Front Immunol. 2020;11:827.
7. Droesch C, Hoang M, DeSancho M, Lee E, Magro C, Harp J. Livedoid and Purpuric Skin Eruptions Associated With Coagulopathy in Severe COVID-19. JAMA Dermatol. 2020;156:1. doi:10.1001/jamadermatol.2020.2800
8. Nicolai L, Leunig A, Brambs S, et al. Immunothrombotic dysregulation in COVID-19 pneumonia is associated with respiratory failure and coagulopathy. Circulation. 2020.
9. Zhang J, Kong W, Xia P, et al. Impaired fasting glucose and diabetes are related to higher risks of complications and mortality among patients with coronavirus disease 2019. Front Endocrinol. 2020;11:525.
10. Chen G, Wu D, Guo W, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620–2629. doi:10.1172/JCI137244
11. Laterre P, François B, Collienne C, et al. Association of interleukin 7 immunotherapy with lymphocyte counts among patients with severe coronavirus disease 2019 (COVID-19). JAMA Network Open. 2020;3(7):e2016485. doi:10.1001/jamanetworkopen.2020.16485
12. Chang D, Zhao P, Zhang D, et al. Persistent viral presence determines the clinical course of the disease in COVID-19. J Allergy Clin Immunol. 2020.
13. Simon D, Tascilar K, Krönke G, et al. Patients with immune-mediated inflammatory diseases receiving cytokine inhibitors have low prevalence of SARS-CoV-2 seroconversion. Nat Commun. 2020;11(1):3774.
14. Carvelli J, Demaria O, Vély F, et al. Association of COVID-19 inflammation with activation of the C5a-C5aR1 axis. Nature. 2020.
15. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382(8):727–733. doi:10.1056/NEJMoa2001017
16. Chang D, Lin M, Wei L, et al. Epidemiologic and clinical characteristics of novel coronavirus infections involving 13 patients outside Wuhan, China. JAMA. 2020;323(11):1092–1093. doi:10.1001/jama.2020.1623
17. Laforge M, Elbim C, Frère C, et al. Tissue damage from neutrophil-induced oxidative stress in COVID-19. Nat Rev Immunol. 2020;20:515–516. doi:10.1038/s41577-020-0407-1
18. Pence BJG. Severe COVID-19 and aging: are monocytes the key? GeroSci. 2020;42(4):1051–1061.
19. Liao M, Liu Y, Yuan J, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26(6):842–844. doi:10.1038/s41591-020-0901-9
20. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi:10.1016/S0140-6736(20)30566-3
21. Duijf P. Baseline pulmonary levels of CD8+ T cells and NK cells inversely correlate with expression of the SARS-CoV-2 entry receptor ACE2. BioRxiv. 2020.
22. Du R, Liang L, Yang C, et al. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study. Eur Respir J. 2020;55(5). doi:10.1183/13993003.00524-2020.
23. Remy KE, Mazer M, Striker DA, et al. Severe immunosuppression and not a cytokine storm characterize COVID-19 infections. JCI Insight. 2020;5. doi:10.1172/jci.insight.140329
24. Argyropoulos KV, Serrano A, Hu J, et al. Withdrawn: association of initial viral load in SARS-CoV-2 patient with outcome and symptoms. Am J Pathol. 2020.
25. Liu Y, Yan L-M, Wan L, et al. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect Dis. 2020;20(6):656–657. doi:10.1016/S1473-3099(20)30232-2
26. Zheng S, Fan J, Yu F, et al. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January-March 2020: retrospective cohort study. BMJ. 2020;369:m1443.
27. Brooks DG, Trifilo MJ, Edelmann KH, Teyton L, McGavern DB, Oldstone MB. Interleukin-10 determines viral clearance or persistence in vivo. Nat Med. 2006;12(11):1301–1309. doi:10.1038/nm1492
28. Liu J, Hill B, Darko S, et al. The peripheral differentiation of human natural killer T cells. Immunol Cell Biol. 2019;97(6):586–596.
29. Ren L, Zhang L, Chang D, et al. The kinetics of humoral response and its relationship with the disease severity in COVID-19. Commun Biol. 2020.
30. Shankar EM, Che KF, Yong YK, et al. Asymptomatic SARS-CoV-2 infection: is it all about being refractile to innate immune sensing of viral spare-parts?—Clues from exotic animal reservoirs. Pathog Dis. 2021;79(1):ftaa076. doi:10.1093/femspd/ftaa076
31. Sharma L, Chang D, Cruz CSD. Does inflammation help during COVID-19? ERJ Open Res. 2020;6(4):00557–2020. doi:10.1183/23120541.00557-2020
32. Girija A, Shankar EM, Larsson M. Could SARS-CoV-2-induced hyperinflammation magnify the severity of coronavirus disease (CoViD-19) leading to acute respiratory distress syndrome? Front Immunol. 2020;11:1206. doi:10.3389/fimmu.2020.01206
33. Group RC. Dexamethasone in hospitalized patients with Covid-19—preliminary report. New England J Med. 2020.
34. Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020;11:1–13. doi:10.1186/s13293-020-00304-9
35. James SL, Abate D, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–1858.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.