Back to Journals » Clinical Epidemiology » Volume 7
Adverse pregnancy outcomes after exposure to methylphenidate or atomoxetine during pregnancy
Authors Bro SP, Kjaersgaard M, Parner E , Sørensen MJ, Olsen J, Bech B , Pedersen LH, Christensen J, Vestergaard M
Received 19 August 2014
Accepted for publication 16 October 2014
Published 29 January 2015 Volume 2015:7 Pages 139—147
DOI https://doi.org/10.2147/CLEP.S72906
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Henrik Sørensen
Søren Pauli Bro,1 Maiken Ina Siegismund Kjaersgaard,2 Erik Thorlund Parner,2 Merete Juul Sørensen,3 Jørn Olsen,4 Bodil Hammer Bech,4 Lars Henning Pedersen,4,5 Jakob Christensen,6,7 Mogens Vestergaard1
1Research Unit and Section for General Practice, Department of Public Health, 2Section for Biostatistics, Department of Public Health, Aarhus University, 3Regional Center for Child and Adolescent Psychiatry, Aarhus University Hospital, 4Section for Epidemiology, Department of Public Health, 5Department of Clinical Medicine – Obstetrics and Gynecology, Aarhus University, 6Department of Clinical Pharmacology, 7Department of Neurology, Aarhus University Hospital, Aarhus, Denmark
Objective: To determine if prenatal exposure to methylphenidate (MPH) or atomoxetine (ATX) increases the risk of adverse pregnancy outcomes in women with attention deficit/hyperactivity disorder (ADHD).
Materials and methods: This was a population-based cohort study of all pregnancies in Denmark from 1997 to 2008. Information on use of ADHD medication, ADHD diagnosis, and pregnancy outcomes was obtained from nationwide registers.
Results: We identified 989,932 pregnancies, in which 186 (0.02%) women used MPH/ATX and 275 (0.03%) women had been diagnosed with ADHD but who did not take MPH/ATX. Our reference pregnancies had no exposure to MPH/ATX and no ADHD diagnosis. Exposure to MPH/ATX was associated with an increased risk of spontaneous abortion (SA; ie, death of an embryo or fetus in the first 22 weeks of gestation) (adjusted relative risk [aRR] 1.55, 95% confidence interval [CI] 1.03–2.36). The risk of SA was also increased in pregnancies where the mother had ADHD but did not use MPH/ATX (aRR 1.56, 95% CI 1.11–2.20). The aRR of Apgar scores <10 was increased among exposed women (aRR 2.06, 95% CI 1.11–3.82) but not among unexposed women with ADHD (aRR 0.99, 95% CI 0.48–2.05).
Conclusion: MPH/ATX was associated with a higher risk of SA, but our study indicated that it may at least partly be explained by confounding by indication. Treatment with MPH/ATX was however associated with low Apgar scores <10, an association not found among women with ADHD who did not use MPH/ATX.
Keywords: attention deficit/hyperactivity disorder, ADHD, methylphenidate, atomoxetine, pregnancy outcomes
Introduction
The central stimulants methylphenidate (MPH) and atomoxetine (ATX) are the most commonly used drugs in the treatment of attention deficit/hyperactivity disorder (ADHD) in Western countries.1,2 The number of children diagnosed with ADHD has risen dramatically over the past two decades, and some remain on treatment into adulthood.3–7 The use of ADHD medication among women of fertile age has increased almost 100-fold during the past 12 years in Denmark, but little is known about the safety of these drugs during pregnancy.2,8,9
Animal studies have shown that high doses of MPH given during pregnancy are associated with an increased risk of malformations in the offspring. Decreased food intake was found among exposed animals, which is consistent with the anorexigenic effect of this medication.10–12 Human studies and case reports of the effects of MPH on pregnancy outcomes have found no major adverse outcomes, but most of these studies are insufficiently powered to identify even strong fetotoxic effects.13–16 A recent meta-analysis of four cohort studies revealed a total of four malformations among 180 first-trimester-exposed children, which did not exceed the expected number.17 A Danish study of 222 pregnancies exposed to MPH during the first trimester showed no increased risk of major malformations.18 A recent Danish study found a two-fold-increased risk of spontaneous abortion (SA) among women who used ADHD medication (MPH, modafinil, or ATX) during pregnancy compared with unexposed pregnancies.9 Therefore, these studies suggest that ADHD medication may be associated with adverse birth outcomes, and warrant further studies of other pregnancy outcomes, such as birth weight, gestational age (GA), and Apgar score.
In pharmacoepidemiological studies, it is always a challenge to disentangle the effects of the medication from the effects of the underlying disease (confounding by indication). We aimed to examine the effects of the underlying disease (ADHD) and its treatment (MPH and ATX) on pregnancy outcomes in a nationwide cohort study from Denmark.
Materials and methods
Study population
The cohort comprised all clinically recognized pregnancies in Denmark with estimated time of conception and an observed pregnancy outcome in the period from February 1, 1997 to December 31, 2008. Information was obtained from Danish administrative health registries, including the Danish Medical Birth Registry (MBR) and the Danish National Hospital Discharge Register (NHDR), and data were linked through the personal identification number unique to each person with a permanent address in Denmark. When investigating birth outcomes, we included only singleton births.
Central stimulant exposure
In Denmark, ADHD medication can only be purchased in authorized pharmacies with a prescription from a physician, usually a psychiatrist. We obtained information on all redeemed prescriptions in Denmark from the Registry of Medicinal Product Statistics, and included information on MPH/ATX on all redeemed prescriptions from January 1, 1997 to December 31, 2008. The pregnancy period was defined from the estimated date of conception based on GA to the date of the outcome. For SA, the exposure window spanned from 30 days before the estimated day of conception until the day prior to abortion or gestational age 152 days (22 completed weeks). For live births and stillbirths, the exposure window spanned from 30 days before the estimated day of conception until the day prior to birth. MPH was defined according to the Anatomical Therapeutic Chemical (ATC) code N06BA04 and ATX with ATC code N06BA09.
Maternal ADHD diagnosis
We received information on hospital contact for psychiatric illnesses from the Danish Psychiatric Central Registry (DPCR). Women were coded as having ADHD if they were recorded with an International Classification of Diseases (ICD)-10 code F90 in the DPCR from the date of conception until the end of the index pregnancy.
Women who had redeemed a prescription for MPH/ATX within the exposure window were defined as “the exposed ADHD cohort”. Women with an ADHD diagnosis who had not redeemed a prescription for MPH/ATX within the exposure window were defined as “the unexposed ADHD cohort”.
Pregnancy outcomes
The following codes were used: abortions (ICD-10 O02.0–O06.9) were coded as spontaneous (ICD-10 O02.0–O03.9), induced (ICD-10 O04.0–O05.2 and O05.5–O06.9), or induced due to fetal disease (ICD-10 O05.3 and O05.4). Molar or ectopic pregnancies (ICD-10 O00.0–O01.9) were excluded from the main analyses. Furthermore, failed induced abortions (ICD-10 O07) were disregarded, as we assumed that a failed induced abortion would be followed by successful induced abortion, a stillbirth, or a live birth.
Live births and stillbirths were identified in the MBR. Stillbirth was defined as an intrauterine death occurring from 22 completed weeks of gestations and onwards. The Danish lower boundary of stillbirth shifted from 28 weeks to 22 weeks in 2004. We therefore recoded all SAs as stillbirths if they occurred from gestational week 22 to gestational week 28, regardless of calendar year. The MBR provided information on Apgar score at 5 minutes, birth weight, gestational age, and neonatal death (defined as death within the first 28 days after birth). Low birth weight was defined as a weight below 2,500 g. Preterm birth was defined as a birth that took place after less than 37 weeks of pregnancy.
We defined those small for GA (SGA) as children with a weight below the 10th percentile for the gestational birth week, and we used the estimated mean birth weight per GA week using the population of children with nonimputed GA. Missing information on GA for abortions, stillbirths, and live births was replaced by the median over the nonmissing values from each category respectively.
We obtained information on major congenital malformations (ICD-10 Q0–Q99, D215, D821, D1810, P350, P351, and P371, except Q25.0 for GA <37 weeks) through the NHDR. For abortions, the NHDR included ICD-10 codes with information on GA. In cases of abortion before 12 weeks, GA is normally based on the last menstrual period, and for late gestations ultrasound scans are used to determine GA. For stillbirths or live births, the MBR contained information on GA at birth, normally based on ultrasound estimates combined with the last menstrual period.
We coded pregnancies hierarchically to avoid misclassification due to repeated contacts. Any stillbirth or live birth resulted in recoding of any other end point in the index pregnancy period. Any coded SA occurring after the diagnosis of an induced abortion was coded as an SA, because the diagnosis of an induced abortion may be coded on outpatient visits days or weeks prior to the surgical procedure.
Covariates
Information on the following variables was obtained from Statistics Denmark and subsequently coded as shown in parentheses: maternal age at conception (continuous), cohabitation at the time of conception (yes/no), income at the time of conception (< median, ≥ median), and education at the time of conception (<10, 10–12, >12 years). The woman’s parity was categorized as either nulliparous or multiparous. From the Registry of Medicinal Product Statistics, we obtained information on the use of other drugs redeemed during the pregnancy period: antipsychotics (yes/no) (ATC code N05A), antiepileptic drugs (yes/no) (ATC code N03A) and antidepressants (yes/no) (ATC code N06A). These drugs were combined into a composite variable for any antipsychotic, antiepileptic, or antidepressant drug use during pregnancy (yes/no). From the DPCR, we obtained information on history of psychiatric comorbidity, defined as having the following diagnosis ever or ongoing: severe mental disorder (yes/no) (ICD-8 296.1–296.8, 298.1, and 295; ICD-10 F20, F30, and F31), depression (yes/no) (ICD-8 296.0, 298.0, or 300.4; ICD-10 F32 and F33), misuse (yes/no) (ICD-10 F10–F19), and epilepsy (yes/no) (ICD-8 345; ICD-10 G40 and G41). These diagnoses were combined into a composite variable for comorbidity (yes/no). Information on maternal smoking during pregnancy (yes/no) was only available if the index pregnancy resulted in a live or stillborn child.
Statistics
Relative risks (RRs) for SA, preterm birth, SGA, low birth weight, and Apgar score <10 were estimated by using binominal regression with robust variance estimation to allow for correlations of pregnancy outcomes in each woman. RRs for SA were adjusted for maternal age, education, cohabitation, comorbidity, and comedication; according to a propensity score-adjustment simulation, this seemed reasonable to do, even though the rule of thumb of a minimum of ten events per parameter was not satisfied (see Supplementary materials). We did not perform RR analysis when fewer than five exposed events were observed; this is indicated with “NA” (not applicable) in Table 3. We used linear regression to study the associations between exposure to ADHD medication and birth weight and GA at birth, respectively. Robust variance estimation was used in the case of birth weight, while confidence intervals (CIs) for GA were bootstrapped based on 1,000 replications, due to departures from the normality assumption.19 Mean differences for birth weight and GA were adjusted for maternal age, smoking, and parity. Analyses were performed using Stata 12 (StataCorp, USA).
Results
The study population consisted of 989,932 pregnant women, of whom 186 (0.02%) used MPH and/or ATX (the exposed ADHD cohort) and 275 (0.03%) who had a history of an ADHD diagnosis but received no treatment with MPH/ATX (the unexposed ADHD cohort) during pregnancy. The reference group consisted of women without MPH/ATX use and no ADHD diagnosis. In the exposed ADHD cohort, 67 (35%) had a hospital diagnosis of ADHD, 166 (89%) used MPH, 18 (9.7%) used ATX, and two (1.1%) used both ATX and MPH.
Compared with the reference group, women in the exposed ADHD cohort and women in the unexposed ADHD cohort were more likely to be younger, single, have less education, low income, take comedication, have comorbidity, smoke, and be nulliparous (Table 1).
Congenital abnormalities occurred in two children (3.8%) from the exposed ADHD cohort, in seven children (6.9%) in the unexposed ADHD cohort, and in 39,557 children (5.7%) in the reference group (Table 2).
Compared to women in the reference cohort, the risk of SA was 55% higher for women in the exposed ADHD cohort ((adjusted relative risk [aRR] 1.55, 95% CI 1.03–2.36) and 56% higher for women in the unexposed ADHD cohort (aRR 1.56, 95% CI 1.11–2.20) after adjustment for maternal age, education, cohabitation, comorbidity, and comedication (Table 3).
Children of women in the unexposed ADHD cohort tended to have a lower mean birth weight than both the reference group of children born to women without an ADHD diagnosis who did not use MPH/ATX and the children born to women who used MPH/ATX, but the difference decreased and was not statistically significant after adjusting for maternal age, smoking, and parity (Table 3). The children of women in the exposed and unexposed ADHD cohorts also tended to have a shorter GA than the reference group, albeit the difference did not reach statistical significance. We found three preterm births among children in the exposed ADHD cohort and ten preterm births among children in the unexposed ADHD cohort. This number was too small to allow calculation of crude RR (cRR) for the exposed ADHD cohort or adjustment for confounding in the unexposed ADHD cohort. However, the unexposed ADHD cohort showed a significantly increased cRR of preterm birth, SGA, and low birth weight (<2,500 g) compared with the reference pregnancies, but after adjustment for smoking, age, and parity, only the aRR for preterm birth was statistically significant. The aRR of Apgar score <10 at 5 minutes was significantly increased in the exposed ADHD cohort, but not in the unexposed ADHD cohort (Table 3).
Discussion
In this nationwide cohort study, we found that women in the exposed ADHD cohort as well as women in the unexposed ADHD cohort had an increased risk of SA. Children born of women using MPH/ATX had an increased risk of being born with a low Apgar score (<10). Children born of women in the unexposed ADHD cohort had a higher crude risk of being born preterm, with a low birth weight or being SGA compared with the reference group of women without an ADHD diagnosis who did not use MPH/ATX. This indicates that the underlying disease or lifestyle factors related to the disease may play an important role for these associations. In the exposed ADHD cohort, we did not have enough events to calculate the cRRs for being born preterm, SGA, or with a low birth weight. A recent Danish study also used nationwide registers to study the risk of SA following exposure to ADHD-medication use during pregnancy.9 The study had more exposed cases than our study, because the study period was longer and because they included modafinil as ADHD medicine, although this medication is only approved in Denmark for the treatment of narcolepsy.20,21 The authors found a two-fold-higher risk of SA among exposed women compared to unexposed women. Case-crossover analyses revealed that SA was not more likely to occur in pregnancies exposed to ADHD medication than in unexposed pregnancies in the same women. The authors concluded that the association may be attributable to factors related to the underlying disorder (ADHD), rather than exposure to the medication itself.9 A recent review, which included four cohort studies and 180 exposed cases, found malformations in four of the children exposed to MPH in fetal life, which yielded an RR of 0.6 (95% CI 0.2–1.6).17 Furthermore, studies on Danish data found no increased risk of malformation, albeit they were both limited by few exposed cases.9,18 We found very few cases of congenital abnormalities, and did not have enough cases to calculate RRs.
The risk of being born with an Apgar score <10 was increased two-fold in the offspring of women in the exposed ADHD cohort; a similar effect was not found in the unexposed ADHD cohort, which suggests that the ADHD medication may play a causal role for the associated risk of low Apgar score. The Apgar score is a measure of heart rate, respiratory efforts, muscle tone, reflex irritability, and color measured 1 and 5 minutes after birth, with a maximum of 2 points for each parameter and a total of 10 points possible. A low Apgar score is associated with increased mortality and adverse health, especially the score at 5 minutes.22,23 Children in the unexposed ADHD group had a higher risk of being born with a low birth weight (<2,500 g), but after adjustment for relevant confounders, this effect was attenuated along with the difference in mean birth weight and no longer significant. Exposure to MPH/ATX during pregnancy or having an ADHD diagnosis during pregnancy was not associated with a lower GA at birth.
We included all pregnancies in Denmark during the study period, and followed all pregnancies without any loss to follow-up. Selection bias is therefore an unlikely explanation for the associations observed. Information on MPH and ATX did not rely on the memory of the women, but was collected automatically from compulsory reporting of medications redeemed at all Danish pharmacies. Therefore, the information about the redeemed drugs was registered accurately. However, we lacked information on the actual usage of the prescribed medication, which could have masked a harmful effect of the medication, although compliance with nervous system drugs appears to be high.24
The information on GA at birth and birth weight in Danish registers is known to have high validity, but the registration of early SA is incomplete.25 The results may be biased if the registration or reporting of early SAs were different among exposed and unexposed women. Underreporting of SA among exposed women would for example lead to an underestimation of risk of SA associated with ADHD treatment.
In spite of being one of the largest epidemiological studies on the adverse effects of MHP and ATX in pregnancy, our study was limited by a small sample size and therefore low statistical precision. We consequently had to keep our statistical models simple, which makes residual confounding an important issue. Due to the small sample size, we had few cases with low birth weight, preterm birth, and Apgar score <10, and we were not able to fully adjust for confounders. However, we found that when adjusting for maternal age, parity, and smoking in the analysis of birth weight and GA at birth, the differences were attenuated compared with the reference group, which indicates that these variables were important confounders for both birth weight and GA at birth. After adjustment, we found that the aRRs for SA decreased in the exposed ADHD cohort, but increased in the unexposed ADHD cohort, although both remained above a 50% increased risk. Furthermore, we had only limited information on potential confounders like lifestyle factors, diet, and somatic and psychiatric comorbidity, which may also influence risks associated with medicine exposure in pregnancy. We had no information on the occurrence or severity of symptoms in our exposed and unexposed ADHD cohort or on the effect of MPH/ATX on these symptoms. If there was a discrepancy of the symptoms between the two cohorts, it could have affected the pregnancy outcomes.
Confounding by indication is a major challenge in pharmacoepidemiological studies, because it is difficult to disentangle the indication for the treatment from the underlying disease. Women in the unexposed ADHD cohort had at least as high a risk of SA as those in the exposed ADHD cohort after adjustment for confounders, which indicates that factors related to the underlying disease may play a causal role in the risk of adverse birth outcome, although confounding from other factors cannot be ruled out.
Conclusion
We found an increased risk of SA among the women in the exposed ADHD cohort as well as among the unexposed ADHD cohort, which suggests that the underlying ADHD disorder may explain at least a part of the association between MPH/ATX and risk of SA. Treatment with MPH/ATX was associated with low Apgar score, an association not found among the unexposed women with ADHD, suggesting a direct effect of MPH/ATX in pregnancy. However, the sample size was small, and larger studies with more clinical information are needed to corroborate these findings.
Acknowledgments
We thank Morten Pilegaard for help with preparation of the manuscript. The study was supported by the Danish Epilepsy Association. Thank you to the Fonden til Styrkelse af Psykiatrisk Forskning ved Børne – Og Ungdomspsykiatrisk Center for financial support.
Disclosure
The authors report no conflicts of interest in this work.
References
Chai G, Governale L, McMahon AW, Trinidad JP, Staffa J, Murphy D. Trends of outpatient prescription drug utilization in US children, 2002–2010. Pediatrics. 2012;130(1):23–31. | |
Pottegård A, Bjerregaard BK, Glintborg D, Kortegaard LS, Hallas J, Moreno SI. The use of medication against attention deficit hyperactivity disorder in Denmark: a drug use study from a patient perspective. Eur J Clin Pharmacol. 2013;69(3):589–598. | |
Sclar DA, Robinson LM, Bowen KA, Schmidt JM, Castillo LV, Organov AM. Attention-deficit/hyperactivity disorder among children and adolescents in the United States: trend in diagnosis and use of pharmacotherapy by gender. Clin Pediatr (Phila). 2012;51(6):584–589. | |
Robison LM, Skaer TL, Sclar DA, Gailin RS. Is attention deficit hyperactivity disorder increasing among girls in the US? Trends in diagnosis and the prescribing of stimulants. CNS Drugs. 2002;16(2):129–137. | |
Winterstein AG, Gerhard T, Shuster J, et al. Utilization of pharmacologic treatment in youths with attention deficit/hyperactivity disorder in Medicaid database. Ann Pharmacother. 2008;42(1):24–31. | |
Hechtman L. Predictors of long-term outcome in children with attention-deficit hyperactivity disorder. Pediatr Clin North Am. 1999;46(5):1039–1052. | |
Hinshaw SP, Owens EB, Zalecki C, et al. Prospective follow-up of girls with attention-deficit/hyperactivity disorder into early adulthood: continuing impairment includes elevated risk for suicide attempts and self-injury. J Consult Clin Psychol. 2012;80(6):1041–1051. | |
McCarthy S, Wilton L, Murray ML, Hodgkins P, Asherson P, Wong IC. The epidemiology of pharmacologically treated attention deficit hyperactivity disorder (ADHD) in children, adolescents and adults in UK primary care. BMC Pediatr. 2012;12:78. | |
Haervig K, Mortensen L, Hansen A, Strandberg-Larsen K. Use of ADHD medication during pregnancy from 1999 to 2010: a Danish register-based study. Pharmacoepidemoiol Drug Saf. 2014;23(5):526–533. | |
Teo SK, Stirling DI, Thomas SD, Hoberman AM, Christian MS, Khetani VD. The perinatal and postnatal toxicity of D-methylphenidate and D,L-methylphenidate in rats. Reprod Toxicol. 2002;16(4):353–366. | |
Teo SK, Stirling DI, Thomas SD, Hoberman AM, Christian MS, Khetani VD. D-methylphenidate and D,L-methylphenidate are not developmental toxicants in rats and rabbits. Birth Defects Res B Dev Reprod Toxicol. 2003;68(2):162–171. | |
Beckman DA, Schneider M, Youreneff M, Tse FLS. Developmental toxicity assessment of d,l-methylphenidate and d-methylphenidate in rats and rabbits. Birth Defects Res B Dev Reprod Toxicol. 2008;83(5):489–501. | |
Smithells RW. Thalidomide and malformations in Liverpool. Lancet. 1962;1(7242):1270–1273. | |
Kopelman AE, McCullar FW, Heggeness L. Limb malformations following maternal use of haloperidol. JAMA. 1975;231(1):62–64. | |
Heinonen OP, Slone D, Shapiro S. Birth Defects and Drugs in Pregnancy: Maternal Drug Exposure and Congenital Malformations. Littleton (MA):Publishing Sciences Group; 1977. | |
Debooy VD, Seisha MM, Tenenbein M, Casiro OG. Intravenous pentazocine and methylphenidate abuse during pregnancy. Am J Dis Child. 1993;147(10):1062–1065. | |
Dideriksen D, Pottegård A, Hallas J, Aagaard L, Damkier P. First trimester in utero exposure to methylphenidate. Basic Clin Pharmacol Toxicol. 2013;112(2):73–76. | |
Pottegård A, Hallas J, Andersen JT, et al. First-trimester exposure to methylphenidate: a population-based cohort study. J Clin Psychiatry. 2014;75(1):e88–e93. | |
Efron B, Tibshirani RJ. Bootstrap methods for standard errors, confidence intervals and other measures of statistical accuracy. Stat Sci. 1986;1(1):54–75. | |
pro.medicin.dk [homepage on the internet]. Modafinil “Mylan”. Copenhagen: Dansk Lægemiddel Information; 2014 [updated January 16, 2014]. Available from: http://pro.medicin.dk/Medicin/Praeparater/6716. Accessed October 7, 2014. | |
pro.medicin.dk [homepage on the internet]. Modiodal: modafinil. Copenhagen: Dansk Lægemiddel Information; 2014 [updated May 21, 2014]. Available from: http://pro.medicin.dk/Medicin/Praeparater/2520. Accessed October 7, 2014. | |
Casey BM, McIntire DD, Leveno KJ. The continuing value of the Apgar score for the assessment of newborn infants. New Engl J Med. 2001;344(7):467–471. | |
Sun Y, Vestergaard M, Pedersen C, Christensen J, Olsen J. Apgar scores and long-term risk of epilepsy. Epidemiology. 2006;17(3):296–301. | |
Olesen C, Søndergaard C, Thrane N, Nilensen GL, Jong-van den Berg L, Olsen J. Do pregnant women report use of dispensed medications? Epidemiology. 2001;12(5):497–501. | |
Kristensen J, Langhoff-Roos J, Skovgaard LT, Kristensen FB. Validation of the Danish Birth Registration. J Clin Epidemiol. 1996;49(8):893–897. |
Supplementary materials
Simulation
The simulation outlined here investigates whether the rule of thumb of a minimum of ten events per predictor variable in a binary regression analysis can be relaxed in the particular covariate pattern seen when studying the effect of attention deficit/hyperactivity disorder (ADHD) medication in pregnancy on spontaneous abortion.1 A study by Vittinghoff and McCulloch suggested that this rule may in some instances be relaxed, although the situations considered in Vittinghoff and McCulloch do not cover the regression models considered in the present study.2 This simulation was done with the dual purpose of assessing whether 1) propensity-score adjustment for a number of covariates is appropriate, and/or whether 2) adjustment for a number of covariates in a binary regression is appropriate when studying the effect of ADHD medication in pregnancy on spontaneous abortion. The question that seeks answering is how many covariates to include in the analysis while ensuring that the procedure for constructing the confidence interval achieves a 95% level of coverage.
A prioritized list of potential confounders to be added one at a time in the two situations outlined is made and simulated as follows: 1) mothers’ age as normally distributed, with a mean of 30 and a standard deviation of 5.5; 2) education as multinomially distributed, with probabilities 0.11, 0.32, and 0.57 corresponding to low, medium, and high education, respectively, and afterward coded as two binary variables for medium versus low and high versus low education; 3) cohabitation as a binary variable with a probability of 0.2; 4) comorbidity as a binary variable with a probability of 0.05; and 5) comedication as a binary variable with a probability of 0.05. All simulated potential confounders are thereafter standardized to have a mean of 0 and a standard deviation of 1.
A logistic regression of ADHD medication on spontaneous abortion is fitted on the real data set, which yields a coefficient value of 0.65. Next, logistic regressions of mothers’ age on ADHD medication and on spontaneous abortion are both fitted on the real data set, which yields coefficient values of -0.20 and 0.24, respectively. Similarly, logistic regressions for the other potential confounders on both ADHD medication and spontaneous abortion are fitted, which yield coefficient values of 0.10 and -0.06 for medium versus low education, -0.38 and -0.08 for high versus low education, 0.54 and 0.12 for cohabitation, 0.33 and 0.05 for comorbidity, and 0.47 and 0.05 for comedication. Baseline coefficients are chosen as -10 and -1.8, respectively.
As one wishes to report risk ratios for spontaneous abortion using binomial regression with log link, data on binary ADHD-medication exposure and binary spontaneous abortion outcome are simulated under this model, while adding covariates successively using the coefficient values reported, ie, the log-probability model for spontaneous abortion firstly consists of only baseline and ADHD medication. Secondly, the model consists of both baseline and ADHD medication and standardized mothers’ age, and so forth, until finally the full model consists of baseline, ADHD medication, standardized mothers’ age, standardized education, standardized cohabitation, standardized comorbidity, and standardized comedication.
Propensity scores were calculated in two ways: 1) as the predicted values from a multiple logistic regression of exposure on standardized covariates, and 2) as the predicted values from a multiple linear regression of exposure on standardized covariates. The propensity-score adjustment was done as a binomial regression, with propensity score adjusted linearly. Coverage probability is calculated based on 1,000 replications.
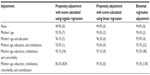
As seen from Table S1, appropriate coverage probabilities are achieved even when all the covariates are included. Propensity scores can thus be calculated using all covariates considered, and can subsequently be used in a propensity score-adjustment analysis of the effect of ADHD medication on spontaneous abortion. Here, both logistic and linear regression can be used to calculate propensity scores; convergence is more often achieved when using linear regression.
However, when simulating data and using the individual covariates instead of combining these into a propensity score, similar appropriate coverage probabilities, as for the propensity score-adjustment simulation. were achieved (Table S1). Therefore, in the setup outlined, relevant for evaluating the effect of ADHD medication in pregnancy on spontaneous abortion, the rule of thumb of a minimum of ten events per predictor variable may be relaxed.
References
Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR. A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol. 1996;49(12):1373–1379. | |
Vittinghoff E, McCulloch CE. Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol. 2007;165(6):710–718. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.