Back to Journals » Nature and Science of Sleep » Volume 13
A Blue-Enriched, Increased Intensity Light Intervention to Improve Alertness and Performance in Rotating Night Shift Workers in an Operational Setting
Authors Sletten TL , Raman B, Magee M , Ferguson SA , Kennaway DJ , Grunstein RR , Lockley SW, Rajaratnam SMW
Received 16 October 2020
Accepted for publication 4 February 2021
Published 24 May 2021 Volume 2021:13 Pages 647—657
DOI https://doi.org/10.2147/NSS.S287097
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Steven A Shea
Tracey L Sletten,1 Bhairavi Raman,1 Michelle Magee,1 Sally A Ferguson,2 David J Kennaway,3 Ronald R Grunstein,4,5 Steven W Lockley,6,7 Shantha MW Rajaratnam1,6,7
1Turner Institute for Brain and Mental Health, School of Psychological Sciences, Monash University, Melbourne, Victoria, Australia; 2Central Queensland University, Appleton Institute, Goodwood, SA, Australia; 3Robinson Research Institute, School of Medicine, Discipline of Obstetrics and Gynaecology, University of Adelaide, Adelaide, SA, Australia; 4Woolcock Institute of Medical Research, University of Sydney, Sydney, NSW, Australia; 5Department of Respiratory & Sleep Medicine, Royal Prince Alfred Hospital, Sydney, NSW, Australia; 6Division of Sleep and Circadian Disorders, Departments of Medicine and Neurology, Brigham and Women’s Hospital, Boston, MA, USA; 7Division of Sleep Medicine, Harvard Medical School, Boston, MA, USA
Correspondence: Tracey L Sletten
Turner Institute for Brain and Mental Health, School of Psychological Sciences, Monash University, Building 2, 270 Ferntree Gully Road, Notting Hill, Victoria, 3168, Australia
Tel + 61 3 990 20734
Email [email protected]
Purpose: This study examined the efficacy of a lighting intervention that increased both light intensity and short-wavelength (blue) light content to improve alertness, performance and mood in night shift workers in a chemical plant.
Patients and Methods: During rostered night shifts, 28 workers (46.0± 10.8 years; 27 male) were exposed to two light conditions each for two consecutive nights (∼ 19:00– 07:00 h) in a counterbalanced repeated measures design: traditional-spectrum lighting set at pre-study levels (43 lux, 4000 K) versus higher intensity, blue-enriched lighting (106 lux, 17,000 K), equating to a 4.5-fold increase in melanopic illuminance (24 to 108 melanopic illuminance). Participants completed the Karolinska Sleepiness Scale, subjective mood ratings, and the Psychomotor Vigilance Task (PVT) every 2– 4 hours during the night shift.
Results: A significant main effect of time indicated KSS, PVT mean reaction time, number of PVT lapses (reaction times > 500 ms) and subjective tension, misery and depression worsened over the course of the night shift (p< 0.05). Percentage changes in KSS (p< 0.05, partial η2=0.14) and PVT mean reaction time (p< 0.05, partial η2=0.19) and lapses (p< 0.05, partial η2=0.17) in the middle and end of night shift, expressed relative to start of shift, were significantly improved during the lighting intervention compared to the traditional lighting condition. Self-reported mood did not significantly differ between conditions (p> 0.05).
Conclusion: Our findings, showing improvements in alertness and performance with exposure to blue-enriched, increased intensity light, provide support for light to be used as a countermeasure for impaired alertness in night shift work settings.
Keywords: shift work, circadian photoreception, alertness, vigilance, light, short-wavelength
Plain Language Summary
Night shift work is often associated with acute and chronic sleep deficiency, impaired alertness and neurobehavioural performance, placing workers at increased risk of injury and accidents. Effective non-pharmacological interventions for alertness and performance impairments during night shifts are needed. This study evaluated the efficacy of a dual-faceted lighting intervention consisting of increased intensity and blue wavelength composition to improve alertness, performance and mood in night workers in an operational setting. The intervention lighting attenuated the deterioration in alertness and performance observed over the night shift under typical lighting. The findings support lighting interventions which employ the principles of circadian photoreception as a practical countermeasure within occupational fatigue management programs. Outcomes from this workplace intervention may inform future lighting standards to improve safety, productivity and wellbeing in operational settings.
Introduction
Due to misalignment of the sleep-wake cycle with the circadian pacemaker, night shift work is often associated with acute and chronic sleep deficiency,1 and impaired alertness and neurobehavioural performance at work.1 This places night workers at increased risk of accidents and injuries in the workplace2 and motor vehicle crashes on the drive home.1 In rapidly rotating shift work, where adaptation of the circadian pacemaker to night shift is not desirable, practical tools for acute alertness management during night shifts are needed. Although pharmacological wake-promoting treatments can be beneficial, they can be associated with side effects, tolerance, sleep disruption and limited duration effect. Effective non-pharmacological interventions for alertness and performance impairments during night shifts are needed.
The development of electric lighting permitted work outside daylight hours, facilitating extended work hours and shift work. In addition to helping people see, exposure to light, especially light at night, has acute alerting effects.3–5 High intensity broad spectrum (ie white) light improves subjective alertness and cortical activation during the day.6,7 Higher intensity broad-spectrum lighting during the night improves subjective alertness and neurobehavioural performance, reduces electroencephalogram (EEG) activity in the theta and low-frequency alpha range (5–9 Hz), reduces slow eye movements, and suppresses the nocturnal secretion of melatonin.6,8,9
These non-visual responses to light are primarily, but not exclusively, driven by specialized intrinsically photosensitive retinal ganglion cells (ipRGCs) containing melanopsin that are sensitive to short wavelength (ie blue) light (λmax~480 nm).10,11 Laboratory studies using monochromatic and narrow-bandwidth light show that short wavelength light is most effective at suppressing melatonin, improving performance,12–15 and enhancing brain responses,16 compared to other wavelengths. Blue-appearing light is impractical for operational use, however, and therefore adjusting the spectral composition of polychromatic (white) light by increasing the short wavelength content is a practical way to improve alertness. Laboratory studies have shown that light sources with high correlated colour temperatures, with increased blue content and melanopic illuminance, the unit which quantifies the amount of light that is stimulating melanopsin, are more effective in improving subjective alertness, neurobehavioural performance and mood, compared to lower colour temperature sources.17,18 Daytime exposure to blue-enriched polychromatic lighting during real-world and simulated work and school-days improves self-reported wellbeing, alertness, concentration and mood.19–22 Few studies have directly tested the effectiveness of blue-enriched polychromatic light to enhance alertness and performance in a night shift work setting.23–26 Based on the well-established principles that both intensity and spectral composition of light independently influence alertness state,8,11,12 we evaluated the implementation of a control room lighting intervention on alertness, performance and mood during night shifts in rapidly rotating shift workers by increasing light intensity and blue light content as compared to the current lighting conditions. The intervention aimed to utilize the acute alerting effects of light directly during the night shifts, and not influence the circadian response to night shifts given that in rapidly rotating shift workers, circadian adaptation to consecutive night shifts is rare,27 and is not advisable due to the quick return to day shifts.
Patients and Methods
Participants
Participants were chemical plant shift workers (Melbourne, VIC), operating on a ten-day shift rotation (2 day shifts, 5 days off, 2 night shifts, 1 day off). Workers spent the majority of their shifts seated at monitors in a single windowless control room. Thirty-five individuals volunteered for the study (58% participation) but only 33 commenced the study due to inappropriate work schedules. Specific exclusion criteria were not applied in this real-world study. Participants provided written informed consent and were reimbursed for time and inconvenience. Study procedures were approved by the Monash University Human Research Ethics Committee (Project number CF11/2342 – 2011001331) and were conducted in accordance with the Declaration of Helsinki.
Study Design
Participants completed questionnaires assessing health status, diurnal preference (Morningness-Eveningness Questionnaire), sleep quality (Pittsburgh Sleep Quality Index), trait sleepiness (Epworth Sleepiness Scale), risk of Obstructive Sleep Apnea (Berlin Questionnaire), and sleep hygiene (Sleep Hygiene Index).
Participants’ sleep-wake behaviour was monitored for 7–10 days prior to and throughout the study using daily sleep diaries and a wrist-worn activity monitor (Actiwatch-L, Actiwatch-64, Actiwatch-2, Philips Respironics, Bend OR, USA). Diaries reported bedtime, rise time, sleep onset latency and duration of awakenings. Activity monitors provided an objective measure of total sleep time (TST). Confirmation of work shifts was obtained from self-reported daily duty diaries.
Participants were monitored during two complete shift rotations, which each included two consecutive night shifts. Night shift duration was 12 hours, scheduled to occur from 19:00 to 07:00 h. Participants were asked to complete assessments of subjective sleepiness, neurobehavioural performance and mood at the start of shift, four hours into shift, then two-hourly until the end of shift.
Subjective sleepiness was assessed using the Karolinska Sleepiness Scale (KSS) representing the level of sleepiness in the preceding 10 minutes. A ten-minute visual Psychomotor Vigilance Task (PVT) was completed on palm pilots as a measure of sustained attention.28 Subjective mood was assessed using 9-point visual analog scales for calmness (1=very calm, 9=very tense), cheerfulness (1=very cheerful, 9=very miserable) and depression (1=very depressed, 9=very elated).29 The depressed-elated scale was reverse-scored such that higher scores represented higher levels of depression. Participants reported where they spent the majority of their time in the two hours preceding each assessment (ie “in the control room”, “not in the control room”, “outdoors”) to ensure they had been exposed to the light condition. At the end of each night shift participants completed a headache and eye strain scale, providing a rating of the extent to which they were experiencing symptoms of irritability, headache, eye strain, general eye discomfort, eye fatigue, difficulty focusing, difficulty concentrating and blurred vision on a scale of 1=absent and 4=severe.20
During two rotations of two consecutive night shifts, participants were exposed to two conditions of overhead lighting in a repeated-measures design (Figure 1). One light condition was maintained for two consecutive nights before changing to the alternative lighting condition. The lamps were changed for both conditions to avoid a potential placebo effect of only changing the light source for the experimental intervention condition. The traditional lighting (43 lux, 4000 K, T5 24W, Philips Lighting, Eindhoven, Netherlands) was designed to match the current lighting conditions in the primary work environment (~40 lux). The intervention condition consisted of higher intensity blue-enriched (increased melanopic illuminance) lighting (100 lux, 17,000 K, Activiva Active TL5 24W, Philips Lighting, Eindhoven, Netherlands) with increased spectral output in the short wavelength range (420–480 nm). Lighting was measured at seated eye level, 122 cm from floor in the horizontal and vertical plane. The intensity of the intervention light source in the horizontal plane was selected based on the range of typical ambient room lighting,8,30 and to avoid a light intensity that may limit visual acuity to effectively monitor screens in the control rooms. The visual illuminance of the intervention condition was 106 lux compared to 43 lux for the traditional lighting condition. When calculated using the International Commission on Illumination (CIE) standard international units calibrated for melanopsin sensitivity,31 the melanopic Equivalent Daylight (D65) Illuminance (EDI) was more than 4 times higher in the intervention condition compared to the traditional light condition (108 compared to 24 lux; Figure 2). The melanopic Daylight Equivalent Ratio (DER) value, an overall indication of the relative melanopic strength of the spectra, was 0.55 and 1.02 for the intervention and traditional lighting conditions, respectively (Figure 2).
 |
Figure 2 Spectral power distribution of the fluorescent light sources used in the intervention (blue line) and traditional (black line) light conditions, expressed relative to total power in the 380–760 nm range. The panel shows mean vertical and horizontal illuminance levels of each light condition in lux, measured across the most populated areas of the control room. Measures of illuminance were taken at a height of 122 cm from the floor level, representative of a workers’ seated position. Simulated, laboratory simulation of light sources where the photopic lux value was scaled to match the observed lux measured on site to provide relative α-opic equivalent (D65) illuminance (EDI, lux) for each of the five photoreceptors and the melanopic daylight efficacy ratio (DER, the ratio of melanopic/photopic lux).31 |
The order of the light conditions was counterbalanced between the five shift roster groups so that the first three shift groups received the traditional light first and last two groups studied received the intervention light first. Unpredictable shift changes resulted in an unequal number of participants in each counterbalance group. Twenty-two participants received the traditional light first; nine received the intervention light first.
Statistical Analyses
Bed and rise times from sleep diaries identified sleep episodes for analysis of actigraphic data. Time in bed and TST in the 24 hours prior to each night shift were calculated. Actigraphic analyses (medium sensitivity) were conducted using Actiware 5.0 software (Philips Respironics, Bend OR).
PVT reaction times <100 ms (0.05% of data) and >10,000 ms (0.007% of data) were excluded from analyses as they represent pre-emptive responses, and distractions, respectively. For each 10-minute PVT, mean reaction time and the number of PVT lapses (reaction time >500 ms) were calculated.
Tests that were completed within 60 minutes of the planned time were binned for analyses. The final test times for analyses were 19:00, 23:00, 01:00, 03:00, 05:00, and 06:00 h. Missing data were replaced with mean values of the two adjacent timepoints, or one adjacent timepoint where the data were missing at the start or end of the shift. The greatest proportion of values were replaced for lapses data; 3.3% on night 1 and 2.4% on night 2. Participants reported that they had not spent the majority of the two hours prior to each assessment within the control room on only 2.4% of occasions on either night shift.
Due to low power for analysis of all timepoints (with variability in test timing and reduced n), and lack of difference in outcome variables between the two night shifts, data points were reduced; for each participant, data were averaged to represent the start (19:00–21:00 h), middle (22:00–01:00 h), and end of shift (02:00–05:00 h), averaged across the two night shifts. Assessment of the impact of the lighting intervention at the middle and end of shift targets the potential to improve the impairments in alertness and performance previously identified at these vulnerable times during the night shift.32 Outcome variables were expressed as percentage change from the start of shift for each individual to account for group differences in alertness and performance levels at shift start. Two-way repeated measures ANOVA was used to compare the effects of traditional vs intervention lighting on change in KSS, mood, transformed mean reaction times [1/RT] and transformed lapses [(√x) + (√(x+1))] (light conditions, time). PVT data were transformed to account for positive skew.33 Mann–Whitney U-tests were used to examine potential lighting condition order effects. Ratings from the headache and eye strain scale were averaged between the two night shifts and compared between light conditions with paired t-tests. Data were analysed using PASW Statistics 26.0 (SPSS Inc, Chicago IL).
Results
Although 33 participants commenced the study, data were only included in the final analyses if participants completed two consecutive night shifts under both light conditions. Demographic characteristics for the final 28 participants included are presented in Table 1. Time in bed (t(22)=1.53, p=0.141) and TST (t(16)=0.78, p=0.448) in the 24 hours prior to exposure to the night shifts did not differ between the two light conditions (Table 1). There was no difference in the amount of sleep obtained between the two consecutive night shifts for the two conditions (t(20)=0.53, p=0.601) (Table 1).
 |
Table 1 Participant Characteristics of Night Shift Workers. Data Presented are Mean ± SD Unless Otherwise Stated |
Due to missing data for one light condition, PVT data were analysed for n=20 on night 1; KSS data were analysed for n=28. For night 2, analysed data were n=25 for KSS, n=19 for PVT mean reaction time and n=17 for PVT lapses. KSS and PVT for night shifts 1 and 2 demonstrated a significant effect of time with an increase in KSS (Night 1 p<0.0001, Night 2 p<0.0001), PVT mean reaction time (Night 1 p=0.003, Night 2 p<0.0001) and number of PVT lapses during each shift (Night 1 p=0.002, Night 2 p=0.002). There was no main effect of light condition or light x time interaction effect for either night shift when analysed separately (p>0.05).
Mann–Whitney U-tests showed no significant differences between the group exposed to traditional lighting first (n=20) or intervention lighting first (n=8) for KSS, PVT reaction time, PVT lapses or subjective mood (p>0.05). Data were subsequently analysed without taking into account the order of exposure to light condition.
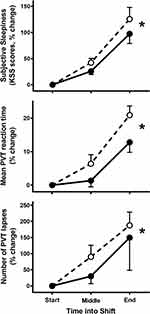
When data across the two night shifts were averaged to compare the start, middle and end of shift, percentage change in mean KSS scores significantly increased between the middle and end of shift (relative to the start) in both light conditions (F1,27=29.63, p<0.0001, partial η2=0.52; Figure 3). The percentage increase in KSS from the middle to end of shift (relative to start) was significantly attenuated in the intervention lighting condition compared to the traditional lighting (F1,27=4.40, p=0.045, partial η2=0.14; Figure 3). No interaction effect for light x time for KSS ratings was observed (F1,27=1.30, p=0.26). From the start to the end of the night shift, KSS increased by 2.85 in the traditional light condition and 2.2 in the intervention condition, representing attenuation of subjective sleepiness by 22.8% with the intervention lighting (Table 2).
For PVT reaction time and lapses, main effects of time were observed, with percentage change in mean reaction times (F1,23=59.21, p<0.0001, partial η2=0.72) and number of lapses (F1,23=12.98, p=0.002; partial η2=0.36) significantly increasing between the middle and end of shift in both light conditions (Figure 3). Significant main effects of light condition were found for percentage change in mean reaction time (F1,23=5.29, p=0.031, partial η2=0.19) and lapses (F1,23=5.00, p=0.035, partial η2=0.18), indicating improved performance under the intervention light condition compared to traditional lighting (Figure 3). For PVT lapses, there was also a significant interaction between time and light condition, where the percentage increase in mean number of lapses between the middle and end of shift was significantly lower during intervention light exposure compared to traditional lighting (F1,23=7.83, p=0.010, partial η2=0.25). There was no significant time x light condition interaction for PVT mean reaction time (F1,23=1.16, p=0.29). The impairment in PVT reaction time at the end of night shift relative to start was reduced by 22.4 ms with the intervention lighting compared to the traditional condition (38.44 ms vs 60.82 ms) (Table 2), representing attenuation of 36.8%. PVT lapses increased by 5.6 at the end compared to the start of the traditional light shift, compared to 3.7 on the intervention night, representing a reduction of 1.9 lapses or 33.9% on average (Table 2).
A significant main effect of time was observed for self-reported mood during the night shift. Participants reported higher levels of tension (lower calmness; F1,27=7.34, p=0.012, partial η2=0.21), misery (lower cheerfulness; F1,27=17.72, p<0.0001; partial η2=0.40) and depression (higher depression; F1,27=10.62, p=0.003; partial η2=0.28) at the middle and end of shift, relative to the start in both conditions (Table 2). There were no main effects of light condition or time x light condition interaction for any of the mood scales (p>0.05). From the headache and eye strain scale, the only symptom that was significantly different between lighting conditions was difficulty concentrating (Supplementary Table 1). The traditional lighting condition was associated with more severe difficulty concentrating (t25 = 2.21, p = 0.026).
Discussion
The current study is the first to systematically test the efficacy and feasibility of a dual-faceted lighting intervention of increased intensity blue-enriched light exposure to enhance alertness in a rapidly rotating night shift working population in an operational occupational setting. The study demonstrated that under typical lighting, subjective sleepiness, PVT reaction time, number of PVT lapses and subjective mood worsened with time into night shift with moderate effect sizes (0.36–0.72). The lighting intervention attenuated this decline in alertness and performance compared to the traditional lighting (effect sizes, 0.14–0.19). While these were relatively small effect sizes overall, the light intervention attenuated the deterioration over the night shift by between 23% and 37% (for subjective sleepiness and PVT reaction time, respectively). Self-reported mood outcomes were not significantly improved by the lighting intervention.
Participants in the study were representative of several shift working populations, reporting high BMI, poor sleep quality, and excessive sleepiness.34 Actigraphic TST in the 24 hours prior to the first night shift averaged between 8.65 h and 9.03 h including napping, which is well above the minimum 7 hours recommended TST for healthy adults,35 suggesting that participants were extending their sleep in preparation for their scheduled night shifts. Between consecutive night shifts, however, participants obtained 5.5 h, indicating considerable sleep deficit between night shifts.
Low levels of subjective sleepiness were evident at the start of the night shift, at a time corresponding to the timing of the wake maintenance zone, during which alertness is expected to be high.36 Subjective sleepiness and neurobehavioural performance worsened during the night, characteristic of the combined effects of homeostatic sleep pressure due to extended time awake and adverse circadian phase.37 The observed increase in sleepiness and impaired performance with time into the night shift is a target for interventions to improve productivity and safety.
The existing lighting in this operational setting on which the control lighting condition was modelled consisted of relatively low intensity (~40 lux) standard lighting (4000 K). By increasing the illuminance, and the melanopic (blue) content of the light, we improved alertness and performance. Under the intervention condition, we observed improvements in alertness and performance which are consistent with previous research demonstrating the alerting effects of enhanced intensity8 and blue-enriched polychromatic light during the night17,38–40 and during the day.19–22
Although the effect size was small, the magnitude of the reduction in subjective sleepiness in our study was 15–20% greater than that reported in previous operational blue-enriched lighting interventions of longer duration.19,20 The prior studies administered similar light intensities between conditions, and therefore we suggest that the increased effect size observed in the current study may be attributed to the increase in overall melanopic illuminance. Our intervention condition provided 4.5 times the melanopic illuminance compared to the traditional lighting (108 compared to 24 in the traditional light condition). By comparison, one of the daytime blue-enriched lighting interventions provided only 1.4 times the melanopic illuminance compared to the typical 4000 K lighting (melanopic illuminance = 348 and 257, respectively).20 Our study was also conducted at night, when sleepiness levels are higher (eg, Cohen et al, 201037) and there is greater potential for improvement due to light.14 Nighttime exposure to blue-enriched polychromatic lighting (17,000 K)41 providing a similar increase in melanopic illuminance to previous daytime exposure20 (1.7 times increase from 711 to 1212 melanopic illuminance) demonstrated as much as five times the potency for melatonin suppression at night compared to lighting of 4000 K. Although it has previously been suggested that the alerting effects of a lighting intervention during the night may be mediated by light-induced suppression of melatonin,42 such improvements in alertness are observed in the absence of circulating melatonin,6,13,20 suggesting that additional neural pathways are activated through light exposure.43,44
The intensity of the intervention condition is consistent with Australian lighting standards for an automatic plant with no or occasional manual control (80 lux), and below that for continuously occupied workplace interiors (160 lux) (Australian/New Zealand Standard 1680). It should be noted, however, that there is no specific standard for lighting for night shift and this light intensity was the highest level that was considered tolerable by staff based on consultation. The intervention intensity was, however, considerably lower than that used in other studies testing the efficacy of blue-enriched lighting interventions during the daytime (lower by ~200–300 lux),19,20 and nighttime (lower by ~400–600 lux).23,24 Previous laboratory comparisons of light sources of similar low intensities (40 lux) have revealed greater alerting responses following evening exposure to blue-enriched polychromatic light compared to standard white light.17 A recent reanalysis of data from several studies of the alerting effects of light in humans reported that exposure to light of <100 lux melanopic EDI can stimulate > 50% of the maximum alerting effects.3 Therefore, high intensity light sources may not be necessary to induce alerting responses, particularly if spectral composition is optimal. When the International Agency for Research on Cancer (IARC) reaffirmed in 2019 that night shift work is “probably carcinogenic to humans”, melatonin suppression was cited as a possible mechanism.45 This raises the need to consider such effects as a possible consequence of light interventions at night compared to the immediate risk of sleepiness-related accidents and injuries, and the need for further research and standards for lighting for night shift.
This study did not find significant differences between the light conditions in three domains of mood (calm-tense, cheerful-miserable, and depressed-elated), which is inconsistent with previous studies.17,19,20 Prior studies have assessed mood as an index of subjective wellbeing19 or general positive and negative affect,20 rather than specific mood domains. Limited effects of the intervention lighting on mood may also be a function of the short duration of light exposure for cumulative mood improvements or the almost all male sample given that males and females differ in patterns of self-reported mood, with females more likely to report depressive symptoms.46 Females are also reported to be more impaired following sleep loss.47
Due to operational demands resulting in differences in test timing and high inter-individual variability between participants, power of the current analyses was limited. Further investigation of the lighting intervention in a larger cohort of night workers is required, to facilitate further assessment of changes in alertness at multiple time points across the shift. Inter-individual differences have been reported in cognitive responses to sleep loss, tolerance to shift work48 and responses to light.49 In view of these findings, future studies should seek to identify individuals who are most vulnerable to alertness impairment due to night shift, for example, the 10–23% of shift workers who are reported to have Shift Work Disorder,50 to receive personalized lighting interventions. Future studies should include circadian phase assessment to account for the impact of individual differences in circadian phase on alertness, neurobehavioural performance and mood.29,51
Conclusions
For the first time, this study systematically tested in a within-subject design the efficacy of increased intensity and blue-enriched light exposure to enhance alertness in rapidly rotating night shift workers in an operational occupational setting. The study has shown that exposure to a higher intensity light with increased blue light content enhances subjective alertness and objective performance at the middle and end of the night shift. We selected a dual-faceted lighting intervention (increased intensity and blue wavelength composition) to induce maximal improvements in alertness, and to be readily translated into practice. Improvements in alertness and performance seen in this study provide support for the potential benefits of light as a countermeasure against sleepiness during night shift in rapidly rotating shift workers. Findings suggest light interventions that capitalize on enhancing melanopsin-based responses may be an effective component of an occupational fatigue management program. Outcomes from this workplace intervention may inform future lighting standards to improve safety, productivity and wellbeing in operational settings. Further studies can examine the benefits of targeted lighting interventions in individuals most vulnerable to alertness impairment during night work.
Acknowledgments
The authors thank Julia Shekleton, DPsych for project establishment and data collection, Dev Kevat, MD for initial data cleaning, Jan White, MD for support to facilitate the project in the operational environment, and Joshua Wiley PhD for comments on the manuscript.
This research was supported by the National Health and Medical Research Council (NHMRC) project grant (# 545871) and was endorsed by the Australasian Sleep Trials Network (NHMRC Enabling Grant # 343020). The study sponsor had no role in the study design or preparation of the manuscript. RRG is a recipient of a NHMRC Senior Principal Research Fellowship (#1106974). aPresent address for Michelle Magee: Centre for Neuroscience of Speech, Melbourne School of Health Sciences, Parkville, The University of Melbourne, Victoria, Australia.
Disclosure
TLS reports her institution has received equipment donations or other support from Philips Lighting, Philips Respironics, Optalert™ and Compumedics and she has received consultancy fees and reimbursement of travel expenses from Vanda Pharmaceuticals; Grants from National Health and Medical Research Council (NHMRC); Endorsement. NHMRC Enabling Grant # 343020 from Australasian Sleep Trials Network.
SAF reports grants from the National Health and Medical Research Council, during the conduct of the study.
BR, MM and DJK report no conflicts of interest in this work.
RRG has provided consulting services to Merck and Teva and has been a medico-legal expert witness for Queensland Health, NSW Nurses Federation, NSW Health and NSW Director of Public Prosecutions.
SWL has no conflicts of interests related to the research or results reported in this paper. In the interests of full disclosure, commercial interests are listed below. SWL has received consulting and personal fees and grants from the Atlanta Falcons, Pegasus Capital Advisors, KBR Wyle Services, EyeJust Inc, Paul, Weiss, Rifkind, Wharton & Garrison LLP, McCullough Hill Leary PS, View Inc, Rec Room, Delos Living, Estee Lauder, Roxbury Latin School, MIT, Stockgrand Ltd, from the CRC for Alertness, Safety and Productivity, Australia (nonprofit), Vanda Pharmaceuticals, Phillips Lytle, Wiley (Journal of Pineal Research), Atlanta Hawks, BHP Billiton, Noble Insights, Slingshot Insights and Team C Racing; honoraria and/or paid travel from BHP Billiton, DIN, Emory University, IES, Ineos, SLTBR, Solemma and Teague; has current consulting contracts with Akili Interactive, Apex2100 Ltd., Consumer Sleep Solutions, Headwaters Inc, Hintsa Performance AG, Light Cognitive, Lighting Science Group Corporation, Mental Workout, PlanLED, Six Senses, Stantec and KBR Wyle Services; has received unrestricted equipment gifts from Bionetics Corporation and F. Lux Software LLC and royalties from Oxford University Press; and has served as a paid expert in legal proceedings related to light, sleep and health. He holds a patent through Harvard University and Brigham and Women’s Hospital for “Systems and Methods for Determining and/or Controlling Sleep Quality.” He has 50% ownership (no monies received to date) from iSleep PTY. He is a member, Scientific Advisory Board (unpaid) for Midwest Lighting Institute.
SWR reports that he has served as a consultant through his institution to Vanda Pharmaceuticals, Philips Respironics, EdanSafe, The Australian Workers’ Union, National Transport Commission, Transport Accident Commission, Teva Pharmaceuticals, and Circadian Therapeutics, and has through his institution received research grants and/or unrestricted educational grants from Vanda Pharmaceuticals, Takeda Pharmaceuticals North America, Philips Lighting, Philips Respironics, Cephalon, Rio Tinto, BHP Billiton and ResMed Foundation, and reimbursements for conference travel expenses from Vanda Pharmaceuticals. His institution has received equipment donations or other support from Optalert™, Compumedics, and Tyco Healthcare. He has also served as an expert witness and/or consultant to shift work organizations. He served as a Program Leader for the CRC for Alertness, Safety and Productivity, Australia, and is the current Chair of the Sleep Health Foundation.
References
1. Barger LK, Lockley SW, Rajaratnam SM, Landrigan CP. Neurobehavioral, health, and safety consequences associated with shift work in safety-sensitive professions. Curr Neurol Neurosci Rep. 2009;9(2):155–164. doi:10.1007/s11910-009-0024-7
2. Rajaratnam SMW, Arendt J. Health in a 24-h society. Lancet. 2001;358(9286):999–1005. doi:10.1016/S0140-6736(01)06108-6
3. Brown TM. Melanopic illuminance defines the magnitude of human circadian light responses under a wide range of conditions. J Pineal Res. 2020;69(1):e12655. doi:10.1111/jpi.12655
4. Cajochen C. Alerting effects of light. Sleep Med Rev. 2007;11(6):453–464. doi:10.1016/j.smrv.2007.07.009
5. Souman JL, Tinga AM, Te Pas SF, van Ee R, Vlaskamp BNS. Acute alerting effects of light: a systematic literature review. Behav Brain Res. 2018;337:228–239. doi:10.1016/j.bbr.2017.09.016
6. Ruger M, Gordijn MC, Beersma DG, de Vries B, Daan S. Time-of-day-dependent effects of bright light exposure on human psychophysiology: comparison of daytime and nighttime exposure. Am J Physiol Regul Integr Comp Physiol. 2006;290(5):R1413–R1420. doi:10.1152/ajpregu.00121.2005
7. Vandewalle G, Balteau E, Phillips C, et al. Daytime light exposure dynamically enhances brain responses. Curr Biol. 2006;16(16):1616–1621. doi:10.1016/j.cub.2006.06.031
8. Cajochen C, Zeitzer JM, Czeisler CA, Dijk D-J. Dose-response relationship for light intensity and ocular and electroencephalographic correlates of human alertness. Behav Brain Res. 2000;115(1):75–83. doi:10.1016/S0166-4328(00)00236-9
9. Phipps-Nelson J, Redman JR, Dijk DJ, Rajaratnam SM. Daytime exposure to bright light, as compared to dim light, decreases sleepiness and improves psychomotor vigilance performance. Sleep. 2003;26(6):695–700. doi:10.1093/sleep/26.6.695
10. Berson DM, Dunn FA, Takao M. Phototransduction by retinal ganglion cells that set the circadian clock. Science. 2002;295(5557):1070–1073. doi:10.1126/science.1067262
11. Gooley JJ, Rajaratnam SMW, Brainard GC, Kronauer RE, Czeisler CA, Lockley SW. Spectral responses of the human circadian system depend on the irradiance and duration of exposure to light. Sci Transl Med. 2010;2(31):31–33. doi:10.1126/scitranslmed.3000741
12. Brainard GC, Hanifin JP, Greeson JM, et al. Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor. J Neurosci. 2001;21(16):6405–6412. doi:10.1523/JNEUROSCI.21-16-06405.2001
13. Phipps-Nelson J, Redman JR, Schlangen LJ, Rajaratnam SM. Blue light exposure reduces objective measures of sleepiness during prolonged nighttime performance testing. Chronobiol Int. 2009;26(5):891–912. doi:10.1080/07420520903044364
14. Rahman SA, Flynn-Evans EE, Aeschbach D, Brainard GC, Czeisler CA, Lockley SW. Diurnal spectral sensitivity of the acute alerting effects of light. Sleep. 2014;37(2):271–281. doi:10.5665/sleep.3396
15. Thapan K, Arendt J, Skene DJ. An action spectrum for melatonin suppression: evidence for a novel non-rod, non-cone photoreceptor system in humans. J Physiol. 2001;535(Pt 1):261–267. doi:10.1111/j.1469-7793.2001.t01-1-00261.x
16. Vandewalle G, Schmidt C, Albouy G, et al. Brain responses to violet, blue, and green monochromatic light exposures in humans: prominent role of blue light and the brainstem. PLoS One. 2007;2(11):e1247. doi:10.1371/journal.pone.0001247
17. Chellappa SL, Steiner R, Blattner P, Oelhafen P, Götz T, Cajochen C. Non-visual effects of light on melatonin, alertness and cognitive performance: can blue-enriched light keep us alert? PLoS One. 2011;6(1):e16429. doi:10.1371/journal.pone.0016429
18. Sletten TL, Ftouni S, Nicholas CL, et al. Randomised controlled trial of the efficacy of a blue-enriched light intervention to improve alertness and performance in night shift workers. Occup Environ Med. 2017;74(11):792–801. doi:10.1136/oemed-2016-103818
19. Mills PR, Tomkins SC, Schlangen LJM. The effect of high correlated colour temperature office lighting on employee wellbeing and work performance. J Circadian Rhythms. 2007;5(1):2. doi:10.1186/1740-3391-5-2
20. Viola AU, James LM, Schlangen LJM, Dijk DJ. Blue-enriched white light in the workplace improves self-reported alertness, performance and sleep quality. Scand J Work Environ Health. 2008;34(4):297–306. doi:10.5271/sjweh.1268
21. Keis O, Helbig H, Streb J, Hille K. Influence of blue-enriched classroom lighting on students’ cognitive performance. Trends Neurosci Educ. 2014;3(3–4):86–92. doi:10.1016/j.tine.2014.09.001
22. Hoffmann G, Gufler V, Griesmacher A, et al. Effects of variable lighting intensities and colour temperatures on sulphatoxymelatonin and subjective mood in an experimental office workplace. Appl Ergon. 2008;39(6):719–728. doi:10.1016/j.apergo.2007.11.005
23. Lowden A, Akerstedt T. Assessment of a new dynamic light regimen in a nuclear power control room without windows on quickly rotating shiftworkers–effects on health, wakefulness, and circadian alignment: a pilot study. Chronobiol Int. 2012;29(5):641–649. doi:10.3109/07420528.2012.675850
24. Motamedzadeh M, Golmohammadi R, Kazemi R, Heidarimoghadam R. The effect of blue-enriched white light on cognitive performances and sleepiness of night-shift workers: a field study. Physiol Behav. 2017;177:208–214. doi:10.1016/j.physbeh.2017.05.008
25. Barger LK, Sullivan JP, Lockley SW, Czeisler CA. Exposure to short wavelength-enriched white light and exercise improves alertness and performance in operational NASA flight controllers working overnight shifts. J Occup Environ Med. 2021;63(2):111–118. doi:10.1097/JOM.0000000000002054
26. Chen Y, Broman AT, Priest G, Landrigan CP, Rahman SA, Lockley SW. The effect of blue-enriched lighting on medical error rate in a university hospital ICU. Jt Comm J Qual Patient Saf. 2021;47(3):165–175. doi:10.1016/j.jcjq.2020.11.007
27. Jensen MA, Garde AH, Kristiansen J, Nabe-Nielsen K, Hansen AM. The effect of the number of consecutive night shifts on diurnal rhythms in cortisol, melatonin and heart rate variability (HRV): a systematic review of field studies. Int Arch Occup Environ Health. 2016;89(4):531–545. doi:10.1007/s00420-015-1093-3
28. Dinges DF, Powell JW. Microcomputer analyses of performance on a portable, simple visual RT task during sustained operations. Behav Res Methods Instrum Comput. 1985;17(6):652–655. doi:10.3758/BF03200977
29. Lockley SW, Dijk D-J, Kosti O, Skene DJ, Arendt J. Alertness, mood and performance rhythm disturbances associated with circadian sleep disorders in the blind. J Sleep Res. 2008;17(2):207–216. doi:10.1111/j.1365-2869.2008.00656.x
30. Zeitzer JM, Dijk DJ, Kronauer R, Brown E, Czeisler C. Sensitivity of the human circadian pacemaker to nocturnal light: melatonin phase resetting and suppression. J Physiol. 2000;526(Pt 3):695–702. doi:10.1111/j.1469-7793.2000.00695.x
31. International Commission on Illumination. CIE System for Metrology of Optical Radiation for ipRGC-Influenced Responses to Light; 2018. CIE S 026/E:2018. doi:10.25039/S026.2018
32. Ganesan S, Magee M, Stone JE, et al. The impact of shift work on sleep, alertness and performance in healthcare workers. Sci Rep. 2019;9(1):4635. doi:10.1038/s41598-019-40914-x
33. Dinges DF, Kribbs NB. Performing while sleepy: effects of experimentally induced sleepiness. In: Monk TH, editor. Sleep, Sleepiness and Performance. Chichester (UK): John Wiley & Sons Ltd; 1991.
34. Rajaratnam SM, Barger LK, Lockley SW, et al. Sleep disorders, health, and safety in police officers. JAMA. 2011;306(23):2567–2578. doi:10.1001/jama.2011.1851
35. Watson NF, Badr MS, Belenky G, et al. Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society on the recommended amount of sleep for a healthy adult: methodology and discussion. J Clin Sleep Med. 2015;11(8):931–952. doi:10.5664/jcsm.4950
36. Shekleton JA, Rajaratnam SM, Gooley JJ, Van Reen E, Czeisler CA, Lockley SW. Improved neurobehavioral performance during the wake maintenance zone. J Clin Sleep Med. 2013;9(4):353–362. doi:10.5664/jcsm.2588
37. Cohen DA, Wang W, Wyatt JK, et al. Uncovering residual effects of chronic sleep loss on human performance. Sci Transl Med. 2010;2(14):14ra13. doi:10.1126/scitranslmed.3000458
38. Chang AM, Aeschbach D, Duffy JF, Czeisler CA. Evening use of light-emitting eReaders negatively affects sleep, circadian timing, and next-morning alertness. Proc Natl Acad Sci U S A. 2015;112(4):1232–1237. doi:10.1073/pnas.1418490112
39. Gronli J, Byrkjedal IK, Bjorvatn B, Nodtvedt O, Hamre B, Pallesen S. Reading from an iPad or from a book in bed: the impact on human sleep. A randomized controlled crossover trial. Sleep Med. 2016;21:86–92. doi:10.1016/j.sleep.2016.02.006
40. Figueiro MG, Rea MS, Bullough JD. Circadian effectiveness of two polychromatic lights in suppressing human nocturnal melatonin. Neurosci Lett. 2006;406(3):293–297. doi:10.1016/j.neulet.2006.07.069
41. Brainard GC, Hanifin JP, Warfield B, et al. Short-wavelength enrichment of polychromatic light enhances human melatonin suppression potency. J Pineal Res. 2015;58(3):352–361. doi:10.1111/jpi.12221
42. Cajochen C, Krauchi K, Danilenko KV, Wirz-Justice A. Evening administration of melatonin and bright light: interactions on the EEG during sleep and wakefulness. J Sleep Res. 1998;7(3):145–157. doi:10.1046/j.1365-2869.1998.00106.x
43. Vandewalle G, Collignon O, Hull JT, et al. Blue light stimulates cognitive brain activity in visually blind individuals. J Cogn Neurosci. 2013;25(12):2072–2085. doi:10.1162/jocn_a_00450
44. Perrin F, Peigneux P, Fuchs S, et al. Nonvisual responses to light exposure in the human brain during the circadian night. Curr Biol. 2004;14(20):1842–1846. doi:10.1016/j.cub.2004.09.082
45. International Agency for Research on Cancer. Night shift work. World Health Organisation; 2019. Available from: https://publications.iarc.fr/593. Accessed April 9, 2021.
46. Smith DJ, Kyle S, Forty L, et al. Differences in depressive symptom profile between males and females. J Affect Disord. 2008;108(3):279–284. doi:10.1016/j.jad.2007.10.001
47. Killgore WD, Muckle AE, Grugle NL, Killgore DB, Balkin TJ. Sex differences in cognitive estimation during sleep deprivation: effects of stimulant countermeasures. Int J Neurosci. 2008;118(11):1547–1557. doi:10.1080/00207450802323970
48. Saksvik IB, Bjorvatn B, Hetland H, Sandal GM, Pallesen S. Individual differences in tolerance to shift work–a systematic review. Sleep Med Rev. 2011;15(4):221–235. doi:10.1016/j.smrv.2010.07.002
49. Vandewalle G, Archer SN, Wuillaume C, et al. Effects of light on cognitive brain responses depend on circadian phase and sleep homeostasis. J Biol Rhythms. 2011;26(3):249–259. doi:10.1177/0748730411401736
50. Wright KP, Bogan RK, Wyatt JK. Shift work and the assessment and management of shift work disorder (SWD). Sleep Med Rev. 2013;17(1):41–54. doi:10.1016/j.smrv.2012.02.002
51. Sletten TL, Segal AY, Flynn-Evans EE, Lockley SW, Rajaratnam SM. Inter-individual differences in neurobehavioural impairment following sleep restriction are associated with circadian rhythm phase. PLoS One. 2015;10(6):e0128273. doi:10.1371/journal.pone.0128273
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.