Back to Journals » International Journal of Women's Health » Volume 11
12-month outcomes of the US patient cohort in the SONATA pivotal IDE trial of transcervical ablation of uterine fibroids
Authors Hudgens J, Johns DA, Lukes AS, Forstein DA , Delvadia D
Received 17 January 2019
Accepted for publication 6 June 2019
Published 5 July 2019 Volume 2019:11 Pages 387—394
DOI https://doi.org/10.2147/IJWH.S201912
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Elie Al-Chaer
Joseph Hudgens,1 D Alan Johns,2 Andrea S Lukes,3 David A Forstein,4 Dipak Delvadia5
1Department of Obstetrics and Gynecology, Eastern Virginia Medical School, Norfolk, VA, USA; 2Baylor Research Institute-Fort Worth, Fort Worth, TX, USA; 3Carolina Women’s Research and Wellness Center, Durham, NC, USA; 4Department of Obstetrics and Gynecology, Touro College of Osteopathic Medicine, New York, NY, USA; 5Department of Obstetrics and Gynecology, Drexel University College of Medicine, Philadelphia, PA, USA
Objective: The prospective SONATA pivotal Investigational Device Exemption (IDE) trial was performed in the United States (US) and Mexico to examine the safety and effectiveness of transcervical fibroid ablation (TFA) in the treatment of symptomatic uterine fibroids. This is an analysis of 12-month clinical outcomes in the US cohort.
Methods: TFA with the Sonata® System was performed on women with symptomatic uterine fibroids. The 12-month co-primary endpoints were reduction in menstrual blood loss and freedom from surgical reintervention. Symptom severity, quality of life, patient satisfaction, safety, and reductions in uterine and fibroid volumes were also evaluated.
Results: One hundred twenty-five patients were enrolled and treated in the US. Both co-primary endpoints were achieved in this US-based cohort, as 65.3% of patients reported ≥50% reduction in menstrual bleeding and 99.2% of patients were free from surgical reintervention. Symptom improvement was noted by 97.4% of patients and 98.3% were satisfied. Ninety-five percent of patients reported reduced menstrual bleeding at 12 months, and 86.8% noted >20% reduction. Significant mean improvements at 12 months were realized in both symptom severity and health-related quality of life (33.8 points and 45.8 points, respectively; all P<0.0001). Mean maximal fibroid volume reduction per patient was 63.8%. There was a 0% incidence of device related adverse events. Mean length of stay was 2.5 hrs and 50% of patients returned to normal activity within 1 day.
Conclusion: This analysis of US patients in the SONATA pivotal IDE trial demonstrates results consistent with those in the full cohort. TFA with Sonata significantly reduced fibroid symptoms with a low surgical reintervention rate through 12 months. These results support the efficacy and safety of the Sonata system as a first-line treatment for women affected by symptomatic uterine fibroids.
Keywords: uterine leiomyoma, transcervical fibroid ablation; TFA, radiofrequency ablation, Sonata system
Introduction
The SONATA pivotal IDE trial (“SONATA trial;” ClinicalTrials.gov Identifier NCT02228174) was a prospective, longitudinal, multicenter, single-arm trial of transcervical fibroid ablation (TFA) with the Sonata® system in a cohort of women with symptomatic uterine fibroids.1 The primary endpoints at 12 months have been reported, and follow up is ongoing through 36 months. The overall trial population of 147 women were made up of patients from the US (n=125) and Mexico (n=22). Previous clinical results with the Sonata system demonstrated significant improvements in menstrual bleeding, quality of life and overall symptom severity in conjunction with excellent safety and surgical reintervention; total fibroid volume was reduced by 62%-67% at 12 months.1,2 This analysis examines the 12-month clinical outcomes of TFA with the Sonata system in the US patient cohort from the full SONATA trial.
Materials and methods
The Sonata system (Gynesonics, Inc; Redwood City, CA, USA), which has received United States Food and Drug Administration (FDA) clearance and Conformité Européene (CE) Mark, has been described in detail.2–5 Previously known as VizAblate™, the Sonata system (Figure 1) consists of a custom RF generator, a laptop-based ultrasound system with custom Graphical Guidance Software (GGS), and a single-use Radiofrequency Ablation (RFA) Handpiece integrally combined with a reusable Intrauterine Ultrasound (IUUS) Probe in a single treatment device. This system provides the gynecologist with a real-time, image-guided transcervical treatment option. Therapeutic radiofrequency energy is delivered to the fibroid according to a treatment cycle that is dependent on ablation size. Each ablation is scalable and determined graphically by the treating physician, maximizing the percentage of each fibroid to be ablated while avoiding thermal injury to adjacent viscera. The anesthesia choice for each patient may be individualized, as there is no requirement for general anesthesia; conscious sedation, and regional anesthesia (all with or without adjuvant paracervical blockade) were options provided within the SONATA clinical trial.
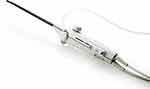 |
Figure 1 The Sonata treatment device, combining an intrauterine sonography probe with a radiofrequency ablation handpiece into a single integrated handpiece. |
Premenopausal women between ages 25–50 with symptomatic uterine fibroids associated with heavy menstrual bleeding were eligible for the SONATA trial if they had up to 10 myomata of International Federation of Gynecology and Obstetrics (FIGO) types 1, 2, 3, 4, and/or 2-5 (transmural) between 1–5 cm in diameter, experienced heavy menstrual bleeding as documented by a pictorial blood loss assessment chart (PBAC) score between 150–500, and had at least one fibroid that either indented (FIGO types 1, 2, 2-5) or abutted (FIGO type 3) the endometrial cavity. While type 5 and type 6 subserous myomata did not count towards the total number of fibroids, they could be ablated at the discretion of the investigator. Women were excluded if they desired fertility, had type 0 fibroids 1 cm or larger, had multiple endometrial polyps or any polyps ≥1.5 cm in diameter, a history of endometrial or fibroid ablation, prior uterine artery embolization (UAE), current tubal implants and clinically significant adenomyosis or substantial fibroid calcification.
Patients underwent screening that included transvaginal sonography, contrast-enhanced magnetic resonance (MR) imaging, assessment of menstrual bleeding via a pictorial blood loss assessment chart (PBAC), symptomatology and quality of life evaluation using the Uterine Fibroid Symptom and Quality-of-Life (UFS-QoL) Questionnaire, and a recording of baseline general health status using the EQ-5D (EuroQoL) questionnaire. All patients underwent cervical cancer and endometrial hyperplasia/carcinoma screening.
The US FDA approved the SONATA Investigational Device Exemption (IDE) trial in the US. Approvals were obtained from local and central institutional review boards (Table S1) prior to patient enrollment. The research was conducted according to the principles stated in the World Medical Association Declaration of Helsinki. Each patient provided her written informed consent to participate in the trial. All studies and records had protected health information deidentified.
The two co-primary endpoints of the SONATA trial, as required by the FDA, consisted of menstrual bleeding reduction at 12 months and freedom from surgical reintervention due to heavy menstrual bleeding (HMB) through 12 months. Overall study success required meeting the individual success criteria for both co-primary endpoints. Each patient was considered to have had a successful treatment outcome if she experienced ≥50% reduction in PBAC score with the score being <250. In terms of study success for this endpoint, the lower confidence limit (LCL) of the percentage of patient success was ≥45%. For the surgical reintervention co-primary endpoint, patient success was defined as a lack of surgical reintervention for HMB due to treatment failure at 12 months, and its study success criterion was a LCL of the percentage of patient success ≥75%.
Secondary endpoints included patient safety, reduction in total and perfused fibroid volumes at 12 months, change in the symptom severity score (SSS) and health-related quality of life (HRQOL) subscales of the UFS-QOL Questionnaire, overall patient treatment outcome using the Overall Treatment Effect (OTE) questionnaire, time to return to normal activity, patient satisfaction, change in general health outcome as determined by the EQ-5D questionnaire, pain and tolerance of the procedure, mean length of stay (LOS) and occurrence of pregnancy with pregnancy outcome. Treated patients were followed at 10 days, 30 days, 3 months, 6 months and at 12 months, with longer-term follow-up planned for 24 months and 36 months.
All statistical analyses were performed with SAS 9.3 (SAS, Cary, NC). Values were considered significant at the level of α=0.05.
Results
Twenty-one clinical study sites in the US enrolled and treated 125 patients. Baseline patient and fibroid characteristics are provided in Table 1 and Table 2, respectively. Mean age of enrolled patients was 43.4±4.0 years with a mean body mass index (BMI) of 29.5±6.4 (kg/m2). Patients were 48% White, 39% Black, and 17% Latina (Table 1).
 |
Table 1 Baseline Patient Characteristics |
 |
Table 2 Baseline PBAC and Fibroid Characteristics, Full Analysis Set (N=122) |
Three patients were excluded from the Full Analysis Set population as they were diagnosed with menopause during the 12 months after TFA with the Sonata system, with a resultant inability to provide a PBAC diary at their 12-month visits. Thus, the Full Analysis Set consists of 122 patients of the 125 patients. For the menstrual bleeding reduction endpoint, 121 of the 122 patients were included in this analysis, as one patient who underwent surgical reintervention prior to her 12-month visit was excluded from the analysis of this endpoint per the study statistical analysis plan. For the surgical reintervention endpoint evaluation, all 122 patients in the Full Analysis Set were included.
All 125 patients were treated in an outpatient setting, including 7 physician practice offices and 4 ambulatory care centers. Sixty-nine patients (55.2%) were treated in an operating room, 33 (26.4%) received their procedures in an ambulatory care center while 23 (18.4%) patients were treated in a physician office. Seventy-four patients (59.2%) received general anesthesia while 51 (40.8%) had their procedures under conscious sedation. Paracervical blockade was co-administered as an ancillary local anesthetic modality in 54 (43.2%) of patients. Mean length of stay (including procedure time) was 2.5±1.3 hrs, with 91 of 125 patients (72.8%) having a length of stay ≤3 hrs. Median length of stay was 2.3 hrs.
Nearly all patients (95%; 115/121) experienced some reduction in menstrual bleeding at 12 months after TFA, with 86.3% experiencing reduced menstrual bleeding by 3 months. Regarding the 12-month PBAC timepoint, 65.3% (79/121) of patients (95% CI 56.1–73.7%) reported at least 50% reduction in menstrual bleeding, which met the endpoint success criterion that the LCL of the 95% CI be ≥45%. Detailed PBAC reduction results are provided in Table 3.
 |
Table 3 Change in PBAC Score by Visit |
The other co-primary endpoint, surgical reintervention for heavy menstrual bleeding at 12 months post-ablation, was also met. Only one patient underwent surgical intervention (elective hysterectomy for abnormal uterine bleeding) within the first 12 months of her post-treatment course. Thus, 99.2% (95% CI 94.3–99.9%) of patients were free from surgical reintervention for heavy menstrual bleeding at 12 months after treatment with the Sonata system. This outcome met the endpoint success criterion that the LCL of the 95% CI be ≥75%. The percentage of patients achieving either co-primary endpoint did not significantly vary by ethnicity and were similar for White, Black and Latina patients.
Treatment with the Sonata system resulted in significant improvements in all patient-reported outcomes, beginning with the 3-month visit (the first post-treatment visit that included these questionnaires). As seen in Figure 2, patients reported a mean SSS reduction of 33.8 points (N=115) at 12 months post-procedure (P<0.0001) and a mean increase of 45.8 points in HRQOL (N=115; P<0.0001). Regarding the OTE questionnaire, 97.4% of patients responding at 12 months (112/115) noted improvement in their fibroid symptoms, while 1.7% (2/115) reported no change in symptoms and 0.9% (1/115) noted a worsening of symptoms.
Patients reported significantly improved health status on the EQ-5D questionnaire at 12 months post-procedure. Self-reported scores on the EQ-5D questionnaire range from values of less than 0, representing health states worse than “death”, to a maximum score of 1.0, representing “perfect health.” At baseline, US patients in the SONATA trial had a mean overall health score of 0.73 (N=122). At 12 months (N=114), their mean EQ-5D score rose to 0.90 (P<0.0001). Of note, health status significantly improved as early as 3 months after treatment with Sonata, with EQ-5D scores rose to 0.88 at 3 months (n=118; P<0.0001).
Of 115 patients reporting at 12 months, 98.3% of patients expressed satisfaction with their treatment, and a similar percentage (96.5%) would also recommend Sonata to a friend or family member. Specifically, 70.4% of reporting patients indicated that they were “very satisfied” with treatment, 18.3% were “moderately satisfied,” 9.6% were “somewhat satisfied,” 0.9% were “somewhat dissatisfied,” and 0.9% were “moderately dissatisfied” at 12 months. No patients were “very dissatisfied” with their treatment. Similarly, 82.6% (95/115) would “definitely recommend” treatment with Sonata, 13.9% would “probably recommend” it, and 3.5% would “probably not recommend” it at 12 months. No patient indicated that she would “definitely not recommend” the treatment.
Pain scores were evaluated on a scale from 0 (“No pain sensation”) to 10 (“Most intense pain imaginable”). Mean procedural pain scores were 0.01±0.1 for procedures under general anesthesia (N=74) and 0.6±1.6 for procedures performed under conscious sedation (N=51). Recovery pain scores were 3.4±2.9 for patients receiving general anesthesia and 2.5±2.7 for those who were treated under conscious sedation. Overall, 98% (122/125) of patients found treatment with the Sonata system to have been tolerable. Specifically, 63.2% (79/125) of patients reported the procedure to have been “Very Tolerable”, 32% (40/147) found the procedure to be “Moderately Tolerable”, 2.4% (3/147) characterized it as “Minimally Tolerable”, and 2.4% (3/147) said it was “Intolerable”. Overall mean pain scores regardless of anesthesia choice were 0.2±1.0 (range 0.0–7.0) during the procedure and 3.0±2.8 (range 0.0–10.0) during recovery (reported from the time between procedure completion and discharge).
Table 4 summarizes characteristics of ablated fibroids. The mean maximal reduction in total leiomyoma volume per patient from baseline to 12 months was 63.8% (N=109). Of the 121 patients included in the analysis of the bleeding reduction co-primary endpoint, there were 33 patients (27.3%) for whom a type 3 myoma was the sole qualifying fibroid type (ie, they did not have an indenting fibroid such as a type 2 myoma). Of these 33 patients, 22 (66.7%) had at least a 50% reduction in PBAC score at 12 months. This is similar to the percentages of patients with indenting fibroids who achieved a 50% or greater reduction in PBAC score at 12 months (58.3%, 67.4% and 63.0% for patients with at least one type 1 fibroid, patients without any type 1 fibroids and at least one type 2 fibroid, and patients without any type 1 and type 2 fibroids and at least one type 2-5 fibroid, respectively).
 |
Table 4 Attributes of Ablated Fibroids* |
Patients reported returning to normal daily activities in 2.1±2.3 days, and half of the patients returned to normal activity the day following the procedure. Patients with employment at the time of procedure indicated having returned to work 3.7±2.6 days post-procedure on average, with half of the patients returning to work within 3 days after the procedure. Patients resumed a normal diet at 0.7±1.3 days, normal sleep at 0.8±1.7 days, and normal urinary and bowel functions at 0.2±0.7 days and 1.5±1.9 days, respectively.
There were no occurrences (0.0%) of device related adverse events, serious or otherwise. There was 1 procedure-related serious adverse events reported in a single subject (0.8%). That single procedure-related serious adverse event involved a deep venous lower extremity thrombus diagnosed 15 days post-procedure, managed as an outpatient without sequelae.
As shown in Table 5, the results of the US cohort from the SONATA trial and that of the original cohort including patients from Mexico are provided.
 |
Table 5 Outcomes of the Full and US-only Cohorts of the SONATA Pivotal IDE Trial |
Discussion
TFA with the Sonata system provides a safe and effective transcervical treatment for symptomatic uterine fibroids that uses RF ablation to treat all types of nonpedunculated fibroids.5 Because it contains an integral intrauterine sonography probe, Sonata can image, target and treat fibroids that are not accessible to other transcervical methods such as hysteroscopic morcellation or resectoscopy. Indeed, in the SONATA clinical trial, 77.3% of treated fibroids in the US cohort were not amenable to operative hysteroscopy, including intramural, transmural and subserous myomata. While it is true that some type 1 and type 2 submucous myomata can be resected hysteroscopically, TFA obviates the inconvenience involved in removal of the resultant fibroid fragments, can address larger and deeper submucous fibroids that are less amenable to resection, and can be used in the same session to ablate other nonresectable fibroids without the need to also employ a resectoscope.
The US cohort represents 85% of patients treated in the SONATA trial, as 22 patients (15%) in the 147-patient SONATA pivotal IDE trial were enrolled in Mexico. Clinical outcomes for US patients in the SONATA trial were consistent with those in the full (US+Mexico) SONATA trial population. The US cohort results also met both co-primary endpoints of the clinical trial.
Results from this US cohort of the SONATA trial as well as those from the full trial are consistent with the results of the previous FAST-EU trial, in which 64.6% of patients (31/48) experienced a >50% reduction in menstrual bleeding and reported a mean reduction of 35.3 points in their SSS, both at 12 months.1,3 In both the full and US-only cohorts of the SONATA trial, 95% of patients had bleeding reduction, with 86.3% of patients experiencing bleeding reduction by 3 months. In the overall SONATA cohort of 147 patients, there were two reported procedure-related serious adverse events, one in Mexico and one in the US. There were no device related adverse events, serious or otherwise in the full SONATA or US cohorts. Patient satisfaction was similar in both groups.
The SONATA clinical trial was noteworthy for its inclusion of a wide variety of fibroid types (every FIGO fibroid type other than pedunculated fibroids [types 0 and 7] are treatable by the Sonata system) and increased number of treatable fibroids/patient compared with the prior FAST-EU clinical trial in Europe and Mexico.2,3 The SONATA trial included a robust patient selection process to minimize confounding factors, excluding patients with other etiologies of abnormal uterine bleeding such as anovulation and bleeding disorders. Furthermore, the study included a mix of patient-reported outcomes to complement the objective reintervention and bleeding primary endpoints.
Patients who expressly desired fertility were excluded. This was secondary to ethical reasons as SONATA was a pivotal safety and effectiveness investigational study. Nonetheless, the SONATA trial includes 3-year follow up of patients and the reporting of any pregnancy outcomes should they occur.
Conclusion
This analysis of US patients in the SONATA pivotal IDE trial demonstrates results consistent with those in the full cohort. TFA with Sonata significantly reduced fibroid symptoms with a low surgical reintervention rate through 12 months. These results support the efficacy and safety of the Sonata system as a first-line treatment for women affected by symptomatic uterine fibroids.
Authors’ Data Sharing Statement
Will individual participant data be available (including data dictionaries)? No.
What data in particular will be shared: Not available.
What other study-related documents will be available? Not available.
When will data be available (start and end dates)? Not applicable.
By what access criteria will data be shared (including with whom, for what types of analyses, and by what mechanism)? Not applicable.
Acknowledgments
David B Toub, MD, MBA, Medical Director of Gynesonics, Inc, contributed to the preparation of this manuscript. Taraneh G Farazi, PhD, Vice-President of Clinical Affairs at Gynesonics contributed to the review of this manuscript. QST Consultations, Ltd provided biostatistical analysis for this study. The abstract of this paper was presented at the 2019 ACOG Annual Clinical and Scientific Meeting as a poster presentation. The poster’s abstract was published in a supplement to the journal Obstetrics & Gynecology (May 2019 - Volume 133 - Issue - p 113S doi: 10.1097/01.AOG.0000558838.56257.35 https://journals.lww.com/greenjournal/Abstract/2019/05001/Transcervical_Radiofrequency_Ablation_of_Uterine.391.aspx).
Disclosure
Joseph Hudgens reports he is one of the members at Speakers Bureau for Applied Medical. Andrea S Lukes reports grants from Gynesonics, Abbvie, Myovant, and Bayer, during the conduct of the study. David A Forstein reports grants from Gynesonics, Inc, during the conduct of the study. The institutions of each author received research support from Gynesonics, Inc. The authors report no other conflicts of interest in this work.
References
1. Chudnoff S, Guido R, Roy K, Levine D, Mihalov L, Garza-Leal JG. Ultrasound-guided transcervical ablation of uterine leiomyomas. Obstet Gynecol. 2019;133:13–22. doi:10.1097/AOG.0000000000003032
2. Brölmann H, Bongers M, Garza-Leal JG, et al. The FAST-EU trial: 12-month clinical outcomes of women after intrauterine sonography-guided transcervical radiofrequency ablation of uterine fibroids. Gynecol Surg. 2016;13:27–35. doi:10.1007/s10397-015-0915-3
3. Bongers M, Brölmann H, Gupta J, Garza-Leal JG, Toub D. Transcervical, intrauterine ultrasound-guided radiofrequency ablation of uterine fibroids with the VizAblate® system: three- and six-month endpoint results from the FAST-EU study. Gynecol Surg. 2015;12:61–70. doi:10.1007/s10397-014-0873-1
4. Garza-Leal JG, Toub D, León IH, et al. Transcervical, intrauterine ultrasound-guided radiofrequency ablation of uterine fibroids with the VizAblate system: safety, tolerability, and ablation results in a closed abdomen setting. Gynecol Surg. 2011;8:327–334. doi:10.1007/s10397-010-0655-3
5. Toub DB. A new paradigm for uterine fibroid treatment: transcervical, intrauterine sonography-guided radiofrequency ablation of uterine fibroids with the sonata system. Curr Obstet Gynecol Rep. 2017;6:67–73. doi:10.1007/s13669-017-0194-2
Supplementary material
 |
Table S1 List of each Institutional Review Board (IRB) |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

