Back to Journals » Cancer Management and Research » Volume 10
ZNF259 promotes breast cancer cells invasion and migration via ERK/GSK3β/snail signaling
Authors Liu B, Xing X, Li X, Guo Q, Xu T, Xu K
Received 20 May 2018
Accepted for publication 29 June 2018
Published 3 September 2018 Volume 2018:10 Pages 3159—3168
DOI https://doi.org/10.2147/CMAR.S174745
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Antonella D'Anneo
Bin Liu,1 Xiaojing Xing,1 Xiang Li,2 Qianxue Guo,1 Tonghong Xu,1 Ke Xu3
1Medical Oncology, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Shenyang, Liaoning Province, People’s Republic of China; 2Breast Surgery, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Shenyang, Liaoning Province, People’s Republic of China; 3Thoracic Surgery, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Shenyang, Liaoning Province, People’s Republic of China
Purpose: Zinc finger protein 259 (ZNF259), also known as ZPR1, is a zinc finger-containing protein that can bind the intracellular tyrosine kinase domain of EGFR. At present, our knowledge on ZNF259 in cancers is limited. Here, we aimed to explore the biological functions of ZNF259 in breast cancer and reveal their mechanisms.
Patients and methods: The expression of ZNF259 was measured in 133 cases of breast cancer by immunohistochemistry. The online database Kaplan–Meier (KM) Plotter Online Tool was used to analyze the relationship between ZNF259 expression and breast cancer patient survival prognosis. Plasmid transfection and small interfering RNA and inhibitor treatments were carried out to explore the functions of ZNF259 in breast cancer cell lines and its potential mechanism. Matrigel invasion and wound healing assays were performed to detect the invasion and migration ability of cancer cells. In addition, protein expressions in tissues and cells were determined by Western blotting.
Results: ZNF259 expression was much higher in breast cancer cells than in the adjacent normal breast duct glandular epithelial cells (75.94% vs 7.52%, P<0.001) and was closely related to the breast cancer patients’ TNM stages (P=0.013) and lymph node metastasis (P=0.021). Knockdown of ZNF259 could downregulate p-ERK, p-GSK3β, and Snail expression, and upregulate the expression of E-cadherin and ZO-1, and then it also inhibited invasion and migration by the breast cancer cell lines MCF-7 and MDA-MB-231. Correspondingly, ZNF259 transfection could upregulate p-ERK, p-GSK3β, and Snail expression, and downregulate E-cadherin and ZO-1 expression, which led to stronger invasion and migration abilities of cancer cells. Furthermore, the ERK inhibitor U0126 could reverse all these effects induced by ZNF259 transfection.
Conclusion: ZNF259 could promote breast cancer cell invasion and migration by activating the ERK/GSK3β/Snail signaling pathway.
Keywords: ZPR1, E-cadherin, ZO-1, U0126, p-ERK, p-GSK3β
Introduction
Despite great advances in diagnostic and therapeutic strategies in recent decades, breast cancer is still listed as the leading cause of cancer-related death in women worldwide,1–3 largely due to breast cancer cell invasion and metastasis.4,5 It has great significance to elucidate the invasion and metastasis mechanism of breast cancer cells, and to find new biological molecules for the development of new targeted drugs.
Zinc finger protein 259 (ZNF259), also known as ZPR1, was first identified in mammalian cells as a cytoplasmic protein that could bind the intracellular domain of the epidermal growth factor receptor (EGFR).6 ZNF259 is an evolutionarily conserved protein with 2 C4 zinc fingers and 2 specific conserved homology domains.6,7 ZNF259 has essential roles in embryonic development and cell apoptosis, and can translocate to the nucleus during the S phase to regulate the cell cycle progression and cell proliferation.8–12 However, so far, our knowledge on ZNF259 expression and biological functions in malignant tumors is limited.
In this study, we explored ZNF259 expression in breast cancer tissue and breast cancer cell lines for the first time, and investigated the roles of ZNF259 in the invasion and migration by breast cancer cells. We found that ZNF259 could activate extracellular signal-regulated kinase (ERK)/glycogen synthase kinase 3β (GSK3β)/Snail signaling, which plays a vital role in breast cancer cell invasion and migration.13,14 In ERK/GSK3β/Snail signaling, activated ERK inhibits the function of GSK3β in promoting Snail degradation and leads to upregulation of Snail.15–17 The inhibition of adhesion factors such as E-cadherin by Snail can directly lead to the invasion and metastasis by cancer cells.18–21 Our results reveal the functions and mechanism of ZNF259 in breast cancer cell invasion and migration, and provide a new potential molecular target for the therapy of breast cancer patients.
Materials and methods
Patients and specimens
This study was approved by the local Institutional Review Board of the Cancer Hospital of China Medical University and Liaoning Cancer Hospital and Institute. The patients whose tissue samples were used in this study provided written informed consent. Primary tumor specimens were obtained from 133 patients who were diagnosed with invasive ductal carcinoma and underwent complete resection at the Cancer Hospital of China Medical University and Liaoning Cancer Hospital and Institute between 2011 and 2013. The patients who underwent neoadjuvant radiotherapy and/or chemotherapy were excluded from the cohort of the present study. All patients received standard chemotherapy after surgery. The mean age of the patients was 51 years (range, 29–83 years). Lymph node metastases were identified in 61 of the 133 patients. The pathological Tumor-Node-Metastasis (p-TNM) staging system of the International Union Against Cancer (seventh edition) was used to classify specimens as stages I (n=29), II (n=50), and III (n=54). In addition, 12 pairs of fresh specimens, including both breast cancer and paired adjacent nontumor tissues, were stored at –80°C immediately after resection, for protein extraction.
Immunohistochemistry
All tissue specimens were fixed in neutral formaldehyde, embedded in paraffin, and sectioned (thickness, 4 µm). The streptavidin-peroxidase immunohistochemical method was used to improve staining. Tissue sections were incubated at 4°C overnight with ZNF259 rabbit monoclonal antibody (1:200, ab134970, Abcam, Cambridge, UK); PBS was used as a blank control. Sections were then incubated with biotin-labeled secondary antibodies (Ultrasensitive; MaiXin, Fuzhou, People’s Republic of China) at 37°C for 30 minutes, followed by diaminobenzidine for coloration. Finally, samples were lightly counterstained with hematoxylin, dehydrated in alcohol, and mounted. Two investigators blinded to the clinical data semi-quantitatively scored the slides by evaluating the staining intensity and percentage of stained cells in representative areas. The staining intensity was scored as 0 (no signal), 1 (weak), 2 (moderate), or 3 (high). The percentage of cells stained was scored as 1 (1%–25%), 2 (26%–50%), 3 (51%–75%), or 4 (76%–100%). A final score of 0–12 was obtained by multiplying the intensity and percentage scores. ZNF259 stain with a score ≥4 was considered positive. Tumor samples with scores between 0 and 3 were defined as negative expression.
Cell culture
MCF-10A, T47D, MCF-7, BT-549, MDA-MB-231, and MDA-MB-468 cell lines were obtained from American Type Culture Collection (Manassas, VA, USA). The cells were cultured in Roswell Park Memorial Institute 1640 (RPMI-1640) (Invitrogen, Carlsbad, CA, USA) containing 10% fetal calf serum (Invitrogen), 100 international units (IU)/mL penicillin (Sigma-Aldrich, St. Louis, MO, USA), and 100 µg/mL streptomycin (Sigma-Aldrich). Cells were grown on sterilized culture dishes and were passaged every 2 days following 0.25% trypsin (Invitrogen) digestion.
Plasmid transfection and small interfering RNA (siRNA) treatment
Plasmids pCMV6-ddk-myc and pCMV6-ddk-myc-ZNF259 were purchased from Origene (RC205721, Rockville, MD, USA). ZNF259-siRNA (sc-35282) and NC-siRNA (sc-37007) were purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Transfection was carried out using the Lipofectamine 3000 reagent (Invitrogen) according to the manufacturer’s instructions.
Western blotting
Total protein was extracted using a lysis buffer (Pierce, Rockford, IL, USA) and quantified with the Bradford method. Total protein samples (30 µg) were separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA). Membranes were incubated overnight at 4°C with the following primary antibodies: ZNF259 (rabbit monoclonal antibody, 1:200, ab134970; Abcam; mouse monoclonal antibody, 1:200, sc-398491; Santa Cruz Biotechnology), phosphorylated mitogen-activated protein kinase (p-MEK), MEK, p-ERK, ERK, p-GSK3β, GSK3β, and Snail (1:500; Cell Signaling Technology, Danvers, MA, USA), E-cadherin (1:500; BD Transduction Laboratories, Lexington, KY, USA), zonula occludens protein 1 (ZO-1) (1:500; Proteintech, Chicago, IL, USA), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:5,000; Sigma-Aldrich). Membranes were washed and subsequently incubated with peroxidase-conjugated anti-mouse or anti-rabbit IgG (Santa Cruz Biotechnology) at 37°C for 2 hours. Bound proteins were visualized using electrochemiluminescence (Pierce) and detected with a bioimaging system (DNR Bio-Imaging Systems, Jerusalem, Israel).
Matrigel invasion
Cell invasion assay was performed using a 24-well Transwell chamber with 8 µm pores (Costar, Cambridge, MA, USA). The inserts were coated with 20 µL Matrigel (1:3 dilution; BD Biosciences, San Jose, CA, USA). Twenty-four hours after transfection, cells were trypsinized, and 3×105 cells in 100 µL of serum-free medium were transferred to the upper Matrigel chamber for 18 hours. Media supplemented with 10% fetal bovine serum were added to the lower chamber as a chemoattractant. After incubation, cells that passed through the filter were fixed with 4% paraformaldehyde and stained with hematoxylin. The invasive cells were microscopically counted in 10 randomly selected high-power fields.
Wound healing assay
In cultures with cell density below 90%, 48 hours after transfection, wounds were created in confluent areas using a 1,000 µL pipette tip. Wound healing within the scrape line was observed at both 0 and 24 hours, and representative scrape lines for each cell line were photographed. Duplicate wells were examined for each condition, and each experiment was repeated 3 times. The optical wound distances were measured using Image J software (National Institute of Health, Bethesda, MD, USA).
Statistical analysis
Statistical Package for Social Science version 23.0 for Mac (IBM Corporation, Armonk, NY, USA) was used for all analyses. The Pearson’s χ2 test was performed to assess possible correlations between ZNF259 and clinicopathological factors. Mann–Whitney U test was performed for the image analysis of Western blotting results and invasive assay results. P<0.05 was considered to indicate statistically significant differences.
Results
ZNF259 is related to the malignant progress of breast cancer
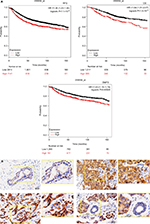
To investigate the functions of ZNF259 in malignant tumors, we first analyzed the relationship between its expression and different cancer patients’ survival prognosis, using an online database Kaplan–Meier (KM) Plotter Online Tool (http://www.kmplot.com). The data showed that ZNF259 gene expression significantly correlated with the breast cancer patients’ relapse-free survival (n=3,951, P=7.1×10-8), overall survival (n=1,402, P=1.5×10-5), and distant metastasis-free survival (n=1,746, P=0.00028) (Figure 1A).
We performed immunohistochemistry staining to analyze the expression of ZNF259 in 133 cases of breast cancer and adjacent noncancerous tissue specimens. The results showed that ZNF259 was very weakly positively stained in normal breast duct glandular epithelium cells (10/133, 7.52%, Figure 1B, Table 1). In breast cancer tissues, ZNF259 stain was much stronger than that in normal breast duct glandular epithelium cells, showing moderate to strong positive stain in the cytoplasm of cancer cells (101/133, 75.94%, P<0.001, Figure 1C and D, Table 1). Scattered nuclear stained cells were rarely visible, but not very obviously (Figure 1D). As Figure 1E shows, ZNF259 expression in breast cancer cells was much higher than that in adjacent normal breast duct glandular epithelium cells in the same field of view. Statistical analysis results indicated that ZNF259 expression was significantly correlated to the breast cancer patient’s TNM stages (P=0.013) and lymph node metastasis (P=0.021) but had no significant association with age (P=0.206, Table 2).
  | Table 1 ZNF259 expression in adjacent normal breast tissue and breast cancer tissue Note: aP<0.001, indicating statistical significance. Abbreviation: ZNF259, zinc finger protein 259. |
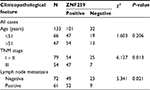  | Table 2 The relevance of ZNF259 expression to clinicopathological characteristics of breast cancer patients Abbreviation: ZNF259, zinc finger protein 259. |
ZNF259 knockdown could inhibit invasion and migration by breast cancer cells
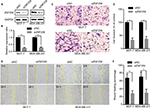
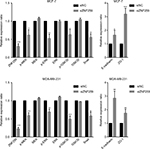
We measured the expression of ZNF259 in breast cancer tissues and cell lines by Western blotting. The results showed that in 12 pairs of fresh tissues, the relative expression rate of ZNF259 in breast cancer tissues was significantly higher than that in adjacent nontumor tissues (P<0.001, Figure 2A and B). In addition, in 5 breast cancer cell lines, T47D, MCF-7, BT-549, MDA-MB-231, and MDA-MB-468, ZNF259 expression was significantly higher than that in normal human mammary epithelial cell line MCF-10A (all P<0.001, Figure 2C and D). Furthermore, we knocked down ZNF259 with siRNAs in MCF-7 and MDA-MB-231 cell lines (MCF7, P=0.0011; MDA-MB-231, P=0.0021, Figure 3A and B). Transwell analysis showed that ZNF259 knockdown could inhibit invasion by the MCF-7 and MDA-MB-231 cells (MCF-7, P=0.0242; MDA-MB-231, P=0.0273, Figure 3C and D). Wound healing assay showed that the migration abilities were also suppressed by ZNF259 knockdown in MCF-7 and MDA-MB-231 cells (MCF-7, P=0.006; MDA-MB-231, P=0.0107, Figure 3E and F).
ZNF259 knockdown could upregulate E-cadherin and ZO-1 expression and downregulate p-MEK, p-ERK, p-GSK3β, and Snail expression
Western blot results showed that ZNF259 knockdown in both MCF-7 and MDA-MB-231 cell lines could downregulate p-MEK, p-ERK, p-GSK3β (Ser 9), and Snail expression, and then upregulate E-cadherin and ZO-1 expression, which could greatly affect the invasion and migration abilities of tumor cells (Figures 4 and S1). The total MEK, ERK, and GSK3β expression showed no obvious change.
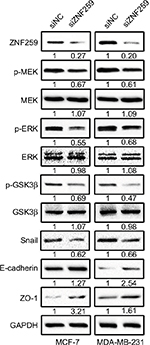  | Figure 4 ZNF259 knockdown could downregulate phosphorylated-extracellular signal-regulated kinase (p-ERK), phosphorylated-glycogen synthase kinase 3β (p-GSK3β), and Snail expression, and upregulate E-cadherin and zonula occludens protein 1 (ZO-1) expression. Notes: Western blotting was performed after ZNF259 knockdown in both MCF-7 and MDA-MB-231 cell lines, and phosphorylated mitogen-activated protein kinase (p-MEK), phosphorylated-extracellular signal-regulated kinase (p-ERK), phosphorylated-glycogen synthase kinase 3β (p-GSK3β) (Ser 9), and Snail expressions were downregulated, while E-cadherin and ZO-1 expression was upregulated. The total MEK, ERK, and GSK3β levels did not show obvious changes. The numbers indicated the relative expression ratios of the protein. At least 3 separate experiments were performed. The statistical analysis histogram is shown in the Figure S1. Abbreviation: ZNF259, zinc finger protein 259. |
ZNF259 could promote the invasion and migration ability of breast cancer cells by activating the ERK/GSK3β/Snail signaling pathway
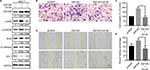
Furthermore, we transfected ZNF259 in MCF-7 cells and treated these cells with the ERK inhibitor U0126. The results showed that U0126 could inhibit the increase in p-ERK, p-GSK3β (Ser 9), and Snail levels induced by ZNF259 transfection, and then restore E-cadherin and ZO-1 expression (Figure 5A). Correspondingly, Transwell and wound healing assays showed that the increased cell invasion and migration caused by ZNF259 were also reversed by U0126 (P=0.0028, Figure 5B and C; P=0.0276, Figure 5D and E).
Discussion
The ZNF family members can regulate gene expression and play important roles in multiple biological processes, including proliferation, development, differentiation, autophagy, metabolism, apoptosis, and stemness maintenance.22–29 ZNF259 has essential roles in embryonic development, cell apoptosis, cycle progression, and proliferation,8–12 indicating that ZNF259 may also play a role in the development of malignant tumors. Our data revealed that ZNF259 was obviously upregulated in the cytoplasm of breast cancer cells, was significantly related to breast cancer patients’ TNM stages (P=0.013) and lymph node metastasis (P=0.021), and could promote invasion and migration by breast cancer cells. The data from KM Plotter Online Tool also showed that ZNF259 gene expression significantly correlated with breast cancer patients’ poor survival (relapse-free survival, n=3,951, P=7.1×10-8; overall survival, n=1,402, P=1.5×10-5; distant metastasis-free survival, n=1,746, P=0.00028), which better supported our results.
Furthermore, we confirmed that ZNF259 could upregulate the expression of p-MEK, p-ERK, p-GSK3β, and Snail, and downregulate E-cadherin and ZO-1 expression, which plays a central role in regulating invasion and migration by tumor cells.30,31 Snail is phosphorylated by GSK3β at the Ser/Thr residues and then degraded.15 Active ERK (p-ERK) can inhibit the function of GSK3β by phosphorylating it at Ser 9, leading to increased Snail levels.13,14,16,17 Meanwhile, both E-cadherin and ZO-1 are classic downstream target genes of Snail.18–21 Thus, our hypothesis was that ZNF259 downregulates E-cadherin and ZO-1 expression via activating p-ERK/p-GSK3β/Snail signaling and promotes invasion and migration by breast cancer cells. The ERK inhibitor U0126 could inhibit the increasing expression of p-ERK, p-GSK3β, and Snail caused by ZNF259 transfection, restored the expression of E-cadherin and ZO-1, and subsequently reversed the invasion and migration abilities of breast cancer cells. These results provide strong support for our above hypothesis, although it is still unclear how ZNF259 regulates p-ERK.
ZNF259 could bind the intracellular domain of the inactive EGFR in the cytoplasm,6 suggesting that ZNF259 may play a role in the EGFR signaling pathway activation process. The role of EGFR signaling in various kinds of malignant tumors has been well elucidated, and Ras/Raf/MEK/ERK is one of the classic downstream pathways of EGFR signaling.32–34 Our results showed that p-MEK could also be upregulated by ZNF259. Thus, ZNF259 may participate in the upstream regulation of Ras/Raf/MEK/ERK pathway. In addition, recently, Shan et al35 showed that ZNF259 plays a tumor suppressor role in non-small-cell lung cancer via focal adhesion kinase (FAK)/protein kinase B (AKT) signaling. In our study, we also observed the effect of ZNF259 on p-FAK and p-AKT; however, in breast cancer cells their expression did not show obvious change (data was not shown). This finding indicates that the function of ZNF259 may be tissue-specific, which is worthy of further research in future studies.
Conclusion
ZNF259 levels increased in breast cancer tissues and cell lines, and its high expression was significantly related to breast cancer patients’ TNM stage and lymph node metastasis. ZNF259 could promote invasion and migration by breast cancer cells via activating ERK/GSK3β/Snail signaling.
Acknowledgment
The authors thank the Department of Pathology of Cancer Hospital of China Medical University and Liaoning Cancer Hospital and Institute for providing tissue samples and technical support. The authors also thank Editage (www.editage.cn) for English language editing.
Disclosure
The authors report no conflicts of interest in this work.
References
Kohler BA, Sherman RL, Howlader N, et al. Annual report to the nation on the status of cancer, 1975–2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107(6):djv048. | ||
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics 2012. CA Cancer J Clin. 2015;65(2):87–108. | ||
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. | ||
Rakha EA. Pitfalls in outcome prediction of breast cancer. J Clin Pathol. 2013;66(6):458–464. | ||
Tremont A, Lu J, Cole JT. Endocrine therapy for early breast cancer: updated review. Ochsner J. 2017;17(4):405–411. | ||
Galcheva-Gargova Z, Konstantinov KN, Wu IH, Klier FG, Barrett T, Davis RJ. Binding of zinc finger protein ZPR1 to the epidermal growth factor receptor. Science. 1996;272(5269):1797–1802. | ||
Ruiz OE, Nikolova LS, Metzstein MM. Drosophila Zpr1 (zinc finger protein 1) is required downstream of both EGFR and FGFR signaling in tracheal subcellular lumen formation. PLoS One. 2012;7(9): e45649. | ||
Galcheva-Gargova Z, Gangwani L, Konstantinov KN, et al. The cytoplasmic zinc finger protein ZPR1 accumulates in the nucleolus of proliferating cells. Mol Biol Cell. 1998;9(10):2963–2971. | ||
Gangwani L, Mikrut M, Galcheva-Gargova Z, Davis RJ. Interaction of ZPR1 with translation elongation factor-1α in proliferating cells. J Cell Biol. 1998;143(6):1471–1484. | ||
Gangwani L, Flavell RA, Davis RJ. ZPR1 is essential for survival and is required for localization of the survival motor neurons (SMN) protein to Cajal bodies. Mol Cell Biol. 2005;25(7):2744–2756. | ||
Dyczkowski J, Vingron M. Comparative analysis of cell cycle regulated genes in eukaryotes. Genome Inform. 2005;16(1):125–131. | ||
Gangwani L. Deficiency of the zinc finger protein ZPR1 causes defects in transcription and cell cycle progression. J Biol Chem. 2006;281(52):40330–40340. | ||
Li S, Lu J, Chen Y, et al. MCP-1-induced ERK/GSK-3β/Snail signaling facilitates the epithelial-mesenchymal transition and promotes the migration of MCF-7 human breast carcinoma cells. Cell Mol Immunol. 2017;14(7):621–630. | ||
Zhang H, Zhou GL. CAP1 (Cyclase-Associated Protein 1) exerts distinct functions in the proliferation and metastatic potential of breast cancer cells mediated by ERK. Sci Rep. 2016;6:25933. | ||
Xu Y, Lee SH, Kim HS, et al. Role of CK1 in GSK3β-mediated phosphorylation and degradation of snail. Oncogene. 2010;29(21):3124–3133. | ||
Ngo HK, Lee HG, Piao JY, et al. Helicobacter pylori induces Snail expression through ROS-mediated activation of Erk and inactivation of GSK-3β in human gastric cancer cells. Mol Carcinog. 2016;55(12):2236–2246. | ||
Wei L, Yao Y, Zhao K, et al. Oroxylin A inhibits invasion and migration through suppressing ERK/GSK-3β signaling in snail-expressing non-small-cell lung cancer cells. Mol Carcinog. 2016;55(12):2121–2134. | ||
Dong C, Wu Y, Yao J, et al. G9a interacts with Snail and is critical for Snail-mediated E-cadherin repression in human breast cancer. J Clin Invest. 2012;122(4):1469–1486. | ||
Li H, Li M, Xu D, Zhao C, Liu G, Wang F. Overexpression of Snail in retinal pigment epithelial triggered epithelial-mesenchymal transition. Biochem Biophys Res Commun. 2014;446(1):347–351. | ||
Ohkubo T, Ozawa M. The transcription factor Snail downregulates the tight junction components independently of E-cadherin downregulation. J Cell Sci. 2004;117(Pt 9):1675–1685. | ||
Wang Y, Shi J, Chai K, Ying X, Zhou BP. The role of snail in EMT and tumorigenesis. Curr Cancer Drug Targets. 2013;13(9):963–972. | ||
Arenzana TL, Schjerven H, Smale ST. Regulation of gene expression dynamics during developmental transitions by the Ikaros transcription factor. Genes Dev. 2015;29(17):1801–1816. | ||
Krebs CJ, Zhang D, Yin L, Robins DM. The KRAB zinc finger protein RSL1 modulates sex-biased gene expression in liver and adipose tissue to maintain metabolic homeostasis. Mol Cell Biol. 2014;34(2):221–232. | ||
Zhang L, Zhang W, Li Y, et al. SHP-2-upregulated ZEB1 is important for PDGFRα-driven glioma epithelial-mesenchymal transition and invasion in mice and humans. Oncogene. 2016;35(43):5641–5652. | ||
Yang L, Wang H, Kornblau SM, et al. Evidence of a role for the novel zinc-finger transcription factor ZKSCAN3 in modulating Cyclin D2 expression in multiple myeloma. Oncogene. 2011;30(11):1329–1340. | ||
Lai KP, Chen J, He M, et al. Overexpression of ZFX confers self-renewal and chemoresistance properties in hepatocellular carcinoma. Int J Cancer. 2014;135(8):1790–1799. | ||
Chauhan S, Goodwin JG, Chauhan S, et al. ZKSCAN3 is a master transcriptional repressor of autophagy. Mol Cell. 2013;50(1):16–28. | ||
Ma X, Huang M, Wang Z, Liu B, Zhu Z, Li C. ZHX1 inhibits gastric cancer cell growth through inducing cell-cycle arrest and apoptosis. J Cancer. 2016;7(1):60–68. | ||
Wang H, Deng X, Zhang J, et al. Elevated expression of zinc finger protein 703 promotes cell proliferation and metastasis through PI3K/AKT/GSK-3β signalling in oral squamous cell carcinoma. Cell Physiol Biochem. 2017;44(3):920–934. | ||
Gheldof A, Berx G. Cadherins and epithelial-to-mesenchymal transition. Prog Mol Biol Transl Sci. 2013;116:317–336. | ||
Polette M, Mestdagt M, Bindels S, et al. β-catenin and ZO-1: shuttle molecules involved in tumor invasion-associated epithelial-mesenchymal transition processes. Cells Tissues Organs. 2007;185(1–3):61–65. | ||
Rajaram P, Chandra P, Ticku S, Pallavi BK, Rudresh KB, Mansabdar P. Epidermal growth factor receptor: role in human cancer. Indian J Dent Res. 2017;28(6):687–694. | ||
Sigismund S, Avanzato D, Lanzetti L. Emerging functions of the EGFR in cancer. Mol Oncol. 2018;12(1):3–20. | ||
Alvarez RH, Valero V, Hortobagyi GN. Emerging targeted therapies for breast cancer. J Clin Oncol. 2010;28(20):3366–3379. | ||
Shan Y, Cao W, Wang T, Jiang G, Zhang Y, Yang X. ZNF259 inhibits non-small cell lung cancer cells proliferation and invasion by FAK-AKT signaling. Cancer Manag Res. 2017;9:879–889. |
Supplementary material
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.