Back to Journals » Drug Design, Development and Therapy » Volume 8
Women taking the “blue pill” (sildenafil citrate): such a big deal?
Authors Lo Monte G, Graziano A, Piva I, Marci R
Received 16 July 2014
Accepted for publication 18 August 2014
Published 7 November 2014 Volume 2014:8 Pages 2251—2254
DOI https://doi.org/10.2147/DDDT.S71227
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 6
Editor who approved publication: Professor Shu-Feng Zhou
Giuseppe Lo Monte, Angela Graziano, Isabella Piva, Roberto Marci
Department of Morphology, Surgery, and Experimental Medicine, University of Ferrara, Ferrara, Italy
Abstract: For years, phosphodiesterase type 5 inhibitors have been used for the treatment of erectile dysfunctions. Due to the similarities between male and female sexual response, several studies have assessed the effects of sildenafil citrate (Viagra®) in women affected by female sexual arousal disorder. The results are still conflicting and the drug is not devoid of adverse effects. Furthermore, female sexual arousal disorder is a heterogeneous condition whose underlying causes are difficult to diagnose and appropriate treatment requires a thorough sexual, psychological, and medical history along with specialist consultations. The clinician should pursue a global approach to the patient with sexual difficulties, while non-hormonal treatment such as phosphodiesterase type 5 inhibitors (ie, sildenafil citrate) should be kept as the last option.
Keywords: phosphodiesterase type 5 inhibitors, female sexual arousal disorder (FSAD), sildenafil citrate
Background
In clinical practice, not only gynecologists but also generalists are often asked to solve specific questions about sexual diseases. According to recent estimates, sexual dysfunction has occurred in 40%–45% of women and 20%–30% of men at least once in their lifetime.1 For years, studies have focused mainly on erectile dysfunction while female sexual disorders (FSDs), although more frequent than male sexual disturbances, have been hardly considered.2 Even general practitioners are used to dealing with male sexual dysfunction and often prescribe easily available drugs such as sildenafil citrate (Viagra®). On the contrary, women have begun to complain about sexual troubles only recently and have caught their doctors off-guard. How should FSD be diagnosed and treated? A more thorough understanding of female sexual problems is needed to ensure appropriate clinical management of sexual difficulties.
Discussion
Clinical features of female sexual arousal disorder
The Diagnostic and Statistical Manual of Mental Disorder, Fifth Edition (DSM-V)2 defines FSDs as disturbances in the female sexual response cycle, resulting in marked distress and interpersonal difficulties. In particular, female sexual arousal disorder (FSAD) belongs to the so-called “female sexual interest/arousal disorder” and is one of the most prevalent subcategories of FSDs. FSAD is traditionally defined as a persistent or recurrent inability to attain or maintain adequate lubrication and genital swelling until completion of sexual activity.3 Such a clinical condition may depend either on local or general factors. In fact, genital congestion and lubrication strictly depend on the hormonal balance (ie, arousal disorders during the menopausal transition) and require adequate vascular function and an efficient nerve transmission of mechanical stimuli.4 Thus, iatrogenic factors (eg, chronic antidepressant treatments, surgical procedures, radiotherapy of the pelvis), along with endocrine, vascular, and neurological disorders can cause female sexual dysfunction. However, a modern definition of FSDs should not focus only on genital phenomena without considering the psychological aspect of arousal. In fact, women very often relate arousal to the subjective feeling of been “sexually involved” more than to the physiological response to erotic stimuli (ie, vaginal lubrication and engorgement of sexual tissues such as nipples, vulva, clitoris, and vaginal walls). Thus, such factors as sexual inhibition, depression or anxiety, inadequate sexual stimulation, or interpersonal problems may impair women’s arousal. Moreover, FSADs are frequently associated with other FSDs concerning desire, orgasm, and pain.4 To sum up, Basson et al5 categorized FSAD into three main classes: subjective, genital, and combined (Table 1). In subjective FSAD, the woman’s emotional response to sexual stimuli is altered but the physical response still occurs, while genital phenomena are impaired in objective FSAD. However, in most cases, both factors contribute to FSAD (combined).
  | Table 1 Categories of female sexual arousal disorder |
Rationale for the use of sildenafil in the treatment of FSAD
In smooth muscle cells, nitric oxide activates the guanylate cyclase enzyme which converts guanosine triphosphate into cyclic guanosine monophosphate. This molecule promotes the relaxation of the smooth muscle cells, causes vasodilatation, and increases blood flow in genital organs. The engorgement of penile corpora cavernosa in men and clitoris and labia minora in women are the main modifications of genital organs during sexual arousal. Furthermore, the ultrafiltration of plasma through capillary vaginal vessels contributes to vaginal lubrication.6 Phosphodiesterase type 5 (PDE5) inhibitors (eg, sildenafil, tadalafil, vardenafil) physiologically enhance the production of guanosine monophosphate from cyclic guanosine monophosphate, thus contrasting the above-mentioned effects (Figure 1). According to emerging data, PDE5 is expressed in vaginal, clitoral, and labial smooth muscles. Thus, PDE5 inhibitors could be used as an easily available medical treatment for genital FSADs.7 Sildenafil citrate (Viagra), a PDE5 inhibitor, has been successfully used for the treatment of male erectile dysfunction since the late 1990s.8 The analogies between male and female physiological response to sexual stimuli have prompted studies over its efficacy in FSAD. However, there are presently very few data regarding the use of sildenafil in women and the results are still conflicting. In a randomized double-blind study, Berman et al administered sildenafil to 202 postmenopausal women with a primary diagnosis of FSAD and possibly associated secondary FSDs. Patients randomly received 12 weeks of treatment with sildenafil citrate 50 mg or placebo. A significant improvement of arousal sensation, lubrication, and orgasm was reported in the study group affected by primary FSAD. On the contrary, women affected by different types of FSDs (eg, hypoactive sexual disorder, dyspareunia due to vaginal dryness) did not experience any significant improvement in desire, pain, or enjoyment.9 In all the studies confirming the positive results of sildenafil, the drug proved to be more effective in the genital subgroup. It can be speculated that sildenafil citrate only acts on the physical phenomena of arousal and does not completely respond to the complexity of FSAD. In fact, encouraging results were achieved in specific groups of patients affected by secondary FSADs (eg, diabetes mellitus, multiple sclerosis, chronic antidepressant users) in which the genital arousal disorder is clearly connected with a neurological or vascular injury.10–12 However, negative results are also reported in literature. Firstly, women affected by FSADs do not benefit from sildenafil citrate as much as men with erectile dysfunction as a result of a lower concentration of PDE5 in vaginal and clitoral tissues than in penile corpora cavernosa.7 Secondly, a randomized double-blind study including both pre- and postmenopausal women did not show any success from sildenafil citrate treatment. In fact, no significant changes in physical response during sexual activity were reported in either group of women.13 It should also be considered that sildenafil citrate is not devoid of side effects. The most common adverse events reported in all studies were headache, flushing, nausea, rhinitis, and visual disturbances.14 In addition, sildenafil is strictly contraindicated in patients using oral or transdermal nitrates, as it dangerously potentiates the hypotensive effect of these drugs.8 Due to these major drawbacks as well as to the lack of conclusive results, non-hormonal treatments for FSAD have not been approved yet.
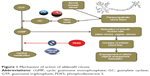  | Figure 1 Mechanism of action of sildenafil citrate. |
Only a few studies have evaluated the role of other PDE5 inhibitors in FSADs. Van der Made et al examined the efficacy of vardenafil alone or combined with testosterone in28 women affected by FSADs and/or hypoactive sexual desire disorder. The combination of testosterone and vardenafil caused an improvement in genital response (ie, vaginal pulse amplitude at vaginal photoplethysmography), especially in a group that initially had low attention for sexual cues.15 The efficacy of tadalafil was evaluated in 33 type 1 premenopausal diabetic women affected by sexual genital arousal disorder. A daily dose of tadalafil 5 mg treatment seemed to improve subjective sexual aspects. It was suggested that this treatment may be effective in improving clitoral blood flow as well as the clitoral functional system, consisting of smooth muscle cells and vascular spaces.16
Practical advice for clinicians
There are a few things that a generalist should bear in mind when addressing a woman with FSAD. Firstly, which type of women may suffer from FASD? Especially postmenopausal patients in whom lubrication and swelling are physiologically impaired. However, even psychological and cultural factors due to aging (ie, change in the familial structure, departure of the siblings, retirement) should be taken into account when addressing an elderly woman with sexual difficulties. In particular, the success of a treatment should be based on a detailed “couple interview.” In case of partnership involvement, the information has to provide the investigator with insight into the level of sexual function and the sexual preference of both partners; otherwise, all therapeutic steps could remain ineffective.17
Secondly, the “blue miracle pill,” although easily available, is not always the right cure. In fact FSAD, as a complex disease, may depend on several underlying medical conditions that need to be appropriately diagnosed and treated. Therefore, the general practitioner should immediately exclude chronic antidepressant treatments, surgical procedures, or radiotherapy of the pelvis and ascertain the presence of possible systemic pathologies such as endocrine, vascular, and neurological disorders. In these cases, the patient should be referred to the appropriate specialist (eg, endocrinologist, surgeon, neurologist). Nevertheless, in the absence of any evident systemic pathology, a generalist should propose both a gynecological and psychological consultation. In fact, objective FSAD (reduced genital physical response to sexual stimuli) may be a sign of various gynecological disorders. As reduced lubrication can cause pain during sexual intercourse, it should be differentiated from other possible causes of dyspareunia such as endometriosis, vulvar vestibulitis syndrome, and recurrent genital or urinary infections. On the contrary, preserved physical response with reduced emotional involvement probably indicates a psychological disturbance that can benefit from cognitive behavioral therapy or traditional sex therapy. However, in most cases, the reasons for FSAD are mixed and difficult to identify. Thus, such a disturbance may persist over time and require the continuous assistance a generalist can provide, unlike a specialist.
Conclusion
FSAD is a complex disease, whose underlying cause is difficult to diagnose. Preliminary evaluation of the sexual, psychological, and medical history is mandatory to exclude possible systemic diseases and to identify the type of FSAD. Generalists should pursue a global approach to the patient with sexual difficulties, while non-hormonal treatment such as PDE5 inhibitors (eg, sildenafil citrate) should be kept as the last option.
Author contributions
IP, GLM, and AG have substantially contributed to the interpretation of data and literature analysis. They also concurred to design, prepare, draft, and revise the final version of the manuscript. RM has substantially contributed to critical literature revision, preparation, drafting, and revising the final version of the manuscript. He also gave extremely important intellectual support and ensured the general supervision of the research group. All authors read and approved the final manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Lewis RW, Fugl-Meyer KS, Bosch R, et al. Epidemiology/risk factors of sexual dysfunction. J Sex Med. 2004;1(1):35–39. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association; 2013. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Health Disorders, Fourth Edition, Text Revision. Arlington, VA: American Psychiatric Association; 2003. | ||
Graziottin A, Leiblum SR. Biological and psychosocial pathophysiology of female sexual dysfunction during the menopausal transition. J Sex Med. 2005;2(Suppl 3):133–145. | ||
Basson R, Leiblum S, Brotto L, et al. Revised definitions of women’s sexual dysfunction. J Sex Med. 2004;1(1):40–48. | ||
Gragasin FS, Michelakis ED, Hogan A, et al. The neurovascular mechanism of clitoral erection: nitric oxide and cGMP-stimulated activation of BKCa channels. FASEB J. 2004;18(12):1382–1391. | ||
Uckert S, Ellinghaus P, Albrecht K, Jonas U, Oelke M. Expression of messenger ribonucleic acid encoding for phosphodiesterase isoenzymes in human female genital tissues. J Sex Med. 2007;4(6):1604–1609. | ||
Licht MR. Sildenafil (Viagra) for treating male erectile dysfunction. Cleve Clin J Med. 1998;65(6):301–304. | ||
Berman JR, Berman LA, Toler SM, Gill J, Haughie S; Sildenafil Study Group. Safety and efficacy of sildenafil citrate for the treatment of female sexual arousal disorder: a double-blind, placebo controlled study. J Urol. 2003;170(6 Pt 1):2333–2338. | ||
Caruso S, Rugolo S, Agnello C, Intelisano G, Di Mari L, Cianci A. Sildenafil improves sexual functioning in premenopausal women with type 1 diabetes who are affected by sexual arousal disorder: a double-blind, crossover, placebo-controlled pilot study. Fertil Steril. 2006;85(5):1496–1501. | ||
Dasgupta R, Wiseman OJ, Kanabar G, Fowler CJ, Mikol D. Efficacy of sildenafil in the treatment of female sexual dysfunction due to multiple sclerosis. J Urol. 2004;171(3):1189–1193. | ||
Nurnberg HG, Hensley PL, Heiman JR, Croft HA, Debattista C, Paine S. Sildenafil treatment of women with antidepressant-associated sexual dysfunction: a randomized controlled trial. JAMA. 2008;300(4):395–404. | ||
Basson R, McInnes R, Smith MD, Hodgson G, Koppiker N. Efficacy and safety of sildenafil citrate in women with sexual dysfunction associated with female sexual arousal disorder. J Womens Health Gend Based Med. 2002;11(4):367–377. | ||
Schoen C, Bachmann G. Sildenafil citrate for female sexual arousal disorder: a future possibility? Nat Rev Urol. 2009;6(4):216–222. | ||
van der Made F, Bloemers J, Yassem WE, et al. The influence of testosterone combined with a PDE5-inhibitor on cognitive, affective, and physiological sexual functioning in women suffering from sexual dysfunction. J Sex Med. 2009;6(3):777–790. | ||
Caruso S, Cicero C, Romano M, Lo Presti L, Ventura B, Malandrino C. Tadalafil 5 mg daily treatment for type 1 diabetic premenopausal women affected by sexual genital arousal disorder. J Sex Med. 2012;9(8):2057–2065. | ||
Beier KM, Loewit KK. Sexual Medicine in Clinical Practice. New York, NY: Springer; 2013. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
