Back to Journals » Cancer Management and Research » Volume 12
WD Repeat Domain 5 Promotes Invasion, Metastasis and Tumor Growth in Glioma Through Up-Regulated Zinc Finger E-Box Binding Homeobox 1 Expression
Authors Dai B, Xiao Z, Zhu G, Mao B, Huang H, Guan F, Lin Z, Peng W , Liang X, Zhang B, Hu Z
Received 6 November 2019
Accepted for publication 13 March 2020
Published 8 May 2020 Volume 2020:12 Pages 3223—3235
DOI https://doi.org/10.2147/CMAR.S237582
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjeev K. Srivastava
Bin Dai, Zhiyong Xiao, Guangtong Zhu, Beibei Mao, Hui Huang, Feng Guan, Zhenyang Lin, Weicheng Peng, Xin Liang, Bolun Zhang, Zhiqiang Hu
Department of Neurosurgery, Beijing Shijitan Hospital, Capital Medical University, Peking University Ninth School of Clinical Medicine, Beijing, People’s Republic of China
Correspondence: Zhiqiang Hu
Department of Neurosurgery, Beijing Shijitan Hospital, Capital Medical University, Peking University Ninth School of Clinical Medicine, No. 10 Tieyi Road, Yangfangdian, Haidian District, Beijing 10038, People’s Republic of China
Tel +86 10 63926627 Email [email protected]
Background: Glioma is one of the important diseases that threaten human survival in today’s society. WD repeat domain 5 (WDR5) belongs to the components of the lysine methyltransferase complex. WDR5 is involved in gene transcription regulation, cell senescence, cancer and other biological events through methylation modification. However, its expression and function in glioma are still unclear.
Materials and Methods: The expression of WDR5 was observed in glioma cells, and then a glioma cell line SW1783 knocked down WDR5 was established. The effects of the decrease of WDR5 expression on proliferation, migration, invasion and EMT of glioma cells were detected, respectively. The downstream regulators of WDR5 were identified by gene expression profiling technology, and the possible molecular mechanisms were identified.
Results: In this study, we found that WDR5 could promote glioma cell’s proliferation, migration, invasion and tumor metastasis. In glioma, especially in metastatic glioma tissues, WDR5 levels were significantly increased, the higher expression level could also cause a significant reduction in overall survival of glioma patients. Second, the ability of cells’ proliferation, migration, invasion and tumor metastasis was significantly reduced in WDR5 knockdown cell lines. We also found a significant change in the expression level of epithelial and mesenchymal markers in WDR5 knockdown cell lines. Furthermore, we found that knockdown of WDR5 could inhibit the expression of zinc finger E-box binding homeobox 1 (ZEB1), and knockdown of ZEB1 could inhibit invasion, migration and epithelial–mesenchymal transformation (EMT) in WDR5 over-expression cell line. We also found that WDR5 may regulate ZEB1’s expression through H3K4me3.
Conclusion: In summary, in this study, we have studied the relationship between WDR5 and glioma, and found that WDR5’s expression is positively correlated with the proliferation, migration, and invasion of glioma cells, which will help find the potential therapeutic target for glioma patients.
Keywords: WDR5, ZEB1, glioma, migration, metastasis
Introduction
Glioma is one of the most common malignancies that seriously endanger human health and is the fourth most common cause of malignancy in the world’s most common malignancy.1 Treatments of glioma patients are various, including surgery, radiotherapy, chemotherapy, biological immunotherapy and Chinese medicine treatment, but the results of these treatments are not satisfactory.2,3 Therefore, to find a suitable therapeutic target is the hot topic of scholars in the worldwide. It is important to look for new targets that contribute to early diagnosis and treatment of glioma, as well as prognostic predictors.
Epithelial–mesenchymal transformation (EMT) refers to a biological process of epithelial cells that are transformed into a stromal phenotype through a specific procedure.4 EMT plays an important role in embryonic development, chronic inflammation, tissue remodeling, cancer metastasis and various fibrotic diseases.5 The main features of EMT are the decrease of cell adhesion molecule, cytokeratin cytoskeleton transformation as the vimentin-based cytoskeleton and morphology with mesenchymal cells and other characteristics.6,7 Through EMT, epithelial cells lose cell polarity and connectivity to the basement membrane and other epithelial phenotypes, but obtain a higher ability of migration and invasion. Moreover, the property of anti-apoptosis, degradation of extracellular matrix and another interstitial phenotype appear.8 Therefore, EMT is an important biological process for epithelial cell derived from malignant tumor cells to obtain ability of migration and invasion.9
WDR5 is one of the components of the lysine methyltransferase complex. The lysine methyltransferase participates in a lysine methylation modification to catalyze the completion of the protein, which is involved in gene’s transcription regulation, cell’s senescence, cancer and other biological events. WDR5 is mainly involved in histone H3K4 dimethyl and trimethylation modification process.10 It has been found that WDR5 plays an important role in the process of embryonic cell self-renewal, bone development and the metastasis and proliferation of some cancer cells.11–14 However, its role in the glioma still unclear.
In this subject, we aimed to study the role of WDR5 in glioma cells. We found that WDR5’s expression is positively correlated with the proliferation, migration, and invasion of glioma cells, and modulate the EMT changes by targeting ZEB1 in glioma cells. These results will help to find the potential therapeutic target for glioma patients.
Materials and Methods
Glioma Tissue Specimens
This study was conducted on 51 glioma tissues and 12 normal brain tissues, which were histopathologically and clinically diagnosed at the Department of Neurosurgery, Beijing Shijitan Hospital between 2012 and 2015. Tumors were classified histologically according to TNM staging criteria from the American Joint Committee on Cancer criteria (Table 1). Clinical information about these patients is shown in Table 1. This was approved by the ethics committee of Beijing Shijitan Hospital, that all patients provided written informed consent, and that this was conducted in accordance with the Declaration of Helsinki.
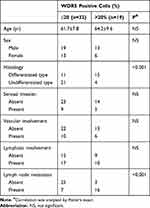 |
Table 1 WDR5 Protein Expression and Clinicopathological Factors in Glioma |
Immunohistochemical Staining
The expression of WDR5 in tissue, carcinoma in situ and distant metastasis was detected by immunohistochemical staining. Paraffin embedded tissue specimens were cut into 5 μm section. After blocking endogenous peroxidase, samples were incubated with primary antibody overnight at 4°C. The next day, samples were incubated with secondary antibody for 1 hour at room temperature. And then, slides were stained with diaminobenzidine and counterstained by hematoxylin.
Cells and Cell Culture
The glioma cell lines, HEB, A 172, U 343, and SW 1783, were got from ATCC, and cultured in DMEM medium supplemented with 10% FBS and 5% CO2 humidified atmosphere at 37°C.
Vectors and Construction of Cell Lines
The shRNA for WDR5 plasmid and the control vector were bought from Sigma and used according to previous report.15 Stable cell lines expression WDR5 silencing SW 1783 cells were generated by retroviral infection and selected by puromycin for 7 days.
RNA Extraction, qRT-PCR
The expression of WDR5 mRNA, epithelial marker mRNA, mesenchymal marker mRNA and ZEB1 mRNA in WDR5 mRNA and WDR5 knockdown cell lines were detected by qRT-PCR. RNA was extracted from the cells using TRIzol reagent (Invitrogen). The qRT-PCR used the SYBR Green Master Mix protocol, with GAPDH as internal control. The primers used were as previously reports.10
Western Blot
The expression of WDR5, epithelial markers, mesenchymal markers and ZEB1 protein were detected by Western blot. The antibodies used in Western blot are anti-WDR5 (ab22512), anti-ZEB1 (ab124512), anti-E-cadherin (ab1416), anti-N-cadherin (ab18203), anti-α-catenin (ab641), anti-vimentin (ab45939), anti-β-actin (ab179467), anti-Fibronectin (ab2413). All the antibodies are purchased from Abcam. The Western blot protocol used in our research were as previously described.16 The full unedited gel of all Western blot results has been shown in the Supplemental Figure 1 (The full unedited gel for Figures 1–8).
MTT Assay
MTT (Invitrogen, Shanghai, China) was used to observe the effect of WDR5 knockdown on cell proliferation. The cells of WDR5 knockdown were inoculated into 96-well plates with 1000 wells. The cells were cultured at 37°C for 12 hours and cultured for 12 h. After adding MTT to the air with a pipette, incubate for 4 hours. After careful removal of the supernatant, 150 μL of DMSO (Invitrogen, Shanghai, China) was added dropwise. And then detected at 0, 24 and 72 h respectively at 570 nm.
Colony Formation Experiments
Colony formation experiments were also used to determine the proliferation of WDR5 knockdown cells. Cells were seeded at 500 densities per well in a 6-well plate, and 3 mL of DMEM (Invitrogen, Shanghai, China) was added. The cells were homogenized by shaking the 6-well plates and incubated at 37°C to form macroscopically Clone, observed under a microscope and counted.
Transwell and Matrigel Assay
Transwell and matrigel experiments were used to observe the effect of WDR5 knockdown on cell migration and invasion. These assays followed previous report.8
Gene Expression Profiling
DNA microarray is a collection of microscopic DNA spots attached to a solid surface. Labeled with targets is prepared from a sample and hybridized to the cDNA sequences on the gene chip. The chip is then scanned for the presence and strength of the fluorescent labels at each spot. Particular spot’s fluorescent level can tell researchers quantitative information about expression of particular gene. Cluster analysis is a multivariate method which aims to classify genome on the basis of measured variables into a number of different groups. Here, we used cluster analysis to analyze the potential target protein of WDR5.
In vivo Glioma Cell Metastasis
The 5-week-old male Balb/c nude mice were purchased from Beijing Institute for Biological Sciences. All animal experiments were approved by the ethics committee of Capital Medical University. And all animal experiments complied with institutional guidelines and the use committee for animal care in Capital Medical University. The SW 1783-shWDR5 and its control vector cells were injected via tail vein as previously described.17 The mice were scarified about 6 weeks after this procedure, and metastatic liver nodules were measured. The in vivo tumor liver metastasis assays were conducted and the liver tissues were detected by H&E staining as previously described.8
Statistical Analysis
The experimental data were processed using SPSS/Win11.0 software (SPSS Inc.). Comparisons between two groups were made using the Student’s t-test. Comparisons among groups were performed by analysis of variance (one-way ANOVA) followed by Bonferroni’s post-hoc test. A P-value of less than 0.05 was considered statistically significant.
Results
Data Analysis Identified the Expression of WDR5 in Glioma
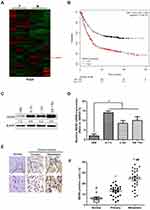
Using the TCGA database, the heat maps of differentially expressed genes in primary glioma (P) and metastatic glioma (M) were drawn. The results showed a higher level of WDR5 in metastatic glioma (Figure 1A). In addition, we also mapped the Kaplan-Meier curve of total survival (OS) in patients with glioma. It was found that the high WDR5 expression level was related to the overall survival of the patients (HR=2.05 (1.69–2.48); logrankP=5.8e-14). The higher the expression level of WDR5, the total survival short (Figure 1B). The above results indicate that WDR5 is associated with glioma. In order to further prove this point, we conducted Western blot analysis experiments to detect the expression of WDR5 in glioma cells. The results showed that compared with normal cell line (HEB), glioma cell lines (A 172, U 343, and SW 1783) WDR5 protein expression increased significantly (Figure 1C). In addition, we also conducted a qRT-PCR experiment, the experimental results consistent with the above results (Figure 1D). Then, we carried out immunohistochemical staining, the normal brain tissue, carcinoma in situ and distant metastasis of glioma tissue WDR5 expression was detected, the results showed that the expression of cancer tissue was higher than the normal brain tissue, the expression of metastatic glioma was the highest among the three (Figure 1E). Finally, we analyzed the number of positive cells in WDR5 in the tissues and found that WDR5 expression in metastatic glioma was significantly higher than that in carcinoma and normal brain tissue (P<0.01) (Figure 1F). In addition, a significant difference in histological type and lymph node metastasis were found between the high WDR5 expression group and the low expression group (P<0.001). On the other hand, no significant difference was seen regarding age, sex, vascular or lymphatic involvement (Table 1). These results indicate that the expression of WDR5 is significantly increased in glioma tissues, and the increased WDR5 expression is also associated with the metastasis of glioma.
WDR5 Knockdown Decreases the Proliferation Abilities of Glioma Cells
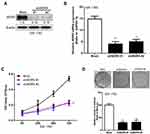
To further confirm the relationship between WDR5 and glioma, we established a WDR5 knockdown SW 1783 cancer cell line. We constructed two shRNAs of WDR5: shWDR5 # 1 and shWDR5 # 2. After the sequencing results were shown correctly, we encapsulated the two shRNAs and control plasmids by lentivirus transfection. In Figure 2A and B show the expression of WDR5 in the cell line. Subsequently, we used MTT assay to detect the proliferation of WDR5 knockdown cell lines. The results showed that cell proliferation was significantly inhibited in WDR5 knockdown cell lines (P<0.01) (Figure 2C). Figure 2D shows the results of the clonal formation experiments, and the results show that knockdown of WDR5 inhibits cell cloning. From the above results, we can see that WDR5 knockdown can significantly inhibit the proliferation of glioma cells, which may be the treatment of glioma cells to provide an important potential treatment target.
WDR5 Knockdown Decreases the Migration and Invasive Abilities of Glioma Cells
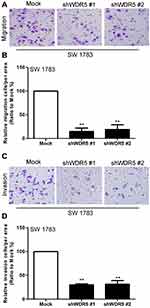
Next, we measured the effect of WDR5 on the migration and invasion of glioma cells. We observed the migration and invasion of cells by Transwell and matrigel experiments. The results showed that WDR5 knockdown cells had a significant decrease in migration and invasion compared with control glioma cells (P<0.01). This result indicates that WDR5 is significantly associated with migration and invasion of glioma cells (Figure 3A–D).
WDR5 Knockdown Changes the EMT Markers of Glioma Cancer Cells
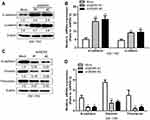
Cancer metastasis is often the leading cause of cancer failure, and this process is often accompanied by epidermal-stromal transition (EMT), so we guess that WDR5 may affect the glioma cell EMT to further affect the migration and invasion of glioma cells capable. We used protein blot and mRNA analysis to observe the expression of epithelial markers in WDR5 knockdown cells. The results showed that the epithelial markers (E-cadherin and α-catenin) were significantly increased after WDR5 knockdown (P<0.01) (Figure 4A and B). We also observed mesenchymal markers expression. The results showed that the mesenchymal markers (N-cadherin, vimentin, and fibronectin) were significantly decreased in WDR5 knockdown cell lines (P<0.01) (Figure 4C and D). These results suggest that WDR5 may promote cell migration and invasion by promoting EMT change in glioma cells.
WDR5 Knockdown Decreases the Metastatic Abilities of Glioma Cells
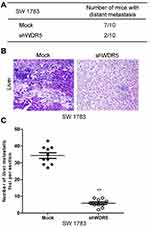
In order to study the relationship between WDR5 and tumor distant metastasis, we observed the effect of WDR5 knockdown on tumor metastasis in mice. The results showed that only 20% of WDR5 knockdown mice had metastasized and WDR5 knockdown mice 70% of the transfer occurred (Figure 5A). Figure 5B shows the transfer of liver tissue, after WDR5 silencing, the metastasis has been significantly improved. Figure 5C shows the number of metastases in live tissue sections. The results showed that the number of metastases was significantly reduced after WDR5 knockdown (P<0.01). The above experiments demonstrate that WDR5 is significantly associated with glioma cells metastases of in vivo.
WDR5 Induces the Expression of ZEB1
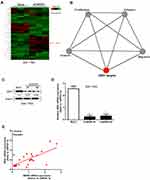
Figure 6A and B is our microarray obtained by the WDR5 knockdown of the cell line thermal map. The figure shows that WDR5 knock down, ZEB1 also significantly reduced. ZEB1 is a zinc finger protein transcription factor, studies have shown that ZEB1 in a variety of tumor tissue has a high expression level. We hypothesize that the effect of WDR5 on glioma may also be associated with ZEB1. Then we carried out the Western blot analysis, the results showed that WDR5 knockdown in the cell line, ZEB1 protein expression was significantly reduced (Figure 6C). The experimental results of qRT-PCR were consistent with the above results (P<0.01) (Figure 6D). Finally, we analyzed the relationship between the expression of ZEB1 and WDR5 in glioma tissues, and the results showed that the two were positively correlated (r=0.512; P<0.0001) (Figure 6E). The above experimental results show that WDR5 can affect the expression of ZEB1 to affect glioma metastasis.
ZEB1 Is a Mediator of WDR5-Induced Malignant Behaviors in Glioma Cells
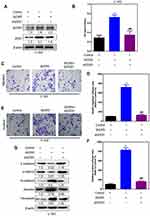
In order to test whether WDR5 induced malignant behavior in glioma cells was mediated by ZEB1, shRNA was used to inhibit the expression of ZEB1 gene in the WDR5 ectopic expression in U 343 cells (Figure 7A). MTT results showed that in WDR5 ectopic, the knockdown of ZEB1 expressed U 343 cells, leading to a decrease in proliferation (P<0.01) (Figure 7B). To verify whether ZEB1 mediated WDR5 induced migration and in vitro invasion, transwell and matrigel analysis. As shown in Figure 7C–F, the knockdown expression of ZEB1 in WDR5 ectopic cells can significantly reduce the migration and invasion ability of U 343 cells (P<0.01). In addition, knockout ZEB1 could save the EMT marker changes caused by excessive expression of WDR5 in U 343 cells (Figure 7G). To sum up, these results indicate that ZEB1 mediated the malignant behavior induced by WDR5 in glioma cells.
WDR5 Induced ZEB1 Up-Regulating Expression by Increasing H3K4me3 Binding to the Promoter Region of ZEB1
Then we discussed the mechanism of WDR5 regulating ZEB1 expression. Histone methyltransferase is often involved in chromatin regulation and histone modification and plays an important role in tumor progression. In order to determine whether WDR5 activity was related to specific histone modifications in glioma cells, the modulated histone modification mode was measured in WDR5 expression. We found that the expression of H3K4me3 protein was affected by WDR5 expression changes, while WDR5 silencing decreased the H3K4me3 protein level (Figure 8A), while WDR5’s ectopic expression increased the modification (Figure 8B). As H3K4me3 was associated with active transcription, we examined whether WDR5 expression was associated with the promoter H3K4me3 of the glioma cell ZEB1 gene. In SW 1783-shWDR5, U343-WDR5 and their control cells, the ChIP-qPCR was detected. H3K4me3 and IgG antibodies were used to inhibit the chromatin complex, and three pairs of primers in the promoter region of ZEB1 gene were used to evaluate the use of ZEB1 gene promoter (Figure 8C). The expression of WDR5 is associated with lower levels of H3K4me3 in the ZEB1 gene promoter region #1 and #3 (P<0.01) (Figure 8D). The occupied rate of H3K4me3 in the promoter region of ZEB1 gene was detected in U343-WDR5 cells (P<0.01) (Figure 8E). These results clearly indicated that WDR5 regulated the transcription activation of ZEB1 expression by regulating H3K4me3 and increased the combination of H3K4me3 and ZEB1 gene promoter in osteosarcoma cells.
Discussion
Glioma is one of the most important tumor diseases in our population, and the mortality rate is high in China.2 China’s early detection rate of glioma is low, until the diagnosis of the disease has often been developed.2 The current treatment of glioma, low cure, and poor prognosis, so looking for a new target for the diagnosis and treatment of glioma is a hot topic in the study of scientists.18 In this subject, we investigated the WDR5 on glioma cell proliferation, migration and invasive ability, as well as possible mechanisms, which provide some theoretical basis for whether WDR5 can serve as a new therapeutic target for glioma patients.
WDR5 is one of the components of the lysine methyltransferase complex, involved in the histone H3K4 dimethylation and trimethylation, which plays an important role in the process of embryo cell self-renewal and skeletal development and is related to the proliferation and migration of various cancer cells.12,19,20 Cancer cell metastasis is often the leading cause of cancer treatment failure, and this process is often accompanied by EMT.9,21 It has been reported that WDR5 plays an important role in the proliferation and metastasis of bladder cancer cells and the proliferation and metastasis of breast cancer cells,11,13 suggesting that WDR5 may serve as a potential biomarker for the treatment or detection of certain cancers. In this study, we first studied the expression of WDR5 in glioma cells. The results showed that the expression of WDR5 in glioma cells was significantly increased, and the normal brain tissue was significantly lower than that in glioma tissues with different degrees of malignancy.
Then, in order to further prove the relationship between WDR5 and glioma, we established the WDR5 knockdown SW 1783 glioma cell line, from the cell proliferation, migration and invasion and other aspects of the study found that WDR5 knockdown cells, the above several capacities has been significantly reduced. We also established a mouse model to observe the WDR5 knockdown in the tumor metastasis, the results show that the reduction of WDR5 expression of the transfer rate of the incidence was significantly reduced.
Cancer metastasis is often accompanied by EMT,22 and in this study, we also observed the effect of WDR5 on EMT. We detected the expression of epithelial markers and mesenchymal markers in WDR5 knockdown cells by Western blot and qRT-PCR. It was found that the knockdown of WDR5 significantly changed the expression of EMT markers, indicating that WDR5 on glioma cell migration and invasive ability, may be affected by glioma cells to achieve the EMT. Finally, the gene map of WDR5 knocked down SW 1783 cells was drawn by microarray measurement. It was found that the decrease of WDR5 and ZEB1 were also decreased. ZEB1 is a zinc finger protein transcription factor, studies have shown that it is associated with the occurrence of a variety of cancers.23,24 We investigated the expression of ZEB1 in WDR5 knockdown cells by Western blot analysis and qRT-PCR. This finding also provides a possible mechanism for the effect of WDR5 on glioma.
In this subject, we conducted an in-depth study of the relationship between WDR5 and glioma. The results showed that WDR5 could promote the proliferation, migration, invasion and tumor metastasis of glioma in a large number of experiments. This conclusion will help in obtaining a new therapeutic target for the detection, treatment and prognosis of glioma.
Acknowledgments
This work was supported by the Beijing Municipal Administration of Hospitals’ Youth Programme (Grant number: QML20170702) and Beijing Shijitan Hospital Research Project Fund (Grant number: 2015-C11).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Liang HX, Sun LB, Liu NJ. Neferine inhibits proliferation, migration and invasion of U251 glioma cells by down-regulation of miR-10b. Biomed Pharmacother. 2019;109:1032–1040. doi:10.1016/j.biopha.2018.10.122
2. Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi:10.3322/caac.21338
3. Jiao Q, Bi L, Ren Y, Song S, Wang Q, Wang YS. Advances in studies of tyrosine kinase inhibitors and their acquired resistance. Mol Cancer. 2018;17:36. doi:10.1186/s12943-018-0801-5
4. Krebs AM, Mitschke J, Lasierra Losada M, et al. The EMT-activator Zeb1 is a key factor for cell plasticity and promotes metastasis in pancreatic cancer. Nat Cell Biol. 2017;19:518–529. doi:10.1038/ncb3513
5. Wang YS, Du L, Liang X, et al. Sirtuin 4 depletion promotes hepatocellular carcinoma tumorigenesis through regulating adenosine-monophosphate-activated protein kinase alpha/mammalian target of rapamycin axis in mice. Hepatol (Baltimore, Md). 2019;69:1614–1631. doi:10.1002/hep.30421
6. Li S, Zhang HY, Du ZX, et al. Induction of epithelial-mesenchymal transition (EMT) by Beclin 1 knockdown via posttranscriptional upregulation of ZEB1 in thyroid cancer cells. Oncotarget. 2016;7:70364–70377. doi:10.18632/oncotarget.12217
7. Wang Y, Liu X, Zheng H, Wang Q, An L, Wei G. Suppression of CUL4A attenuates TGF-beta1-induced epithelial-to-mesenchymal transition in breast cancer cells. Int J Mol Med. 2017;40:1114–1124. doi:10.3892/ijmm.2017.3118
8. Wang Y, Wen M, Kwon Y, et al. CUL4A induces epithelial-mesenchymal transition and promotes cancer metastasis by regulating ZEB1 expression. Cancer Res. 2014;74:520–531. doi:10.1158/0008-5472.CAN-13-2182
9. Agajanian M, Runa F, Kelber JA. Identification of a PEAK1/ZEB1 signaling axis during TGFbeta/fibronectin-induced EMT in breast cancer. Biochem Biophys Res Commun. 2015;465:606–612. doi:10.1016/j.bbrc.2015.08.071
10. Kim JY, Banerjee T, Vinckevicius A, et al. A role for WDR5 in integrating threonine 11 phosphorylation to lysine 4 methylation on histone H3 during androgen signaling and in prostate cancer. Mol Cell. 2014;54:613–625. doi:10.1016/j.molcel.2014.03.043
11. Chen X, Gu P, Li K, et al. Gene expression profiling of WDR5 regulated genes in bladder cancer. Genomics Data. 2015;5:27–29. doi:10.1016/j.gdata.2015.05.003
12. Carugo A, Genovese G, Seth S, et al. In vivo functional platform targeting patient-derived xenografts identifies WDR5-Myc association as a critical determinant of pancreatic cancer. Cell Rep. 2016;16:133–147. doi:10.1016/j.celrep.2016.05.063
13. Dai X, Guo W, Zhan C, Liu X, Bai Z, Yang Y. WDR5 expression is prognostic of breast cancer outcome. PLoS One. 2015;10:e0124964. doi:10.1371/journal.pone.0124964
14. Chen X, Xie W, Gu P, et al. Upregulated WDR5 promotes proliferation, self-renewal and chemoresistance in bladder cancer via mediating H3K4 trimethylation. Sci Rep. 2015;5:8293. doi:10.1038/srep08293
15. Malek R, Gajula RP, Williams RD, et al. TWIST1-WDR5-hottip regulates hoxa9 chromatin to facilitate prostate cancer metastasis. Cancer Res. 2017;77:3181–3193. doi:10.1158/0008-5472.CAN-16-2797
16. Wang Y, Ma G, Wang Q, et al. Involvement of CUL4A in regulation of multidrug resistance to P-gp substrate drugs in breast cancer cells. Molecules. 2013;19:159–176. doi:10.3390/molecules19010159
17. Wang YS, Du L, Liang X, et al. SIRT4 depletion promotes HCC tumorigenesis through regulating AMPKalpha/mTOR axis. Hepatology. 2018.
18. Sun Y, Yu J, Liu X, et al. Oncosis-like cell death is induced by berberine through ERK1/2-mediated impairment of mitochondrial aerobic respiration in gliomas. Biomed Pharmacother. 2018;102:699–710. doi:10.1016/j.biopha.2018.03.132
19. Thomas LR, Foshage AM, Weissmiller AM, Tansey WP. The MYC-WDR5 nexus and cancer. Cancer Res. 2015;75:4012–4015. doi:10.1158/0008-5472.CAN-15-1216
20. Tan X, Chen S, Wu J, et al. PI3K/AKT-mediated upregulation of WDR5 promotes colorectal cancer metastasis by directly targeting ZNF407. Cell Death Dis. 2017;8:e2686. doi:10.1038/cddis.2017.111
21. Zhang S, Hong Z, Chai Y, et al. CSN5 promotes renal cell carcinoma metastasis and EMT by inhibiting ZEB1 degradation. Biochem Biophys Res Commun. 2017;488:101–108. doi:10.1016/j.bbrc.2017.05.016
22. Wu CW, Peng ML, Yeh KT, Tsai YY, Chiang CC, Cheng YW. Corrigendum to “Inactivation of p53 in pterygium influence miR-200a expression resulting in ZEB1/ZEB2 up-regulation and EMT processing” [Exp. Eye Res. 146 (2016) 206-211]. Exp Eye Res. 2016;151:256. doi:10.1016/j.exer.2016.06.005
23. Yu JM, Sun W, Hua F, et al. BCL6 induces EMT by promoting the ZEB1-mediated transcription repression of E-cadherin in breast cancer cells. Cancer Lett. 2015;365:190–200. doi:10.1016/j.canlet.2015.05.029
24. Kumar K, Chow CR, Ebine K, et al. Differential regulation of ZEB1 and EMT by MAPK-interacting protein kinases (MNK) and eIF4E in pancreatic cancer. Mol Cancer Res. 2016;14:216–227. doi:10.1158/1541-7786.MCR-15-0285
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.