Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Validation of the Chinese Version of the 16-Item Negative Symptom Assessment
Authors Huang B , Wang Y, Miao Q, Yu X, Pu C, Shi C
Received 25 February 2020
Accepted for publication 8 April 2020
Published 4 May 2020 Volume 2020:16 Pages 1113—1120
DOI https://doi.org/10.2147/NDT.S251182
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Bing-jie Huang,1– 4 Yong Wang,1– 4 Qi Miao,1– 4 Xin Yu,1– 4 Cheng-cheng Pu,1– 4 Chuan Shi1– 4
1Clinical Research Department, Peking University Sixth Hospital, Beijing, People’s Republic of China; 2Institute of Mental Health, Peking University, Beijing, People’s Republic of China; 3NHC Key Laboratory of Mental Health (Peking University), , Beijing, People’s Republic of China; 4National Clinical Research Center for Mental Disorders (Peking University Sixth Hospital), Beijing, People’s Republic of China
Correspondence: Cheng-cheng Pu; Chuan Shi Email [email protected]; [email protected]
Purpose: The Negative Symptom Assessment-16 (NSA-16) is an instrument with significant validity and utility for assessing negative symptoms associated with schizophrenia. This study aimed to validate the Chinese version of the NSA-16.
Patients and Methods: A total of 172 participants with schizophrenia were assessed with the NSA-16, Scale for Assessment of Negative Symptoms (SANS), Positive and Negative Syndrome Scale (PANSS), Calgary Depression Scale for Schizophrenia (CDSS) and Rating Scale for Extrapyramidal Side Effects (RSESE). The factor structure of the NSA-16 was evaluated via exploratory and confirmatory factor analysis. Cronbach’s α and intraclass correlation coefficients were computed. Correlations were evaluated via Spearman correlation coefficient.
Results: The original five-factor model of the NSA-16 did not fit our sample. Exploratory factor analysis followed by confirmatory factor analysis suggested a three-factor structure, consisting of communication, emotion and motivation, with 15 items. The NSA with 15 items was termed as the NSA-15. The NSA-15 showed excellent convergent validity by high correlations with the SANS and PANSS total and negative factor scores and good divergent validity by independence from the PANSS positive factor, CDSS and RSESE. The NSA-15 showed good internal consistency, interrater reliability and test–retest reliability.
Conclusion: The NSA-15 is best characterized by a three-factor structure and is valid for assessing negative symptoms of schizophrenia in Chinese individuals.
Keywords: negative symptoms, schizophrenia, NSA, reliability, validity
Introduction
As a core dimension of schizophrenia, negative symptoms are associated with poor functional outcomes and decreased quality of life for patients.1,2 However, treatment along this dimension remains an unmet need.3 Suitable assessments are needed to explore the psychopathology of negative symptoms as well as interventions for their treatment.
The Negative Symptom Assessment-16 (NSA-16) examines the presence, severity and range of negative symptoms in schizophrenia.4 The NSA-16 has strong psychometric properties in terms of interrater reliability and convergent and divergent validity,5 has good clinical utility in different cultures and patient settings,6,7 and has sensitivity to changes in negative symptoms over time.4 Changes in ratings of the NSA-16 are associated with changes in functional outcomes.8
Initially, confirmatory factor analysis of 223 untreated in-patients suggested a five-factor structure: communication, emotion/affect, social involvement, motivation and retardation.4 However, Popp and colleagues identified a three-factor structure via confirmatory factor analyses: communication/social involvement, emotion/retardation and motivation.9 Recently, factor analyses of 274 Asian outpatients suggested a four-factor structure, consisting of restricted speech, poor quality of speech, affective blunting and motivation, with 12 items.7
Remarkably, the NSA-16 is the only scale that includes a single item measuring the global severity of negative symptoms based on the interviewer’s global impression of the patients. The item provides a comprehensive estimation of the global negative symptoms of participants that integrates the information of the interview and the interviewer’s clinical experience. This item was reported to be highly associated with component factors of the NSA-16 and other negative symptom assessments.5,7
The NSA-16 has several advantages over existing instruments that assess negative symptoms. It is an explicit instrument with well-defined items and detailed anchoring rating criteria. It is an easy-to-use instrument for which raters only need brief training to administer.4 The standardized semi-structured interview makes ratings from different raters and sites easier to compare. Compared to older instruments such as the Scale for Assessment of Negative Symptoms (SANS) and the Positive and Negative Syndrome Scale (PANSS), the NSA-16 has better content validity as it does not encompass symptoms from other dimensions such as the cognitive dimension.10 Newer instruments, such as the Brief Negative Symptom Scale (BNSS) and the Clinical Assessment Interview for Negative Symptoms (CAINS), were designed to overcome the limitations of older instruments and to assess the five agreed-upon domains of negative symptoms that arose from the Consensus Development Conference on Negative Symptoms.11 Most of the validation studies suggested good validity, but Kring and colleagues12 reported low convergent validity of the CAINS motivation/pleasure subscale. Additionally, the sensitivity to changes and utility in diverse cultures have rarely been reported. In contrast, as one of the most widely used scales, these characteristics have been confirmed in the NSA-16 by various studies.
The NSA-16 is a classical instrument with good validity and clinical utility for measuring negative symptoms in schizophrenia. However, its validity in Chinese settings remains unknown. This study aimed to introduce a Chinese version of the NSA-16 and perform validation of this version.
Patients and Methods
Participants
The participants of this cross-sectional study were recruited from the Peking University Sixth Hospital in Beijing (the Community Health Service Centre of the Institute of Mental Health). The inclusion criteria were a diagnosis of schizophrenia and an age of 16–60 years. The exclusion criteria were comorbidities consisting of other Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) Axis I disorders; a history of head injury or neurological disorder; severe or unstable somatic disease; women who were pregnant or breastfeeding; and patients who had received modified electroconvulsive therapy in the previous three months. Diagnoses of schizophrenia were determined using the Structured Clinical Interview for DSM-IV (SCID-I).13
A total of 172 in- and outpatients with long-standing schizophrenia were recruited. All patients completed the assessments. The participants had an average age (SD) of 30 (10) years and an average disease duration of 103 (94) months (disease duration was defined as duration since the first reported symptoms), and 55% of the subjects were male. All participants provided written informed consent (a parent or legal guardian provided written informed consent for participants under the age of 18 years). This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committees of the Institute of Psychology and the Institute of Mental Health (Peking University Sixth Hospital).
Measures
Chinese Version of the NSA-16
The NSA-16 contains 16 items rated on a six-point Likert scale (1–6 points), with higher scores reflecting greater impairment, along with a global negative symptom rating based on the interviewer’s impression of patient’s negative symptoms. The rating of the scale is based on a 15–30 minute semi-structured interview.4 We developed the Chinese version of the NSA-16 via a forward-backward translation procedure. Two psychiatrists translated the instrument into Chinese, and then another two translators retro-translated the Chinese version back into English, which was confirmed by the primary author of the NSA-16. The final Chinese version was integrated and approved by a team of clinicians and scientific methodologists.
Scale for Assessment of Negative Symptoms (SANS)
The SANS is a classical instrument that assesses negative symptoms of schizophrenia. The scale contains 25 items rated on a six-point Linkert scale (0–5 points), where higher scores reflect greater impairment. The Chinese version of the SANS has been validated.14 We used the Chinese version of the SANS to assess the convergent validity of the NSA-16.
Positive and Negative Syndrome Scale (PANSS)
The PANSS is a comprehensive scale that assesses the severity of schizophrenia psychopathology.15 The Chinese version of the PANSS contains 30 items rated on a seven-point Likert scale and is comprised of three subscales: positive, negative and general psychopathology factors. Good reliability and validity were reported by Chinese researchers.16 The PANSS total and negative factor scores were used to assess the convergent validity of the NSA-16, while the PANSS positive and general psychopathology factor scores were used to assess the divergent validity of the NSA-16.
Calgary Depression Scale for Schizophrenia (CDSS)
The CDSS was designed to assess depressive symptoms of schizophrenia.17 It contains nine items rated on a four-point Linkert scale. The validated Chinese version of the CDSS was adopted.18
Rating Scale for Extrapyramidal Side Effects (RSESE)
The RSESE was used to assess extrapyramidal side effects of pharmacotherapy.19 The Chinese version of the RSESE contains 10 items rated on a five-point Linkert scale (0–4 points) and has acceptable validation.20 The CDSS and RSESE were used to assess the divergent validity of the NSA-16.
Three research clinicians were raters for this study. The three raters all had at least three years of experience in conducting patient symptom interviews on individuals with schizophrenia. They attended a training session on the NSA-16 and other instruments, and good intraclass correlation coefficients (ICCs) were obtained for the ratings on scales used in this study.
Data Analyses
Data were analyzed with SPSS 17.0 and AMOS 22.0. We used confirmatory factor analysis (CFA) to evaluate whether the five-factor structure of the original NSA-164 could fit our version of the assessment. The goodness of fit of the CFA was evaluated using the comparative fit index (CFI, >0.90), the root-mean-square error of approximation (RMSEA, <0.80) and the ratio of the chi-square statistic to the degrees of freedom (a ratio between one and three is acceptable).21 The fit indices on CFA were not acceptable. Hence, we randomly split the sample into two datasets to explore the scale structure. We used the Kaiser criterion (eigenvector >1) to explore the number of factors to extract, followed by exploratory factor analysis (EFA) using principal axis extraction with VARIMAX rotation on the first half of the sample (n=86). Only items loaded greater than 0.40 were included, and items with cross-loading with loadings greater than 0.40 on two factors or more were excluded. Then, we conducted CFA on the remaining half of the sample (n=86) to confirm the replication of the model suggested by the EFA. We reran the models according to modification indices that could improve model fit.
Internal consistency was appraised by computing the Cronbach’s α coefficient. ICCs were computed for a subsample (N=18) for which the NSA-16 ratings were conducted by three raters to determine interrater reliability. Test–retest reliability coefficients were computed for a subsample (N=28) for which the NSA-16 was administered twice with an interval of 2–4 weeks. Correlations between the NSA-16 and other instruments were assessed using Spearman correlation coefficient because the distribution of the scores on most of the instruments was skewed.
Results
The demographic characteristics of participants and the distribution of rating scores for the main instruments are presented in Table 1. Ratings on the NSA-16 and the PANSS seemed to be low to moderate.
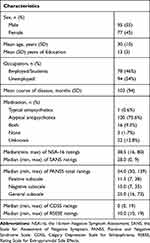 |
Table 1 Demographic and Clinical Characteristics of the Sample (n=172) |
Construct Validity
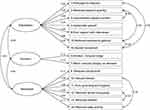
The fit indices of the five-factor structure were not acceptable: CFI=0.892, RMSEA=0.107, and the ratio of the chi-square statistic to the degrees of freedom=1.974. The EFA of the first half of the sample suggested a three-factor solution: the Kaiser-Meyer-Olkin value was 0.87, Bartlett’s Test of Sphericity showed statistical significance (p<0.001), and the model explained 64.44% of the variance in total. The factor loadings for the three-factor solution are presented in Table 2. We excluded item 6 because it had variable loadings (the factor loadings on expression and motivation were both >0.4). The result of CFA for the remaining half of the sample is shown in Figure 1. CFA revealed that the three-factor structure with 15 items fit well with our sample: CFI=0.948, RMSEA=0.074, and the ratio of the chi-square statistic to degrees of freedom=1.468. This version of the NSA with 15 items was termed the NSA-15, and the three factors were termed as communication, emotion and motivation.
 |
Table 2 Factor Loadings of Exploratory Factor Analysis of the NSA-16 (n=86) |
Convergent and Divergent Validity
Correlations between the NSA-15 and other clinical ratings are exhibited in Table 3. The NSA-15 total scores were highly correlated with the SANS total and PANSS negative scores (r>0.5, p<0.001) and were weakly correlated with the PANSS positive scores and general psychopathology scores and the RSESE (r<0.5, p<0.001). Correlations between the NSA-15 and the CDSS did not reach statistical significance. Correlations between the NSA-15 global negative symptom rating and other instruments were similar to those for the NSA-15 total score.
 |
Table 3 Convergent and Divergent Validity of the NSA-15: Spearman Correlations (n=172) |
The NSA-15 expression factor was most strongly associated with the SANS alogia factor, and the emotion factor was strongly associated with the SANS affective blunting factor but weakly associated with the SANS avolition factor. The motivation factor was strongly associated with the SANS asociality and avolition factors.
Internal Consistency and Test–Retest and Interrater Reliability
Cronbach’s α for the NSA-15 was 0.918 for the total score, 0.878 for communication, 0.700 for emotion, and 0.845 for motivation. The intraclass correlations were 0.959 for the total scale, 0.893 for communication, 0.893 for emotion, and 0.958 for motivation. Test–retest reliability was 0.917 for the total scale, 0.782 for communication, 0.822 for emotion and 0.932 for motivation.
Discussion
This study aimed to develop a Chinese version of the NSA-16 and provide valid evidence for its use in Chinese settings. Our results suggested that the Chinese version of NSA-16 had good construct, convergent and divergent validity, as well as good reliability.
Regarding the factor structure of the Chinese version of the NSA-16, factor analyses suggested a three-factor model, consisting of communication, emotion and motivation, with 15 items. This three-factor model was different from the five-factor model suggested by Axelrod et al4 and the four-factor model suggested by Rekhi et al.7 It has been suggested that variant analytic methods and sample differences might contribute to variability in factor structure exploration.22 The analytic methods that we used were similar to the validation studies of the original NSA-16 mentioned above (CFA and EFA). With respect to sample differences, the samples of this and previously mentioned two studies had different characteristics with regard to ethnicity, disease duration, medication and whether the participants were in- or outpatients. There is some evidence supporting that these characteristics are associated with the structure of negative symptoms. A follow-up study examined the structure of negative symptoms in first-episode schizophrenia with the SANS and found that the structure of negative symptoms was associated with the disease duration and could change as the disease progressed.23 A study examined the structure of the CAINS with CFA in Asian settings and established a two-factor model,24 while another study conducted CFA on a Western sample and suggested a five-factor model.25 These findings imply that the structure of negative symptoms may be heterogeneous in samples with various characteristics. Future studies aimed to evaluate the structure of negative symptoms in specific subgroups are needed.
The communication factor was composed of items assessing a prolonged response interval, poor speech quantity, content and prosody, reduced gestures, slowed movement and poor rapport. Ratings of these items are dependent on participants’ explicit performance, and impairment of response interval, speech quantity and quality and body language could lead to poor rapport with the interviewer. These may explain why these seven items were loaded onto the same factor.
The emotion factor consisted of items measuring reduced emotional range and impairment of affect display. This factor is similar to the emotion/affect factor suggested by Axelrod and colleagues4 except that item 6 (Affect: Reduced modulation of intensity) was excluded due to double loading. The two items respectively measure two important aspects of affect: appraisal and expression; therefore, they loaded on a single factor.
The motivation factor contained items assessing social drive, intimacy interest, reduced purpose and interest, poor appearance and activity. The items that were loaded on this factor are similar to those in the factor model reported by Popp and colleagues.9 These six items assess participants’ motivation in three aspects: social, interpersonal and functional aspects.
Regarding convergent validity, relatively higher correlations were observed between the NSA-15 and similar instruments, indicating that the NSA-15 is consistent with other similar instruments. Divergent validity was demonstrated by lower correlations with dissimilar instruments, indicating that the NSA-15 has little overlap with positive symptoms, depression and medication side effects. Our results of convergent and divergent validity are similar to those of a previous validation study of the NSA-16.5
Our results also suggested good internal consistency of the NSA-15 with high α values of total scores and subscale scores, similar to the original validation study of the NSA-16,4 indicating that the 15 items are strongly related to each other without redundancy. Good test–retest reliability was demonstrated for the NSA-15, indicating that the assessment of the NSA-15 is stable over a period of 2–4 weeks and that good replicability across assessments can be obtained. Regarding interrater reliability, high ICCs among three raters were demonstrated in our study, indicating good rater agreement for the scale. Remarkably, our results for test–retest and interrater reliability of the NSA-15 were comparable or greater than those of similar instruments such as the BNSS and the CAINS,12,24,26 indicating that the NSA-15 may be a more reliable instrument for the Chinese population. Further studies should compare the validation of the NSA-15, BNSS and CAINS in a single sample.
Notably, we found that the global negative symptom rating item was highly associated with the NSA-15 total score and the three factors. Additionally, this item was moderately-highly associated with similar instruments and independent of irrelevant instruments, similar to the NSA-15 total score. Similar results were reported by previous studies.5,7 High consistency between the NSA global rating and the total scale suggested that this item is predictive of the severity of negative symptoms measured by the NSA-15. As previously mentioned, the NSA global rating combines objective sources from the interview with the interviewer’s subjective experience. This may provide evidence for the validity of integrating objective information with clinicians’ experience when developing new assessment tools for negative symptoms.
There are several limitations to the present study. The sample size of this study was not large compared to that of other similar studies.5,7 The ratings of our sample on the NSA and PANSS were low to moderate; therefore, the generalizability of our findings could be limited by the severity of illness in the sample. Another limitation of this study was that we did not address some important psychometric characteristics of the NSA-15, such as the ability to detect changes in negative symptoms in a clinical trial.1,27 In addition, all rating scales for each participant were completed by the same rater in our study, which may lead to bias by shared rater variance. Future studies with better methodological designs that concentrate on relationships between the NSA-16 and other important characteristics are needed.
Conclusion
In conclusion, the Chinese version of the NSA-15 shows satisfying psychometric properties in evaluating negative symptoms associated with schizophrenia in Chinese patients. The NSA-15 is best characterized by a three-factor model consisting of communication, emotion and motivation.
Acknowledgments
This work was supported by Capital’s Funds for Health Improvement and Research (CFH, No.2018-4-4116). The authors would like to thank all participants for their involvement.
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
1. Fervaha G, Foussias G, Agid O, Remington G. Impact of primary negative symptoms on functional outcomes in schizophrenia. Eur Psychiatr. 2014;29:449–455. doi:10.1016/j.eurpsy.2014.01.007
2. Robertson BR, Prestia D, Twamley EW, Patterson TL, Bowie CR, Harvey PD. Social competence versus negative symptoms as predictors of real world social functioning in schizophrenia. Schizophr Res. 2014;160:136–141. doi:10.1016/j.schres.2014.10.037
3. Kantrowitz JT. Managing negative symptoms of schizophrenia: how far have we come. CNS Drugs. 2017;31:373–388. doi:10.1007/s40263-017-0428-x
4. Axelrod BN, Goldman RS, Alphs LD. Validation of the 16-item negative symptom assessment. J Psychiatr Res. 1993;27:253–258. doi:10.1016/0022-3956(93)90036-2
5. Alphs L, Morlock R, Coon C, Cazorla P, Szegedi A, Panagides J. Validation of a 4-item negative symptom assessment (NSA-4): a short, practical clinical tool for the assessment of negative symptoms in schizophrenia. Int J Methods Psychiatr Res. 2011;20:e31–e37. doi:10.1002/mpr.339
6. Daniel DG, Alphs L, Cazorla P, Bartko JJ, Panagides J. Training for assessment of negative symptoms of schizophrenia across languages and cultures: comparison of the NSA-16 with the PANSS negative subscale and negative symptom factor. Clin Schizophr Relat Psychoses. 2011;5:87–94. doi:10.3371/CSRP.5.2.5
7. Rekhi G, Alphs L, Ang MS, Lee J. Clinical utility of the negative symptom assessment-16 in individuals with schizophrenia. Eur Neuropsychopharmacol. 2019;29:1433–1441. doi:10.1016/j.euroneuro.2019.10.009
8. Velligan DI, Alphs L, Lancaster S, Morlock R, Mintz J. Association between changes on the negative symptom assessment scale (NSA-16) and measures of functional outcome in schizophrenia. Psychiatr Res. 2009;169:97–100. doi:10.1016/j.psychres.2008.10.009
9. Popp D, Williams J, Cohen EA, Detke MJ. Poster #176 NSA-16 revisited: identifying latent factors of negative symptoms in schizophrenia. Schizophr Res. 136:S344. doi:10.1016/S0920-9964(12)71008-3
10. Garcia-Portilla MP, Garcia-Alvarez L, Saiz PA, et al. Psychometric evaluation of the negative syndrome of schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2015;265:559–566. doi:10.1007/s00406-015-0595-z
11. Kirkpatrick B, Fenton WS, Carpenter WT, Marder SR. The NIMH-MATRICS consensus statement on negative symptoms. Schizophr Bull. 2016;32:214–219. doi:10.1093/schbul/sbj053
12. Kring AM, Gur RE, Blanchard JJ, Horan WP, Reise SP. The clinical assessment interview for negative symptoms (CAINS): final development and validation. Am J Psychiatr. 2013;170:165–172. doi:10.1176/appi.ajp.2012.12010109
13. First MB, Gibbon M, Spitzer RL, JBW W. Structured Clinical Interview for DSM-IV Axis I Disorders.
14. Xia M. Scale for Assessment of Negative Symptoms (SANS). Shanghai Archives of Psychiatry; 1990.
15. Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13:261–276. doi:10.1093/schbul/13.2.261
16. Shu L, Si T, Li X, Kong Q. The Reliability, Validity of PANSS and its Implication. Chinese Mental Health Journal. 2004;45–47. doi:10.3321/j.issn:1000-6729.2004.01.016
17. Addington D, Addington J, Maticka-Tyndale E. Assessing depression in schizophrenia: the calgary depression scale. Br J Psychiatr Suppl. 1993;39–44. doi:10.1192/S0007125000292581
18. Zhang H, Xiao W. Assessing Dpressive Symptoms of Schizophrenia-- Calgary Depression Scale for Schizophrenia. Journal of International Psychiatry. 2006;8–12.
19. Simpson GM, Angus JW. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand Suppl. 1970;212:11–19. doi:10.1111/j.1600-0447.1970.tb02066.x
20. Zhang M. Handbook of Rating Scales in Psychiatry. Hu nan: Hunan Science & Technology press; 1998:202–205.
21. Bentler PM. Comparative fit indexes in structural models. Psychol Bull. 1990;107:238–246. doi:10.1037/0033-2909.107.2.238
22. Tibber MS, Kirkbride JB, Joyce EM, et al. The component structure of the scales for the assessment of positive and negative symptoms in first-episode psychosis and its dependence on variations in analytic methods. Psychiatr Res. 2018;270:869–879. doi:10.1016/j.psychres.2018.10.046
23. Ergül C, Üçok A. Negative symptom subgroups have different effects on the clinical course of schizophrenia after the first episode: a 24-month follow up study. Eur Psychiatr. 2015;30:14–19. doi:10.1016/j.eurpsy.2014.07.005
24. Xie DJ, Shi HS, SSY L, et al. Cross cultural validation and extension of the clinical assessment interview for negative symptoms (CAINS) in the Chinese context: evidence from a spectrum perspective. Schizophr Bull. 2018;44:S547–547S555. doi:10.1093/schbul/sby013
25. Strauss GP, Nuñez A, Ahmed AO, et al. The latent structure of negative symptoms in schizophrenia. JAMA Psychiatr. 2018;75:1271–1279. doi:10.1001/jamapsychiatry.2018.2475
26. Kirkpatrick B, Strauss GP, Nguyen L, et al. The brief negative symptom scale: psychometric properties. Schizophr Bull. 2011;37:300–305. doi:10.1093/schbul/sbq059
27. Harvey PD, Koren D, Reichenberg A, Bowie CR. Negative symptoms and cognitive deficits: what is the nature of their relationship. Schizophr Bull. 2006;32:250–258. doi:10.1093/schbul/sbj011
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.