Back to Journals » Open Access Journal of Sports Medicine » Volume 10
Use of viscosupplementation for the recovery of active football players complaining of knee pain
Authors Migliore A , Giannini S , Bizzi E, Massafra U , Cassol M, Michael Abilius MJ , Boni G
Received 6 February 2018
Accepted for publication 24 September 2018
Published 24 December 2018 Volume 2019:10 Pages 11—15
DOI https://doi.org/10.2147/OAJSM.S164693
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Andreas Imhoff
Alberto Migliore,1 Silvana Giannini,2 Emanuele Bizzi,1 Umberto Massafra,1 Maurizio Cassol,3 Martin Jose Michael Abilius,1 Giovanni Boni4
1Operative Unit of Rheumatology, San Pietro Fatebenefratelli Hospital, Rome 00189, Italy; 2Department of Radiology, Villa Stuart Sport Clinic, Rome 00136, Italy; 3Department of Internal Medicine, San Pietro Fatebenefratelli Hospital, Rome 00189, Italy; 4ANTIAGE Sportive Medicine Ambulatory Department (Ambulatorio di Medicina dello Sport Antiage), Foligno, Italy
Objective: The aim of this study is to investigate the effect of intra-articular hyaluronic acid administration in active football players complaining of knee pain after sports activity. Efficacy and safety profiles of intra-articular hyaluronic acid and time needed for football players to recover and restart sports activity were examined.
Methods: Clinical data of active football players reporting knee pain after sports activity were included in this retrospective study. All patients who received an intra-articular injection at time 0 and after 2 weeks were included in the study. Patients underwent laboratory examination, knee X-ray, ultrasound, and clinical examination before receiving the intra-articular injection. Effusions or cysts were drained before injections. Lequesne index score, pain visual analog scale (VAS) score, and patient’s global assessment score were recorded at time 0 (day of the first injection), 1 and 2 days after the first injection, at 2 weeks (day of the second injection), and at follow-up visits. Only data from patients completing the follow-up were analyzed.
Results: Data from 17 patients were analyzed: 16 males and one female, of which three were professional players (two males and one female) and 14 were nonprofessional players. The mean age of patients was 39.8±11.8 years. Two patients (one male and one female) showed joint effusion. Two patients reported relevant joint pain after injection that regressed without any medication. At the first week, all parameters examined indicated improvement that was maintained until the end of follow-up. One day after the first and second injection, patients reported a slight increase in pain VAS score, which was not statistically significant, and the pain resolved after 1 day. All patients successfully restarted playing after the first injection within 3.1±2.0 days and kept playing after the second injection following our indication (1 day of break).
Conclusion: The use of a medium-molecular weight hyaluronic acid in football players affected by knee osteoarthritis seems efficacious and safe and resulted, in our experience, a stable improvement of symptoms; moreover, it allowed a rapid restart of sports activity. Larger studies on larger populations are needed to confirm these findings.
Keywords: sports, hyaluronic acid, intra-articular, injection, football, trauma
Corrigendum for this paper has been published
Introduction
Active football players may experience a limitation in their recreational/agonist activity due to knee pain, especially after sporting activity. The relationship between OA and repeated traumas is also well-known.1,2 Repeated small traumas involving joints may result in cartilage lesions and joint instability, concurring to precocious development of OA.3 In this sense, sports activity may represent a cause of repeated traumas and therefore a cause of precocious OA. Several studies had reported how sports such as football may represent a cause of early-onset OA, especially after major traumas in the dominant or nondominant leg, involving usually knee, ankle, and hip joints.3 Athletes affected by knee pain usually continue practicing their sports, often with difficulties and decrease in quality of performance. Intra-articular hyaluronic acid treatment represents a well-known therapeutic option for the management of OA.4–6 Several studies have reported on the good efficacy and safety profiles of intra-articular hyaluronic acid and its effects on the various joints affected by OA, such as hip, ankle, shoulder, and above all knee.7–9 As a side effect, several studies have reported how in the hours or days following the intra-articular injection of hyaluronic acid some patients may experience a transient pain in the injected joint.10 Usually, this pain is reported as transient and regresses without any need of medications. The use of intra-articular hyaluronic acid has not been reported for professional athletes, but has only been reported for patients affected by symptomatic OA, independent of the cause of OA. A major attention has been recently given to platelet-rich plasma for intra-articular and peri-articular or intra-lesional use,11–13 but still we lack data regarding the use of hyaluronic acid in professional athletes. Interestingly, OA is a common finding in former professional athletes and early-onset OA may be found in active professional athletes,2,3 but no data regarding the use of hyaluronic acid have been reported till now. In this study, we aimed to investigate the efficacy and safety profiles of intra-articular hyaluronic acid in active football players with symptomatic knee pain and to establish whether it may represent a suitable therapeutic option for managing OA at the early stages and consequent algofunctional impairment.
Methods
This is a multicentric, retrospective study. Outpatients’ data from the rheumatology operative unit of San Pietro Fatebenefratelli Hospital, ANTIAGE sports medicine ambulatory, and Villa Stuart radiology department in Rome, Italy, were evaluated, and only data of active football players complaining of monolateral knee impairment were taken into account for the eventual inclusion in this study.
Eligible patients were as follows: active football players who were 18 years of age and older with knee pain evaluated clinically and a consequent radiological examination of affected knee. Selected patients underwent a laboratory examination (erythrocytes sedimentation rate, C-reactive protein, uric acid, complete blood count) that was commonly performed before the injection of hyaluronic acid. Similarly, only data from patients who underwent knee ultrasound examination before injection were taken into account, while effusion or cysts was drained before intra-articular injection. Only data from patients showing grade I–II knee OA according to Kellgren–Lawrence classification were analyzed independently from pain VAS (1–10 scale).
Excluded from the study were patients affected by a different form of arthritis other than OA involving knee or other joints, patients less than 18 years of age, and patients with a history of allergic reaction to hyaluronic acid. All patients who received an intra-articular dose of medium molecular weight (800–1,200 kDa) hyaluronic acid, SINOVIAL HL 8 mg/mL (IBSA Pharma Inc., Lugano, Switzerland), 2×2 mL/vial for each injection, at time 0, and after 2 weeks were included for data analysis. For all patients, pain VAS score was recorded at time 0, 1 day after the first infusion, 2 days after the first infusion, at 2 weeks (day of the second infusion), 1 day after the second infusion, 2 days after the second infusion, and at 4, 12, and at 24 weeks, respectively. Index of severity of OA was expressed as Lequesne index score,14 and PGA (1–10 scale)15 was performed at baseline, at 1 week, and at 4, 12, and 24 weeks. Data of evaluations performed 1 and 2 days after each injection were obtained by phone interview. Only patients who completed follow-up were included for further data analysis. All data were gathered and analyzed to determine if hyaluronic acid was effective and safe for the treatment of algofunctional impairment in active football players affected by knee OA. Statistical significance of all observed parameters vs baseline values was evaluated. The study was approved by the Institutional Review Board of San Pietro Fatebenefratelli Hospital, and a signed informed consent was obtained from all subjects in accordance with the Declaration of Helsinki and its subsequent modifications.
Results
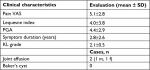
Data from 17 active football players were analyzed in this study. Demographic characteristics of patients are reported in Table 1. The group of patients whose data were analyzed was composed of 16 males and one female. This inhomogeneity of distribution of the two genders is to ascribe the sociocultural factors, as football is a sport more common among males.16 Mean age of patients was 39.8 years. Three patients were professional football players, while 14 patients practiced football for recreational purposes. Of the 14 nonprofessional players, two were formerly professional players. Baseline disease characteristics of patients are also reported in Table 1. Kellgren–Lawrence grading of knee OA attested on low levels (2.1 mean value), but interestingly duration of symptoms was not short (2.8 years as mean value). Two patients (1 male and one female, both professional players) showed joint effusion during ultrasound examination of the knee, which was drained before injection. No patients showed Baker’s cysts. Change in parameters (pain VAS, PGA, and Lequesne index scores) (Figure 1) after 1 week of follow-up was found to be significant when compared with baseline values (P<0.05). Two weeks after baseline injection, a second injection was administered. At 4 weeks of follow-up, all parameters exhibited further improvement that was found to be significant when matched with baseline values (P<0.05). After second day of each injection, a transient increase in pain VAS score was observed, but it did not reach statistical significance (Figure 2). All patients restarted practicing sports in a short time (2.1±2 days), and their physical activity was not burdened by adverse events; instead, it was characterized by symptom relief. Assessments revealed that findings at weeks 12 and 24 remained invariant starting from week 4 as shown in Figure 1.
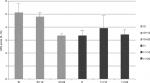  | Figure 2 Pain VAS score measured on the day of injections (t0) and at 1 (t1) and 2 (t2) days after each injection. Abbreviations: VAS, visual analog scale; d, days. |
Discussion
Hyaluronic acid for knee OA represents a common therapeutic option. The use of intra-articular hyaluronic acid in active sports players remains a controversial issue, and no scientific reports on its use are available till now in scientific literature. In this study, we aimed to investigate the effects of the use of hyaluronic acid in active football players complaining of knee pain. In our experience, hyaluronic acid administered in knee joints of active football players affected by OA resulted in a rapid relief of symptoms in terms of pain and functionality of the affected knee. Hyaluronic acid allowed not only symptom relief but also a rapid restart of sports activity, and this was especially relevant in professional players who involved in sports activity frequently and found it challenging. This is the first report on the use of intra-articular hyaluronic acid in active football players with knee OA, and it adds evidence on the efficacy and safety of this therapeutic option. Whether intra-articular hyaluronic acid only results in symptom relief and a rapid restart of sports activity or its cyclic use may also reduce the risk of developing early-onset OA remains to be investigated. This hypothesis can only be confirmed with further close follow-ups of athletes, and a longitudinal study can reveal whether some of the athletes will develop OA. Furthermore, we plan to recruit athletes from other sports fields and study the influence of hyaluronic acid treatment on their quality of life and sports activities.
Conclusion
The use of intra-articular hyaluronic acid in active football players with knee OA seems to be effective and safe and allows a rapid restart of sporting activity in both professional and nonprofessional players, independent of the severity of symptoms and radiological classification. Larger studies with longer follow-up times are needed to confirm the consistency of these findings and to explore if the use of hyaluronic acid may exert protective effects on the progression of disease.
Abbreviations
OA, osteoarthritis; PGA, patient’s global assessment; VAS, visual analog scale.
Disclosure
The authors report no conflicts of interest in this work.
References
Gouttebarge V, Inklaar H, Frings-Dresen MH. Risk and consequences of osteoarthritis after a professional football career: a systematic review of the recent literature. J Sports Med Phys Fitness. 2014;54(4):494–504. | ||
Dvorak J. Osteoarthritis in football: FIFA/F-MARC approach. Br J Sports Med. 2011;45(8):673–676. | ||
Krajnc Z, Vogrin M, Recnik G, Crnjac A, Drobnic M, Antolic V. Increased risk of knee injuries and osteoarthritis in the non-dominant leg of former professional football players. Wien Klin Wochenschr. 2010;122(Suppl 2):40–43. | ||
Migliore A, Giovannangeli F, Bizzi E, et al. Viscosupplementation in the management of ankle osteoarthritis: a review. Arch Orthop Trauma Surg. 2011;131(1):139–147. | ||
Migliore A, Bizzi E, Massafra U, et al. Viscosupplementation: a suitable option for hip osteoarthritis in young adults. Eur Rev Med Pharmacol Sci. 2009;13(6):465–472. | ||
Migliore A, Giovannangeli F, Granata M, Laganà B. Hylan g-f 20: review of its safety and efficacy in the management of joint pain in osteoarthritis. Clin Med Insights Arthritis Musculoskelet Disord. 2010;3:55–68. | ||
Bannuru RR, Vaysbrot EE, Sullivan MC, McAlindon TE. Relative efficacy of hyaluronic acid in comparison with NSAIDs for knee osteoarthritis: a systematic review and meta-analysis. Semin Arthritis Rheum. 2014;43(5):593–599. | ||
Reid MC. Viscosupplementation for osteoarthritis: a primer for primary care physicians. Adv Ther. 2013;30(11):967–986. | ||
Roque V, Agre M, Barroso J, Brito I. Managing knee ostheoarthritis: efficacy of hyaluronic acid injections. Acta Reumatol Port. 2013;38(3):154–161. | ||
Miller LE, Block JE. US-approved intra-articular hyaluronic acid injections are safe and effective in patients with knee osteoarthritis: systematic review and meta-analysis of randomized, saline-controlled trials. Clin Med Insights Arthritis Musculoskelet Disord. 2013;6:57–63. | ||
Miller LE, Block JE. US-Approved Intra-Articular Hyaluronic Acid Injections are Safe and Effective in Patients with Knee Osteoarthritis: Systematic Review and Meta-Analysis of Randomized, Saline-Controlled Trials. Clin Med Insights Arthritis Musculoskelet Disord. 2013;6:57–63. | ||
Chang KV, Hung CY, Aliwarga F, Wang TG, Han DS, Chen WS. Comparative effectiveness of platelet-rich plasma injections for treating knee joint cartilage degenerative pathology: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95(3):562–575. | ||
Khoshbin A, Leroux T, Wasserstein D, et al. The efficacy of platelet-rich plasma in the treatment of symptomatic knee osteoarthritis: a systematic review with quantitative synthesis. Arthroscopy. 2013;29(12):2037–2048. | ||
Lequesne MG, Mery C, Samson M, Gerard P. Indexes of severity for osteoarthritis of the hip and knee. Scand J Rheumatol. 1987; (Suppl 65):85–89. | ||
Hoeksma HL, van den Ende CH, Breedveld FC, Ronday HK, Dekker J. A comparison of the OARSI response criteria with patient’s global assessment in patients with osteoarthritis of the hip treated with a non-pharmacological intervention. Osteoarthritis Cartilage. 2006;14(1):77–81. | ||
Patience MA, Kilpatrick MW, Sun H, Flory SB, Watterson TA. Sports game play: a comparison of moderate to vigorous physical activities in adolescents. J Sch Health. 2013;83(11):818–823. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.